Abstract
Higher body mass index (BMI) is associated with greater cardiovascular disease (CVD) risk, in part due to aortic stiffening assessed by carotid-femoral pulse wave velocity (cfPWV). Importantly, greater cardiorespiratory fitness (CRF; VO2peak) decreases CVD risk, and is associated with reductions in aortic stiffness. We tested the hypothesis that young adult overweight (OW, n=17) compared with healthy-weight (HW, n=17) men will have greater resting aortic stiffness, reduced CRF and an impaired post-exercise hemodynamic response. Resting cfPWV was greater in OW versus HW individuals (5.81 ± 0.13 vs 4.81 ± 0.12 m/sec, p<0.05). Relative CRF (VO2peak; mL/kg/min) was lower in OW compared with HW individuals (49.4 ± 1.3 vs 57.6 ± 1.0 mL/kg/min, p<0.05), and was inversely related with cfPWV (p<0.05). However, CRF as absolute VO2peak (L/min) was not different between groups and there was no relation between cfPWV and absolute VO2peak (L/min), indicating reduced relative CRF in OW men is due to greater body mass. Following the maximal treadmill exercise test, cfPWV was greater in OW compared with HW subjects from rest to 60 minutes post-exercise (p<0.05). Compared with HW, OW individuals had higher systolic blood pressure (main effect, p<0.05) and diastolic blood pressure was selectively increased for up to 60 minutes following exercise (p<0.05). Overweight individuals had an attenuated post-exercise decrease in mean arterial pressure (p<0.05). Collectively, these results indicate that young, apparently healthy, OW men have greater resting aortic stiffening and an impaired post-exercise hemodynamic response.
Introduction
Epidemiological evidence has shown mortality due to cardiovascular diseases (CVD) has decreased in recent years. 1 Despite this recent trend, CVD still remains the leading cause of death in the United States at an alarming rate, with approximately one death occurring every 30 seconds.1–3 Prevalence of CVD is projected to increase over the next 20 years with estimates indicating nearly half of the adult population in the United States will suffer from CVD. 1 Importantly, aortic stiffness assessed by carotid-femoral pulse wave velocity (cfPWV) is a manifestation of large artery dysfunction that has been identified as an independent risk predictor of major CVD-related events, including premature mortality.1,4 Thus, detection of elevated aortic stiffness in young, apparently healthy adults may prevent or delay future CVD events by identifying individuals in need of early intervention and treatment.
Obesity contributes to greater CVD risk, and current estimates indicate that approximately 30% of adults in the United States suffer from its related adverse consequences. 5 Furthermore, aortic stiffness is accelerated by obesity, which is defined as a body mass index (BMI) of >30 kg/m2. 5 This relationship of excessive body mass relative to height is therefore an important link to CVD progression and pathology. Being overweight (OW), defined as a BMI of 25–29.9 kg/m2, also confers a higher CVD risk and is associated with enhanced aortic stiffening.5,6 As such, it is clinically relevant to elucidate factors that are associated with OW-related aortic stiffness in order to identify modifiable targets for intervention to reduce CVD risk.
Lower cardiorespiratory fitness (CRF) is a predictor of cardiovascular mortality independent of body mass in both healthy and diseased adult populations.7,8 For instance, greater CRF in obese adults results in reduced mortality; while, conversely, lower CRF in healthy-weight adults results in increased CVD risk.9,10 Importantly, several investigations have demonstrated positive effects of chronic exercise training in obese adults by increasing CRF and reducing aortic stiffness, which was associated with weight loss and improved body composition.7,11 Despite these findings for the relation of body mass to arterial stiffness, it is unclear if maximal CRF is inversely related to aortic stiffening in young healthy-weight (HW; BMI of 18.5–24.9 kg/m2) and OW adults.5,12 In addition, the effect of acute maximal exercise on arterial stiffness in an overweight population has potential to elucidate increased CVD risk not detectable at rest. 13 For example, in young healthy adults, the post-exercise arterial stiffness response has been shown to be above or below baseline values following maximum aerobic exercise.13,14 In contrast, aortic stiffness has also been demonstrated to not change acutely following a graded exercise test.15–17 Therefore, it is important to further elucidate the relative changes of aortic stiffness in response to maximal exhaustive exercise in OW individuals.
Being OW raises cardiovascular risk and promotes aortic stiffening. In contrast, greater CRF is associated with reduced cardiovascular risk, and lower aortic stiffness. Whether CRF is related to aortic stiffness, or if aortic stiffness is influenced by a maximal graded exercise test (GXTmax) in HW and OW young adults is equivocal. In the current study, we tested the hypothesis that CRF will be inversely associated with aortic stiffness in young apparently HW and OW adults at rest. Additionally, we hypothesized the post-exercise arterial stiffness and hemodynamic response to maximal treadmill testing would be impaired in OW versus HW young men.
Methods and procedures
Subjects
Subjects for this study included 34 young men aged 18–35 years of age. Potential participants were recruited by advertisement flyers that were posted within the university community. Subjects were grouped by BMI status and included 17 HW (BMI <25 kg/m2) and 17 OW adults (BMI 25.0–29.9 kg/m2). All volunteers were non-smokers and free from any known CVD, diabetes mellitus, pulmonary disease, and/or orthopedic limitations. None of the subjects were taking medication affecting cardiovascular function. Prior to participation, written consent was obtained for each subject in accordance with the policies and procedures of the University of Kentucky Office of Research Institutional Review Board. Each subject completed a Physical Activity Readiness Questionnaire and Health History Form to identify any existing contraindications to the maximal graded exercise testing as specified in the ACSM’s Guidelines for Exercise Testing and Prescription (American College of Sports Medicine). Additionally, the inclusion criteria included engagement in resistance training equal to or greater than 3 days per week, involving full body movement lifts using major muscle groups, lasting at least 45 minutes per session. Each subject underwent a resting electrocardiogram (ECG) to ensure normal cardiac rhythms. No subjects were excluded due to an abnormal ECG. All procedures were completed in a single testing session, and all subjects were asked to refrain from alcohol and caffeine for at least 12 hours and strenuous exercise for at least 24 hours prior to data collection. The single testing session consisted of resting measures, followed by a maximal graded exercise test, and concluded with recovery measures at regular intervals for 60 minutes (e.g. 2, 5, 10, 20, 30, 45, and 60 minutes). The resting measures included waist and hip circumferences, brachial blood pressure, heart rate, total body composition and cfPWV measurements. The recovery measures included brachial (peripheral) blood pressure and cfPWV measurements.
Anthropometric and body composition measures
Standing height (to the nearest 0.1 cm) and body mass (to the nearest 0.1 kg) were determined using a wall-fixed stadiometer (Model 216; Seca North American West, Ontario, CA, USA) and a calibrated electronic scale (American Digital Scale, model DI-10; Teraoka Weigh-System Pte Ltd, Singapore), respectively. The standing height and body mass measurements (and calculated BMI; kg/m2) were determined for all subjects while wearing light-weight clothing and no shoes. Waist circumference (WC) and hip circumference (HC) were measured three times each using a standard anthropometric measuring tape (Creation Health Products, Orlando, FL, USA) in accordance with the ACSM guidelines to the nearest 0.1 cm and mean measurement used for analyses.
A Bodystat QuadScan 4000 (Bodystat, Isle of Man, British Isles) bioelectrical impedance analysis (BIA) device was used to determine the total body composition (absolute and relative fat and fat-free masses) for each subject using a two-compartment model. During the measurement procedure, all subjects were placed in a supine position on a non-conductive surface, using previously reported standardized methodology, 18 and the manufacturer’s proprietary equation was used to determine the absolute and relative fat and fat-free masses.
Resting heart rate and blood pressure
Resting heart rate and blood pressure were determined for each subject following a minimum of 10 minutes of supine rest. Heart rate was determined using a heart rate monitoring device (Polar A3; Lake Success, NY, USA) and blood pressure was determined by manual auscultation using an appropriate sized cuff and hand-held aneroid (Model DS58-MCCB; Welch Allyn, Bangor, ME, USA) and stethoscope (3M, St Paul, MN, USA) by a trained investigator.
Electrocardiogram
A resting 12-lead ECG was acquired in the supine position using a Nihon Kohden ECG-1550A (Irvine, CA, USA). Each resting ECG was reviewed to ensure no abnormalities were present.
Maximal graded exercise test
Each subject performed a maximal graded exercise test (GXT; 3-minute progressive speed and grade stages) on a treadmill with simultaneous indirect calorimetry testing (Parvo Medics TrueOne 2400 metabolic cart; Parvo, Sandy, UT, USA). A similar protocol was administered in young healthy subjects previously and indicated differences in aortic stiffness following exhaustive aerobic exercise.13,14 Specifically, the gas analyzers were calibrated with known concentrations of 4.0% O2 and 16.0% CO2. The flowmeter was calibrated with a 3-Liter syringe at a range of ventilatory flow rates. During testing, continuous measurements of oxygen consumption and heart rate were monitored and/or recorded, and the oxygen consumption measures were averaged each minute. At the final 30 seconds of each stage, heart rate, blood pressure, and rating of perceived exertion (RPE) were taken and recorded. The test was terminated upon volitional fatigue. Each subject obtained VO2 peak (mL/kg/min) determined by the participant’s ability to obtain a minimum of two of the following criteria: (1) respiratory exchange ratio (RER) of ⩾1.15; (2) no increase in heart rate despite an increase in workload or age predicted heart rate max (220 – age) achieved; or (3) a RPE ⩾17 using the Borg scale.
Aortic stiffness
The cfPWV, the gold-standard measure of aortic stiffness, was assessed with a SphygmoCor high-fidelity applanation tonometer probe (AtCor; Itasca, IL, USA). Both baseline and post-GXT measurements were taken at the carotid and femoral arteries on the right side of the body. Pressure waves were recorded sequentially from the two sites, and transit times were calculated for both the carotid and femoral arteries from the foot of each acquired pressure wave to the R-wave of the ECG. The cfPWV was calculated by dividing distance (m) by the time delay (transit time) between each foot of the carotid and femoral pressure waveforms. Distance was measured from the carotid measurement site to the suprasternal notch and from the suprasternal notch to the femoral measurement site using a tape measure, taking care not to follow the contours of the body. The path length, defined as our aortic distance component, was the difference between these two measurements. Immediately following exercise, each subject returned to a supine position. The cfPWV and BP measurements were obtained at 2, 5, 10, 20, 30, 45, and 60 minutes post-GXT. All cfPWV measurements were taken on the right side of the body, and brachial BP was assessed on the left arm of the subject.
Statistical analysis
All data are presented as mean ± standard error of the mean (SEM) with statistical analyses completed using JMP 10 software (SAS Institute, Cary, NC, USA) and SigmaPlot 13 (Systat Software Inc., San Jose, CA, USA). Two-tailed independent samples t-tests were used to compare baseline subject characteristics between groups. Individual bivariate correlations between all cardiovascular and anthropometric variables were determined with Pearson product-moment analyses. A two-way repeated measures analysis of variance (ANOVA) was used to evaluate changes in cfPWV, systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), and pulse pressure (PP) for post-exercise test data. Area under the curve (AUC) was assessed for cfPWV across all time points following (i.e. does not include baseline measures) acute exercise (2–60 minutes post-GXT) using the trapezoidal rule and between-group differences assessed with a paired t-test. Adjusted means were determined with analysis of covariance (ANCOVA). Linear regression was used to determine R and R2. Post hoc analyses were used when appropriate. Statistical significance was set at p<0.05 for all analyses.
Results
Resting subject characteristics and cardiorespiratory fitness
Demographic and descriptive baseline and resting characteristics are presented in Table 1. Compared to HW, OW individuals had greater body mass, BMI, absolute fat mass and fat-free mass, relative fat mass, WC, HC, W:H (waist to hip ratio), SBP, PP and MAP measures, pulsatile stress (defined as pulse pressure × heart rate (HR), a validated measure of ‘pulsatile stress’ 19 ), and lower relative fat-free mass (p<0.05, all; Table 1). Resting cfPWV was greater and relative VO2peak was lower in OW compared with HW individuals (p<0.05, both; Figures 1A and 1B). Bivariate correlation analysis demonstrated an inverse correlation for cfPWV and relative VO2peak (p<0.05, r2=0.29; Figure 1C). There were no differences in absolute VO2peak between groups (Figure 2A), and no association between cfPWV and absolute VO2peak (r2=0.0004; Figure 2B). The remaining bivariate associations are presented in Supplemental Table 1. Additionally, resting cfPWV had a positive relation to body mass, and absolute fat mass and fat-free mass (p<0.05, all; Figures 3A, 3B, 3C, respectively). Collectively, these data demonstrate increased resting cfPWV in young OW men; reduced relative CRF in young OW adults was largely due to greater body mass.
Subject characteristics.
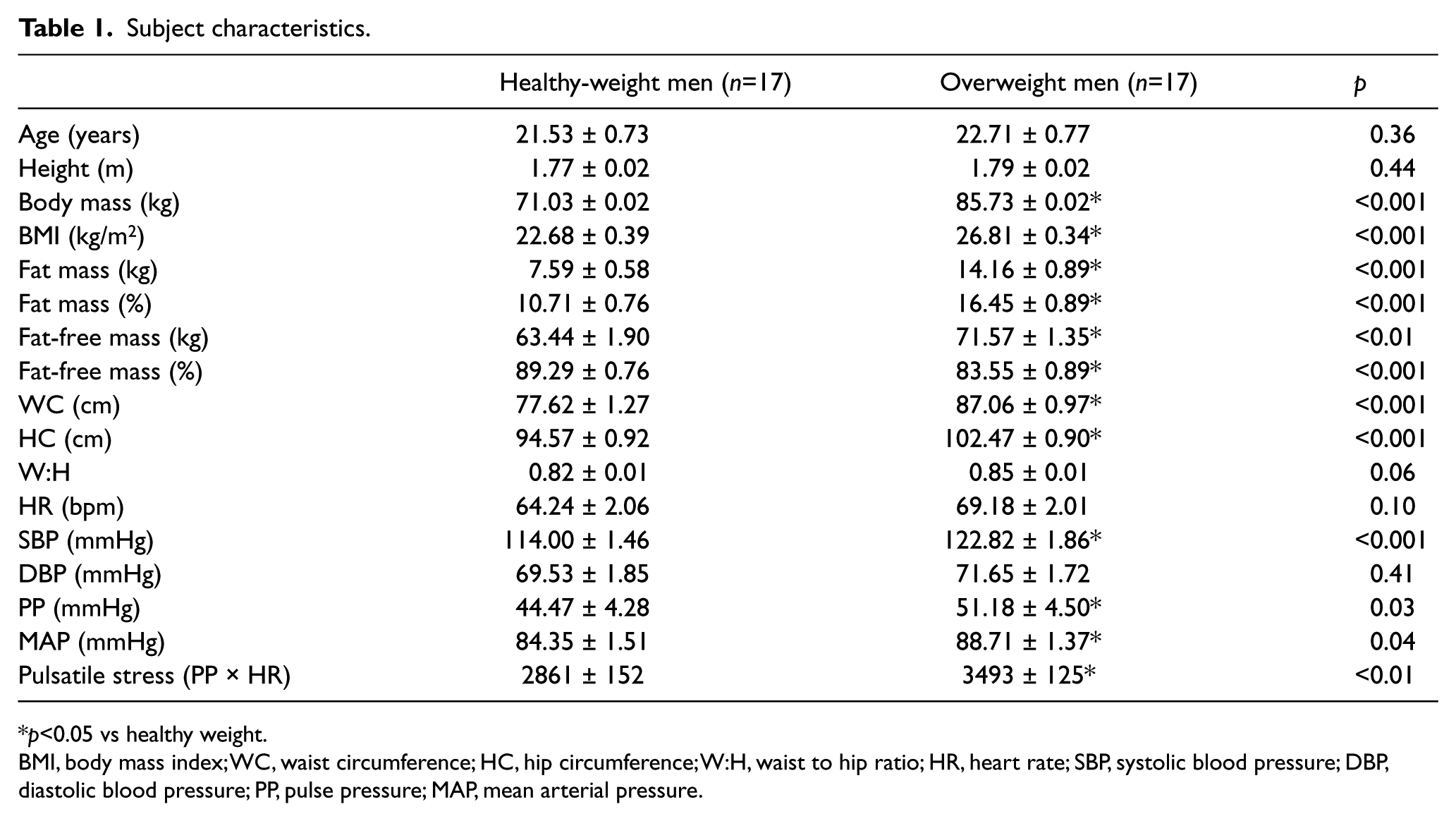
p<0.05 vs healthy weight.
BMI, body mass index; WC, waist circumference; HC, hip circumference; W:H, waist to hip ratio; HR, heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; PP, pulse pressure; MAP, mean arterial pressure.
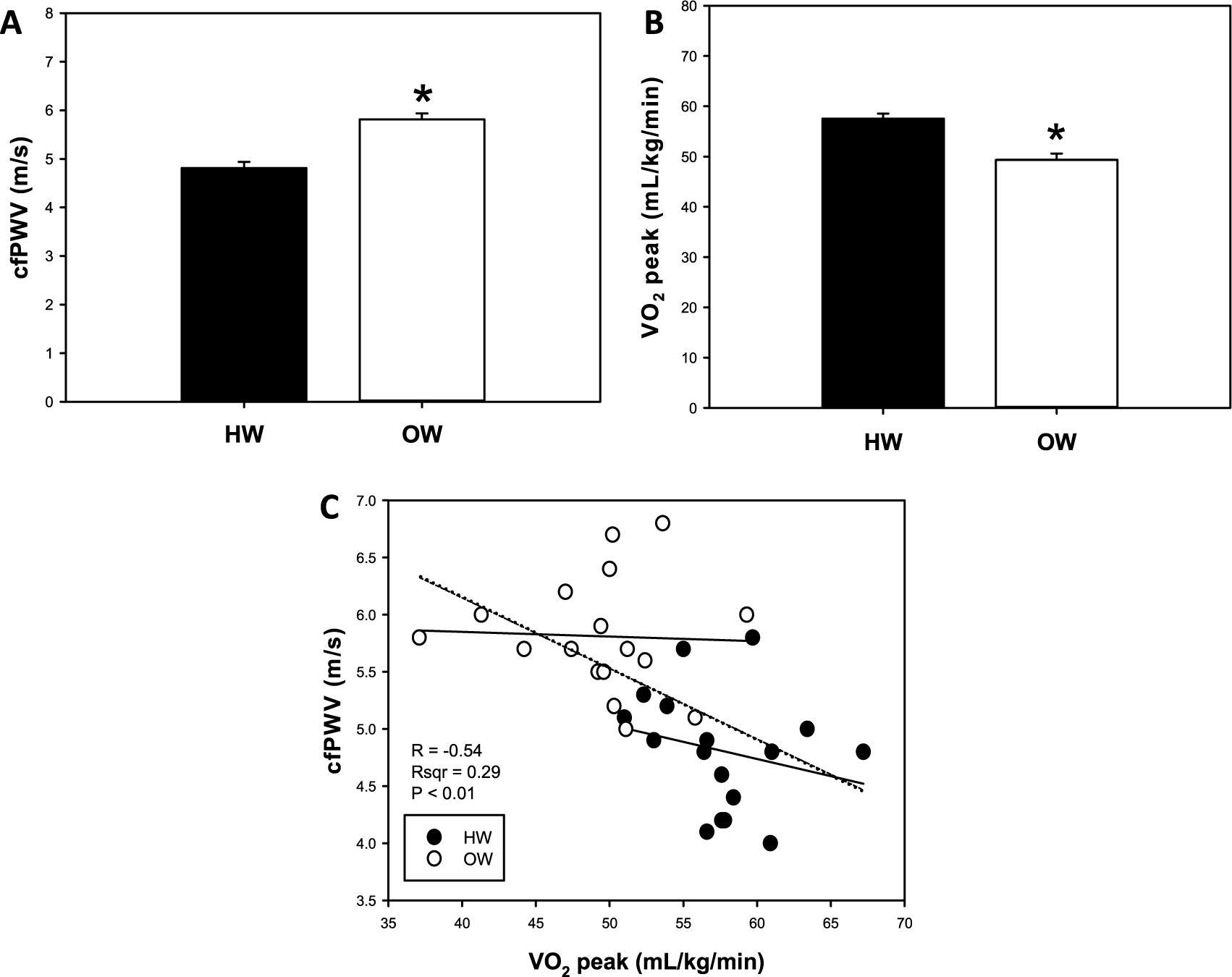
Resting carotid-femoral pulse wave velocity (cfPWV; A); peak relative oxygen consumption (VO2peak; B); and bivariate correlation between cfPWV and relative VO2peak (C) in young healthy-weight (HW) and overweight (OW) men. Data are mean ± SEM; *p<0.05 versus HW.
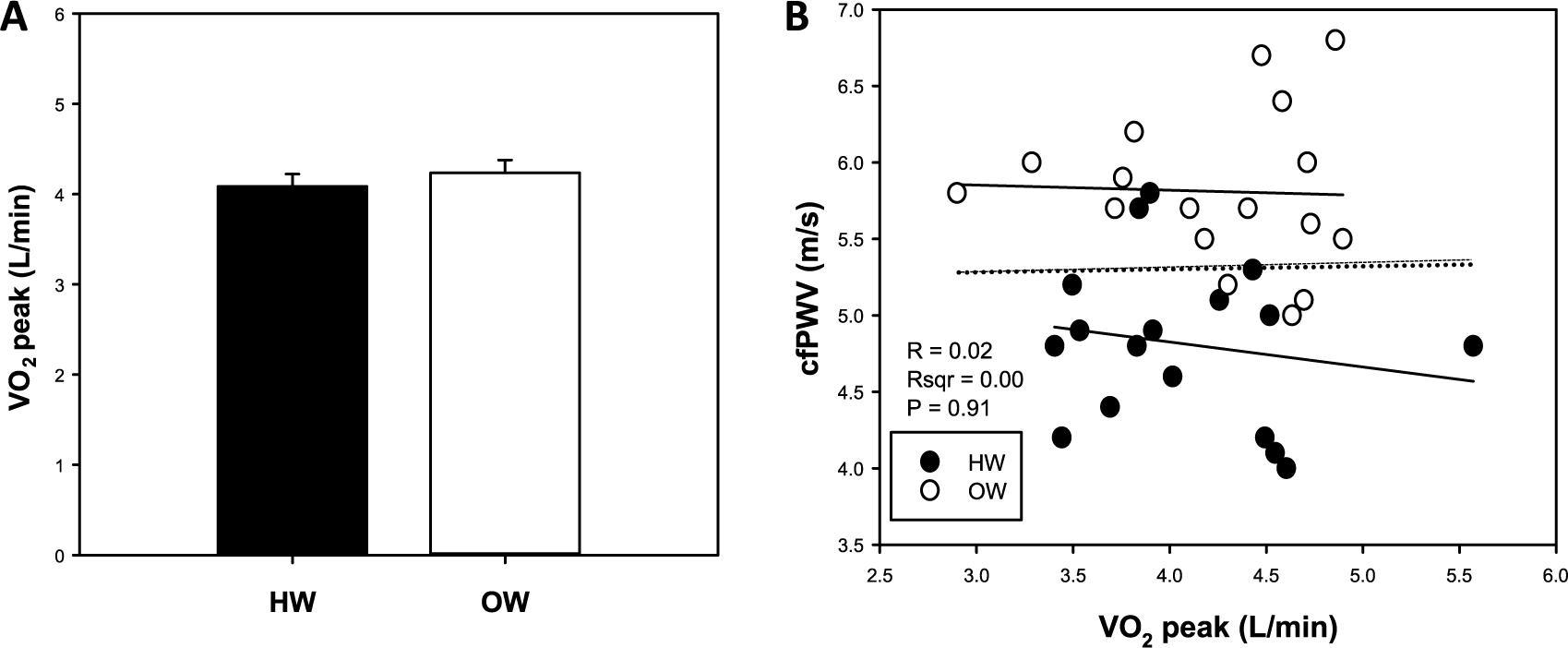
Peak absolute oxygen consumption (VO2peak; A) and bivariate correlation between resting carotid-femoral pulse wave velocity (cfPWV) and absolute VO2peak (B) in young healthy-weight (HW) and overweight (OW) men. Data are mean ± SEM.
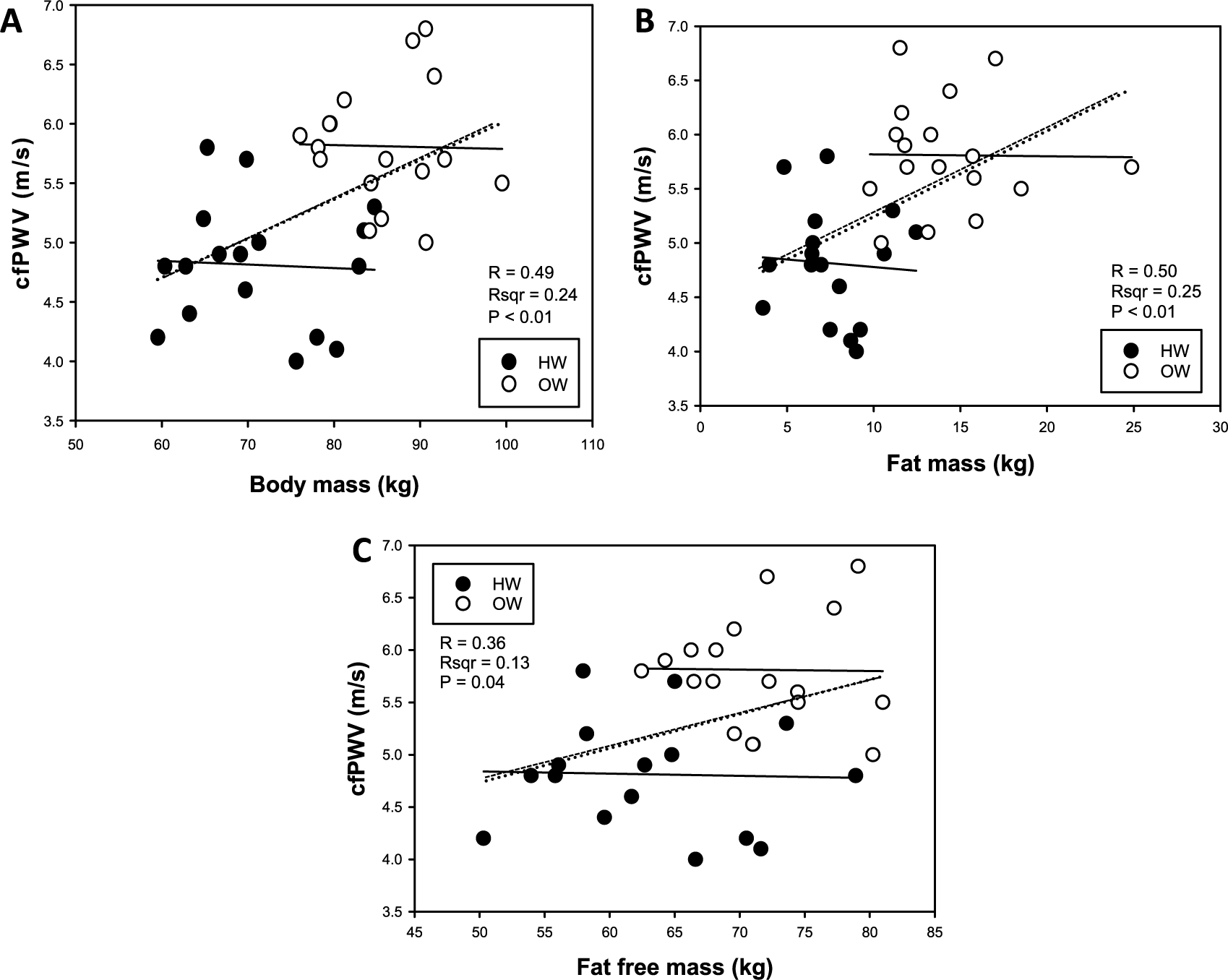
Individual bivariate correlations between resting carotid-femoral pulse wave velocity (cfPWV) and body mass (A), absolute fat mass (B), and absolute fat-free mass (C) in young healthy-weight (HW) and overweight (OW) men.
Post-GXT cardiovascular hemodynamics
Aortic stiffness assessed by cfPWV was greater in OW compared with HW after the GXT (p<0.05, main effect of weight; Figure 4A). Area under the curve (AUC) was greater for OW compared with HW (p<0.01; Figure 4B), further indicating greater aortic stiffness during the post-GXT recovery period. Additionally, for brachial SBP, there were main effects of weight (HW vs OW) and time (p<0.05, both; Figure 5A), indicating that SBP was greater for OW compared to HW individuals, and that SBP decreased in both groups over time. Brachial DBP was decreased at 2 and 5 minutes post-GXT in both HW and OW compared to resting levels (p<0.05; Figure 5B). Compared with HW, OW had an increased brachial DBP from 10 to 60 minutes post-GXT (p<0.05; Figure 5B). In addition, OW brachial DBP was greater than resting values at 20 and 30 minutes following exercise (p<0.05; Figure 5B). MAP was greater in OW compared to HW from 10 to 60 minutes post-exercise (p<0.05; Supplemental Figure 1A). Additionally, a main effect of time was observed for brachial PP, which also decreased during the post-exercise recovery period (p<0.05; Supplemental Figure 1B).
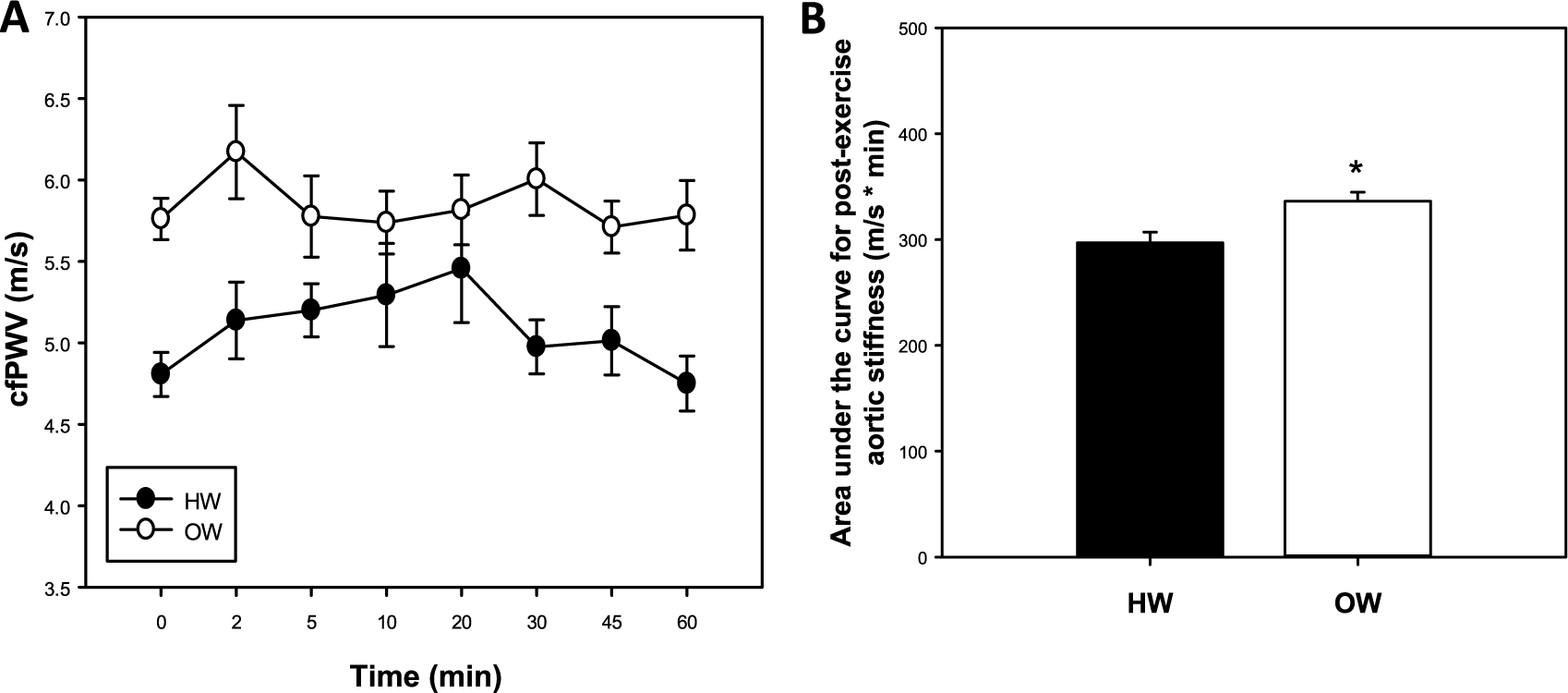
Post-exercise carotid-femoral pulse wave velocity (cfPWV) response following maximal treadmill exercise in healthy-weight (HW) and overweight (OW) young adults (A). The post-exercise cfPWV response presented as area under the curve (AUC; B). Data are mean ± SEM; p<0.05, main effect of weight for cfPWV; *p<0.05, versus HW.
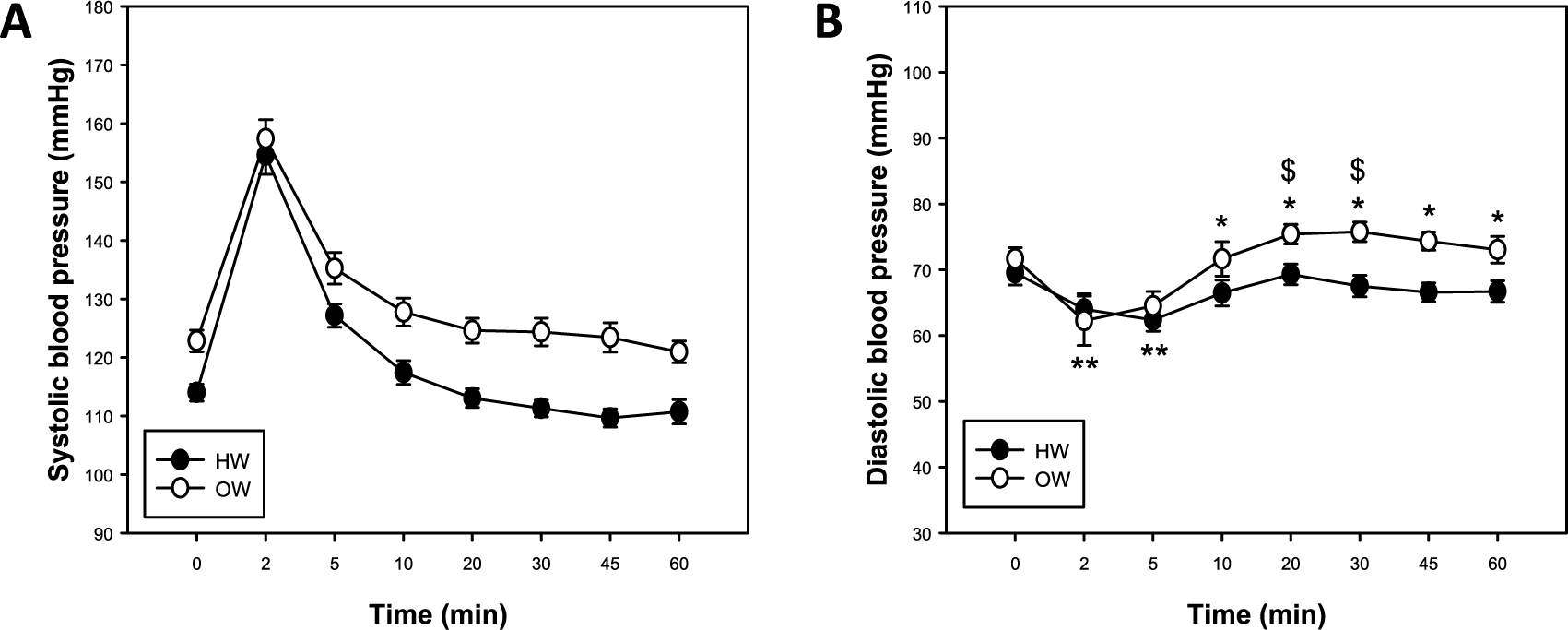
Systolic (A) and diastolic (B) blood pressure responses following maximal treadmill exercise in healthy-weight (HW) and overweight (OW) adults. Data are mean ± SEM; p<0.05, main effect of time and weight for systolic blood pressure; *p<0.05, HW versus OW at 10, 20, 30, 45 and 60 minutes; **p<0.05, within HW and OW groups versus 0 minutes; $ within OW versus 0 minutes.
Statistical adjustment of cfPWV
After controlling for SBP, an important covariate that may influence cfPWV, the group differences in aortic stiffness were not changed (p<0.05; Table 2). In addition, BMI, MAP and HR, and VO2 peak did not influence the group differences in cfPWV, as each covariate was not related to the dependent variable in an analysis of variance interaction test (p>0.05, all).
Analysis of covariance (ANCOVA) results of adjusted means for aortic stiffness (m/s).
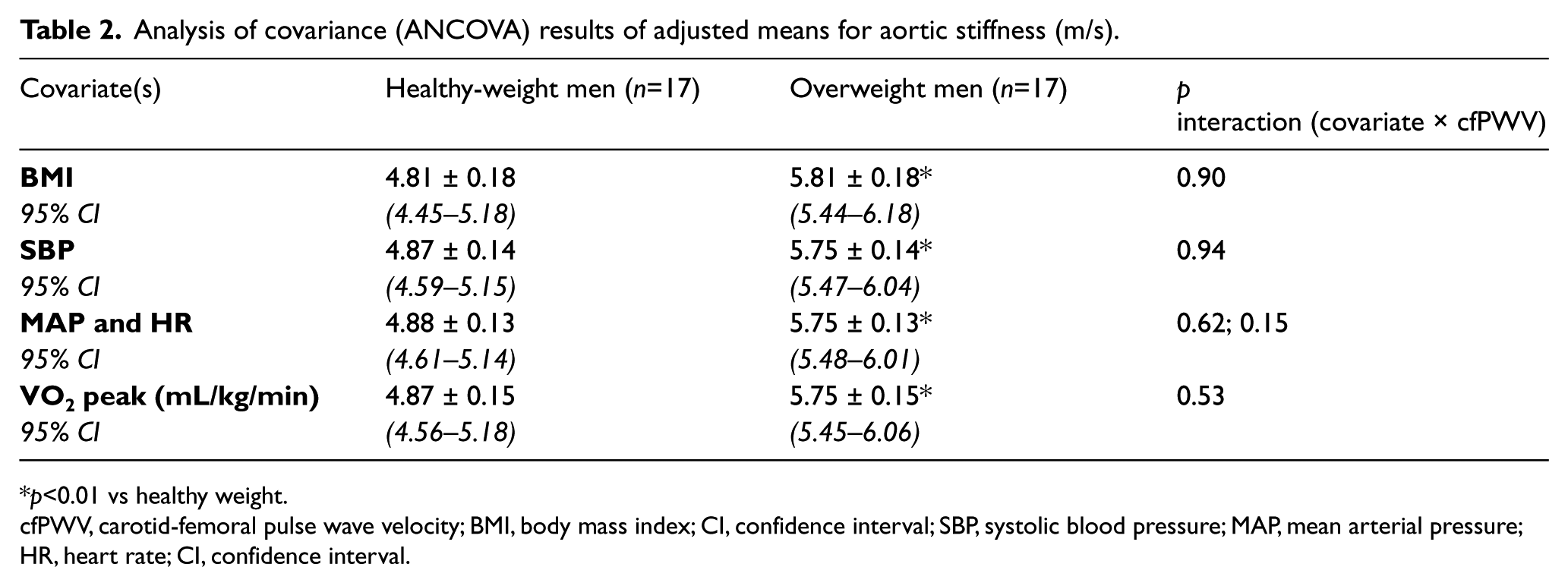
p<0.01 vs healthy weight.
cfPWV, carotid-femoral pulse wave velocity; BMI, body mass index; CI, confidence interval; SBP, systolic blood pressure; MAP, mean arterial pressure; HR, heart rate; CI, confidence interval.
Discussion
Identifying and treating modifiable risk factors early in life that may prevent or delay future CVD-related events has important clinical implications. Obesity is a global epidemic concern of modern societies, which significantly increases CVD risk. Overweight adults have been shown to have a greater CVD risk that is associated with increased aortic stiffness. 20 Importantly, greater aortic stiffness is an emerging risk factor and independent predictor of major CVD-related events.21,22 Thus, the results of our study have revealed important arterial stiffness-related findings in young OW adults, which include: (1) compared with young HW adults, young OW men have greater resting aortic stiffness and reduced relative peak aerobic capacity; (2) an inverse relation for cfPWV and CRF in a young apparently healthy population, which is largely due to increased body mass; and (3) impaired post-GXT response of aortic stiffness and peripheral blood pressure in young, healthy, OW adults. Collectively, these findings highlight the importance of elucidating modifiable cardiovascular risk factors in apparently healthy, young, OW adult populations.
Aortic stiffness, BMI and SBP
In the present study, OW compared with HW subjects had greater body mass, BMI, absolute fat and fat-free masses, relative fat mass, WC, HC, W:H, SBP, PP, MAP, pulsatile stress, and cfPWV, whereas relative fat-free mass and relative VO2peak were lower (Table 1 and Figure 1). These findings are supported by previous studies demonstrating that elevated BMI and relative body fat are associated with increased blood pressure, WC, HC, W:H, and reductions in CRF.23,24 Recent investigations support our current findings that increased SBP is associated with increased aortic stiffening.24–26 The detrimental effects of a greater BMI have also been related to higher aortic stiffness in apparently healthy adults, 27 and that greater CRF may attenuate these negative effects of elevated BMI. 28 Additionally, chronically elevated SBP is detrimental to the arterial system as greater intravascular pressures damage the elastic properties of vessels. Structurally, elastin is major elastic protein within the artery that provides the arterial system the spring-like properties providing buffering against arterial blood pressure. 26 As such, modestly increased blood pressure in young OW adults may promote aortic stiffness resulting in early vascular aging and a greater likelihood for developing future hypertension. 29 It is important to note, however, that in the present study we statistically adjusted for SBP and BMI, and the group differences in aortic stiffness remained. Our data thus suggest increased BMI is related to greater aortic stiffness, which may be independent of higher SBP in this young adult, male population.
Aortic stiffness and CRF
Previous studies support the current findings (Figure 1C) that aortic stiffness is inversely associated with relative VO2peak.12,30 Individuals with greater CRF possess favorable arterial adaptations 12 such as increased elastic properties. 30 Our findings also demonstrate that increased aortic stiffness is coupled with an increase in SBP. These data indicate that even a modest increase in resting SBP (~6 mmHg) may have potential detrimental effects on the arterial system, resulting in aortic stiffness. Thus, improving CRF has potential to reduce SBP and aortic stiffness in apparently healthy young adults. In addition, our data highlight that body mass is critical for the inverse relation between cfPWV and relative VO2peak (Figures 1C, 2 and 3). Importantly, greater total body mass, fat mass and fat-free mass were all positively related to cfPWV (Figure 3). Previous reports have shown reductions in carotid compliance in young and middle-aged resistance-trained adults who have greater body mass due to resistance training, 31 supporting our findings that increased overall body mass and even fat-free mass may promote aortic stiffness. Moreover, the correlation matrix showed that a higher fat-free mass percentage was associated with reduced cfPWV and greater CRF. However, when the body mass becomes excessive, as was the case in many of our resistance-trained subjects, the absolute fat mass may be detrimental to cardiovascular function. While the finding of increased fat-free mass and greater aortic stiffness is not supported in the literature, this association may be a result of our two-compartment body composition assessment. In other words, perhaps due to greater body water, the subjects who were excessively muscular presented with ‘artificially inflated’ fat-free masses. This method of body composition was a limitation in this study. In any regard, our findings indicate that total body mass largely explains the relative reductions of VO2peak in young, apparently healthy, OW adults.
Acute exercise and aortic stiffness
One proposed mechanism responsible for the chronic reduction in aortic stiffness as a result of a sustained exercise program involves a greater peripheral vasodilation following exercise.7,8 The greater vasodilation of the peripheral arteries would reduce augmentation indices by slowing down the reflected wave to the central arteries, and thereby reduce arterial stiffness. During acute submaximal and maximal exercise, oxygen demand is increased and the arteries are required to alter basal tone. Therefore, elevated heart rate, blood pressure, and aortic stiffness responses lead to greater cardiac output beyond resting conditions.8,32 Our data thus suggest that HW young men who have greater relative CRF may have increased peripheral vasodilation compared with young OW men, which may influence the central arterial stiffness response.
Many investigations have focused on the cardio-protective effects of exercise on CVD. Studies have shown acute all-out or maximal bouts of exercise affect many aspects of arterial function in young HW men.32,33 As such, blood pressure increases transiently after short maximal bouts of exercise. 32 However, this blood pressure response returns to baseline approximately 15 minutes after termination of exercise, suggesting aortic stiffness may have a similar response. 32 The results of our study, however, demonstrate that post-exercise cfPWV was greater in OW individuals from rest to 60 minutes post-GXT (Figure 4). In addition, the AUC was higher for OW individuals, suggesting that HW men had a lower aortic stiffness burden from baseline throughout the post-GXT recovery period (Figure 4). Others have shown that cfPWV in young, healthy adults returns to baseline values after approximately 20 minutes, and is ~8% below baseline up to 60 minutes post-exercise, 32 which was not observed in the current study. Collectively, these findings indicate that young OW compared with HW adult males have greater aortic stiffness at rest and following maximal exercise.
Post-exercise blood pressure
The current study demonstrates a main effect of time and weight for SBP after exercise (Figure 5A). As previously discussed, SBP increases with bouts of acute exercise due to increased oxygen demand and greater basal tone.8,32 Yet, SBP returns to baseline by 15 minutes post-exercise, and continues to decrease up to 60 minutes post-exercise. 32 Owing to excessive adiposity, BP is often higher in OW compared with HW adults.20,34 Thus, our data suggest that even modest increases in body mass, fat mass, and fat-free mass in OW adults may all contribute to greater resting and post-exercise SBP responses.
DBP was also elevated in OW compared with HW individuals from 10 to 60 minutes post-exercise (Figure 5B). There is evidence that while SBP increases acutely with exercise, DBP decreases initially due to peripheral dilation, 35 which was observed in both HW and OW individuals in the current study. In HW men, however, DBP returned to baseline values by 10 minutes post-exercise, but were lower compared with OW men for the remaining resting period. Importantly, DBP increased in the OW group above resting values at 20 and 30 minutes post-GXT (Figure 5B). This phenomenon suggests that the peripheral dilation may be impaired in our OW subjects.20,34
MAP was greater in OW versus HW individuals following maximal exercise (Supplemental Figure 1A). This finding further indicates an overall impairment in blood pressure responsiveness post-exercise. MAP is the product of SBP and DBP, which also had attenuated responses in the OW men. Both SBP and DBP are negatively influenced by BMI,8,20 and greater CRF is associated with improved arterial function including SBP and aortic stiffness. Thus, young OW adult males with increased BMI and reductions in CRF may explain the impairments in MAP.7,8,34
In the present study, SBP, DBP, MAP, and PP were measured from a peripheral site (i.e. brachial artery). As blood flows distally, arterial properties naturally become stiffer due to structural protein changes that result from increased branching and musculature.21,36 Thus, peripheral BP is greater when comparing to central blood pressure. Previous studies have concluded that central SBP and central PP are better predictors of CVD when compared to peripheral, and our peripheral blood measures may be a limitation of the current investigation.37,38
Limitations
The principal finding of this study was the association of body mass, fat mass, and fat-free mass with greater aortic stiffness, which was independent of cardiorespiratory fitness. It should be recognized, however, that other unmeasured cardiometabolic factors, including dysglycemia, insulin resistance, unfavorable lipid concentrations and lifestyle habits (e.g. drugs, alcohol, nutrition), may also influence the inverse relation between cfPWV and CRF. In addition, the findings of this study are derived from a limited population comprised of young, college-aged men who are apparently healthy. This small, homogenous population limits the generalizability of the findings, but provides important information for this limited cross-section of the overall population. Importantly, while a relatively small sample size of n=17 per group was examined, the effect size (Cohen’s d) was large, at 1.70, with 99% power. Therefore, despite the relatively small sample size of this study the group differences in aortic stiffness are statistically reliable.
Conclusions
Supporting the hypotheses, it was found that increased aortic stiffness in young OW adult males is associated with lower levels of relative VO2peak, which is largely due to greater body mass. In addition, post-GXT arterial hemodynamics are impaired in young adult OW males. Therefore, the burden of elevated aortic stiffness seen in OW men during resting conditions is carried into the post-exercise period of recovery, during which time the leaner HW men demonstrate a comparatively lower aortic stiffness and blood pressure. Our results indicate that young, OW, apparently healthy adult men maintain a greater aortic stiffness and blood pressure response at rest and following a maximum aerobic test that is related to reduced cardiorespiratory fitness, which is associated with greater body mass, fat mass and fat-free mass.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
