Abstract
Purpose:
Familial history of aortic disease has been associated with increased risk of aneurysm-related complications after standard endovascular aortic repair. The aim of this study was to evaluate outcomes of fenestrated-branched endovascular aortic repair (FB-EVAR) for complex aortic aneurysms in patients with or without familial aortic aneurysms (FAAs).
Materials and Methods:
We reviewed the clinical data of consecutive patients enrolled in a prospective non-randomized study to investigate outcomes after FB-EVAR for treatment of complex aortic aneurysms between 2013 and 2022. Familial aortic aneurysm was defined as occurrence of an aortic aneurysm or dissections in a first- or second-degree relative and compared with patients with sporadic aortic aneurysms (SAAs). Endpoints included procedural metrics, 30-day mortality and major adverse events (MAEs), mid-term patient survival, freedom from reintervention and endoleak, and aneurysm sac shrinkage.
Results:
There were 499 patients treated with FB-EVAR (70% males, mean age was 74±7.7 years) including 66 (13%) patients with FAAs and 434 (87%) with SAAs. Both groups had similar demographics, clinical characteristics, aneurysm extent, and prior history of aortic dissection or aortic repair. Patients with FAAs had longer fluoroscopy time (82 vs 71 minutes) and radiation exposure (1856 vs 1213 mGy; each p<0.05), with no difference in technical success (FAA, 94% vs SAA, 97%; p=0.51). There was no difference in 30-day mortality (1.5% vs 0.9%; p=1.00) and MAEs (23% vs 18%; p=0.44) among patients with FAA and SAA, respectively. Median follow-up was 28 months. At 3 years, patient survival, freedom from secondary interventions, and cumulative incidence of sac shrinkage were 76±2.5%, 74±2.5%, and 32±2.6% for all patients, respectively, with no difference between FAA and SAA groups.
Conclusion:
Fenestrated-branched endovascular aortic repair was performed with high technical success, low mortality, and low incidence of MAEs regardless of the presence of FAA. There was no difference in mid-term patient survival, secondary interventions, and sac changes among patients with or without family history of aortic disease.
Clinical Impact
This study demonstrates that fenestrated-branched endovascular aortic repair (FB-EVAR) can be performed with high technical success and comparable early and mid-term outcomes in patients with or without a familial history of aortic aneurysm, after exclusion of genetically triggered aortic diseases. These findings challenge the assumption that family history alone should preclude complex endovascular repair. Clinically, this supports broader use of FB-EVAR in carefully selected patients with familial aortic disease, emphasizing anatomical suitability rather than pedigree alone. The innovation lies in validating FB-EVAR as a safe alternative to open repair for this understudied population, informing patient selection, counseling, and surveillance strategies.
Keywords
Introduction
Familial aortic aneurysms (FAAs), defined as the occurrence of an aortic aneurysm or dissection in a first- or second-degree relative, have been described as an important determinant for the development of aneurysms and dissections, as well as for treatment outcomes. 1 Familial aggregation of aortic disease is well established. First-degree relatives of patients with thoracic or abdominal aortic aneurysm (AAA) or dissection have a significantly higher risk—ranging from 2-fold to 7-fold—of developing concordant aortic disease and a greater risk of aortic-related mortality.2–8 The most common familial form of transmission is autosomal dominant, although specific genetic mutations are identified in a minority of cases. 9 Familial aneurysms have been associated with a higher frequency of multifocal aneurysm distribution, increased risk of disease progression, and in some reports adverse outcomes following endovascular aortic repair (EVAR).2,10
The development of fenestrated-branched endovascular aortic repair (FB-EVAR) led to expansion in the indications of EVAR to include patients with complex aneurysms involving the aortic arch and thoracoabdominal aorta.1,11 These techniques potentially decrease the risk of early morbidity and mortality compared with open surgical repair, but the need for secondary interventions and continued surveillance represents important limitations.12,13 Among patients with genetically triggered aortic diseases, open surgical repair remains the first line of treatment. 1 In these cases, FB-EVAR may be considered for treatment of recurrent aneurysms or in high-risk patients. Concerns with the use of endovascular stents include the potential risk of arterial injury and progressive enlargement due to continued radial expansion of a native aorta, which may result in loss of sealing zone, retrograde dissection, or rupture. 14 This study aimed to investigate the early and mid-term outcomes of FB-EVAR for the treatment of complex aneurysms involving the renal and mesenteric arteries among patients with or without familial history of aortic aneurysm or dissection in the absence of genetically triggered aortic diseases.
Materials and Methods
Study Design
The study is a retrospective review of patients enrolled in 2 prospective non-randomized physician-sponsored investigational device exemption (PS-IDE) studies by a single primary investigator in 2 academic centers (G130030 and G130266; ClinicalTrials.gov NCT01937949 and NCT02089607) between November 2013 and December 2022. The study was approved by the Institution Review Board of the UTHealth Houston and Mayo Clinic with data sharing agreement and all patients consented for inclusion in the PS-IDE and retrospective analysis. We included all consecutive patients treated by FB-EVAR for complex abdominal aortic aneurysms (CAAAs) and thoracoabdominal aortic aneurysms (TAAAs) using off-the-shelf or patient-specific fenestrated-branched stent-grafts manufactured by Cook Medical Inc (Bloomington, Indiana). Patients with heritable thoracic aortic disease (HTAD), such as Marfan syndrome (MFS), Loeys-Dietz syndrome (LDS), and vascular Ehlers-Danlos syndrome (EDS), confirmed by genetic assessment, were excluded from the analysis.
The family history of aortic disease—defined as the presence of thoracic or AAA or dissection in a first- or second-degree relative—was obtained through direct interviews with the patient and/or their family members, conducted by the primary physician, advanced clinical practitioner, or study research coordinator. Demographics, clinical, anatomical variables, procedural variables, imaging, and postoperative outcomes were collected prospectively in case report forms and entered into Medidata Rave electronic database (Medidata Solutions Inc, Boston, Massachusetts). All patients were followed per PS-IDE protocol with clinical examination, laboratory tests, quality of life questionnaires, duplex renal-mesenteric ultrasound, and computed tomography angiography (CTA) at 6 to 8 weeks, 6 months, 12 months, and annually thereafter for 5 years. The imaging studies were assessed by a vascular radiologist and study team. The clinical data and events were monitored by an independent regulatory team, adjudicated by the clinical event committee and data safety monitoring board (DSMB), and reported annually to the Food and Drug Administration (FDA).
Device Planning
All procedures were planned by the principal investigator using the centerline of flow analysis of preoperative CTA. Specifics of stent-graft design have been previously reported but included either an off-the-shelf multibranched stent-graft or a patient-specific stent-graft with any combination of fenestrations or directional branches, with or without concurrent iliac branch devices if needed.
Endpoints
The clinical data and endpoints were analyzed in all patients and patients with or without a history of aortic aneurysm and/or dissection affecting a first- or second-degree relative. Patients with HTAD such as MFS, LDS, and vascular EDS, were excluded from the analysis. The reporting standards of the Society for Vascular Surgery (SVS) for endovascular repairs requiring incorporation of renal and mesenteric arteries were used to define variables, aneurysm extent, and endpoints. 15 Technical success was defined as successful deployment of the fenestrated-branched stent-graft with successful stenting of all intended target vessels and no evidence of type I or III endoleak on completion angiography. Primary endpoints were rates of major adverse events (MAEs), which were defined as a composite of any-cause mortality, acute kidney injury, major stroke, paraplegia, myocardial infarction, respiratory failure requiring prolonged (>24 hours from anticipated) mechanical ventilation or reintubation, and bowel ischemia within 30 days or hospital stay if longer than 30 days. Aneurysm sac dynamics, endoleaks, and stent-graft-related outcomes were assessed using the proposed SVS reporting standard definitions.
Statistical Analysis
Continuous variables are reported as the mean±standard deviation (SD) or the median with interquartile range (IQR) for the description of normally or non-normally distributed parameters. The categorical variables are reported as proportions. Preoperative characteristics, device and procedural data, and early outcomes were compared between 2 cohorts using the 2-sample t or Mann-Whitney U-tests for continuous variables, and Pearson’s chi-square tests or Fischer’s exact test for categorical variables. Mid-term time-dependent outcomes were calculated using the Kaplan-Meier method, and a log-rank test was used to determine the differences. A p-value<0.05 was considered as statistical significance. All statistical analyses were performed using SPSS statistics (Version 28.0, IBM Corp, Armonk, New York) and R version 2022.07.2 (RStudio Inc, Boston, Massachusetts). Accordingly, the primary and secondary endpoints were compared between patients with and without FAA.
Results
Study Patients
There were 506 consecutive patients enrolled in the study, including 350 male (69%) and 156 female (31%) patients with a mean age of 74±8 years old. Seven patients (1.4%) had HTADs with specific mutations and were excluded from the analysis. Among 499 patients with no diagnosis of HTAD, there were 66 patients (13%) with FAAs, and 433 patients (87%) with sporadic aortic aneurysms (SAAs). Clinical characteristics were similar in both groups (Table 1) with a non-significant trend toward younger age at presentation (72±8.6 vs 74±8.1 years old; p=0.09) and a lower rate of coronary artery disease (38% vs 49%; p=0.09) in patients with FAAs compared with SAAs, respectively. There were no significant differences in aneurysm extent and distribution of aortic disease, maximum aneurysmal sac diameter, and incidence of prior aortic dissection between the 2 groups. History of any prior aortic repair (open or endovascular) was present in 39 patients (59%) with FAAs and 204 patients (47%) with SAAs (p=0.11).
Clinical Characteristics, Cardiovascular Risk Factors, Aortic History, and Anatomical Features of 499 Patients Undergoing Fenestrated-Branched Endovascular Aortic Repair.
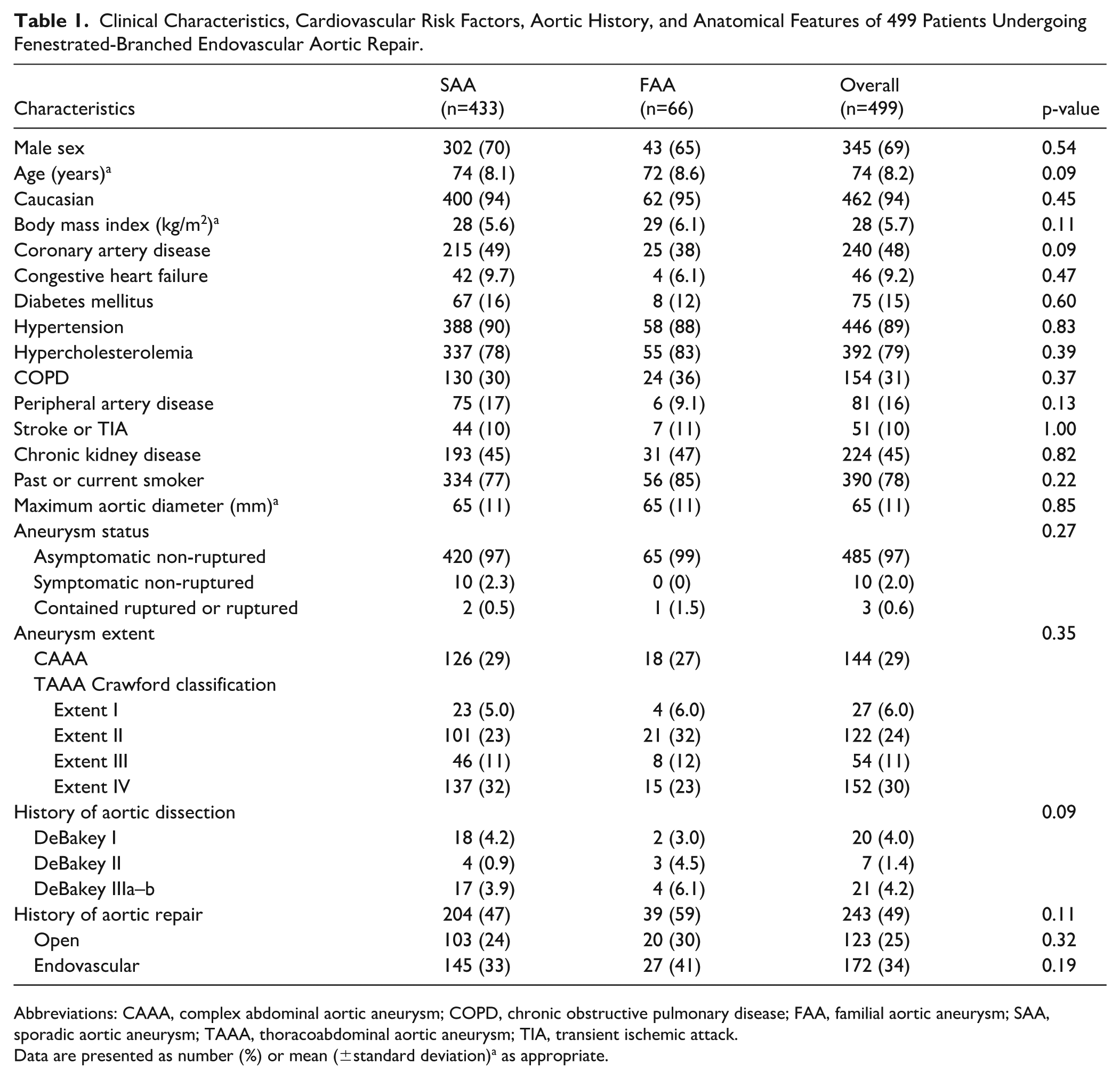
Abbreviations: CAAA, complex abdominal aortic aneurysm; COPD, chronic obstructive pulmonary disease; FAA, familial aortic aneurysm; SAA, sporadic aortic aneurysm; TAAA, thoracoabdominal aortic aneurysm; TIA, transient ischemic attack.
Data are presented as number (%) or mean (±standard deviation)a as appropriate.
Procedural Data
A total of 1835 renal and mesenteric arteries were incorporated using 1274 reinforced fenestrations, 561 directional branches, and 74 double-wide scallops. Differences in stent-graft design between the 2 groups (Table 2) included slightly lower usage of reinforced fenestrations among patients with FAAs (68% vs 70%, p<0.01) with no difference in the use of directional branches and double-wide scallops. Both groups had similar procedural variables, except for higher total fluoroscopy time and radiation dose among patients with FAAs. Overall technical success was 96% with no difference among patients with FAAs or SAAs (94% vs 97%; p=0.22), respectively.
Device Characteristics and Procedural Data of 499 Patients Undergoing Fenestrated-Branched Endovascular Aortic Repair.
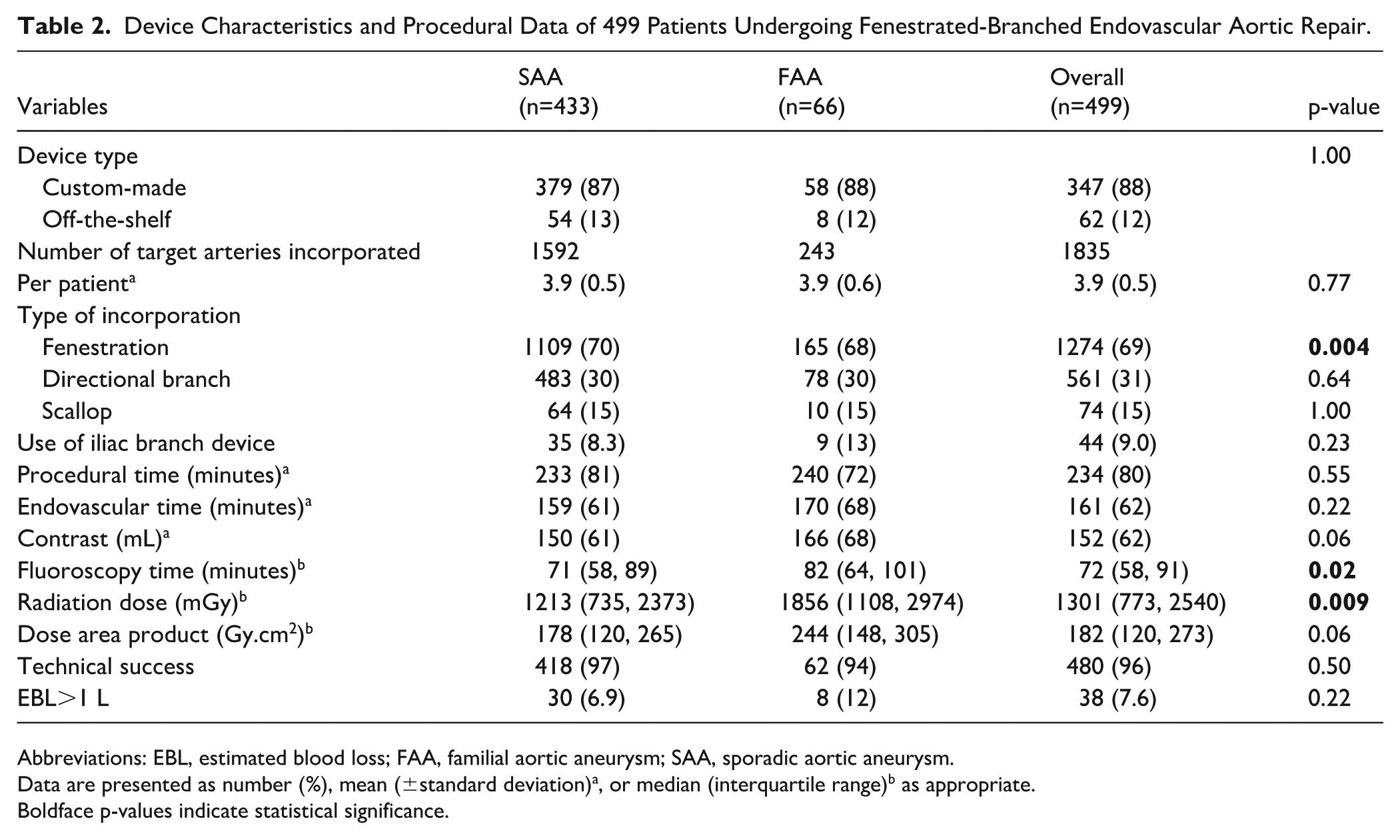
Abbreviations: EBL, estimated blood loss; FAA, familial aortic aneurysm; SAA, sporadic aortic aneurysm.
Data are presented as number (%), mean (±standard deviation)a, or median (interquartile range)b as appropriate.
Boldface p-values indicate statistical significance.
Early Outcomes
The 30-day or in-hospital mortality was 1% (n=5/499) for the entire cohort with no difference among patients with FAAs or SAAs (0.9% vs 1.5%, p=1.0). At least 1 MAE occurred in 92 patients (19%), with no differences between the 2 groups (Table 3). The need for secondary intervention within the first 30 days occurred in 50 patients (10%), with no difference between groups. The median (IQR) length of stay in the intensive care unit (ICU) and hospital were 3 (2, 5) and 5.5 (5, 10) days, respectively, with slightly longer ICU stay lengths for the FAA group. Hospital discharge to home occurred in 414 patients (84%), with no difference between groups.
Thirty-Day Outcomes of 499 Patients Following Fenestrated-Branched Endovascular Aortic Repair.
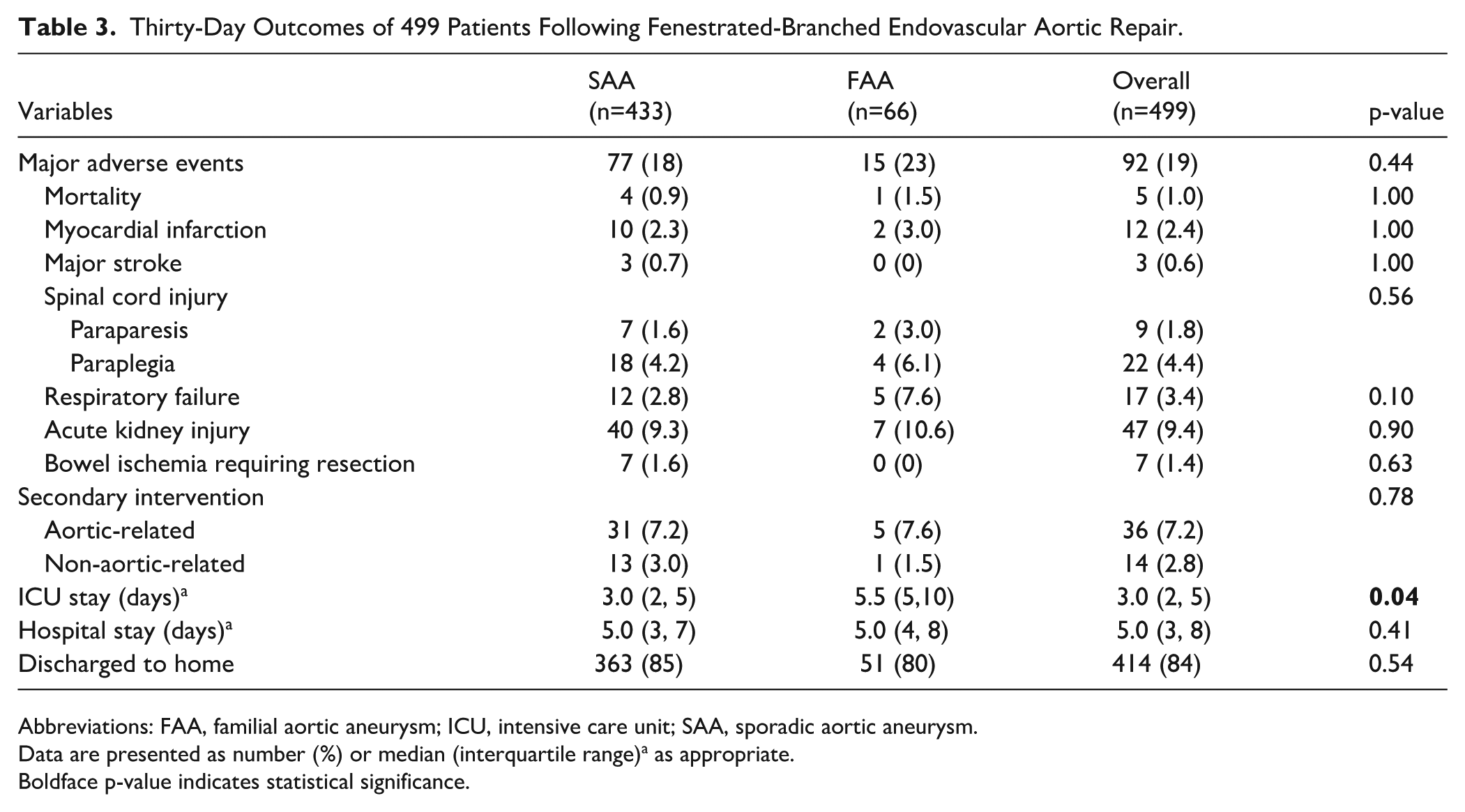
Abbreviations: FAA, familial aortic aneurysm; ICU, intensive care unit; SAA, sporadic aortic aneurysm.
Data are presented as number (%) or median (interquartile range)a as appropriate.
Boldface p-value indicates statistical significance.
Mid-term Outcomes
The median (IQR) follow-up was 28 (4, 66) months for the entire cohort. Patient survival at 1, 3, and 5 years was 91±1.4%, 76±2.5%, and 48±4.4%, respectively, for all patients with no difference between groups (Figure 1A). Freedom from any secondary intervention at 1, 3, and 5 years was 83±1.8%, 74±2.5%, and 63±4.4%, respectively, for all patients with no difference between groups (Figure 1B). The cumulative incidence of aneurysm sac shrinkage >5 mm was 9±1.4%, 32±2.6%, and 69±3.1% for all patients at 1, 3, and 5 years, respectively, with no difference between groups (Figure 2A). Freedom from type I or III endoleak at 1, 3, and 5 years was 89±1.5%, 85±1.9%, and 79±3.0%, respectively, for all patients with no difference between groups (Figure 2B).
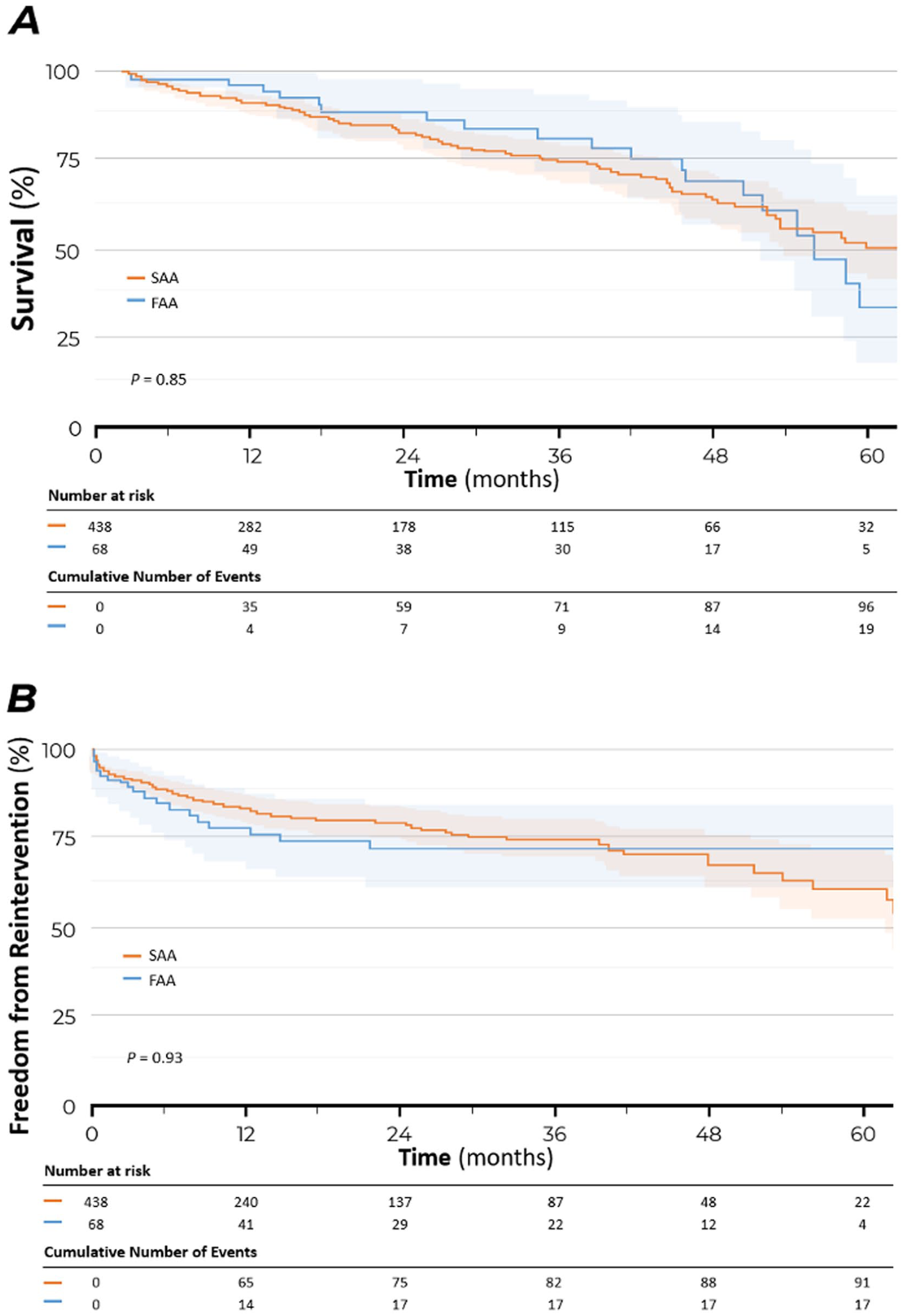
Kaplan-Meier survival estimates for patient survival (A) and freedom from secondary intervention (B) after fenestrated-branched endovascular aortic repair in patients with familial aortic aneurysm (FAA, blue) as compared with patients with sporadic aortic aneurysm (SAA, orange).
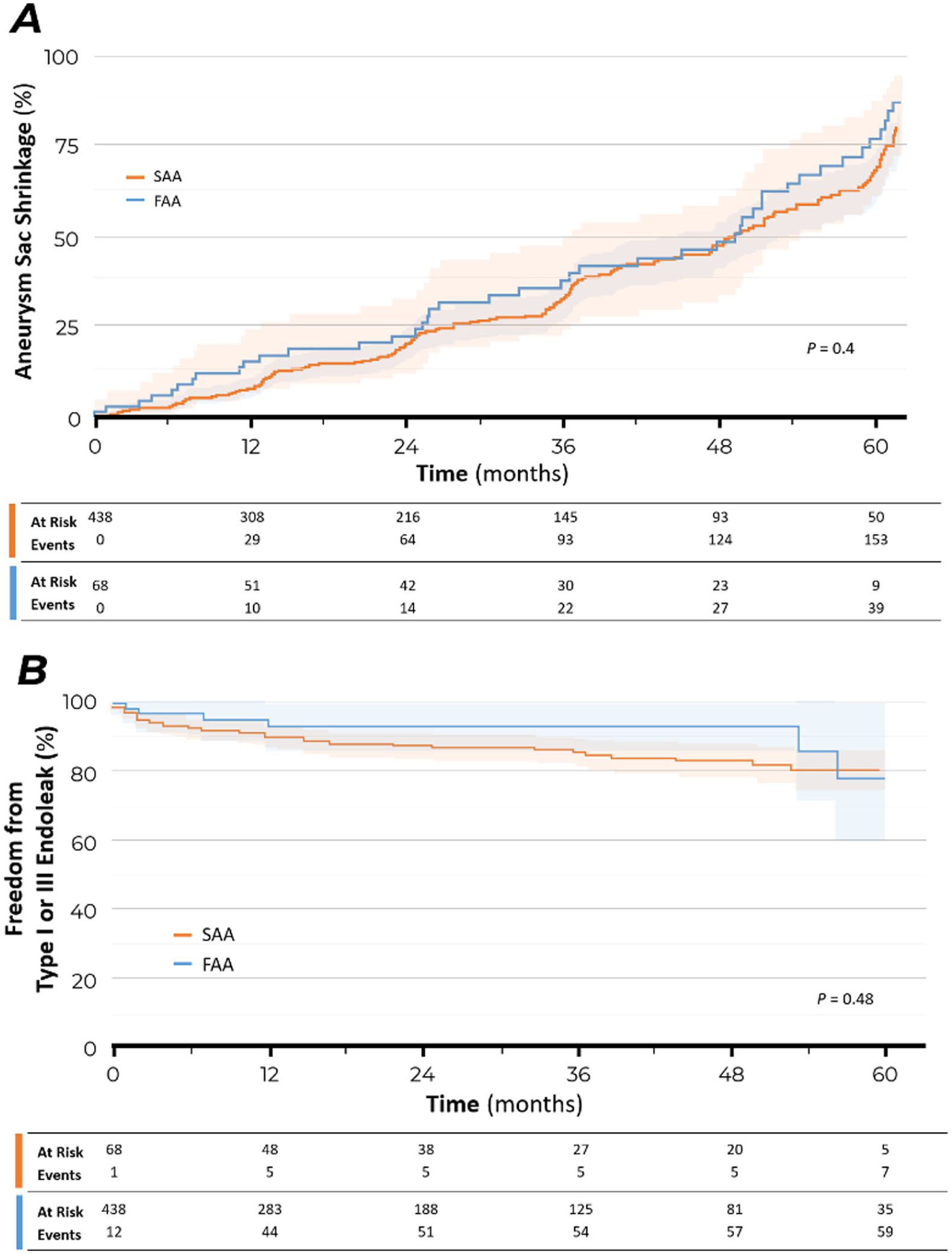
Kaplan-Meier survival estimates for the cumulative rate of sac shrinkage (A) and freedom from type I or III endoleak (B) after fenestrated-branched endovascular aortic repair in patients with familial aortic aneurysm (FAA, blue) as compared with patients with sporadic aortic aneurysm (SAA, orange).
Discussion
This study highlights the benefits of FB-EVAR including low early and late aortic-related mortality, irrespective of the presence of a familial history of aortic aneurysm and/or dissection, after excluding patients with known genetic mutations and diagnosis of HTAD. There were no significant differences with respect to clinical characteristics, aneurysm extent, most of the procedural variables, and outcomes in the early and mid-term follow-up interval. Overall, FB-EVAR was an effective technique in patients with FAAs and SAAs with a low cumulative incidence of aortic-related mortality up to 5 years of follow-up. Despite continued improvements in genetic testing, most patients with FAAs (97%) do not have identifiable genetic mutations or syndromes. Among patients with genetically triggered aortic diseases, FB-EVAR was used selectively with favorable results.
In our study population, there were no significant differences in demographics and cardiovascular risk factors, except for a slight non-significant trend toward younger age among FAA patients. This is similar to the findings of prior reports, including the analysis of a larger cohort in the United States Aortic Research Consortium (US-ARC).2,16 Brown and associates demonstrated the initial presentation of aortic aneurysms in patients with a positive family history at younger ages. Besides, they reported this group to have more frequent proximal aortic involvement. 10 In the US-ARC study, findings were also nearly identical to our study. 16 There was no significant difference in aneurysm extent and distribution of aortic disease between patients with and without FAA. In another study on participants screened for AAA in the VIVA trial, first-degree male relatives of AAA patients were shown to have wider aortas and a 2-fold higher prevalence of AAA compared with the age-adjusted background population. 17
The similar aneurysm extent and target-related outcomes observed in our cohort suggest that fenestrated-branched stent-graft design can generally be similar for patients with FAA or SAA. Procedural planning should focus on patient-specific anatomical characteristics, including gap distance, tortuosity, access, and luminal diameter, rather than family history alone. However, this approach may not apply to patients with genetically triggered aortic diseases, who are at higher risk for complications, such as stent-induced new entry tears or target vessel endoleaks. In these cases, stent-graft selection and design—such as type of incorporation or bridging stent—should be individualized, and open surgical repair may be preferred due to concerns regarding tissue fragility and long-term durability.18,19
A continued concern with endovascular approaches and FB-EVAR is the high rate of secondary interventions. A recent review of the US-ARC indicated that freedom from any secondary intervention was 88% at 5 years, 20 although the vast majority of procedures were classified as minor and low clinical magnitude and these have not affected survival. These rates are very similar to our findings both in the FAA and SAA cohorts. A continued concern is that patients with FAAs may have unidentified genetic mutations and may be more predisposed to secondary failures from the progression of aortic disease. 10 Traditionally, genetic disorders were excluded from pivotal trials evaluating the effectiveness of stent-grafts because of potential risks of arterial trauma or late enlargement of sealing zones due to radial force of self-expandable stent-grafts or direct injury related to active fixation mechanisms. In this study, we only had 7 patients with genetically triggered aortic diseases, most often after failed proximal aortic repairs. Among these patients, we had 1 retrograde aortic dissection when the stent was placed in native aortic tissue. After excluding the patients with genetically triggered aortic diseases, subgroup analysis identified similar results with no difference between FAAs and SAAs.
Patients with HTAD and those with FAAs without identifiable mutations represent a broad spectrum of heritable aortic conditions with variable effects on aortic wall fragility.10,21,22 These patients should be analyzed and grouped into different categories. Within the group of patients with HTAD, prognosis differs greatly depending on specific mutation, with similar survival in patients with MFS and LDS, but markedly decreased survival in those with EDS. 23 In addition to survival, vulnerability to the aorta and branches to the radial force exerted by self-expandable stent-grafts is also variable. 24 Patients with EDS, MFS, and LDS exhibit distinct phenotypic differences in their vascular manifestations. In MFS, aortic dilation predominantly affects the sinuses of Valsalva and the ascending aorta, forming a characteristic “pear-shaped” annuloaortic ectasia, leading to progressive aortic root enlargement and dissection, emphasizing the need for surgical intervention.25,26 Conversely, LDS is characterized by a more aggressive vascular phenotype, with aortic aneurysms growing at rates exceeding those seen in MFS, resulting in a younger age of death primarily due to aortic dissections. Vascular EDS presents a different challenge, with a more dire prognosis compared with other EDS subtypes, often associated with visceral artery pathology.25,27 Despite these distinctions, FAA without identifiable mutations may represent a less aggressive variant, albeit distinct from severe aortic aneurysms, such as those observed in LDS. 25
Few reports have analyzed the effects of FAA on outcomes of endovascular repair. van de Luijtgaarden and colleagues identified higher 30-day mortality among EVAR patients with FAAs compared with SAAs, but this early difference did not affect late survival in both groups. 28 Ryer and colleagues conducted a retrospective single-institution review of 392 patients, including 89 patients with FAA, undergoing open and endovascular AAA repair. They reported an increased risk of delayed type I endoleak in patients with FAA, regardless of the specific stent-graft used. Although FAA group did not incur any significant differences in MAEs after EVAR or open surgical repair, patients with FAA did experience a significantly higher rate of endoleak and secondary intervention after EVAR compared with open surgical repair. 19 In this study, we found no difference in rates of type I endoleak with more extensive endovascular repairs. Moreover, changes in sac dynamics were nearly identical between patients with FAA and SAA in our study. These differences in our study compared with prior reports may be explained by the more extensive nature of the repairs with liberal use of extensive supra-celiac sealing zones to minimize risks of disease progression and late failure.
This study has several major limitations. Routine genetic testing was not used in our cohort and was selectively indicated according to guidelines in patients who presented with aneurysms or dissections before 50 years of age or had a first- or second-degree relative with identified genetically triggered aortic disease. It is possible that additional patients with genetically triggered aortic diseases would have been identified with routine testing. Follow-up interval beyond 5 years is still limited, and therefore, conclusions about the durability of FB-EVAR are limited beyond this interval. Although the clinical data and outcomes were prospectively collected and adjudicated, these outcomes represent the late experience of a single surgeon in 2 tertiary care aortic centers, and should not be generalized.
Conclusion
The FB-EVAR appears to be safe and effective, with low early and mid-term cumulative incidence of aortic-related mortality in patients with FAAs or SAAs. The presence of a prior familial history of aortic aneurysm or dissection did not affect technical outcomes, MAEs, mortality, patient survival, secondary interventions, and changes in sac dynamics. Although further studies are required to more clearly define the role of familial aortic disease, FB-EVAR could be considered a valuable alternative to open surgical repair in patients who meet clinical and anatomical criteria.
Footnotes
Authors’ Note
This study was presented in the International Poster Competition at the 2023 Vascular Annual Meeting (VAM) of the Society for Vascular Surgery, National Harbor, Maryland, June 14 to 17, 2023.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Bernardo C. Mendes received consulting fees and research grants from Cook Medical and W.L. Gore, and the scientific advisory board of Medtronic. Dr Gustavo S. Oderich has received consulting fees and grants from W.L. Gore and GE Healthcare with personal reimbursement. He has consulting agreements with Cook Medical and Centerline Biomedical with no personal reimbursement. GE Healthcare has provided research support to The University of Texas Health Science at Houston and Baylor College of Medicine. Other co-authors: none.
