Abstract
Sex affects the presentation, treatment, and outcomes of abdominal aortic aneurysm (AAA). Although AAAs are less prevalent in women, at least in the general population, women with an AAA have a poorer prognosis in comparison to men. Sex differences in the genetic predisposition for aneurysm disease remain to be established. In this study we investigated the familial risk of AAA for women compared to men. All living AAA patients included in a 2004–2012 prospective database were invited to the multidisciplinary vascular/genetics outpatient clinic between 2009 and 2012 for assessment of family history using detailed questionnaires. AAA risk for male and female relatives was calculated separately and stratified by sex of the AAA patients. Families of 568 AAA patients were investigated and 22.5% of the patients had at least one affected relative. Female relatives had a 2.8-fold and male relatives had a 1.7-fold higher risk than the estimated sex-specific population risk. Relatives of female AAA patients had a higher aneurysm risk than relatives of male patients (9.0 vs 5.9%, p = 0.022), corresponding to 5.5- and 2.0-fold increases in aneurysm risk in the female and male relatives, respectively. The risk for aortic aneurysm in relatives of AAA patients is higher than expected from population risk. The excess risk is highest for the female relatives of AAA patients and for the relatives of female AAA patients. These findings endorse targeted AAA family screening for female and male relatives of all AAA patients.
Keywords
Introduction
Cardiovascular disease (CVD) is the leading cause of morbidity and mortality, accounting for one of every three deaths. 1 The unique aspects of cardiovascular health in women as well as the sex differences in the prevention, diagnosis, and treatment of CVD are increasingly being recognized. Sex specific data on CVD thus far have mainly focused on ischemic heart disease, demonstrating a unique female pattern characterized by a higher prevalence of angina, a lower burden of obstructive coronary artery disease, and a poorer prognosis in comparison to males. 2
Sex differences in other vascular diseases, such as aortic aneurysm, have gained much less attention. Abdominal aortic aneurysm (AAA) occurs in approximately 6% (range 4–8%) of men and 1% (range 1–2%) of women in the general population above the age of 55 years.3–8 Although AAAs are about four times less common in women than in men, those who develop an AAA seem to have a worse outcome than men. 9 AAAs in women have faster growth rates, rupture at smaller diameters, and have higher rupture rates at any given diameter.9–11 In case of rupture, women are less likely to undergo aortic repair and have inferior outcomes after open or endovascular emergency repair.12–15 Women also have higher complication rates for elective endovascular aneurysm repair (EVAR). 16
While family history, age, smoking, and hypertension are the main clinical risk factors for developing an AAA, 17 sex specific differences in determinants of the pathophysiology of AAA are largely unknown. Depending on the method of assessment, in 15–25% of AAA patients familial clustering of aneurysm disease is reported (Tables S1 and S2, Supplemental data). However, the majority of these family studies that indicate a clear genetic predisposition for aneurysm disease were directed at finding aneurysms in the male relatives of mostly male patients. Very little is known about familial risk for female relatives or if there are different familial risks for female and male AAA patients.
Since AAAs are still underdiagnosed and undertreated in women, identification of a specific high-risk category of women that benefits from selective family screening may be a cost-effective way to reduce AAA related morbidity and mortality in the female population. Therefore, the aim of the current study was to answer two questions using family history cohort analysis. First, is there a difference in the risk of developing an aneurysm between female and male relatives of an AAA patient (i.e. do female relatives of an AAA patient have a higher risk of developing an AAA than male relatives)? Second, is there a difference in familial clustering of aortic aneurysm disease between female and male AAA patients (i.e. do family members of a female AAA patient have a higher risk of developing an AAA than relatives of a male AAA patient)?
Methods
Patients and design
In line with the recommendations of the Society for Vascular Surgery (SVS) and European Society for Vascular Surgery (ESVS) guidelines,18,19 to include screening of relatives in routine clinical care of AAA patients, a multidisciplinary vascular/genetics outpatient clinic was set up in 2009. To determine the risk for relatives of AAA patients, a cross-sectional, observational, single-centre cohort study was performed on a prospective database of all consecutive patients who presented with an AAA at the vascular surgical outpatient clinic of the Erasmus University Medical Centre for AAA between 2004 and 2012. AAA was defined as an external maximum transverse abdominal aortic diameter ⩾30 mm. 18 No routine molecular testing was performed for known genetic aortic aneurysm disorders (e.g. Marfan, Loeys–Dietz or vascular Ehlers–Danlos), but patients already diagnosed with one of these syndromes were excluded. The Dutch civil registry was consulted at the start of 2009 to establish whether patients eligible for inclusion were alive and to exclude deceased patients. All living AAA patients were invited to the multidisciplinary vascular/genetics outpatient clinic between 2009 and 2012 for routine assessment of family history. Patients who did not respond after one reminder were contacted and interviewed by phone (KvdL). The medical ethics committee of the Erasmus University Medical Centre (MEC-2012-078) approved this study and no informed consent was required according to local directives. The study complies with the Helsinki declaration on research ethics.
Family history questionnaire
Family history of aortic aneurysms in first-, second-, and third-degree relatives was assessed using a semi-structured questionnaire. The questionnaire requested information about the presence of aortic aneurysms in relatives, the types of aneurysms and the age at aneurysm diagnosis. We defined aortic aneurysm as abdominal as well as thoracic aortic aneurysms (TAA). We chose to include TAA since it has been reported that more than one-fourth of the patients with an abdominal aneurysm have a concomitant TAA. 20 The questionnaire also included questions on the medical histories of individual first-degree relatives, with a specific focus on the occurrence of cardiovascular disease. All AAA patients with a first-degree relative with an aortic aneurysm were subsequently invited for genetic counseling (DMK). The diagnosis of AAA in relatives was routinely verified at the department of clinical genetics after written permission was obtained to retrieve the medical records.
Classification of familial AAA
Patients were classified as familial AAA (fAAA) when at least one first-degree relative (i.e. parent, sibling or child) was reported with an aortic aneurysm. Patients who did not report a first-degree relative with an aortic aneurysm were classified as sporadic AAA (spAAA). Since the reporting of medical information of second- or third-degree relatives is considered less reliable and it is difficult to retrieve the medical records of distant relatives, 21 patients reporting only affected second- or third-degree relatives were also classified as spAAA.
Clinical characteristics
The clinical characteristics of the index AAA patients were obtained from medical files and included sex, age at diagnosis, age at surgery, body mass index (BMI), as well as AAA specifics, cardiovascular comorbidities and risk factors. AAA specifics included location of the aortic aneurysm (i.e. suprarenal or infrarenal), and whether the AAA was ruptured or not. Cardiovascular comorbidities included congestive heart failure, ischemic heart disease (history of myocardial infarction, angina pectoris, coronary revascularization or pathologic Q-waves on the electrocardiogram), and cerebrovascular disease (history of ischemic/hemorrhagic stroke or transient ischemic attack). Cardiovascular risk factors included kidney disease (serum creatinine ⩾2.0 mg/dL), diabetes (fasting plasma glucose ⩾7.0 mmol/L, non-fasting glucose ⩾11.1 mmol/L, or use of anti-diabetic medication), hypertension (blood pressure ⩾140/90 mmHg in non-diabetics, ⩾130/80 mmHg in diabetics, or use of antihypertensive medication) and hypercholesterolemia (low-density lipoprotein (LDL) cholesterol ⩾3.5 mmol/L, or use of lipid lowering medication). Smoking status included current smoking and ever smoking. Prescription medications included the use of statins, beta-blockers, renin-angiotensin system inhibitors, diuretics, and antiplatelets.
Statistical analysis
Dichotomous data are described as counts and percentages. Continuous variables are described as means and standard deviations. Categorical data were compared using chi-squared tests. Continuous variables were compared using ANOVA. Increase in risk was estimated for male and female relatives of AAA patients. Sex specific AAA risks were compared to the pooled estimate AAA risk in the general population (i.e. 5.6% for men and 1.4% for females over the age of 55 years).3,5–8 Multivariable logistic regression analysis was used to investigate sex differences. The dependent variable in the model was sex, and female sex was used as the reference category. Independent variables included in the model were age ⩽65 years at diagnosis, BMI, location of the aneurysm, congestive heart failure, ischemic heart disease, cerebrovascular disease, kidney disease, diabetes mellitus, hypertension, hypercholesterolemia, and ever smoking. Variables were chosen on the basis of biological plausibility. A two-sided p < 0.05 was considered significant. All analyses were performed using IBM SPSS Statistics, version 20.0 (IBM Corp., Armonk, NY, USA).
Results
Participants
Between 2004 and 2012, 780 patients were diagnosed with an AAA at the outpatient clinic. Of the 600 patients who were alive between 2009 and 2012, 482 (80.3%) returned the family history questionnaire after one reminder, while an additional 108 questionnaires were administered by telephone. Family data were missing for 10 patients, representing a response rate of 98.3% (590/600). Pedigree analysis of the 590 completed questionnaires revealed that 22 patients were family members of other participating AAA patients. In these cases, the first family member who was diagnosed with AAA was considered as the index patient, while the other relatives were defined as affected family members. Thus, 568 index patients with an AAA were included for family history analysis. The clinical characteristics of these patients are displayed by sex in Table 1.
Clinical characteristics of all AAA patients.
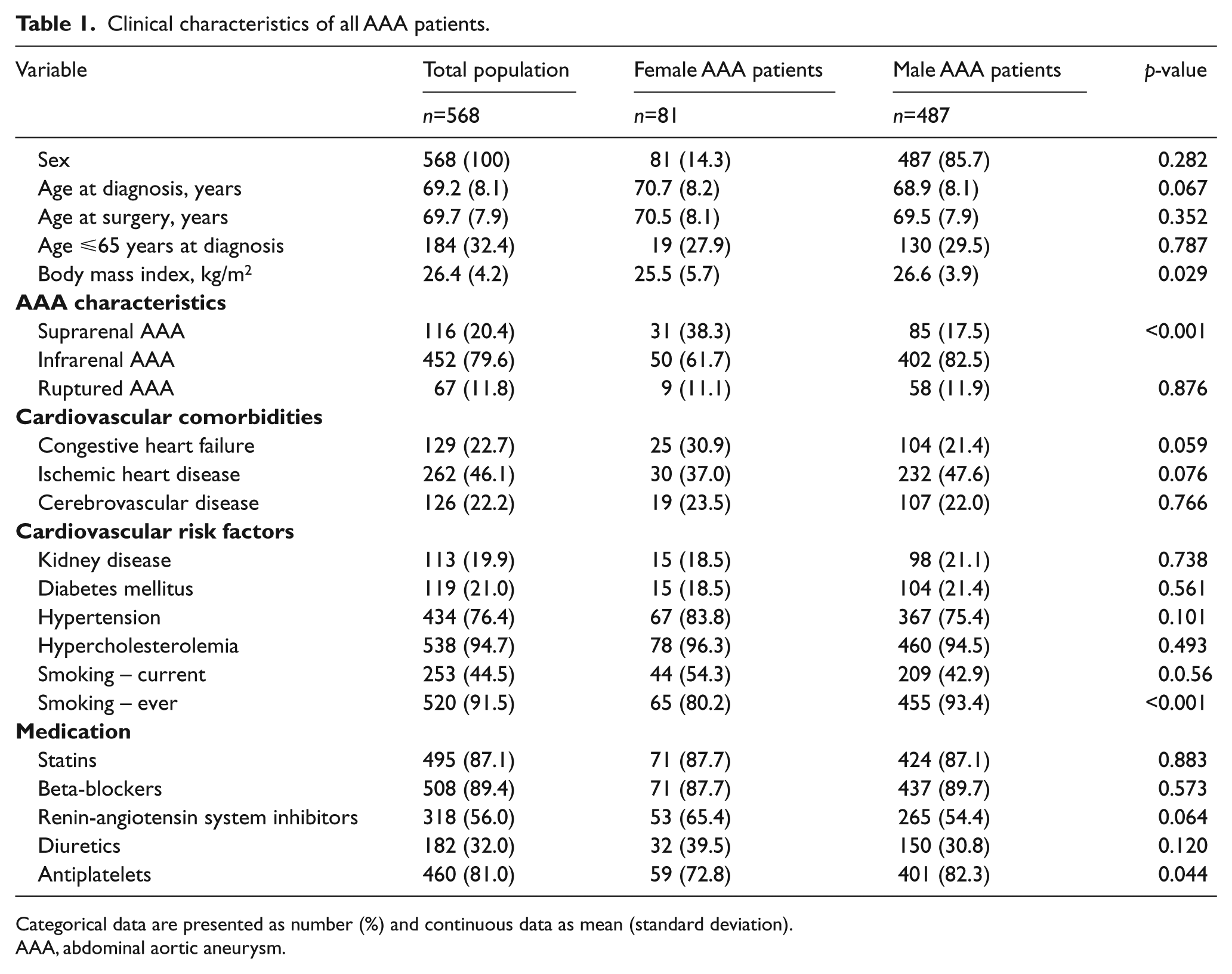
Categorical data are presented as number (%) and continuous data as mean (standard deviation).
AAA, abdominal aortic aneurysm.
Familial AAA
Familial occurrence of AAA was reported by 128 of the 568 index AAA patients, indicating a prevalence of 22.5% in this study population. In 66% of these cases the invitation for genetic counseling was accepted, allowing a review of the medical records of the relatives of 72 fAAA patients (56.3%). In all of these verified cases, the positive reports of aortic aneurysms in relatives of AAA patients were confirmed (i.e. there were no false positive reports). All verified relatives who were affected by aneurysm disease were diagnosed with an AAA and not a TAA. In 42 families, the medical records of relatives were not available for review because they could not be retrieved (n=13) or because the index patient died during the course of the study (n=29). Fourteen fAAA patients refused genetic counseling.
Since the occurrence of AAA is age-related, only three children (two sons and one daughter) with an aortic aneurysm were identified; hence, offspring was not included in our analyses. Of the 128 index patients with fAAA, 87 (68.0%) reported one affected relative, 30 patients (23.4%) reported two affected, and 11 patients (8.6%) reported three or more affected relatives. There was a small but significant difference in average family size between fAAA and spAAA patients (6.4 vs 4.7, p < 0.001).
Sex differences
In the overall study population, 81 of the 568 index patients (14.3%) were women. In univariable analysis, sex differences were observed for BMI, location of aortic aneurysm, ever smoking, and antiplatelet use (Table 1). Multivariable logistic regression analysis showed that female AAA patients had a higher rate of suprarenal AAA compared to male patients (38.3 vs 17.5%, odds ratio (OR) 3.04, confidence interval (CI) 1.8–5.26; p < 0.001), a lower BMI (25.5 vs 26.6, OR 0.93, CI 0.88–0.99; p = 0.033), more congestive heart failure (30.9 vs 21.4%, OR 1.91, CI 1.06–3.44;p = 0.032), and fewer women had ever smoked (80.2 vs 93.4%, OR 0.21, CI 0.10–0.42; p < 0.001).
The prevalence of fAAA was comparable for female and male index patients (27.2 vs 21.8%, p = 0.28). In the overall study population of 568 AAA patients, 6.4% of all parents and siblings were reported to have an aortic aneurysm (Table 2). When stratified for sex of the relatives, 4.0% (53/1331) of the female relatives and 9.5% (130/1375) of the male relatives were affected with aortic aneurysm (Table 3). This corresponded with an unadjusted 2.8-fold increase in AAA risk for female relatives and a 1.7-fold increase in risk for male relatives as compared to the estimated general population risk. The increase in risk was larger for female relatives than male relatives because the population risk for AAA is lower for women than for men. Next we examined if the sex of the index case (i.e. the patient who presented with an AAA at the outpatient clinic) had an effect on risk for family members. AAA was significantly more common among the relatives of female AAA patients as compared to the relatives of male cases (9.0 vs 5.9%, p = 0.022; Table 2 and Figure 1). Compared with the estimated general population risk, the female and male relatives of female patients had 5.5- and 2.0-fold increases in AAA risk, respectively. The relative risks of AAA were 2.4-fold higher for female and 1.6-fold higher for male relatives of male AAA patients (Table 3 and Figure 1).
First-degree relatives with aortic aneurysm of 568 AAA patients.

AAA, abdominal aortic aneurysm.
Specified data were available for 1571 siblings.
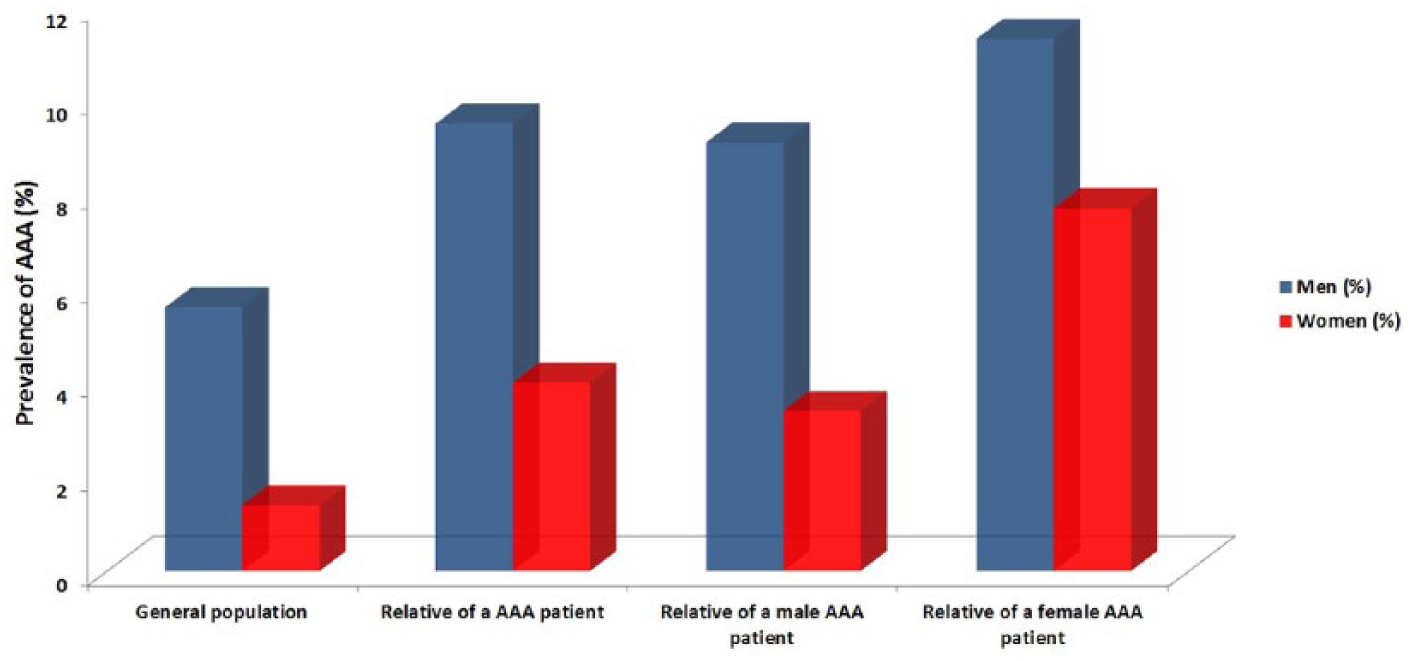
Sex-related risk for relatives of abdominal aortic aneurysm (AAA) patients.
Sex-specific risk for relatives of AAA patients.
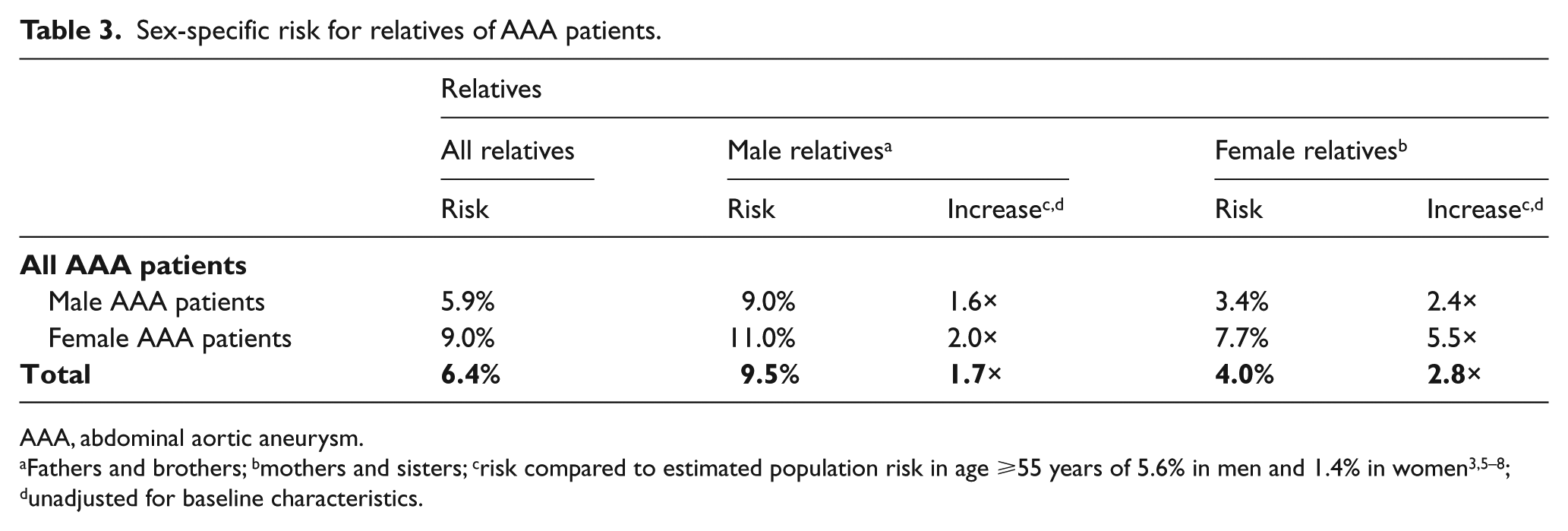
AAA, abdominal aortic aneurysm.
Discussion
This study shows that familial occurrence of AAA is common, affecting one in four patients with an AAA. We found that relatives of AAA patients have an increased risk for aneurysms compared to the population risk. Female relatives have a much higher increase in risk compared to male relatives (2.8- vs 1.7-fold, respectively), although still more male relatives are affected. In addition, relatives of female AAA patients carry a higher risk of developing AAA than relatives of male AAA patients. This stresses the need to focus on targeted family screening, in particular for families of women who present with an AAA.
The AAA risk of family members in this Dutch study was compared to the estimated population prevalence of AAA as derived from the large European screening programs that were used in the current European aneurysm guideline. 18 Some more recent studies report a lower population prevalence of AAA,22–24 possibly due to differences in the methodology of AAA measurement. Since we did not want to overestimate our results with regard to increases in aneurysm risk for relatives as compared to the general population, we chose the population data from the current guideline as a reference.
The prevalence of an aortic aneurysm in 4% of all female relatives of AAA patients, irrespective of the presenting patient’s sex, is much higher than the prevalence of AAA among women in the general population. Previous reports on aneurysm risks for female relatives are scarce. Only five of the 13 family history studies and two of the 20 studies on ultrasound AAA family screening reported sex-specific risks (Tables S1 and S2, Supplemental data).25–31 In line with our study, they all showed higher AAA occurrence in families of female patients and among female relatives.
It is well known that fewer women have AAA than men, suggesting a sex specific susceptibility for AAA in men. However, as hypothesized for other complex genetic disorders occurring more frequently in men (e.g. pyloric stenosis), the genetic susceptibility that could be described as a genetic ‘burden’ necessary to develop the disease, is lower for men than for women. According to this theory, affected women are expected to have a higher genetic ‘burden’. This may explain that relatives of female patients are at a higher risk to develop aortic aneurysms than relatives of male patients, and underlines the importance of including families of female patients and female relatives in screening programs. 32 The male preponderance in familial AAA may reflect that similar sex related risk factors contribute to familial AAA and sporadic AAA. Alternatively, it may indicate the occurrence of phenocopies (i.e. the occurrence of a sporadic form of AAA in families with familial AAA).
The findings of our study have important clinical implications. First, there are diverging recommendations in the current aneurysm guidelines on the targeted family screening of relatives of AAA patients. For example, the American College of Cardiology (ACC)/American Heart Association (AHA) guideline recommends screening of only male relatives, whereas the ESVS guideline recommends screening of all relatives in case of a family history of AAA (Table S3, Supplemental data).18,33 Considering their significantly increased risks, our data suggest focusing on AAA risk for female relatives and relatives of female AAA patients. Thus, our findings support targeted family screening of all (first-degree) relatives of all AAA patients, irrespective of sex. Second, based on trials showing that screening of men over the age of 65 years was effective in reducing mortality from aortic aneurysm rupture,18,34–36 population screening programs have been initiated in several countries, focusing on the male elderly population. 37 Recent data from these programs across the United Kingdom, Sweden and United States, however, showed a lower incidence of AAA than expected, raising questions on the efficacy of population screening.22–24 Given the discussions about the cost-effectiveness of nationwide screening programs, our data suggest that screening for aneurysms in family members of AAA patients may be a valuable alternative to screening in the general population.
No conclusions can be drawn about the risks for children from our data, since we did not include offspring of AAA patients because these relatives were generally too young to have aortic aneurysms yet. 18 European guidelines advise starting screening from the age of 50 years. 18 This threshold is based on the finding that 7% of AAA patients worldwide are younger than 60 years, 38 and an increase in abdominal aortic diameter is expected to start several years prior to the average age of presentation. 39 Since it is unknown whether familial abdominal aneurysms have different expansion rates than the average of 2–3 mm annually, 18 it seems reasonable to keep relatives with a small aneurysm under surveillance as proposed in the current guidelines.
Limitations
Our study has several limitations. First, the data are based on family history and not on aortic imaging of complete families. Validation of the family history data in over half of our study population, however, showed that no relatives were incorrectly reported as affected, indicating that the AAA risk in relatives was not overestimated. On the contrary, since AAA is usually asymptomatic, underreporting of familial disease is more likely. Few patients in this study reported only second- or third-degree relatives with an aneurysm. In the absence of reliable hospital records of distant relatives, these patients were classified as sporadic AAA. Exclusion of these patients from the familial AAA group may have introduced underreporting of familial disease rather than an overrepresentation of the sex specific familial risk. Second, since there is no population-wide screening program for AAA in the Netherlands, the reported AAA prevalence in the presenting group might be an underestimation of the true aneurysm prevalence in the Dutch population. However, in terms of sex, AAAs were incidental findings in both female and male index patients and their relatives. Third, underreporting of AAA in females may be related to the fact that given the smaller body size, a dilatation of the aorta in a woman represents a greater relative dilatation of the aorta compared with the same diameter in a man. However, since none of the methods to index aortic diameter to some measure of body size has been incorporated into clinical practice yet, we defined AAA as an aortic diameter ⩾3 cm for both men and women. Size adjustment would potentially increase the already higher AAA risk for female relatives. Last, the predominance of individuals of European ancestry in our population may limit the generalizability of our results to other ethnicities and other parts of the world.
Conclusion
The risk for aortic aneurysm in relatives of AAA patients is much higher than expected from the population risk. This excess risk is highest for the female relatives of AAA patients and for the relatives of female AAA patients. Our findings may lead to the adjustment and expansion of the current recommendations for targeted family screening in order to improve early detection of aneurysms in both female and male relatives of all AAA patients. However, the cost-effectiveness of such an approach as well as its effect on aneurysm-related mortality remains to be determined.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KM van de Luijtgaarden is supported by an unrestricted grant from the Lijf & Leven Foundation, Rotterdam, the Netherlands.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
