Abstract
Pathogenesis of abdominal aortic aneurysm (AAA) is unclear. The aim of this study was to evaluate inflammatory and hemodynamic plasma biomarkers as predictors for AAA in the prospective longitudinal cohort of middle-aged individuals from the cardiovascular cohort of the Malmö Diet and Cancer Study (n=5551; 1991–94). C-reactive protein, cystatin C, copeptin, N-terminal pro-B-type natriuretic peptide (N-BNP), midregional pro-atrial natriuretic peptide (MR-proANP) and conventional risk factors at baseline were measured in patients with incident AAA during follow-up and compared to individuals without a diagnosis of AAA. Subjects were followed until 31 December 2013. Multivariable analyses were expressed in terms of hazard ratios (HR) per 1 standard deviation increment of each respective log-transformed plasma biomarker in the Cox proportional hazard models. Mean follow-up time was 20.7 years. Cumulative incidence of AAA was 1.5% (men 2.9%, women 0.5%). Mean age of individuals with incident AAA was 59.7 years at study entry and AAA was diagnosed on average 14 years later. Adjusting for age, sex, smoking, body mass index, hypertension and diabetes mellitus, N-BNP (HR 1.29; 95% CI 1.03–1.62), but not MR-proANP (HR 1.20; 95% CI 0.95–1.50), was independently associated with incident AAA. In conclusion, the plasma biomarker N-BNP was associated with future development of AAA, which implies that this marker is a sensitive indicator of early subclinical cardiovascular disease.
Introduction
The natural history of abdominal aortic aneurysm (AAA) is progressive growth and subsequent rupture in a proportion of patients with a very high overall mortality exceeding 80%. 1 The pathogenesis of AAA features several hallmarks of atherosclerotic and atherothrombotic disease, such as inflammation with leukocyte infiltration, degradation of extracellular matrix and depletion of vascular smooth muscle cells. 2 It also compromises an additional, predominant feature of proteolysis resulting in degradation and destabilization of the aortic wall, with accumulation of neutrophils in the intraluminal thrombus. 3 AAA has been strongly associated with conventional risk factors for atherosclerosis such as age, tobacco use, male sex and hypertension, 4 whereas diabetes mellitus seems to be a protective factor. 5
Plasma biomarker studies of AAA pathogenesis have often been restricted by their cross-sectional design, not allowing differentiation between biomarkers related to AAA development and those associated with presence of AAA. To date, meta-analysis of cross-sectional case–control studies suggests that increased levels of several proteases, inflammatory markers, acute-phase reactants and some lipids, as well as decreased levels of two other lipids (apolipoprotein A and high-density lipoprotein), are all associated with AAA development, but there is significant heterogeneity between studies. 6
Plasma biomarkers associated with heart and renal failure have been established in cardiovascular disease research. Cystatin C is a marker of both vascular inflammation and renal function. 7 The plasma biomarkers copeptin (a stable vasopressin marker), 8 and the natriuretic peptides N-terminal pro-B-type natriuretic peptide (N-BNP) 9 and midregional pro-atrial natriuretic peptide (MR-proANP) 9 have all been proposed as markers for development of cardiovascular disease. Data on associations between these biomarkers and incident AAA has, however, been lacking.
The main aim of the present study was to evaluate a set of inflammatory and hemodynamic plasma biomarkers together with conventional risk factors for prediction of incident AAA in a large Swedish longitudinal, population-based, prospective cohort study of middle-aged individuals.
Materials and methods
Study sample
The population-based Malmö Diet and Cancer Study (MDCS) 10 included 30,447 individuals: men born 1923–1945 and women born 1923–1950 from Malmö, Sweden. Participants attended baseline examinations between 1991 and 1996, with sampling of peripheral venous blood and collection of clinical characteristics. Participants underwent anthropometric measurements, blood pressure (BP) measurement using a mercury-column sphygmomanometer in the supine position after 10 minutes of rest, and completed a questionnaire including questions about smoking and medication. Smoking was defined as self-reported regular smoking or smoking cessation within the last year. Diabetes mellitus was defined as self-reported physician’s diagnosis, use of anti-diabetic medication, or fasting whole blood glucose >6.0 mmol/L when available. Hypertension was defined as use of antihypertensive medication or BP ≥140/90 mmHg. From this cohort, a random sample, examined between November 1991 and February 1994 was included in the MDCS CV cohort, 11 of whom 5551 individuals underwent risk factor evaluation and blood sampling under standardized fasting conditions (Figure 1). Individuals (n=29) with known AAA at baseline were excluded. No screening of AAA with ultrasound was performed in the study participants at study entry.
Descriptive flow diagram of study participants and plasma biomarker data. AAA, abdominal aortic aneurysm; CRP, C-reactive protein; N-BNP, N-terminal pro-B-type natriuretic peptide; MR-proANP, midregional pro-atrial natriuretic peptide.
Informed consent was obtained from all participants and the study was approved by the regional ethical review board in Lund, Sweden (Dnr 2013/566). The investigation conforms to the principles outlined in the Declaration of Helsinki.
Endpoint ascertainment
Individuals from the MDCS with a first registered diagnosis of AAA, ruptured AAA, or surgical procedure for AAA were identified from the Swedish national registers (the Inpatient and Outpatient Register and the Cause of Death Register) by linkage of the 10-digit personal identification number unique to each Swedish resident. All subjects were followed up from the baseline examination until first AAA event, emigration from Sweden, mortality, or end of follow-up at 31 December 2013. The Inpatient Register includes information on dates of admission and discharge as well as diagnostic and procedural codes from all hospitalizations in Sweden. The Cause of Death Register includes diagnoses of underlying and contributing causes of death from death certificates. In both validated 12 registers, diagnoses are coded using a Swedish revision of the International Classification of Disease (ICD), versions 8, 9 and 10, and surgical procedures are coded using a Swedish classification system. 13
Laboratory measurements
The blood samples collected in ethylenediaminetetraacetic acid (EDTA) tubes were centrifuged at 300 × g for 10 minutes with removal of plasma, followed by a second centrifugation at 2000 × g for 10 minutes to remove platelets and then banked in 2 × 2 mL of plasma samples. Plasma biomarkers were measured from fasting plasma samples that had been frozen at −80°C immediately after collection. 14 C-reactive protein (CRP) was measured by a high-sensitivity Tina-quant® latex assay (Roche Diagnostics, Basel, Switzerland). Cystatin C was measured using a particle-enhanced immune-nephelometric assay (N Latex Cystatin C; Siemens Diagnostics, Dade Behring, Deerfield, IL, USA). Copeptin was measured using a commercially available assay in the chemiluminescence/coated tube format (BRAHMS AG, Henningsdorf, Germany). The lower detection limit was 0.4 pmol/L and the functional assay sensitivity (< 20% interassay coefficient of variation) was < 1 pmol/L. 15 MR-proANP were measured using immunoluminometric sandwich assays targeted against amino acids in the midregion of the peptide (BRAHMS AG, Berlin, Germany). N-BNP was measured using the automated Dimension Vista Intelligent Lab System method (Siemens Diagnostics, Nürnberg, Germany). 16 The mean interassay coefficients of variation were 4.6% for CRP, 4.3% for cystatin C, ≤ 10% for MR-proANP and 2.7% for N-BNP. At baseline, fasting total cholesterol and triglycerides were measured according to standard procedures at the Department of Clinical Chemistry, Skåne University Hospital, Malmö. HbA1c was determined by ion exchange chromatography using the Swedish Mono-S standardization system; reference values were 3.9–5.3% in non-diabetic individuals.
Statistical analysis
Quantitative normal and skewed distributed variables are presented as mean with standard deviation and median with interquartile range (IQR), respectively. Dichotomous variables are presented as count and proportion. Differences between groups were assessed with the Mann–Whitney U test for continuous non-parametric variables, the t-test for continuous parametric variables and with the chi-squared test for nominal variables. The endpoint studied in subsequent prospective analyses was incident AAA. Plasma biomarkers for incident AAA were assessed using Cox proportional hazards regression models, tested in age- and sex-adjusted models. Owing to skewed distributions, log-transformed values of CRP, cystatin C, copeptin, MR-proANP and N-BNP were used in the Cox models. Conventional risk factors for AAA, such as age at study entry, sex, body mass index (BMI), current smoking, diabetes mellitus, hypertension and each plasma biomarker, were included in a multivariable-adjusted model where all variables were simultaneously entered into the model. Hazard ratios (HRs) with 95% confidence intervals (CI) were expressed per 1 standard deviation (SD) increment of each respective log-transformed plasma biomarker in the Cox proportional hazard models. Analyses were performed using SPSS for Windows, version 23.0 (SPSS Inc., Chicago, IL, USA). A two-sided p < 0.05 was considered significant.
Results
Baseline conventional risk factor assessment
The mean follow-up time was 20.7 years. The cumulative incidence of AAA was 1.5% (84/5551): 2.9% (67/2308) for men and 0.5% (17/3243) for women. Of the 84 individuals with incident AAA during follow-up, 22 (26.2%) were operated on and 16 (19.0%) had ruptured. Baseline risk factor characteristics for individuals with or without AAA in the cohort are shown in Table 1.
Baseline characteristics in subjects with and without later development of AAA.

AAA, abdominal aortic aneurysm; SD, standard deviation; IQR, interquartile range; CRP, C-reactive protein; N-BNP, N-terminal pro-B-type natriuretic peptide; MR-proANP, midregional pro-atrial natriuretic peptide.
Plasma biomarkers
Baseline values of the evaluated plasma biomarkers in individuals with and without incident AAA are shown in Table 1. In the age- and sex-adjusted model, CRP (HR 1.40 per 1 SD increment of log-transformed CRP; 95% CI 1.12–1.73) and N-BNP (HR 1.28; 95% CI 1.02–1.60) were strongly associated with incident AAA (Table 2). In the multivariable-adjusted analysis, only N-BNP (HR 1.29; 95% CI 1.03–1.62) was independently associated with incident AAA (Table 3).
Age- and sex-adjusted hazard ratios for inflammatory and hemodynamic plasma biomarkers at baseline in subjects who later developed AAA.
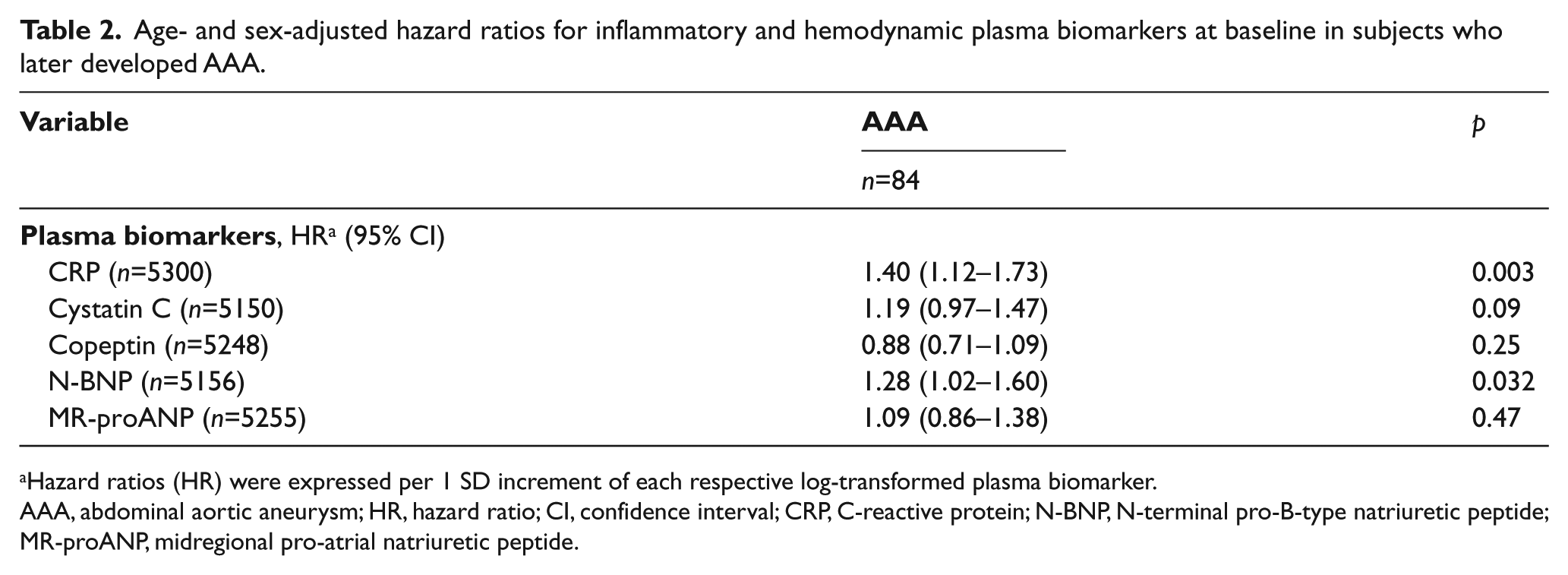
Hazard ratios (HR) were expressed per 1 SD increment of each respective log-transformed plasma biomarker.
AAA, abdominal aortic aneurysm; HR, hazard ratio; CI, confidence interval; CRP, C-reactive protein; N-BNP, N-terminal pro-B-type natriuretic peptide; MR-proANP, midregional pro-atrial natriuretic peptide.
Multivariable analysis of inflammatory and hemodynamic plasma biomarkers at baseline in subjects who later developed AAA.
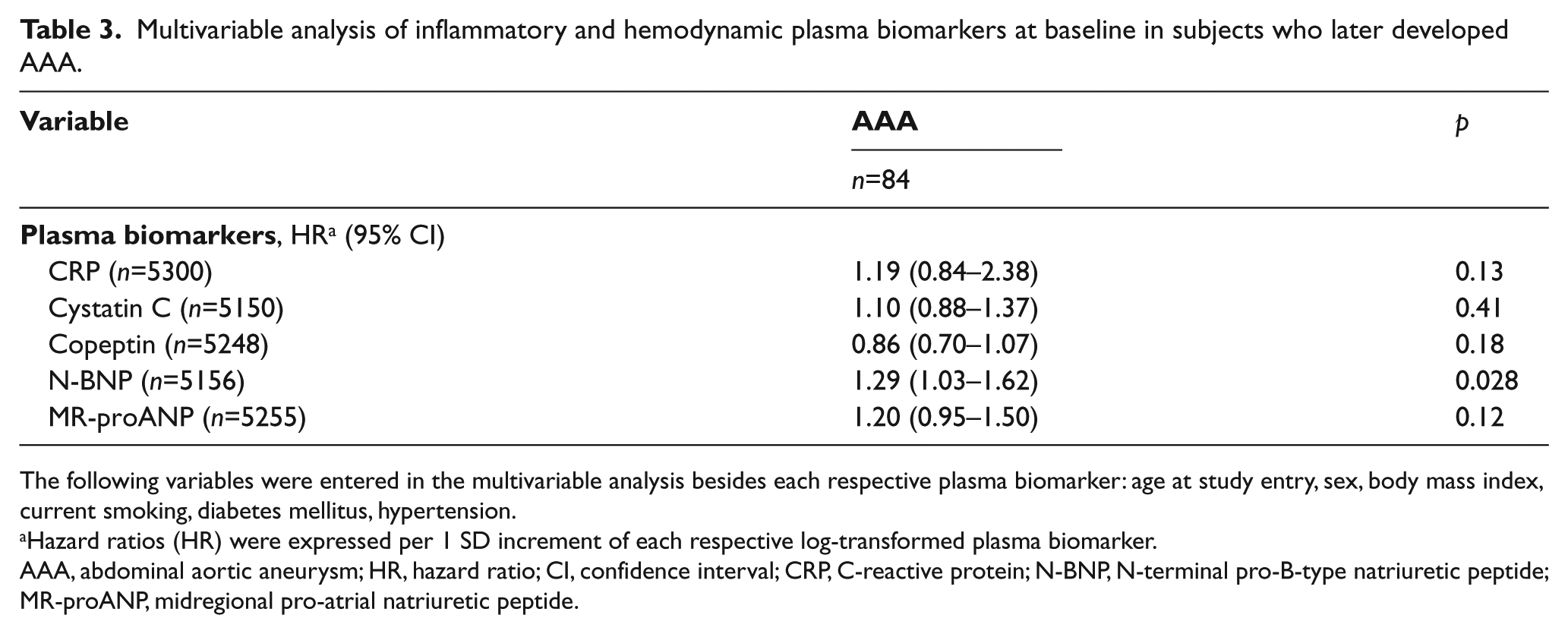
The following variables were entered in the multivariable analysis besides each respective plasma biomarker: age at study entry, sex, body mass index, current smoking, diabetes mellitus, hypertension.
Hazard ratios (HR) were expressed per 1 SD increment of each respective log-transformed plasma biomarker.
AAA, abdominal aortic aneurysm; HR, hazard ratio; CI, confidence interval; CRP, C-reactive protein; N-BNP, N-terminal pro-B-type natriuretic peptide; MR-proANP, midregional pro-atrial natriuretic peptide.
Discussion
In the present study, levels of N-BNP adjusted for conventional confounders were independently associated with incident AAA during long-term follow-up. This suggests that elevated N-BNP levels reflect susceptibility for later development of clinically relevant AAA. As a high proportion of study individuals with incident AAA were hospitalized due to large AAA, underwent operation, or suffered rupture, they might have been exposed to pathophysiological mechanisms promoting AAA growth for a long time before diagnosis of AAA. In a recent report, it was found that the same plasma biomarkers at baseline were not correlated with abdominal aortic diameters after 14–19 years of follow up, 16 probably reflecting the small difference in risk factor exposure in individuals with aortas within the normal diameter range and the presence of very few identified individuals with AAA. 17
Plasma N-BNP is a well-established biomarker for diagnosis and prognosis of heart failure, 18 whereas potential associations between N-BNP and aortic pathology have been very sparsely investigated. A cross-sectional study showed positive associations between N-BNP levels and aortic diameters in women with and without Turner syndrome. 19 In accordance with the present study, the longitudinal Atherosclerosis Risk in Communities (ARIC) study, which included re-invitation with ultrasound examination of the abdominal aorta, showed that N-BNP was associated with an increased risk of AAA well before the clinical recognition of the disease. 20 Interestingly, the ARIC investigators reported that the more markers an individual had in the highest quartiles of a set of six plasma biomarkers, reflecting inflammation, thrombin generation, cardiac injury and stretching, the greater was his or her future AAA risk, reflecting the multiple pathways in the origin of AAA. 20 The view that multiple biomarkers may identify a subgroup of individuals with a high risk of AAA has further been strengthened by previous findings of elevated plasma levels of lipoprotein-associated phospholipase A2 21 and adrenomedullin 22 in individuals later developing AAA.
N-BNP is recognized as a marker of increased cardiac ventricular and auricular filling pressure 23 and vascular resistance. 24 The positive association between N-BNP and incident AAA implicates a potential role for increased peripheral vascular resistance in AAA development and a secondary plasma elevation of N-BNP due to secretion by cardiomyocytes under stretched condition. This view is supported by the fact that individuals with elevated N-BNP and a normal blood pressure at baseline are at an increased risk of developing hypertension, 25 a well-known risk factor for AAA development. 13 Another natriuretic peptide, MR-proANP, was not associated with AAA risk. In relation to N-BNP, MR-proANP secretion is more strongly related to atrial than ventricular strain and is a better biomarker for incident atrial fibrillation, whereas BNP is a better marker of heart failure and coronary disease.9,26 Whereas atrial strain is reflective of filling pressures and volume overload, ventricular strain is more highly dependent on cardiac afterload and arterial hypertension, a disease process more strongly related to AAA than volume overload. At the extreme, war veterans who had undergone an above-knee amputation, a condition associated with increased peripheral resistance, arterial hypertension and cardiac afterload, indeed developed a fivefold higher incidence of ultrasound-verified AAA, compared to an evenly matched control group of non-amputees. 27 It is worth noting, however, that MR-proANP displayed a similar but slightly weaker effect estimate for AAA in our study than N-BNP (HR 1.20 vs 1.29 per SD) that may not have reached significance due to limitations in sample size. Until proven otherwise, middle-aged individuals with greater N-BNP are seemingly at increased risk of AAA as part of their generally high risk of incident heart failure 9 and other cardiovascular diseases. Whether serial N-BNP measurements may be used to monitor aortic diameter and/or detect aortic pathology in individuals at risk for aortic disease remains to be evaluated. Although N-BNP was statistically independently associated with incident AAA, the small difference between median N-BNP plasma levels in AAA and non-AAA individuals, with overlapping interquartile ranges in the descriptive statistics, makes this biomarker a poor clinical determinant of future AAA development.
The MDCS is a very large prospective population-based cohort originally designed to explore the effects of diet on cancer risk. 10 The major strength of the present longitudinal study design is the mean age of 59.7 years at study entry among individuals later developing AAA, and the average 14-year time-lag between collection of plasma samples and diagnosis of AAA. The possibility of study entrance of a few individuals with undiagnosed small-sized AAA, however, was not excluded, since ultrasound examination of the abdomen not was performed at baseline. In addition, we relied upon administrative databases and not upon results of ultrasound examination for endpoint ascertainment of AAA, which might have led to misclassification of individuals. The cumulative incidence of mainly large-sized AAA (1.5%) in the MDCS cohort was relative high considering that most individuals in the cohort are women with a much lower AAA incidence 1 and a peak of AAA one decade later 28 than men. Furthermore, the declining autopsy rate, especially among elderly, in the population has most certainly resulted in an underestimation of AAA incidence. 29 Future cohort studies with the aim to study the pathogenesis of AAA might need to have a higher proportion of men, which might, for instance, be achieved by implementing biomarker research in the nationwide screening cohorts in Sweden 30 and the United Kingdom. 31 In Sweden, where all 65-year-old men are invited for ultrasound AAA screening, individuals with dilated aortas are serially examined, 30 enabling better opportunities to perform repeated measurements of biomarkers in relation to AAA growth. 32 In such studies, it would also be interesting to evaluate biomarker profile differences in subjects with large and small aneurysms.
Furthermore, it is highly probable that changes in risk factor status might have occurred during the long follow-up period in our study. During the last decade, decreasing smoking prevalence 33 and improved coverage of cardiovascular medication 34 among individuals with, or at high risk for, cardiovascular disease might well have influenced the incidence of AAA. 29
Conclusion
Elevated levels of the plasma biomarker N-BNP was a marker of increased risk for future development of AAA, implying that this marker is a sensitive indicator of early subclinical cardiovascular disease.
Footnotes
Acknowledgements
The authors wish to thank all participants in the Malmö Diet and Cancer Study for making this study possible.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Malmö Diet and Cancer study was made possible by grants from the Swedish Cancer Society, the Swedish Medical Research Council, the Swedish Dairy Association, the Albert Påhlsson and Gunnar Nilsson Foundations and the Malmö city council. GE and JGS were supported by the Swedish Heart-Lung Foundation (2016-0315) and the Medical Faculty of Lund University. OM, GE and JGS were supported by the Swedish Medical Research Council (2014-2265). OM was supported by grants from the Medical Faculty of Lund University, Skåne University Hospital in Malmö, the Albert Påhlsson Research Foundation, the Crafoord Foundation, the Swedish National Health Service, the Hulda and Conrad Mossfelt Foundation, the Ernhold Lundströms Research Foundation, the King Gustaf V and Queen Victoria Fund, the Lennart Hanssons Memorial Fund, the Marianne and Marcus Wallenberg Foundation and the Knut and Alice Wallenberg Foundation. AG was supported by grants from the Ernhold Lundström Foundation, Research Funds at Skåne University Hospital and at Region Skåne (430751), the Hulda Ahlmroth Foundation and from the Swedish Government under the LUA/ALF agreement. JGS was supported by grants from the European Research Council, Wallenberg Center for Molecular Medicine in Lund, the Crafoord Foundation, Skåne University Hospital and the Swedish National Health Service. PMN was supported by the Swedish Medical Research Council (2013-2756).
