Abstract
Aortic diseases are a heterogeneous group of disorders, including atherothrombotic conditions like aortic atheroma, cholesterol embolization syndrome, aortic mural thrombus, thrombus within an aneurysm, and large vessel vasculitis. In this review, we provide a summary of the current evidence regarding atherothrombotic diseases of the aorta, focusing on therapeutic avenues. In patients with previous stroke, aortic arch atheroma is recognized as a strong predictor of recurrent atheroembolism, and antiplatelet therapy alone is still associated with a high (11.1%) residual risk of recurrent stroke. In secondary prevention, the use of dual antiplatelet therapy or moderate intensity anticoagulation with warfarin may lower the risk of recurrent stroke at a cost of increased life-threatening bleeding. Thrombi adherent to the aortic wall are generally associated with underlying atherosclerosis or aneurysmal disease. Primary aortic mural thrombus is a rare condition, sometimes related with systemic prothrombotic or inflammatory diseases. Retrospective studies suggest that anticoagulation is beneficial in patients with mobile mural thrombus. The pathogenesis and consequences of thrombus in an aortic aneurysm, or in an endograft following endovascular aneurysm repair, have been studied, but the role of antiplatelet therapy in those two conditions is still unclear and should be driven by general cardiovascular risk prevention. The benefit of anticoagulation to reduce thrombus load is uncertain. Patients with large vessel vasculitis experience increased cardiovascular events secondary to inflammation-driven atherothrombotic processes. Antiplatelet therapy is recommended as part of the therapy for prevention of cardiovascular disease. Anticoagulation with warfarin has shown limited benefit in few retrospective studies.
CME Accreditation Statement
The University of Virginia School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™ per article. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Learners are expected to read this article along with any references and supporting material as appropriate, and complete the online post-test questions with an 80% pass rate to receive credit. Post-test questions are accessed through the member portal on the Society for Vascular Medicine (SVM) website (www.vascularmed.org). Please note that CME credits are only available to members of the SVM. This activity expires two years after the publication date, on February 13, 2019.
The faculty, staff and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose.
The CME planning committee of Vascular Medicine disclosed the following:
Heather Gornik: Astra Zeneca, research support; Summit Doppler Systems, Inc., intellectual property rights; FlexLife Health, intellectual property rights and stock/ownership (proceeds donated).
Aditya Sharma: National Institute of Health Sciences, research support; AstraZeneca, research support; Biomet Biologics, research support; Portola Pharmaceuticals, research support; Pedra Technology, research support.
Valerie Clark: None.
Case report
A 54-year-old male who was a former smoker, with a history of hypertension, stage 3 chronic kidney disease and dyslipidemia, presented with complaints of progressive left foot pain and discoloration over the past 1 week. On examination, his vital signs were stable and he was in sinus rhythm. The popliteal and distal pulses and Doppler signals were absent in the left extremity. There was marked cyanosis of the left foot digits, with a capillary refill increased over 5 seconds. The patient had no history of intermittent claudication, connective tissue disease, thrombotic disorder or myeloproliferative neoplasm. His medications were aspirin, ramipril and hydrochlorothiazide, and rosuvastatin. An echocardiogram showed a normal ejection fraction and no valvular disease or intracardiac thrombus. A computed tomography (CT) angiogram revealed an acute thrombosis of the popliteal artery and an adherent thrombus in the supra- and infra-renal portions of the abdominal aorta (arrows in Figure 1). What is the appropriate antithrombotic strategy in this patient?
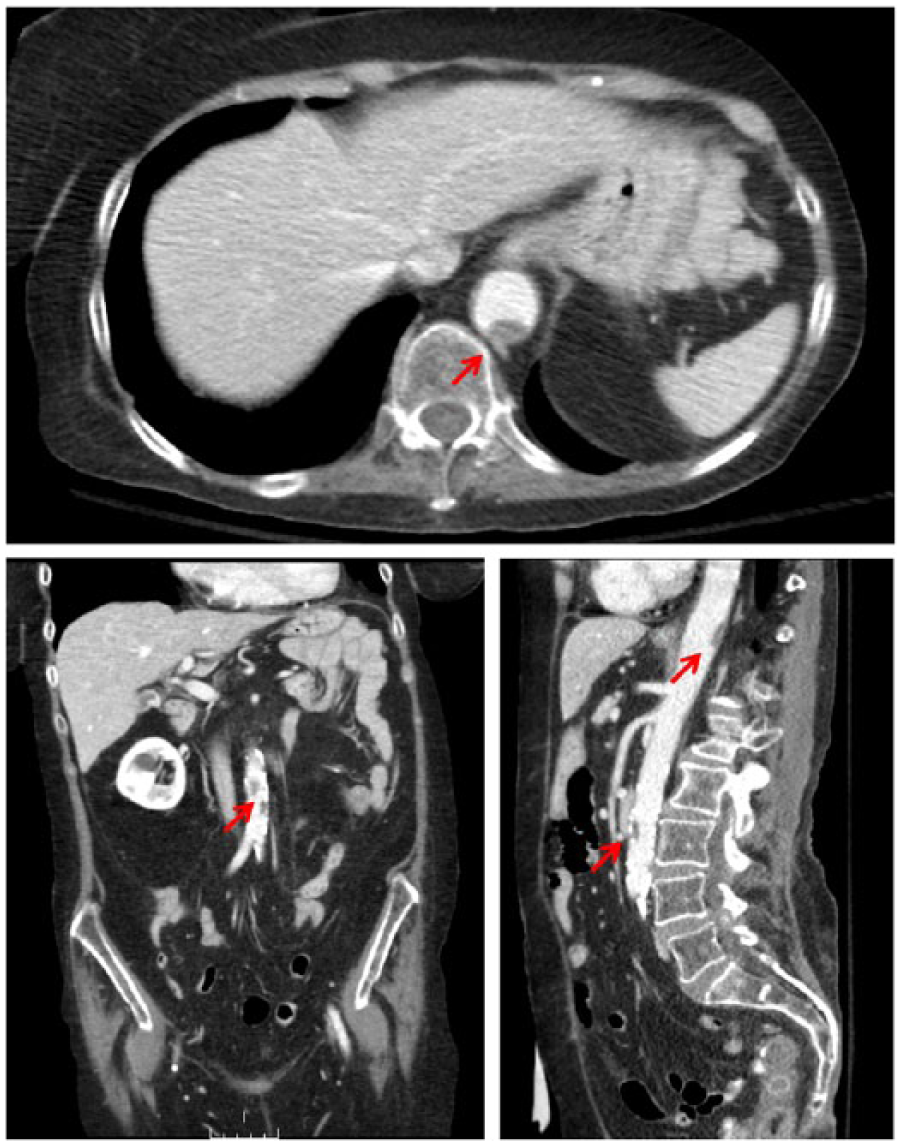
Supra- and infra-renal aortic mural thrombi (arrows). (Image reproduced with permission from http://www.angiologist.com/arterial-disease/mural-thrombus/.)
Aortic atheroma
Aortic atherosclerosis involves mechanisms that are in many ways similar to coronary atherosclerosis, but the large diameter of the vessel and generous arterial blood flow allows larger plaques to protrude into the arterial lumen long before occluding it. 1 Vulnerable plaques in the aorta can remain clinically silent for long periods of time, as demonstrated by post-mortem studies reporting 8% of ruptured or ulcerated peri-renal aortic plaques in apparently healthy organ donors, with increasing prevalence with age. 2 Observational data from cardiac surgery, autopsy and advanced imaging studies demonstrate the presence of thrombi superimposed on complex or ulcerated aortic plaques.3,4 This process is well described in patients with coronary atherosclerosis who present with acute coronary syndromes and undergo coronary angiography. 5 Thromboembolism resulting from occlusion of distal arteries by thrombotic material manifests clinically as ischemic stroke, transient ischemic attack, renal or bowel infarction, or acute limb ischemia.6,7
Atheromatous disease of the aorta is a relatively common finding in the general population. The Stroke Prevention: Assessment of Risk in a Community (SPARC) study8,9 was a cross-sectional analysis of transesophageal echocardiography (TEE) in 581 participants over 45 years of age, and showed a prevalence of simple (<4 mm of maximal thickness) and complex (⩾4 mm of maximal thickness) aortic plaques of 51.3% and 7.6%, respectively. The APRIS study, a prospective cohort study, found a similar prevalence of aortic arch plaques (62.2%) in 209 stroke-free subjects. The Framingham Offspring Heart Study performed cardiac magnetic resonance imaging (MRI) on 1763 participants, of which 200 had cardiovascular disease (CVD). Aortic plaques were identified in 46% of CVD-free participants. 10
Risk factors for aortic atherosclerosis include increasing age (odds ratio (OR) 3.56 per 10-year increase; 95% confidence interval (CI): 2.57–4.92), smoking (OR 2.18; 95% CI: 1.38–3.46), diabetes (OR 2.98; 95% CI: 1.58–5.61), and hypertension (OR 1.18 per 10 mmHg increase in out-of-bed systolic pressure; 95% CI: 1.01–1.38). 8 Among CVD-free participants of the Framingham Offspring cohort, the presence of hypertension increased the risk of thoracic aortic plaques on cardiac MRI threefold. 10 Increased concentrations of fibrinogen (OR 1.18; 95% CI: 0.96–1.12), 11 homocysteine (OR 1.21; 95% CI: 1.05–1.41) 12 and an elevated peripheral leukocyte count 13 (adjusted OR 1.38; 95% CI: 1.05–1.79 per unit increase in leukocyte count) have also been associated with aortic atherosclerosis but may represent markers rather than mediators of this disease process.
Aortic atherosclerosis has been associated with CVD in studies using various methods for diagnosis. In the SPARC study,8,9 the presence of aortic atherosclerosis was associated with coronary artery disease (adjusted OR 2.99; 95% CI: 1.47–6.10; p=0.003), but the association with ischemic stroke and transient ischemic attack was not significant after adjustment. In the APRIS study, 6 the presence of aortic plaques was not associated with incident myocardial infarction and ischemic stroke. In the Framingham Offspring Heart Study, 10 aortic plaque prevalence on cardiac MRI was significantly higher in patients with known CVD and this difference was more marked for thoracic plaques (relative risk (RR) 2.9; p<0.0001).
Aortic arch disease, due to its proximity with the cerebral vessels, has a stronger association with cerebrovascular disease. In an autopsy study of 500 patients with stroke or other neurologic diseases, Amarenco and colleagues reported a strong correlation between the presence of aortic arch plaques and stroke (26% vs 5%; age-adjusted OR 4.0; 95% CI: 2.1–7.8). 14 Ulcerated plaques were found more frequently in patients with cryptogenic stroke (61% vs 22%). A similar study with TEE found atherosclerotic plaques with a width of over 4 mm in 14.4% of patients admitted for stroke, 28.2% of patients with cryptogenic stroke, but only in 2% of the controls (OR 9.1; 95% CI: 3.3–27.5; p<0.001). 7 In a prospective study of 331 consecutive stroke patients aged 60 or above, the French Study Group 15 found a fourfold increase for stroke recurrence in patients with aortic plaques of ⩾4 mm in thickness. The annual incidence of stroke recurrence in that group was 11.9%, despite the fact that more than 70% were treated with antiplatelet or anticoagulant therapy.
The natural evolution of aortic atheroma over time is variable, dynamic, and dependent on concomitant diseases (i.e. diabetes), medication use (i.e. statin use), and risk factors (i.e. smoking, hypertension). In TEE retrospective studies, most atheroma remain stable at 1 year, 20–40% progress in size, and regression is observed in about 10%.16,17 In a prospective MRI study, aortic plaques regressed by 15% after 6 months of simvastatin 80 mg in atherosclerotic patients. 18 In patients with stroke or transient ischemic attack, the progression of atheroma is associated with an increased risk of death or cardiovascular event (adjusted hazard ratio (HR) 5.8; 95% CI: 2.3–14.5; p=0.0002). 19
Diagnosis of aortic atheroma
Despite its convenience and technical progress with harmonic imaging, transthoracic echocardiography (TTE) has a low sensitivity when compared with other techniques.20,21 In the absence of a gold standard, calculation of sensitivity can hardly be done, but when considering TEE as the reference, we approximate the sensitivity of TTE to be around 62%. 21 TEE was traditionally the most frequently used imaging modality for imaging of the aortic arch.20–22 A study of pathological correlation 23 showed a sensitivity of 91% and a specificity of 90% for identification of thrombus and an agreement of 93% for complex aortic plaque. TEE informs on mobility, ulceration and composition of the plaque with good reliability (kappa: 0.58–0.87) and has very good inter- and intra-observer reliability for plaque thickness (intraclass correlation coefficient: 0.85–0.97). 24 Its limitations are the necessity of conscious sedation and the invasive nature of the test for patients. Furthermore, a small proportion of the aortic arch adjacent to the innominate artery is masked by the tracheal air column, resulting in approximately 2% of missed atheroma. 25
Non-enhanced CT has an 87% sensitivity when compared with TEE, and can identify plaques in the innominate artery missed by TEE. 26 However, it has limited ability to identify heavily calcified plaques and should be complemented with a TEE or contrast-enhanced CT.
Contrast-enhanced CT and magnetic resonance angiography (MRA) provide the most detailed information about plaque distribution, morphology and composition. 27 MRA has a 100% sensitivity to detect plaques visualized on TEE 28 and has an 80% agreement with TEE for plaque characterization. Technological advances in the precision of CT angiograms and MRAs and their ability to image the innominate artery make them the modality of choice for imaging of the aorta and its branches. One common limitation to those two techniques is their inability to identify a mobile thrombus, which the TEE can identify very reliably.
Primary prevention
Given that aortic atheroma is modestly prevalent in the general population, it is a frequent fortuitous finding on imaging tests. Aortic atherosclerosis is correlated with CVD in retrospective studies, but whether it is an independent predictor remains unconfirmed. Recent guidelines29–33 make no recommendation on how to prevent CVD in patients with aortic atheroma on imaging. They do, however, unanimously recommend the use of aspirin in patients with asymptomatic peripheral artery disease, as defined by an ankle–brachial index lower than 0.90. Altogether, while the presence of aortic plaques in an asymptomatic patient should not mandate antiplatelet therapy on its own, it should lower the threshold to initiate it for the primary prevention of cardiovascular events. Dual antiplatelet therapy is not recommended in primary prevention of cardiovascular events, as it is associated with an increased risk of bleeding without reducing cardiovascular events in patients with peripheral artery disease 34 and cerebrovascular disease. 35 Furthermore, anticoagulation with warfarin together with antiplatelet therapy is not recommended in primary prevention, as it has been shown to increase the rate of bleeding with no benefit in ischemic events when compared with aspirin in patients with peripheral artery disease. 36
Secondary prevention
In patients with a previous history of stroke with thoracic aortic plaques, the results from the French Study Group suggest a high residual risk of recurrence in patients on single antiplatelet therapy. 15 The Patent Foramen Ovale in Cryptogenic Stroke Study (PICSS) 37 randomized 516 patients with ischemic stroke to warfarin or aspirin and followed them over 2 years, evaluating them at baseline with TEE for aortic plaques. Large plaques were associated with an increased risk of recurrent ischemic stroke or death (HR 2.12; 95% CI: 1.04–4.32), but event rates were similar in the warfarin and aspirin groups (16.4% vs 15.8%; p=0.43). Thus, the selection of antithrombotic therapy in secondary prevention should be made according to current stroke guidelines, irrespective of the presence of aortic plaques. 38 Thus, based on the present evidence, short-term dual antiplatelet therapy is a reasonable option after an acute atheroembolic event for patients at low bleeding risk in whom full dose anticoagulation with warfarin is not indicated.
Cholesterol embolization syndrome
Atheroembolism, also known as cholesterol crystal embolism, refers to embolization of cholesterol crystals from a large proximal artery to smaller distal arteries, causing end-organ damage. The most frequent etiology of cholesterol embolization syndrome (CES) is instrumentation of the aorta (i.e. intra-aortic balloon pump, catheter intervention), which breaks the plaque and releases the cholesterol emboli. 39 The incidence of clinically apparent atheroembolism is less than 1% in patients with documented aortic atherosclerosis and less than 2% in patients undergoing cardiac catheterization. 40 Clinical manifestations of CES are acute progressive renal failure, gastrointestinal symptoms from mesenteric infarctions, ‘blue toe’ syndrome and livedo reticularis. 41 Crystals can be documented in retinal vessels (Hollenhorst plaques) and contribute to the diagnosis. Patients with CES have a poor prognosis, with a mortality rate of 17% to 75%, depending on the absence or presence of end-stage renal failure, respectively. This high mortality probably reflects the high baseline risk of the population who undergo aortic instrumentation.
CES has been reported in association with the use of anticoagulants such as warfarin. 42 However, in contemporary cohorts of patients with aortic plaques, it remains a very rare event. In three studies including more than 1000 patients,15,43,44 CES occurred in four patients on warfarin and three patients off warfarin. For that reason, we would not suggest to hold warfarin in patients with CES and a clear indication for anticoagulation.
While there is no evidence to use antiplatelet agents to prevent CES, they are recommended for prevention of cardiovascular events in patients with known peripheral artery disease.
Primary aortic mural thrombus
Thrombotic material adherent to the aortic wall is associated with atherosclerosis or aortic aneurysms in the vast majority of cases.45,46 Primary aortic mural thrombus occurs in the absence of the aforementioned conditions, has been reported in case series,47–50 and is associated in the majority of cases with systemic conditions like heparin-induced thrombocytopenia,51,52 antiphospholipid syndrome,53,54 underlying malignancy,55–57 chemotherapy,58,59 myeloproliferative neoplasms, 46 or thrombophilias.56,60,61
In a retrospective study of 88 patients with acute limb ischemia, 19 were found to have an underlying aortic mural thrombus. 47 The most frequent sites of thrombus in this series were the distal aortic arch and descending thoracic aorta, abdominal aorta and ascending aorta, found in 74%, 14% and 12% of cases, respectively. The patients did not exhibit the risk factors typically associated with atherosclerotic disease: only 5.3% were smokers and none of the 19 patients had any other comorbidity. Aorta mural thrombi are also occasionally identified during the investigation of peripheral arterial embolism. In a large retrospective cohort, 62 the source of acute lower limb ischemia was embolic in 40% of cases, of which 78% were of cardiac origin and non-cardiac or unknown embolic sources accounted for the remaining 22%. In a series of 556 consecutive patients undergoing TEE, Karalis and colleagues 63 found an aortic thrombus in 7% of the patients. Pedunculated and highly mobile thrombi were associated with a higher risk of embolic events (73%) than were layered and immobile thrombi (12%). With the advent of imaging techniques allowing visualization of the aorta, we estimate that 10% of non-cardiac emboli originate from the aorta, half from aneurysmal disease and half from atherothrombosis.64–66
The management of aortic mural thrombi relies largely upon experience and mechanistic understanding of the disease, as evidence is limited to case reports. While some authors suggest that anticoagulation alone is a reasonable solution that leads to complete resolution of the mural thrombus,67,68 other studies report a high risk of recurrence and suggest a surgical approach in young patients with a mobile thrombus located in the aorta. 45 An analysis of 200 published cases up to 2014 compared surgical management with anticoagulation. 46 One in four patients initially anticoagulated required a secondary aortic procedure for persistent thrombus or recurrent peripheral arterial embolization, whereas only 4.5% of patients in the primary surgery group required a second procedure. Factors associated with a higher risk of recurrent embolization were location of the thrombus in the ascending aorta (OR 12.7; 95% CI: 2.3–38.8) or aortic arch (OR 18.3; 95% CI: 2.6–376.7), stroke as a presenting symptom (OR 11.8; 95% CI: 3.3–49.5) and atherosclerotic aortic wall (OR 2.5; 95% CI: 1.0–6.4). Figure 2 suggests a diagnostic and therapeutic algorithm for patients with acute limb ischemia and aortic mural thrombus. The duration of antithrombotic treatment in patients treated with an anticoagulation-only approach should be guided by the evolution of the thrombus on repeated imaging and a consideration of the patient’s bleeding risks. Complete resolution of the thrombus in a patient without an ongoing prothrombotic condition makes it a reasonable option to discontinue anticoagulation, depending on the patient’s clinical risk factors and severity of presenting thrombosis, and considering the patient’s values and preferences.
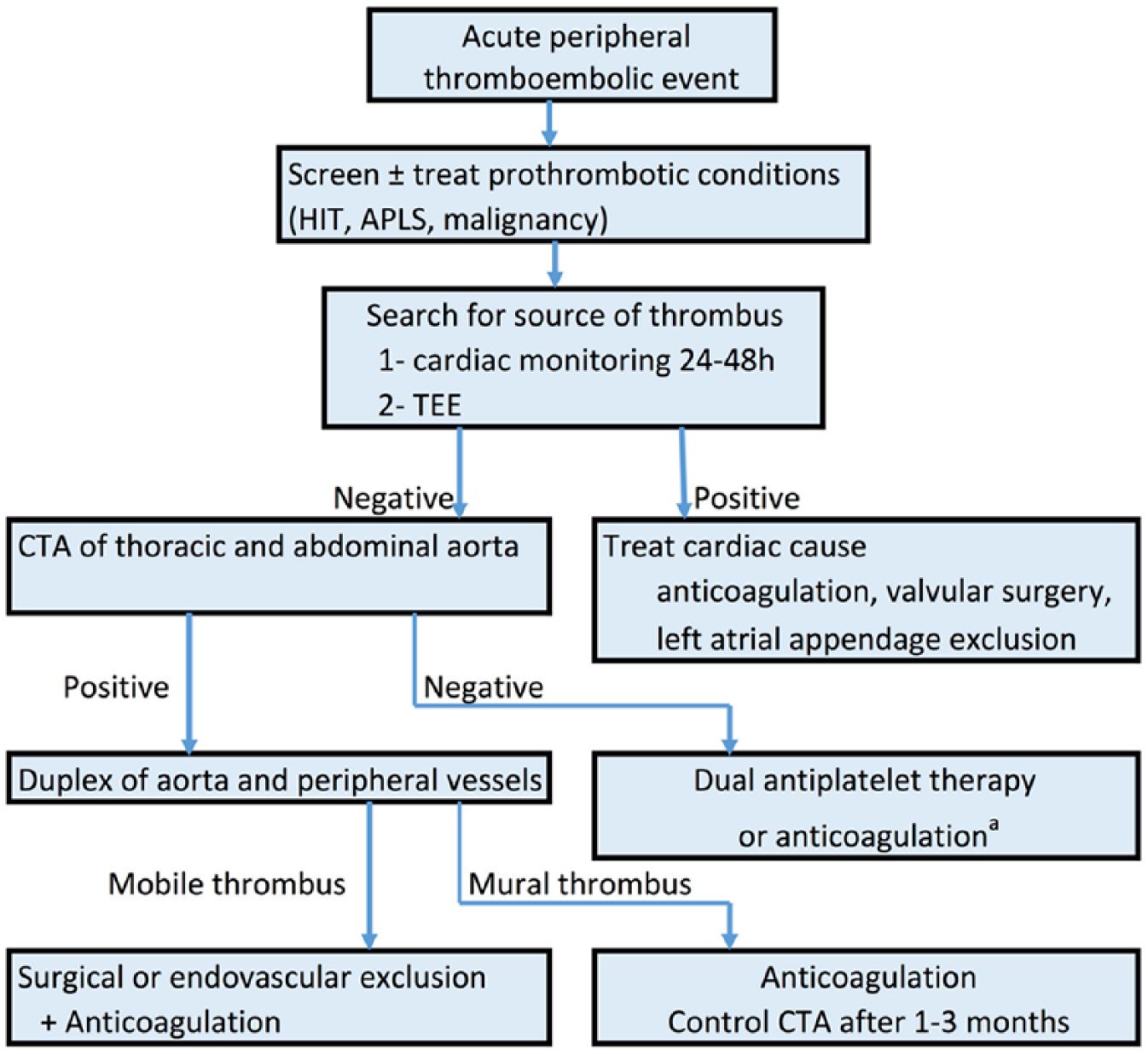
Diagnostic algorithm for acute limb ischemia of thromboembolic origin. (HIT, heparin-induced thrombocytopenia; APLS, antiphospholipid syndrome; TEE, transesophageal echocardiogram; CTA, computerized tomography-angiogram; aLow level of evidence.)
Aortic aneurysm-related thrombus
Abdominal aortic aneurysms (AAAs) are frequently lined with a layer of intraluminal thrombus. 69 This thrombus has mixed effects on the evolution of the aneurysms. In studies using CT-generated three-dimensional reconstructions on the aorta, 69 it appeared that the presence of thrombus reduced wall stress by up to 38%, thus potentially reducing the risk of rupture. The thrombus was also associated with further progression of the aneurysm by enhancing the proteolytic process within the aortic wall. 70
Whether antiplatelet therapy affects the rate of growth or rupture of aneurysms is a matter of debate. In a meta-analysis of individual data from 4137 patients included in six studies, 71 the use of antiplatelet agents was not associated with a significant difference in the rate of growth of AAAs after multivariable adjustment (–0.123 mm/year, standard error 0.106, p=0.24). Furthermore, a Danish case–control study 72 compared 4010 patients with a ruptured AAA with patients with an unruptured AAA, and found no difference in the risk of rupture between patients taking aspirin and those who did not (adjusted OR 0.97; 95% CI: 0.86–1.08). However, the case fatality rate was higher in patients on aspirin (adjusted RR 1.16; 95% CI: 1.06–1.27).
Endovascular aneurysm repair (EVAR) is gaining in popularity for the management of AAAs in higher-risk patients. Mural thrombus develops as a result of rheologic factors and properties of the device, but whether the presence of thrombus leads to thrombotic consequences is uncertain. In a retrospective cohort of 414 post-EVAR patients, 68 developed thrombus over a median follow-up of 3.4 years, and were compared with a control group of patients without thrombus. 73 The incidence of peripheral thromboembolic events did not differ between the group with thrombus and the group without thrombus (4.4% vs 3.5%, p=0.70). A majority of patients in both groups received antiplatelet therapy (91.2% vs 85.4%), and a minority received oral anticoagulation (10.3% vs 15.4%). Oral anticoagulation at time of implantation was not associated with the rate of thrombus accumulation in the endograft (HR 0.63; 95% CI: 0.27–1.46). These results do not support intensification of antithrombotic therapy in patients with mural thrombus within an aortic endograft.
In patients with AAA and post-EVAR, general cardiovascular risk prevention should be the main driver of antiplatelet therapy prescription. The high prevalence of atherosclerosis in other vascular beds warrants single antiplatelet therapy for patients without contraindications.
Large vessel vasculitis
Giant cell arteritis (GCA) and Takayasu arteritis, the large vessel vasculitides, are the most common non-infectious causes of aortitis, although other rheumatic diseases have been associated with aortic involvement, namely HLA-B27-associated spondyloarthropathies, Cogan’s syndrome, rheumatoid arthritis, systemic lupus erythematosus, sarcoidosis and anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides. 74 In the last few years, recognition of immunoglobulin (Ig)G4-related systemic disease has brought a potential explanation for an important proportion of aortitis and periaortitis cases formerly labeled as ‘isolated aortitis’. 41 While the clinical presentation is generally representative, GCA and Takayasu’s share many aspects of their pathogenesis. Vascular injury results from influx of leukocytes and macrophages through the vasa vasorum into the lumen intima. Inflammatory mechanisms involve tumor necrosis factor (TNF)-alpha, interleukin (IL)-6, B-cell activating factor, and matrix metalloproteases, leading to angiogenesis, myointimal proliferation and, eventually, stenosis or occlusion of the lumen, and/or destruction of elastic fibers resulting in aneurysm formation. Granuloma and multinucleated giant cells, although generally attributed to GCA, can be observed in both diseases.75,76 Clinical manifestations differ: occlusive or stenotic manifestations are encountered more commonly in patients with Takayasu aortitis, whereas patients with GCA have a much higher likelihood of developing aneurysmal disease. 77 Despite the lower age and baseline cardiovascular risk of patients with Takayasu arteritis, intermittent ischemic symptoms are very common (62% in the upper limb and 32% in the lower limb), and cardiac manifestations occur in almost 40% of cases, mainly driven by aortic regurgitation. In patients with GCA, a large population-based retrospective study where 28% of patients were taking aspirin, compared with 22% of controls, demonstrated an increased risk of cardiovascular events including myocardial infarction, stroke, peripheral artery disease, aneurysm or aortic dissection (adjusted HR 2.1; 95% CI: 1.5–3.0). 78
Aspirin for GCA
The use of antithrombotic therapy for primary prevention of vascular events in GCA was addressed in a recent Cochrane review. 79 While three retrospective studies80–82 have found a correlation between low-dose aspirin use and a reduced incidence of ischemic events, others have not.83,84 In 175 consecutive patients with GCA, of which 36 were on aspirin at the time of diagnosis and 41 were started on aspirin by the treating physician, aspirin prevented cranial ischemic complications with an odds ratio of 0.18 (95% CI: 0.04–0.84; p=0.03). 80 Another retrospective case series of 143 patients with GCA recorded a relative risk of ischemic complications of 16.2% in patients on aspirin versus 48% in patients without aspirin. 81 Two later studies comparing patients already on antiplatelet agents at the time of diagnosis with controls found no statistically significant difference in the incidence of ischemic events.83,84 Finally, a recent Brazilian retrospective cohort with a high proportion of aspirin-treated patients (71.1%) suggested a strong preventive effect of aspirin against relapses (OR 0.19; 95% CI: 0.04–0.80; p=0.024). 82 Despite this uncertainty about the preventive effect of aspirin on the evolution of the inflammatory disease, low-dose aspirin is advocated as part of the treatment in GCA,85,86 based on the increased risk of CVD in that population.
Animal studies suggested that high-dose aspirin can inhibit interferon-gamma and block the inflammatory process of GCA. 87 However, the doses of aspirin used in most patients in the above-mentioned studies are antiplatelet doses and may not reach the appropriate concentration to inhibit inflammation. On the other hand, thrombocytosis is associated with a higher incidence of ocular complications of GCA, suggesting a role for antiplatelet therapy. 88
High doses of corticosteroids are the cornerstone of therapy for GCA, but their association with aspirin may raise concern over the risk of gastrointestinal bleeding. In the general population (n=22,049), the association of high-dose corticosteroids with low-dose aspirin increases the risk of upper gastrointestinal bleeding (RR 4.43; 95% CI: 2.10–9.34). 89 However, in a cohort of 143 patients with GCA, there was no increase in bleeding in patients on concomitant aspirin and corticosteroids (3%) when compared to patients on prednisone alone (8.8%, p>0.2). 81
Aspirin for Takayasu’s arteritis
Fundamental studies suggest a theoretical benefit for antiplatelet agents in Takayasu’s patients90–92 and there is a small amount of evidence from retrospective studies to support it. 93 In the evolution of their disease, patients with Takayasu’s arteritis often undergo percutaneous interventions to restore patency in occluded vessels. The rate of restenosis following angioplasty in retrospective series varies between 48% and 78%.94–97 While there is no satisfying evidence for the use of antiplatelet therapy after aortic angioplasty, experience from other populations requiring arterial stents suggests that, at a minimum, single-agent antiplatelet therapy is beneficial.31,98,99 A retrospective review of 97 patients with Takayasu’s arteritis suggested that patients on immunosuppressive therapy at the time of the intervention had an increased rate of successful endovascular intervention (85% vs 24%; p=0.001), but this may be biased by the fact that patients off-therapy probably have a burned-out phase, which reflects a heavier arterial burden and higher risk. 94
Anticoagulants
Few studies have assessed anticoagulant medications in large vessel vasculitis. In a retrospective cohort study of 143 patients with GCA by Lee and colleagues, 81 warfarin use had a protective effect against ischemic events without increasing bleeding after adjustment for age, sex, and previous cerebrovascular risk factors (OR 0.17; 95% CI: 0.03–0.92; p<0.04). This finding should be reproduced in randomized studies before it is translated to practice.
Direct oral anticoagulants (DOACs)
Currently, no study has assessed the newer anticoagulants in aortic arch atheroma, aortic thrombus or aortitis. Two phase 3 randomized trials are currently assessing dabigatran (RE-SPECT ESUS, NCT: 02239120) and rivaroxaban (NAVIGATE ESUS, NCT: 02313909) in patients with embolic stroke of undetermined source (ESUS). The results of these large trials will help inform future treatment choices in patients with aortic thrombosis.
Back to the patient
In the case of our patient, since the decreased renal function is a relative contraindication to endovascular intervention, anticoagulation with unfractionated heparin was started, and the patient was bridged to warfarin with a target international normalized ratio (INR) between 2 and 3. The ischemic symptoms in the leg improved promptly in the first week of treatment. The patient was kept on warfarin alone, and had no recurrence of limb ischemia at 1-year follow-up. The duration of warfarin therapy in this case is unknown, and until directed by future trials should be individually tailored to the patient’s risk profile, presence of persistent aortic thrombi on repeat imaging, and preference for anticoagulants.
Conclusion
As the scientific knowledge of atherothrombosis advances, the pathogenesis of aortic atherosclerosis still raises unresolved questions. We have summarized the data available for antithrombotics in aortic diseases (Table 1). For three decades, TEE has allowed an in vivo description of aortic atherosclerosis and identification of high-risk patients. The risk of CVD should be assessed in asymptomatic patients with aortic arch atheroma, and complex plaques should be considered to double the risk of cardiovascular events. Patients with cryptogenic stroke in which a complex aortic arch atheroma is identified remain at high risk of subsequent cerebrovascular events, despite single antiplatelet therapy. The presence of an aortic thrombus superimposed on an atherosclerotic plaque makes it a complex lesion, but does not immediately mandate anticoagulation. However, patients with a thrombus with a mobile component should be anticoagulated, and surgery should be considered, depending on the patient’s surgical risk, and the location and size of the thrombus. Future studies of DOACs compared to antiplatelet therapy will add to the evidence base to guide clinicians regarding antithrombotic therapy in patients with aortic atherosclerosis.
Recommendations for antithrombotic medications in aortic atherothrombotic conditions.
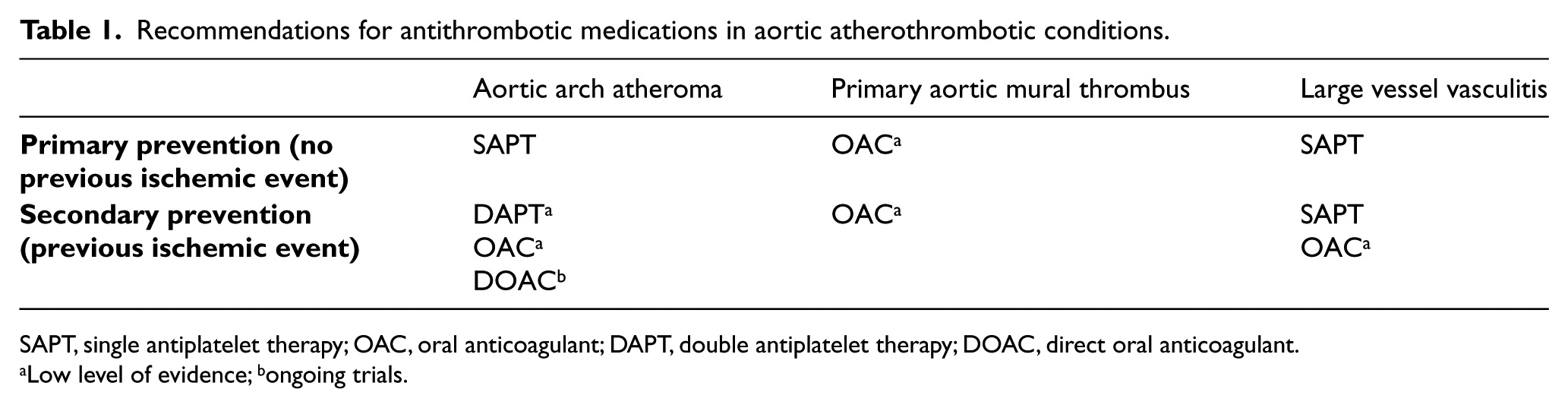
SAPT, single antiplatelet therapy; OAC, oral anticoagulant; DAPT, double antiplatelet therapy; DOAC, direct oral anticoagulant.
Low level of evidence; bongoing trials.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
