Abstract
Keywords
What is a thoracic aortic aneurysm (TAA)?
An aneurysm occurs when the walls of a blood vessel weaken, causing it to enlarge or dilate. Aneurysms can form in any blood vessel in the body, but they are most common in the aorta. 1
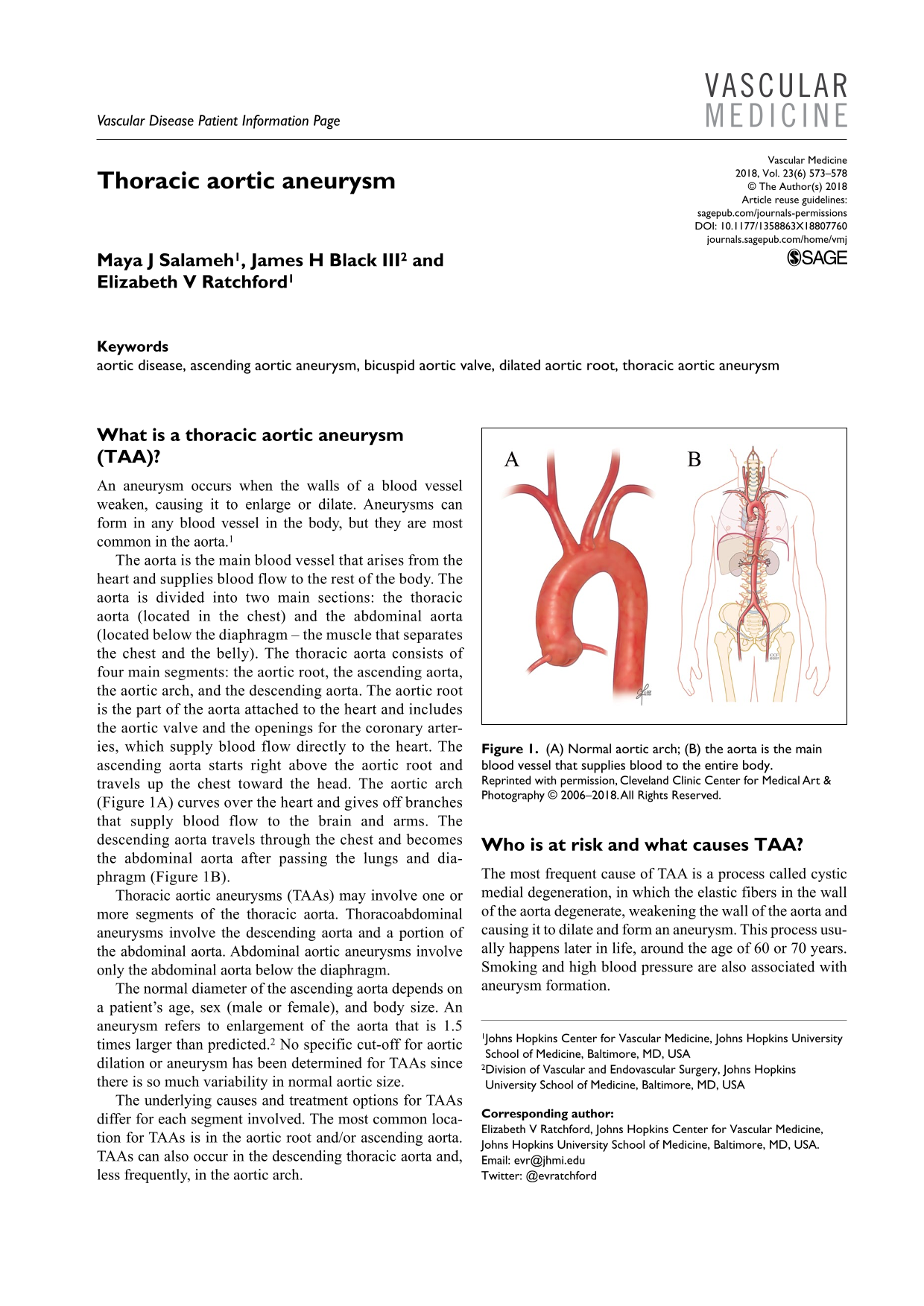
The aorta is the main blood vessel that arises from the heart and supplies blood flow to the rest of the body. The aorta is divided into two main sections: the thoracic aorta (located in the chest) and the abdominal aorta (located below the diaphragm – the muscle that separates the chest and the belly). The thoracic aorta consists of four main segments: the aortic root, the ascending aorta, the aortic arch, and the descending aorta. The aortic root is the part of the aorta attached to the heart and includes the aortic valve and the openings for the coronary arteries, which supply blood flow directly to the heart. The ascending aorta starts right above the aortic root and travels up the chest toward the head. The aortic arch (Figure 1A) curves over the heart and gives off branches that supply blood flow to the brain and arms. The descending aorta travels through the chest and becomes the abdominal aorta after passing the lungs and diaphragm (Figure 1B).
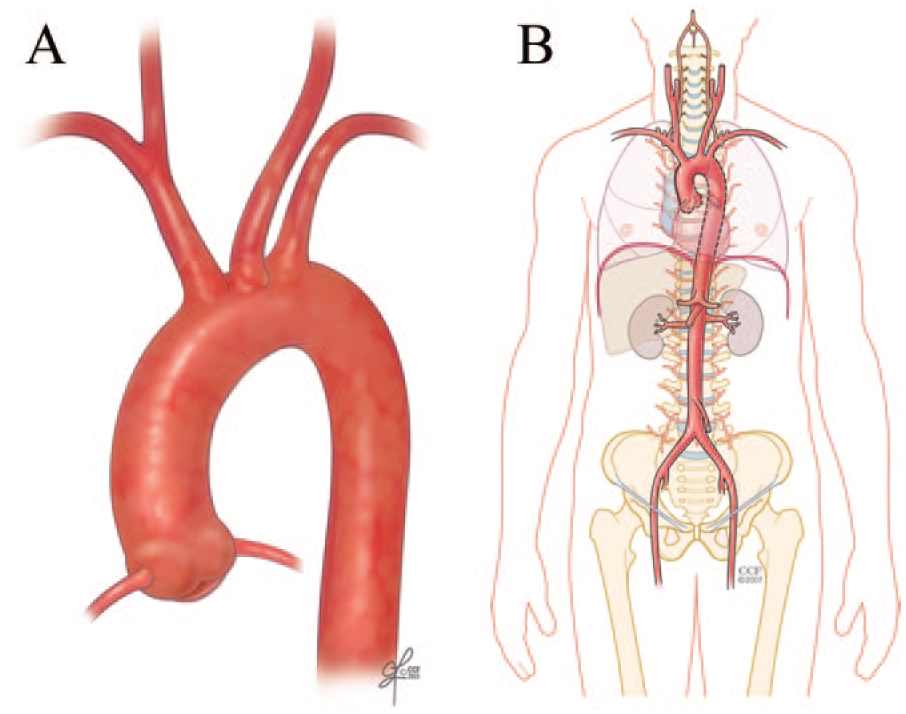
(A) Normal aortic arch; (B) the aorta is the main blood vessel that supplies blood to the entire body.
Thoracic aortic aneurysms (TAAs) may involve one or more segments of the thoracic aorta. Thoracoabdominal aneurysms involve the descending aorta and a portion of the abdominal aorta. Abdominal aortic aneurysms involve only the abdominal aorta below the diaphragm.
The normal diameter of the ascending aorta depends on a patient’s age, sex (male or female), and body size. An aneurysm refers to enlargement of the aorta that is 1.5 times larger than predicted. 2 No specific cut-off for aortic dilation or aneurysm has been determined for TAAs since there is so much variability in normal aortic size.
The underlying causes and treatment options for TAAs differ for each segment involved. The most common location for TAAs is in the aortic root and/or ascending aorta. TAAs can also occur in the descending thoracic aorta and, less frequently, in the aortic arch.
Who is at risk and what causes TAA?
The most frequent cause of TAA is a process called cystic medial degeneration, in which the elastic fibers in the wall of the aorta degenerate, weakening the wall of the aorta and causing it to dilate and form an aneurysm. This process usually happens later in life, around the age of 60 or 70 years. Smoking and high blood pressure are also associated with aneurysm formation.
In younger patients, TAA is often due to a genetic cause. These include connective tissue disorders such as Marfan syndrome, Loeys–Dietz syndrome, and vascular Ehlers–Danlos syndrome. Cystic medial degeneration can also be seen in TAAs that are not due to a specific connective tissue disorder but still have a genetic link. This is referred to as familial TAA syndrome. Turner syndrome is another genetic disease, affecting women, that is associated with TAAs. TAAs with a genetic cause usually involve the aortic root and/or ascending aorta. Other genetic mutations associated with TAA have also been identified. In many of these cases, multiple family members have had an aortic aneurysm and/or dissection (tear in the aorta).
In addition, TAAs can be associated with a bicuspid aortic valve. This condition is present from birth (congenital), and is when the aortic valve of the heart has only two cusps (bicuspid; Figure 2A) instead of the normal three (tricuspid; Figure 2B). Cystic medial degeneration is also the main cause of TAAs associated with a bicuspid aortic valve. About 44% of those aneurysms involve the ascending aorta, and 20% involve the aortic root. 2 The aorta can still dilate even if the bicuspid valve is functioning normally.
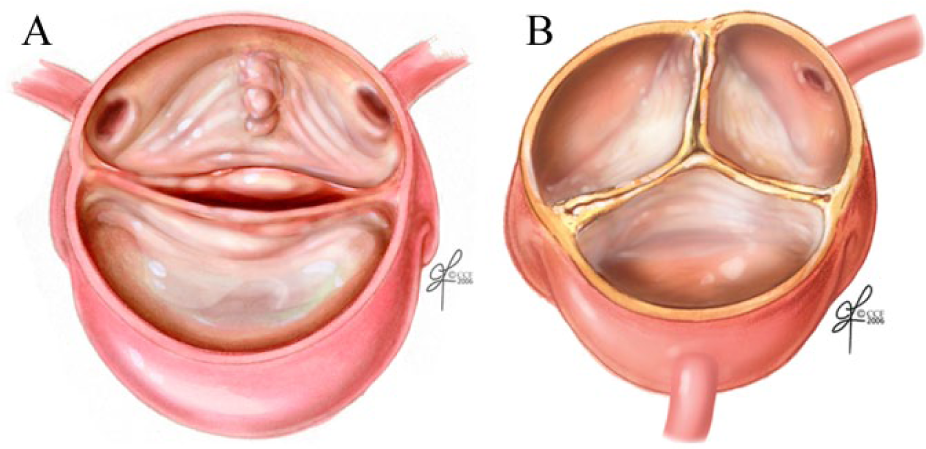
(A) A bicuspid aortic valve (only two cusps), a condition present from birth that is linked to thoracic aortic aneurysms; (B) a normal tricuspid aortic valve with three cusps.
Aortitis, or inflammation of the aorta, can also lead to TAA. Aortitis can be due to an underlying infection or can be non-infectious. Syphilis is an example of infectious aortitis, which causes TAA by destroying collagen and elastic tissues within the walls of the aorta. Syphilis was once the most common cause of TAAs involving the ascending aorta; however, with modern antibiotics, syphilitic aneurysms are rare today. Non-infectious causes of aortitis include giant cell arteritis and Takayasu’s arteritis, which are both uncommon diseases that cause inflammation in the blood vessel wall.
Atherosclerosis, or plaque build-up in the arteries, may lead to aneurysm formation over time, especially in the descending aorta and aortic arch. Trauma from deceleration injuries, such as from major car accidents or injury from contact sports, may also lead to TAA. The typical location for these aneurysms is at the part of the aorta just after the aortic arch.
The causes of TAA are summarized in Table 1.
Causes of TAA.
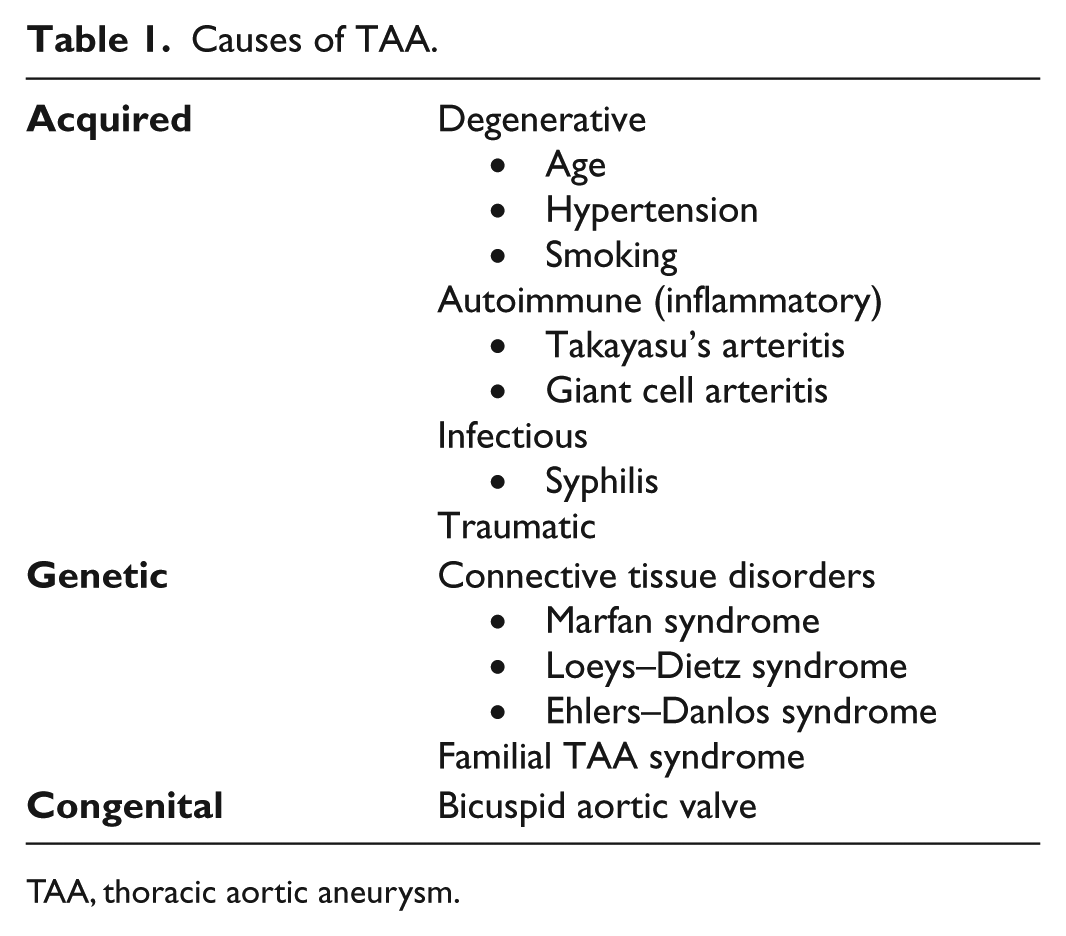
TAA, thoracic aortic aneurysm.
Is TAA inherited? What about genetic testing?
Patients with TAA may be referred to genetic counselors or medical genetic specialists to look for an inherited cause, particularly if several family members have had problems with the aorta or if the aneurysm involves the ascending aorta. Certain physical features may suggest a genetic cause of TAA, such as cleft palate, widely spaced eyes, long fingers, and abnormally flexible joints.
Finding a genetic mutation can be helpful not only for the patient but also for family members who can then also be tested. For example, if Marfan syndrome is suspected based on physical features, then testing for the FBN1 gene can be performed. Genetic testing can help to diagnose or rule out other conditions such as Loeys–Dietz and vascular Ehlers–Danlos syndromes. Even if the tests are negative (normal), there still may be a genetic cause but it may be from a gene that has not yet been discovered.
First-degree relatives (parents, brothers, sisters, and children) of patients with TAA should be screened because family studies have found an approximately 20% chance of another first-degree relative having a TAA. 3 Therefore, even if genetic testing does not reveal an identifiable mutation, first-degree relatives should have their aorta checked. An echocardiogram is usually recommended, especially in relatives of patients with Marfan syndrome or bicuspid aortic valve. Other imaging may be needed to see the entire aorta in certain situations (see below).
What are the signs and symptoms of TAA?
Most people with TAA have no symptoms. TAAs are typically found incidentally when the patient is undergoing an imaging study for another reason. Aneurysms of the aortic root may lead to leakage of the aortic valve, so a heart murmur may be heard on physical examination. The most serious consequence of TAA is aortic dissection or aortic rupture. Ruptured TAAs are most often fatal, but symptoms of rupture include severe chest or back pain with severely low blood pressure and shock.
Most patients with an aortic dissection experience severe and sudden chest pain that is sharp or tearing, particularly in dissections involving the ascending aorta (type A). With dissections in the descending aorta (type B), the pain may be more in the upper back or in the abdomen. In both types of aortic dissection, the pain is often described as the worst pain a patient has ever experienced. Other symptoms of aortic dissection caused by a decrease in blood flow to the body include fainting, nausea/vomiting, sweating, shortness of breath, or leg pain. 4
Aortic dissection can also cause a heart attack, heart failure, or stroke due to lack of blood flow to the heart or brain. Unfortunately, the diagnosis of aortic dissection may be delayed when patients do not have typical symptoms. Other signs of aortic dissection include a new heart murmur, weak pulses, or a difference in the blood pressure measurement between the right and left arms. 4
It is important to recognize the symptoms of aortic rupture or dissection as quickly as possible. Any of these symptoms should prompt immediate evaluation in the emergency room. The patient should inform the emergency room physician that he or she has a known TAA and that a computed tomography (CT) scan of the chest is needed to rule out aortic dissection or rupture.
How is TAA diagnosed?
Computed tomography angiography (CTA) or magnetic resonance angiography (MRA) are the imaging tests of choice for diagnosing and measuring TAAs. Disadvantages of CTA include radiation exposure and the need for using intravenous (IV) contrast dye, which some patients cannot receive due to allergy or reduced kidney function. Disadvantages of MRA include the length of the test (45–60 minutes), which can be uncomfortable, especially for patients who are claustrophobic or have trouble lying flat. MRA generally requires the use of gadolinium for contrast, which could be a problem for patients with reduced kidney function. Figure 3 shows a CTA image and Figure 4 shows an MRA image.
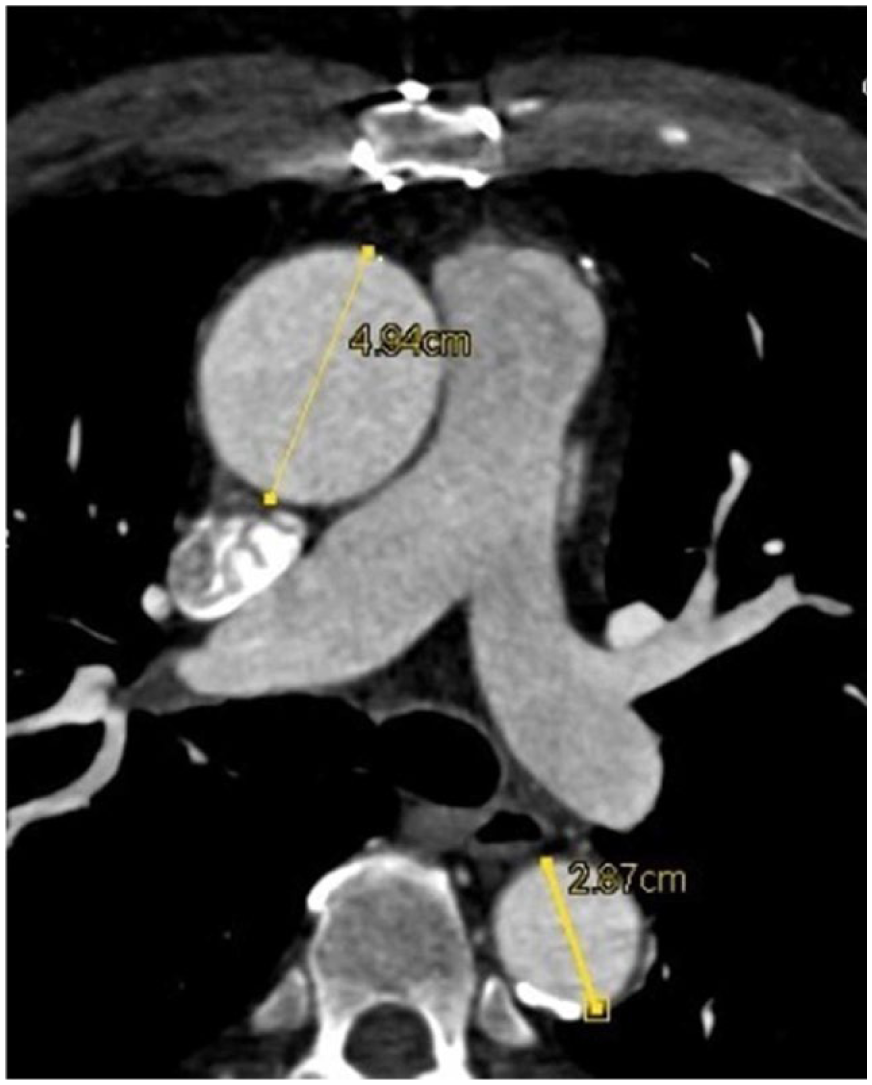
CTA image of a 4.94-cm ascending aortic aneurysm in a 57-year-old man with familial thoracic aortic aneurysm syndrome. Note: the descending aorta seen in the same image is normal, measuring 2.87 cm in diameter.
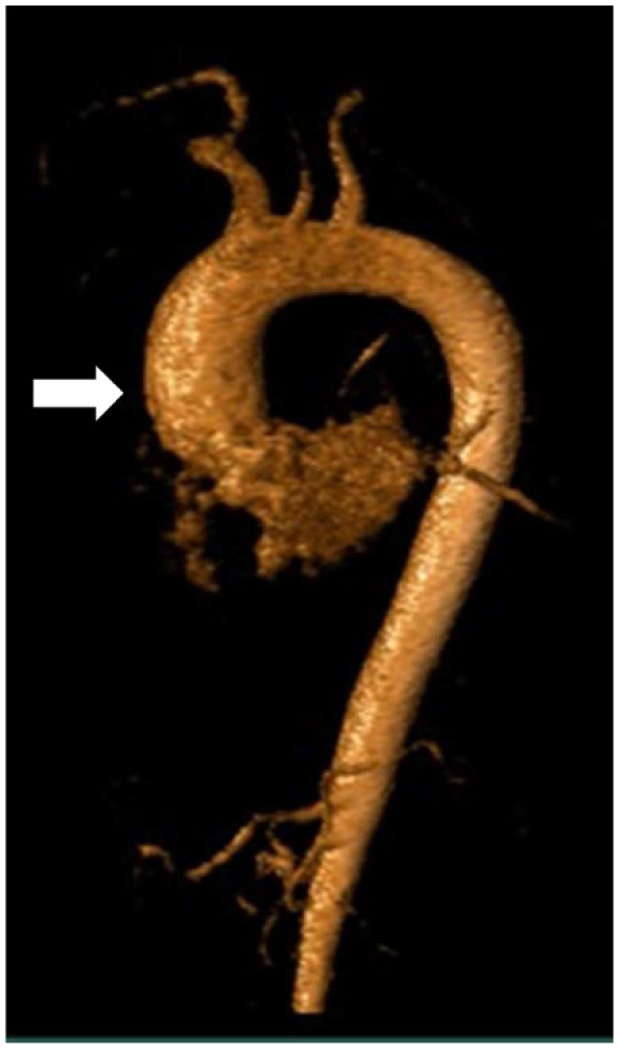
MRA image of a 4.5-cm ascending aortic aneurysm in a 79-year-old woman with hypertension and a history of heavy smoking. Note the dilated appearance of the ascending aorta (arrow) relative to the arch and the descending aorta.
Echocardiography (an ‘echo’) may help in the diagnosis of TAA. An echo is an ultrasound of the heart and aorta using a probe with gel over the chest. Echo is useful to look at the aortic root but may miss aneurysms farther away from the heart in the ascending aorta and aortic arch. The descending aorta cannot be easily seen by echo. An ascending aortic aneurysm detected by echo is shown in Figure 5.
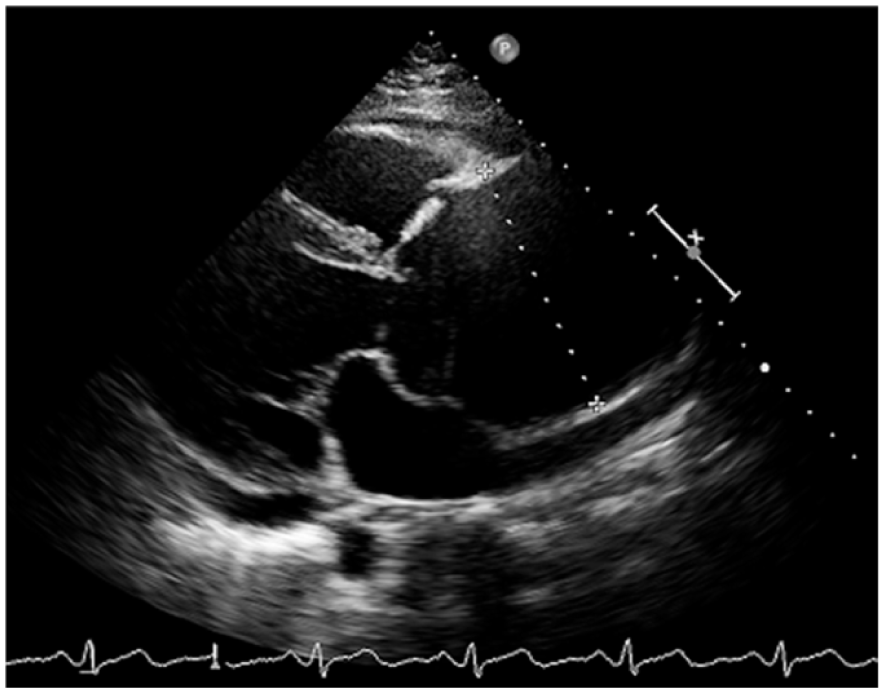
Echocardiography image showing a large aneurysm of the ascending aorta measuring 8.2 cm in a 54-year-old man with a bicuspid aortic valve.
If a TAA is detected, the abdominal aorta should be imaged (by ultrasound, CTA, or MRA) to look for an abdominal aortic aneurysm. Patients with TAA are also at increased risk for developing brain aneurysms. However, the role of brain aneurysm screening is unclear. Each patient should discuss screening for other aneurysms with his or her doctor.
What is the medical treatment for TAA?
The medical treatment for TAA depends on its location and suspected cause. In general, medications to slow the growth of TAAs and prevent dissection and rupture are limited. Beta-blocker blood pressure medications (e.g. propranolol or metoprolol) have been shown to slow the rate of growth and decrease the chances of death for certain patients with TAA. Beta-blockers work by reducing stress on the wall of the aorta. Other blood pressure medications such as losartan, an angiotensin-receptor blocker, have been shown to slow the growth rate of aortic aneurysms in patients with Marfan syndrome by interfering with signaling of the TGF-beta protein. 5 Blood pressure should be maintained as close to normal (i.e. ⩽ 120/80 mmHg) as possible.6,7
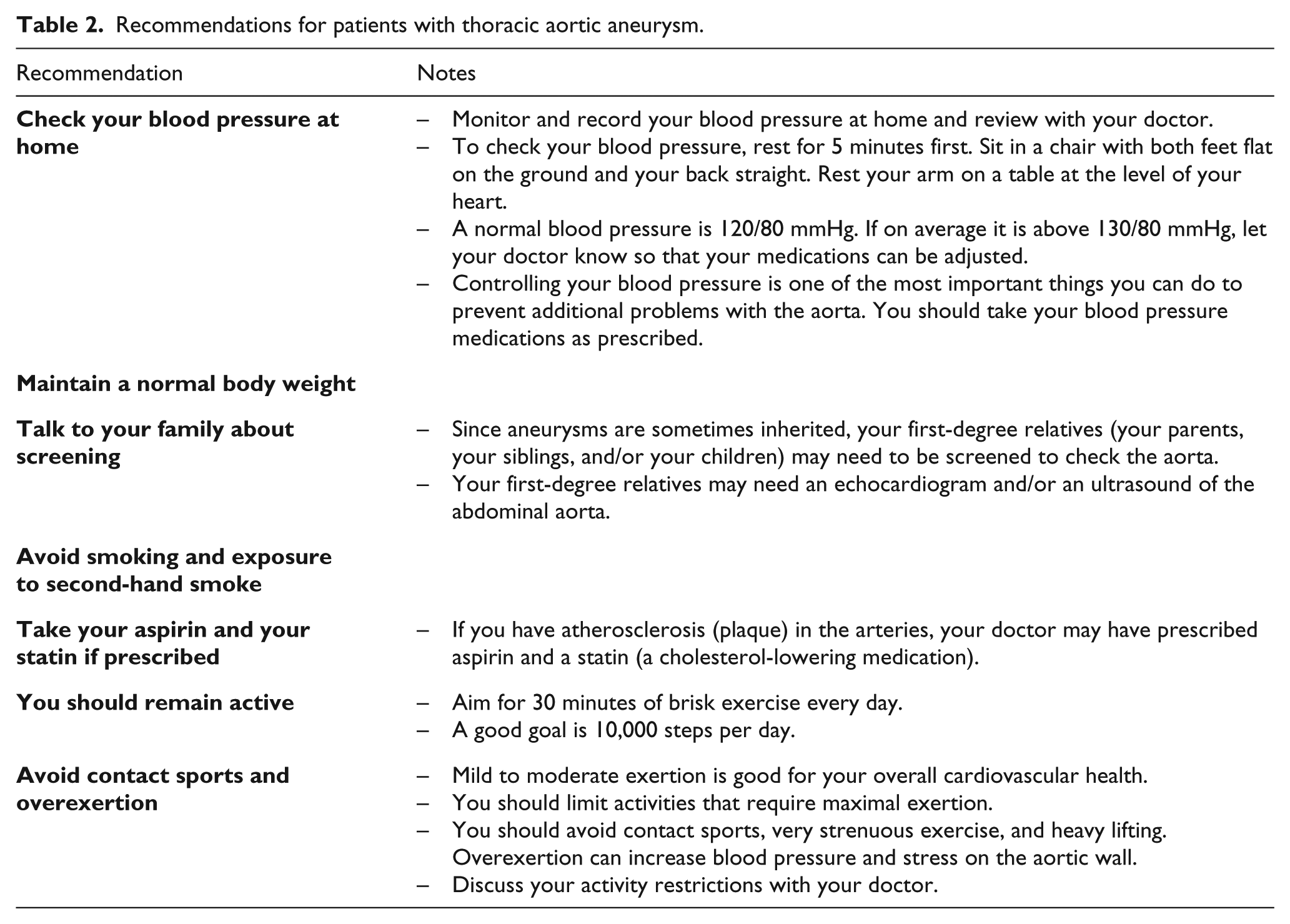
Aneurysms of the aortic arch and descending aorta are most often associated with atherosclerosis. In patients with plaque (atherosclerosis) in the arteries, additional medical treatment is recommended to reduce the risk of heart attack and stroke. A blood-thinning medication such as aspirin is usually prescribed to reduce this risk. A statin is also prescribed to reduce cholesterol and to reduce the risk of heart attack and stroke. Quitting smoking is essential. Diabetes should be controlled, and a normal body weight should be maintained (Table 2). 4
Recommendations for patients with thoracic aortic aneurysm.
How often does TAA need to be monitored?
Surveillance imaging is typically performed 6 months after the initial TAA diagnosis and then annually thereafter, with more frequent imaging in high-risk cases.
TAAs usually get bigger over time. The average rate of growth is approximately 0.1 cm per year. 8 However, certain aneurysms may grow more quickly. For example, TAAs in Loeys–Dietz syndrome can enlarge by up to 1 cm per year. 6 Larger TAAs tend to expand faster than smaller TAAs. The growth rate of individual aneurysms varies, so all TAAs should be monitored regularly with imaging. If the TAA is expanding or is linked to a genetic condition, then more frequent monitoring may be needed. 9
When is surgery recommended for TAA?
The annual risk of rupture or dissection for TAAs is approximately 2% for TAAs < 5 cm in diameter, 3% for TAAs 5–5.9 cm, and 7% for TAAs > 6 cm. 8 Balancing these risks against the risk of the procedure, surgery is recommended for TAAs ⩾ 5.5 cm in most cases.
However, the risk of rupture may be much higher at lower diameters in patients with certain genetic disorders. For example, surgical repair is typically recommended once the TAA reaches 4.5 cm for patients with Marfan syndrome and 4.0 cm for patients with Loeys–Dietz syndrome. 6 For patients with bicuspid aortic valve, surgery is recommended for aortic diameters ⩾ 5.5 cm but a lower threshold of 5.0 cm may be used in patients at higher risk for dissection, such as those with a family history of dissection or those with a rapidly expanding aneurysm. 10 For most descending TAAs, the typical surgical repair threshold is about 6 cm.
For any patient with a TAA growth rate exceeding 0.5 cm per year, surgical repair is often recommended given the high risk of rupture of aneurysms that are rapidly expanding.
What are the surgical options for TAA?
Surgical options for TAA depend on which part of the aorta is dilated. For ascending aortic aneurysms, repair requires a sternotomy (opening up the chest) and placing the patient on cardiopulmonary bypass. The diseased segment of the aorta is replaced with a tube graft. When the aneurysm involves the aortic root and is affecting aortic valve function (e.g. significant leakage of the valve), a procedure called the Bentall procedure is typically performed using a tube graft with a prosthetic aortic valve sewn into one end. 6 Valve-sparing surgeries can also be performed if the aortic valve function is adequate. The mortality rate of elective (not emergency) surgical repair of ascending aortic aneurysms is about 3–5% and usually requires a prolonged hospital stay after surgery, often up to 10 days.
For TAAs involving the descending or thoracoabdominal aorta, thoracic endovascular aortic repair (TEVAR) is a less invasive option with lower mortality rates and shorter hospital stays compared to traditional open surgical repair. The rates of spinal cord complications may also be lower with TEVAR. With TEVAR, a device called a stent graft is used. This device is covered with fabric and is advanced over wires into the aorta and then deployed.
The main complications of TAA repair (from either endovascular or the open surgical approach) include stroke and damage to the spinal cord (paralysis or paraplegia). Rarely, a TEVAR procedure may need to be converted to open surgery.
The decision to treat with a stent or open surgery depends on the aneurysm anatomy and whether it is conducive to a stent graft or open surgery and is generally made in consultation with a team of surgical and medical specialists. Sometimes a patient may not be fit for open surgery due to other medical problems. In addition, patients with a genetic cause for the TAA may have complications associated with stenting, such as increased rupture risk. Thus, some surgeons elect to treat those patients with traditional open surgery instead of stenting.
How is TAA monitored over the long term after surgery?
After TAA repair, lifelong medical follow-up helps prevent later problems with the aorta. Several specialists may be involved at the hospital, such as a cardiothoracic surgeon, a cardiologist, an intensive care unit specialist, a vascular surgeon, and/or a vascular medicine specialist. After hospital discharge, regular outpatient follow-up should occur with at least one physician with experience in the care of patients with TAA. After repair of ascending aortic aneurysms, imaging is recommended with echo before discharge and yearly thereafter. 9 After TEVAR, a CTA is recommended at 1 month, 6 months, and then annually to check the graft position and make sure it is not leaking (called an endoleak).
What precautions are needed with exercise with TAA?
Regular exercise is important for everyone. Adults should aim for at least 30 minutes per day of brisk exercise. Walking is a great way to start, with an ultimate goal of 10,000 steps per day. Mild to moderate exertion poses no problem in patients with TAA. However, activities involving extreme or maximal exertion, contact sports, isometric exercises (such as sit-ups, push-ups, or pull-ups), and heavy lifting should be avoided. Over-exertion or straining can increase the blood pressure and the stress on the aortic wall. Exercise should not be performed to the point of exhaustion. All patients with TAA should discuss their exercise program and precautions with their physician.
What about TAA and pregnancy?
When patients with TAA become pregnant, complications may occur due to the release of hormones (estrogen and progesterone). The increased heart rate and volume of blood pumped by the heart may speed up the breakdown of the arterial wall, leading to increased dilation. Guidelines recommend that all pregnant women with TAA undergo monthly echo to look at the ascending aorta. 8 Strict blood pressure control with a goal of < 120/80 mmHg is recommended. 8 In patients with TAA who are planning to become pregnant, prophylactic surgery may be recommended to prevent complications such as dissection or rupture. For example, prophylactic repair is typically recommended at 4–4.5 cm in patients with Marfan syndrome and 4.5–5 cm in patients with bicuspid aortic valve who are planning pregnancy. 2
Summary
TAA is an abnormal enlargement of the aorta that can affect the aortic root/ascending aorta, aortic arch, and/or descending aorta. TAA is associated with older age, high blood pressure, and smoking. In some cases, genetic testing may be helpful to look for a connective tissue disorder or other inherited cause. Most TAAs are discovered incidentally and do not cause symptoms, but sudden chest or back pain should prompt emergency evaluation for possible dissection or rupture. Medical treatment consists of aggressive blood pressure control, often with beta blockers and/or angiotensin-receptor blockers. Surgical repair is typically recommended at diameters ⩾ 5.5 cm for ascending aortic aneurysms and ⩾ 6 cm for descending aortic aneurysms, with a lower threshold in certain cases. Echocardiography, CTA, or MRA can all be useful in the diagnosis and monitoring of TAA.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
