Abstract
We investigated whether family history (FHx) of atherosclerotic cardiovascular disease (ASCVD) was associated with presence of abdominal aortic aneurysm (AAA). The study cohort comprised of 696 patients with AAA (70±8 years, 84% men) and 2686 controls (68±10 years, 61% men) recruited from noninvasive vascular and stress electrocardiogram (ECG) laboratories at Mayo Clinic. AAA was defined as a transverse diameter of abdominal aorta ⩾ 3 cm or history of AAA repair. Controls were not known to have AAA. FHx was defined as having at least one first-degree relative with aortic aneurysm or with onset of ASCVD (coronary, cerebral or peripheral artery disease) before age 65 years. FHx of aortic aneurysm or ASCVD were each associated with presence of AAA after adjustment for age, sex, conventional risk factors and ASCVD: adjusted odds ratios (OR; 95% confidence interval): 2.17 (1.66–2.83, p < 0.01) and 1.31 (1.08–1.59, p < 0.01), respectively. FHx of ASCVD remained associated with AAA after additional adjustment for FHx of aortic aneurysm: adjusted OR: 1.27 (1.05–1.55, p = 0.01). FHx of ASCVD in multiple arterial locations was associated with higher odds of having AAA: the adjusted odds were 1.23 times higher for each additionally affected arterial location reported in the FHx (1.08–1.40, p = 0.01). Our results suggest both unique and shared environmental and genetic factors mediating susceptibility to AAA and ASCVD.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is a permanent dilatation of abdominal aorta conventionally defined as a transverse diameter ⩾ 3.0 cm. 1 It is often asymptomatic until rupture, which is associated with a mortality rate as high as 80%. The prevalence of AAA increases with age, and has been reported to be 12.8% in men and 4.1% in women aged > 65 years. 2 No pharmacological treatment is available to effectively limit disease progression. Early identification followed by elective aneurysm repair has been shown to reduce aneurysm-related mortality. 3 Given the significant disease burden and paucity of treatment options to reduce aneurysm formation and growth, identifying individuals at high risk for AAA may allow tailored screening and improve outcomes. Family history (FHx) is a useful tool for risk assessment, serving as a proxy for genetic predisposition as well as shared environmental factors that contribute to disease development. 4 A positive FHx is a risk factor for coronary heart disease (CHD), cerebrovascular disease (CVD) and peripheral artery disease (PAD).1,5–7 AAA is a multifactorial disease with a significant genetic component8–10 and risk factors that are shared across subtypes of atherosclerotic cardiovascular disease (ASCVD).11,12 Whether FHx of ASCVD is associated with presence of AAA is unknown. We hypothesized that FHx of ASCVD is a risk factor for AAA. To test this hypothesis, we investigated the association of FHx of ASCVD with presence of AAA in a case–control study of patients referred to the Mayo Clinic. A secondary aim of the study was to assess whether FHx of different subtypes of ASCVD and parental vs. sibling history were differentially associated with presence of AAA.
Methods
All participants were from the Mayo Clinic Vascular Disease Biorepository (VDB) initiated in May 2006 to identify genetic susceptibility variants for vascular diseases. The design and selection criteria for VDB have been reported previously. 13 Briefly, participants included patients who underwent noninvasive vascular evaluation or stress electrocardiogram (ECG) at the Mayo Clinic. A questionnaire was given to each participant at the time of consent and scanned into the database after completion. Until August 2013, we had recruited 11,814 participants. The biorepository comprises 8062 participants who had given blood samples, including 1493 individuals without AAA, ASCVD, or rare vascular diseases such as vasculitis, fibromuscular dysplasia, etc. Study questionnaires were available in 5146 out of 8062 participants, including 1015 controls. We excluded 203 participants who were adopted by self-report. A total of 696 participants met the criteria for being AAA cases. As controls we included 1671 participants from the vascular disease group who had ASCVD but not AAA, and 1015 without ASCVD or other vascular disease.
This resulted in a sample of 696 cases and 2686 controls for the analyses (Figure 1). All participants gave informed consent. The study protocol was approved by the Institutional Review Board of the Mayo Clinic.
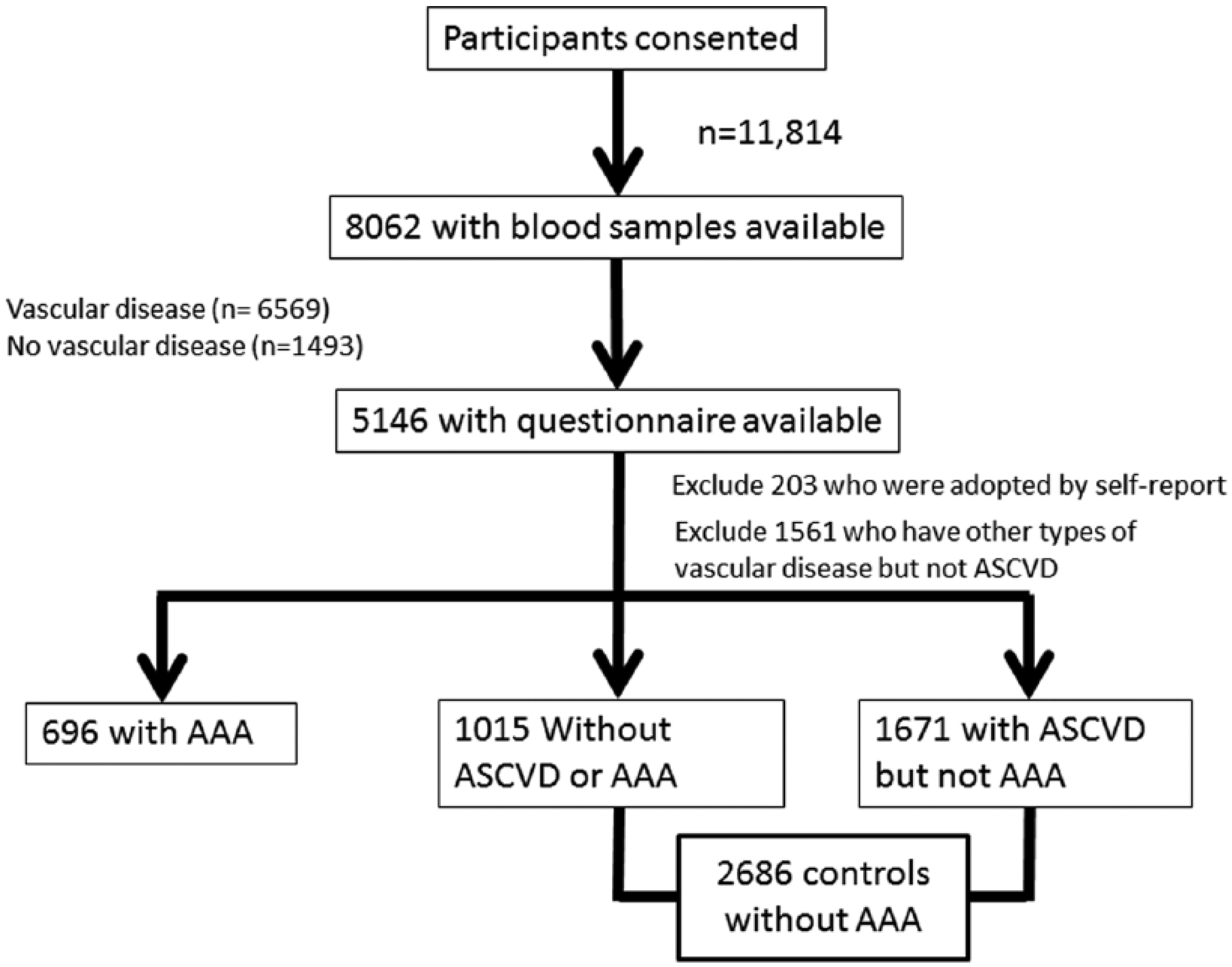
Flow chart of ascertainment of AAA cases and controls in the Vascular Disease Biorepository (VDB). (AAA, abdominal aortic aneurysm; ASCVD, atherosclerotic cardiovascular disease.)
We sampled patients based on their AAA status before 31 December 2014. AAA cases were defined as: (1) a distal, infrarenal or juxtarenal abdominal aortic transverse diameter ⩾ 3 cm, or (2) history of AAA repair. Controls were patients not known to have AAA. Case status was confirmed by manual review. Controls had no ICD-9 (International Classification of Diseases, Ninth Revision) diagnosis codes for AAA. Prevalent ASCVD, family history and conventional risk factors were ascertained from the study questionnaire. ASCVD was considered present based on physicians’ diagnoses of CVD, CHD or PAD, or history of procedures including carotid stenting or endarterectomy, percutaneous coronary intervention or bypass, or revascularization or bypass due to lower extremity arterial stenosis. Hypertension, diabetes and hyperlipidemia were based on self-report (patients were asked if they were ever diagnosed by a physician or were taking antihypertensive, lipid-lowering or hypoglycemic medication), while ever-smoking was defined as a lifetime use of ⩾ 100 cigarettes. Patients were asked if their first-degree relatives – father, mother, full sibling, sons and daughters – previously had a myocardial infarction, coronary revascularization or bypass, stroke, carotid endarterectomy, or lower-extremity revascularization or bypass before age 65, and if they had an aortic aneurysm. Details of the questionnaire have been reported previously.5,6
Statistics
Descriptive statistics were used to compare demographic and conventional cardiovascular risk factors between cases and controls. Continuous variables were presented as mean (with SD) and dichotomous variables were presented as percentages. Comparisons were performed after adjustment for age and sex. To assess the association of FHx of ASCVD with AAA, logistic regression analysis was performed using the presence of AAA as the dependent variable, first without adjustment and then adjusting for age, sex, body mass index (BMI), hypertension, diabetes, smoking, hyperlipidemia and ASCVD. Analyses were performed stratifying by sex as well (see supplementary material). Additionally, we stratified patients based on (1) FHx of CHD, PAD or CVD and (2) parental and sibling history. We repeated the analyses to (1) compare the association of FHx of subtypes of ASCVD and (2) parental/sibling history with presence of AAA. Interactions between prevalent ASCVD and FHx of ASCVD/aortic aneurysm/CHD/CVD/PAD were assessed and included in the multivariable regression analyses. A two-sided p < 0.05 was considered statistically significant. All analyses were performed using the JMP 11.0 (SAS Institute, Cary, NC, USA) software.
Results
Patient characteristics are shown in Table 1. Hypertension, hyperlipidemia, history of smoking, FHx of aortic aneurysm and FHx of ASCVD were present more often in patients with AAA than in controls after accounting for differences in age and sex. Prevalence of ASCVD was similar between AAA cases and controls, while diabetes was less prevalent in cases than in controls.
Patient characteristics.
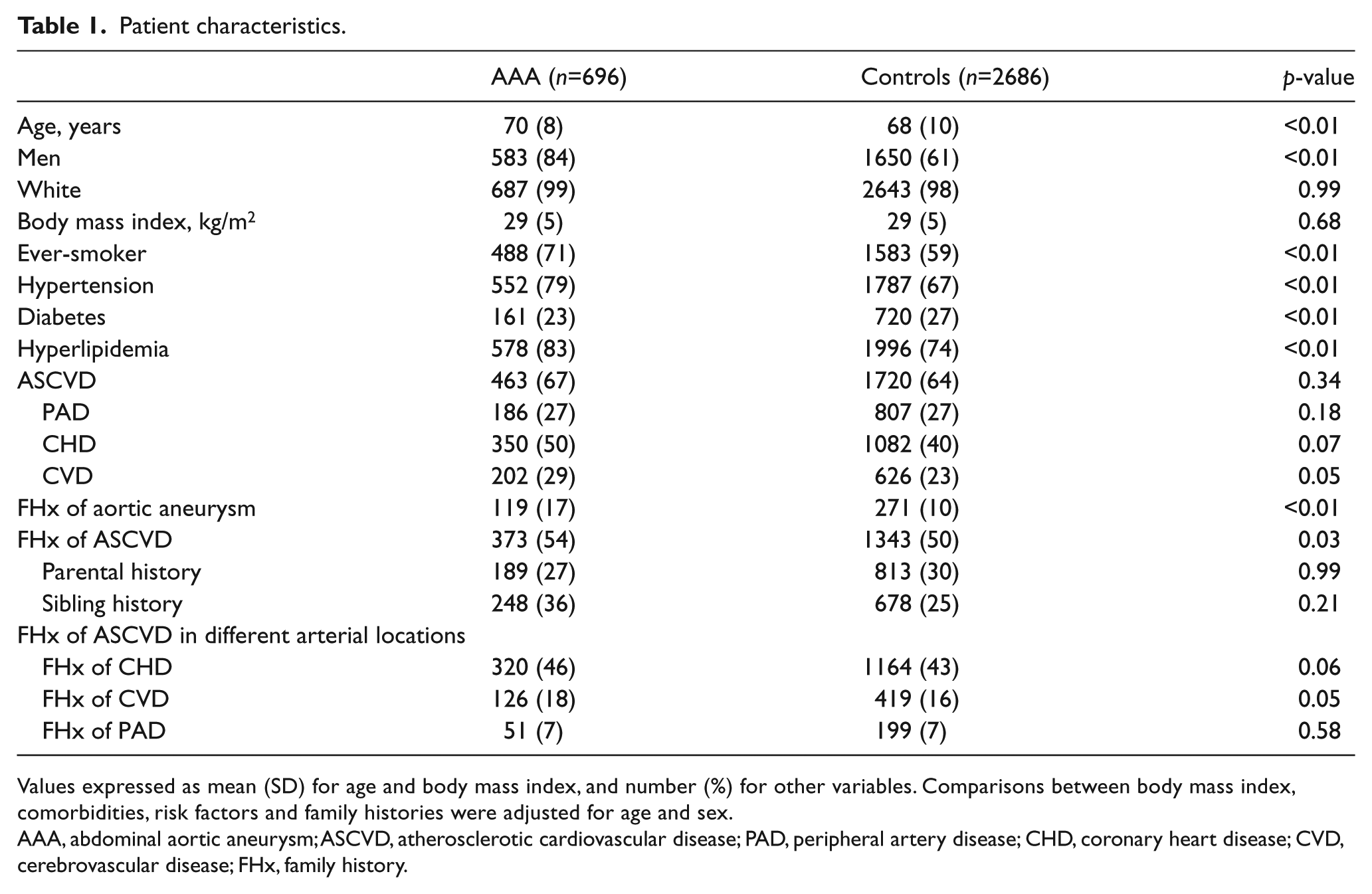
Values expressed as mean (SD) for age and body mass index, and number (%) for other variables. Comparisons between body mass index, comorbidities, risk factors and family histories were adjusted for age and sex.
AAA, abdominal aortic aneurysm; ASCVD, atherosclerotic cardiovascular disease; PAD, peripheral artery disease; CHD, coronary heart disease; CVD, cerebrovascular disease; FHx, family history.
FHx of aortic aneurysm and ASCVD were each associated with presence of AAA after adjustment for age and sex. The associations of FHx with aortic aneurysm and ASCVD remained significant after further adjustment for BMI, hypertension, diabetes, smoking, hyperlipidemia, and ASCVD (Table 2). Patients with FHx of ASCVD had a 27% higher likelihood of having AAA after additional adjustment for FHx of aortic aneurysm (adjusted OR, 95% CI: 1.27, 1.05–1.55, p = 0.01).
Associations of FHx of ASCVD and aortic aneurysm with presence of AAA.
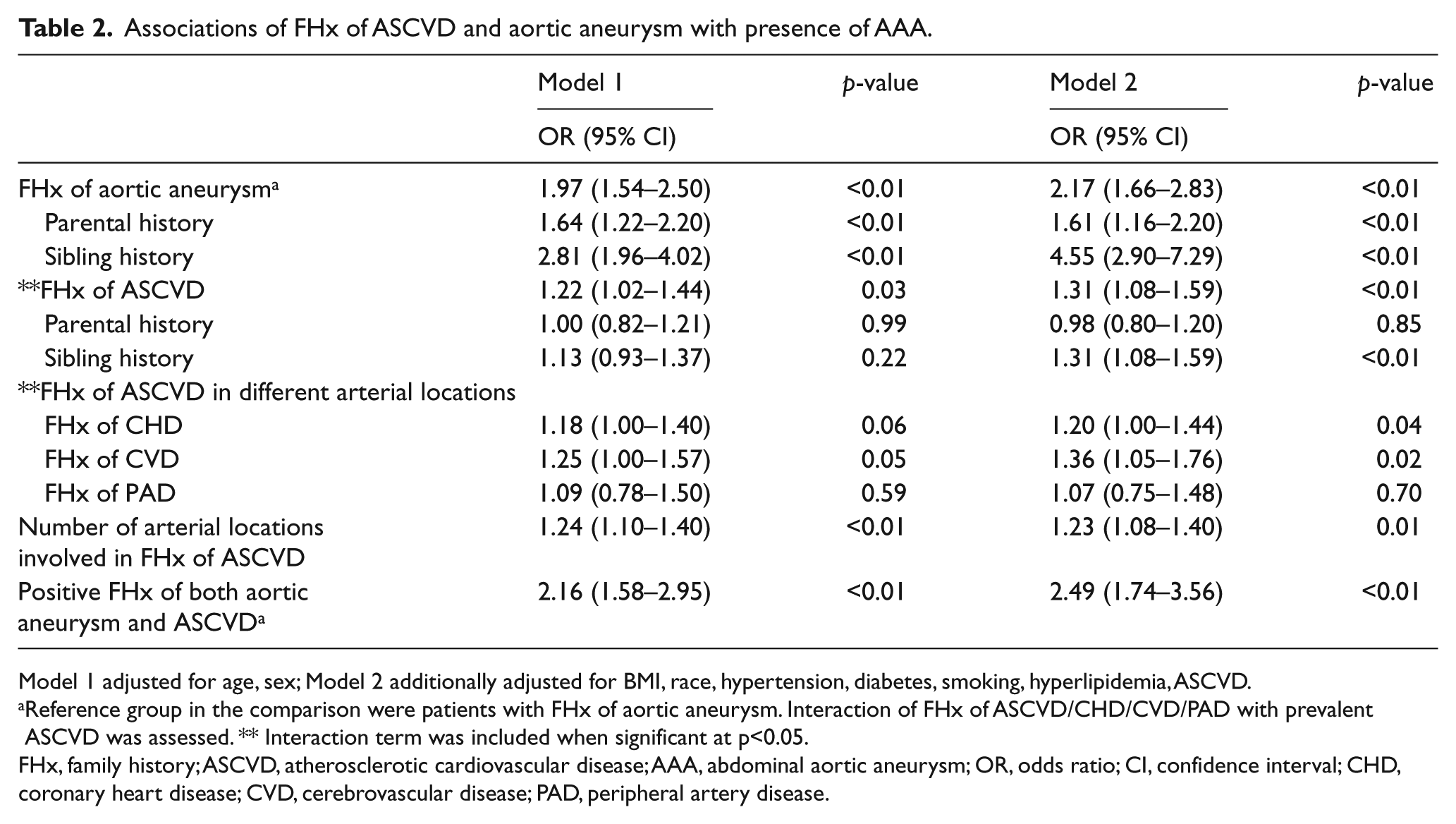
Model 1 adjusted for age, sex; Model 2 additionally adjusted for BMI, race, hypertension, diabetes, smoking, hyperlipidemia, ASCVD.
Reference group in the comparison were patients with FHx of aortic aneurysm. Interaction of FHx of ASCVD/CHD/CVD/PAD with prevalent ASCVD was assessed. ** Interaction term was included when significant at p<0.05.
FHx, family history; ASCVD, atherosclerotic cardiovascular disease; AAA, abdominal aortic aneurysm; OR, odds ratio; CI, confidence interval; CHD, coronary heart disease; CVD, cerebrovascular disease; PAD, peripheral artery disease.
FHx of CHD and CVD were each associated with presence of AAA in models adjusted for BMI, hypertension, type 2 diabetes, smoking, hyperlipidemia, and ASCVD, whereas FHx of PAD was not associated with presence of AAA (Table 2). In addition, FHx of ASCVD in multiple arterial locations was associated with presence of AAA, with a 23% higher likelihood of having AAA for each additionally affected arterial location present in the FHx (Table 2).
Parental and sibling history of aortic aneurysm was associated with presence of AAA after adjustment for age, sex, BMI, hypertension, diabetes, smoking, hyperlipidemia and ASCVD (Table 2). Sibling history of ASCVD was associated with presence of AAA after adjustment for age and sex and additional covariates listed above, while there was no statistically significant association of parental history of ASCVD with AAA (Table 2).
When we categorized participants based on FHx of aortic aneurysm in addition to ASCVD, using patients with FHx of aortic aneurysm as the reference group, patients with FHx of both aortic aneurysm and ASCVD had higher odds of having AAA (OR, 95% CI: 2.00, 1.48–2.70, p <0.01). The association remained significant after adjustment for age and sex and additional covariates (Table 2).
Given the sex difference in AAA, we assessed whether sex was a modifier for the association of FHx with AAA. We did not find that sex modified the association of FHx of ASCVD with presence of AAA. We did not find FHx of ASCVD to be additive to risk factors for AAA as assessed by corrected Akaike information criterion from logistic regression models (see supplementary material).
Discussion
The major findings of our study are: (1) FHx of ASCVD was associated with presence of AAA independent of conventional cardiovascular risk factors and FHx of aortic aneurysm; (2) sibling history of ASCVD had a stronger association with AAA than parental history; and (3) FHx of ASCVD in multiple arterial locations increased the odds of having AAA. Our results suggest that both unique and shared environmental and genetic factors mediate disease susceptibility to AAA and ASCVD.
A positive FHx was associated with a two-fold risk of having AAA, with ORs of 1.6–2.5 reported in population-based studies. 11 We found an OR of ~ 2.0 for a positive FHx consistent with previous reports. A novel finding of our study is the association of FHx of ASCVD with presence of AAA. Previous studies have shown several biological pathways to be associated with both FHx of ASCVD and presence of AAA, including inflammatory markers such as C-reactive protein,10,14–16 interleukin-617–20 and impaired endothelial function.21–24 Recent genome-wide association studies (GWAS) have revealed several genes to be associated with both ASCVD and AAA, including SORT1 at 1p13.3 (mediating triglyceride metabolism), DAB2IP at 9p33.2 (mediating cell apoptosis and survival), CDKN2A-2B at 9p21 (mediating atherosclerotic plaque formation) and LDLR at 19p13.2,25–28 suggesting pleiotropic effect of these loci on disease development and common susceptibility genes for both traits. We found that a positive FHx of both ASCVD and aortic aneurysm was associated with higher odds of having AAA than FHx of aortic aneurysm alone (Table 2), consistent with shared genetic susceptibility and environmental risk factors between ASCVD and AAA.
We found a stronger association of sibling history of ASCVD with presence of AAA than parental history. The association remained significant after further adjustment for numbers of full brothers and full sisters (analyses not shown). A stronger sibling–sibling association with the presence of CHD and stroke than parental–offspring association has been reported previously.29,30 We demonstrate for the first time that a similar pattern exists for AAA. Siblings are more likely to have common environmental factors than parent–offspring pairs. Adverse environment in childhood has been reported to affect risk of atherosclerosis 31 and death due to cardiovascular disease later in adulthood. 32 Alternatively, sibling history may be more easily recalled than remote medical history of parents.
We found a differential association of FHx of CHD and CVD with presence of AAA versus that of FHx of PAD with AAA. This could be due to the small number of patients with AAA who had a FHx of of PAD. Recent GWAS reported shared genetic susceptibility variants for ASCVD in different arterial locations. 33 Whether ASCVD in a particular arterial bed is differentially associated with AAA is unclear. The Tromsø study found a carotid atherosclerosis and CHD to be associated with presence of AAA.34,35 Our results suggest that FHx of atherosclerosis in different arterial locations is differentially associated with presence of AAA. Further studies are needed to assess shared and unique genetic susceptibility to ASCVD in different locations and AAA.
Limitations
Our study included a large cohort of AAA cases and controls with comprehensive assessment of family history by questionnaire. However, several limitations of our study need to be mentioned. First, all subjects were referred to Mayo Clinic, a tertiary care center, which may limit the generalization of our results. Almost all of our participants were white (>98%) and generalizability to other ethnic groups is unclear. Second, owing to the retrospective nature of our study, we cannot rule out recall bias. However, given the natural history of AAA, it is not feasible to conduct a prospective study in a large cohort and collect relevant information efficiently. Ascertainment of family history was based on participant self-report and recall bias may be present. However, we found a similar rate of self-reported family history as that in published population-based studies. Third, not all of the controls underwent ultrasound screening, and we cannot rule out presence of AAA in controls. However, in a random set of controls (n=50) with at least one abdominal imaging study in the electronic health records (EHR), none had AAA identified. The association of FHx of ASCVD with presence of AAA did not change when we limited the controls to those with an abdominal imaging study in the EHR (n=2221 analysis not shown). Fourth, the response rate to the study questionnaire was 67%. We compared risk profiles of responders versus non-responders (data not shown). We found that, compared to responders, non-responders were younger, with a higher BMI, and more often were hypertensive or diabetic. However, proportions of men and women and patients with hyperlipidemia or ASCVD were similar between the two groups.
Conclusion
We report an association of FHx of ASCVD with presence of AAA in a large cohort of AAA cases and controls. We found that: (1) FHx of ASCVD was associated with presence of AAA independent of conventional risk factors and FHx of aortic aneurysm; (2) sibling history of ASCVD had a stronger association with AAA than parental history; (3) FHx of ASCVD in multiple arterial locations was associated with higher odds of having AAA. Our results suggest that FHx of ASCVD is a risk factor for AAA, and that shared environmental and genetic factors mediate disease susceptibility to both AAA and ASCVD. The presence of FHx of ASCVD may identify patients at increased risk of having AAA, and provide insights on genetic risk for disease development for further investigation.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Kullo was supported by grant U01 HG-06379 from the National Human Genome Research Institute. The publication was made possible by the Center for Translational Science Activities Grant UL1 TR000135 from the National Center for Advancing Translational Sciences, a component of the NIH. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIH.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
