Abstract
Peripheral artery disease (PAD) is associated with increased mortality and concomitant coronary artery disease (CAD). However, it is unclear whether uncovering the presence of functional coronary ischemia by single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) would further help stratifying that excess risk. From January 2000 to 2009, 4294 individuals underwent cardiac stress testing within 180 days of ankle–brachial index (ABI) measurements. Of these, 645 had PAD and SPECT MPI stress testing. Abnormal ABI was defined as ⩽0.9 or prior lower extremity arterial revascularization. Myocardial ischemic burden and total jeopardized myocardium were represented by the summed difference score (SDS) and summed stress score (SSS), respectively. Univariate and multivariable Cox proportional hazard analyses were used to study the impact of SDS and SSS on all-cause mortality. Additionally, using a hierarchical approach, we examined the step-wise addition of post-stress test coronary and lower extremity arterial revascularizations as time-varying covariates on outcomes. We found no significant difference in all-cause mortality between patients with ischemic myocardium (SDS>0) and those without (SDS=0) (adjusted HR: 0.94, 95% CI: 0.53–1.69; p=0.84). Similarly, the presence of jeopardized myocardium (SSS>0) did not have a significant impact on mortality (adjusted HR: 1.16, 95% CI: 0.67–2.00; p=0.59). Moreover, adjustment for post-testing coronary and lower extremity arterial revascularizations did not affect our results. In conclusion, ischemic and jeopardized myocardia are not predictors of all-cause mortality in PAD; thus, SPECT MPI does not appear to be a useful risk stratification tool in these patients.
Introduction
Peripheral artery disease (PAD) and coronary artery disease (CAD) coexist. The ankle–brachial index (ABI) is a commonly used test to diagnose PAD and is associated with all-cause mortality; 1 moreover, measuring ABI in CAD patients has an incremental prognostic value.2–4 Single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) is also used to evaluate for CAD, and its scores are known to be associated with high risk of all-cause mortality in general.5–7 Given the high prevalence of CAD and cardiovascular morbidity and mortality in patients with PAD,8,9 it remains unclear whether the identification of functional coronary ischemia allows better risk stratification in these patients. 10 We therefore examined the impact of myocardial ischemic burden and total jeopardized myocardium on all-cause mortality among patients with PAD.
Methods
Study population
During the period from January 2000 to January 2009, we identified all patients (n = 4294) who had a cardiac stress test 180 days before or after assessment for PAD with ABI measurement at our tertiary care institution. Of these, the 645 who had PAD (an abnormal ABI or a history of lower extremity arterial intervention) and underwent cardiac stress testing with SPECT MPI comprised the study population. Others who underwent imaging with echocardiography or positron emission tomography (PET) were excluded to avoid differences related to imaging modality. The ABI measurements were performed for evaluation of lower extremity symptoms, signs, or asymptomatic screening in a laboratory accredited by the Intersocietal Accreditation Commission (IAC) in vascular testing and by certified technologists. Cardiac stress testing was performed for cardiac symptom evaluation in 43%; the remaining underwent testing for either resting electrocardiogram abnormalities or pre-operative assessment. The baseline characteristics of the study population, cardiac medication use, and cardiac stress test data were obtained from the prospectively collected stress test database at our institution. 11 Electronic medical records were reviewed to capture coronary revascularizations (open heart coronary bypass or percutaneous coronary intervention) if there was any pre- and/or post-cardiac stress testing. Similarly, lower extremity arterial revascularization procedures (open or endovascular) were obtained pre- and post-ABI measurement. These data were specifically captured to determine whether any revascularization as the result of cardiac stress testing would impact long-term outcome. The left ventricular ejection fraction (LVEF) was obtained from transthoracic echocardiography or SPECT MPI data during the same period. The study was approved by the Institutional Review Board, data were de-identified and informed consent was waived.
The majority of patients (90%) underwent pharmacologic vasodilator stress testing at the discretion of the clinician. On SPECT MPI, using the 17-segment model and 5-point scale (0 = normal, 1 = mild, 2 = moderate, 3 = severe, 4 = absence of uptake), the summed rest score (SRS) and summed stress score (SSS) were obtained by addition of the scores of the 17 segments at rest and peak stress, respectively. There was a maximum possible overall score of 68. 12 The summed difference score (SDS) was defined as the difference between the SSS and SRS. An SDS > 0 indicated the presence of stress-induced reversible myocardial ischemia, which was used to define a positive stress test. The SDS represented total ischemic myocardium (myocardial ischemic burden), while the SSS signified total myocardium that was ischemic and infarcted (total jeopardized myocardium).
For ABI, systolic arm and leg blood pressures were first obtained from all extremities using a Doppler-based technique with the patient at rest and in the supine position. For each lower extremity, the ABI was calculated by dividing the higher of two ankle systolic blood pressures (measured at dorsalis pedis or posterior tibial artery) by the higher of the two brachial systolic blood pressures. The lower of the two ABIs, right or left leg, was the ABI used for analysis. All ABIs reported in this study were performed at rest. Patients were classified as having PAD if they had an ABI ⩽ 0.9 13 or a prior history of lower extremity arterial intervention regardless of their ABI values (n = 293). Death was defined as all-cause and obtained using the Social Security Death Index (SSDI) with a closing date of 25 February 2011.
Statistical analysis
Simple descriptive statistics were used to summarize the data. Continuous variables are presented as mean ± standard deviation. Comparisons were made using the Wilcoxon rank-sum non-parametric test. Categorical data are described using frequencies and percentages. Comparisons were made using the chi-squared test or Fisher’s exact test if the frequency was less than 5. Mean imputation was used to impute the missing values (only race and body mass index variables had missing values) with < 1% of the data missing. The distribution of SDS and SSS among the study population was left skewed with a large proportion having SDS = 0 (454 (70%) patients) and SSS = 0 (357 (55%) patients) (Figure 1). Plots of SDS and SSS against survival were conducted to examine the optimal cutoff for these variables; however, linear relationships were observed. Therefore, we considered both elements of SDS > 0 and the continuous SDS (likewise with the SSS elements) in the analyses because the data were zero inflated.
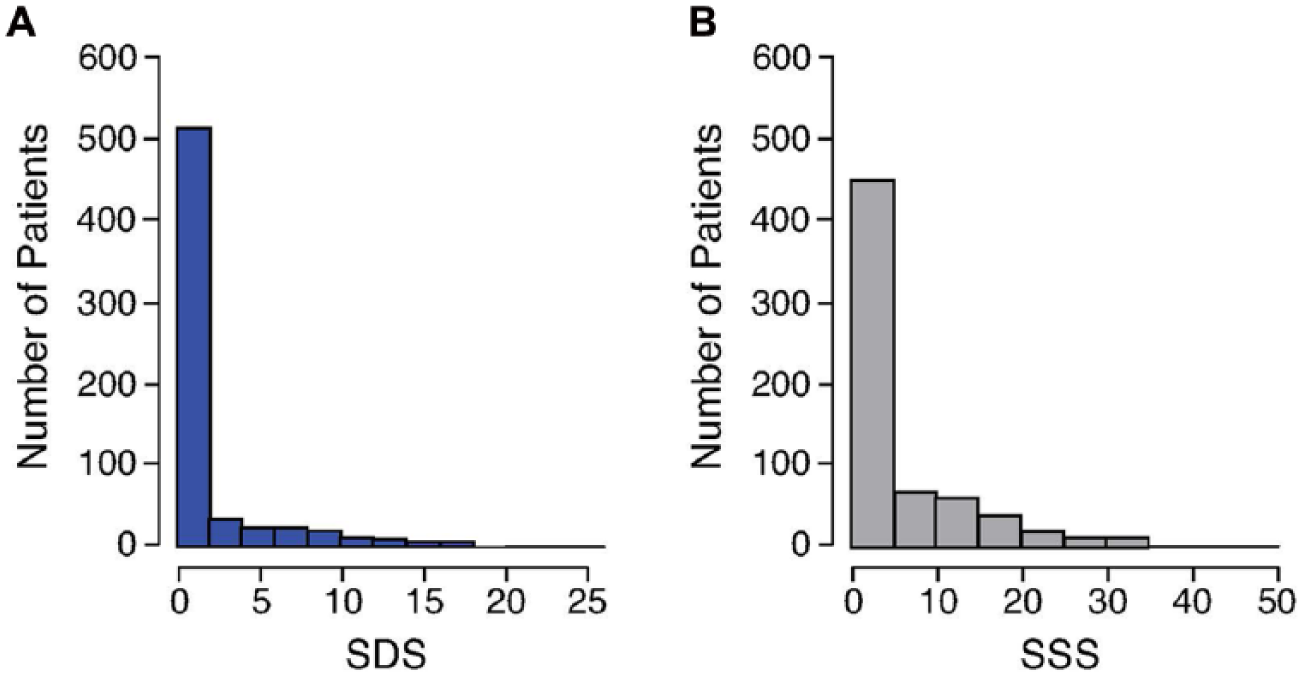
Histogram of SDS and SSS on SPECT MPI in peripheral artery disease patients. Histograms of the summed difference score (SDS) and summed stress score (SSS) for the entire study population are depicted in panels A and B, respectively. The dataset is ‘zero-inflated’ with the majority of SDS and SSS values equal to zero. (MPI, myocardial perfusion imaging; SPECT, single-photon emission computed tomography.)
We employed a multivariable Cox proportional hazard regression model using reliable predictors obtained by bootstrapping methods to examine whether ischemic or jeopardized myocardium had any impact on the occurrence of subsequent (post-stress test) coronary revascularization in PAD patients. We adjusted for history (pre-stress test) of coronary and lower extremity arterial interventions.
All-cause mortality was studied non-parametrically by the Kaplan–Meier method with the date of cardiac stress test as time zero. Univariate and multivariable Cox proportional hazard models using reliable predictors obtained by bootstrapping methods were also employed to examine the association of all-cause mortality in PAD patients with SDS and SSS. Again, we adjusted for a pre-test history of coronary and lower extremity arterial interventions. In addition, we repeated all-cause mortality analyses after excluding ejection fraction from the models. Furthermore, we re-examined the impact of SDS and SSS on all-cause mortality only in those whose cardiac stress testing was performed for evaluation of cardiac symptoms. In the univariate models, each SDS, SDS > 0, SSS, and SSS > 0 was modeled individually, while all of them were included together in the multivariable models. Importantly, a hierarchical approach examined the step-wise addition of post-testing coronary and lower extremity arterial interventions to the full mortality models to understand their impact on the above associations. Bootstrap bagging methods14,15 were used to determine reliable predictors for the above models with random re-sampling and automated step-wise selection (entry criteria of p ⩽ 0.1 and retention criteria of p ⩽ 0.05). Variables or clusters of variables which entered more than 50% of 1000 models were chosen for the final model. The results were presented as unadjusted and adjusted hazard ratios (HRs) with 95% confidence intervals (CIs). The proportional hazards assumption was tested. We used SAS (v9.2; SAS Inc., Cary, NC, USA) for statistical analyses with a p-value < 0.05 for statistical significance.
Results
Patient characteristics
Of the 645 patients with PAD, 288 (45%) had an abnormal SPECT MPI stress test (SSS > 0). The baseline characteristics of patients stratified by SSS are presented in Table 1. Patients with an abnormal stress test (SSS > 0) were more likely to be male with a significantly higher prevalence of myocardial infarction, heart failure, coronary bypass grafting, prior coronary intervention, and cerebrovascular accident. Patients with abnormal stress tests were also more likely to be on baseline aspirin, statin, or angiotensin-converting enzyme inhibitors, and tended to have lower ejection fractions (Table 1). Pharmacologic stress was performed in the majority of patients without significant difference between the two groups (Table 1). We also presented baseline characteristics of the study population based on the indication of cardiac stress testing. Those with cardiac symptoms were more likely to have cardiac morbidities, abnormal or positive stress tests, or higher stress testing scores, while those with no cardiac symptoms were more likely to have end-stage renal disease, aortoiliac aneurysm, or cancer, which possibly reflects the indication for pre-operative stress testing in these patients (Appendix, Table 1). The mean ± standard deviation follow-up among the study population was 3.2 ± 1.8 years (median = 3.0 years). A total of 2036 patient years were available for analysis. Ten percent had follow-up of nearly 6 years (5.7 years) or more.
Selected baseline characteristics of the study peripheral artery disease population according to SSS a .
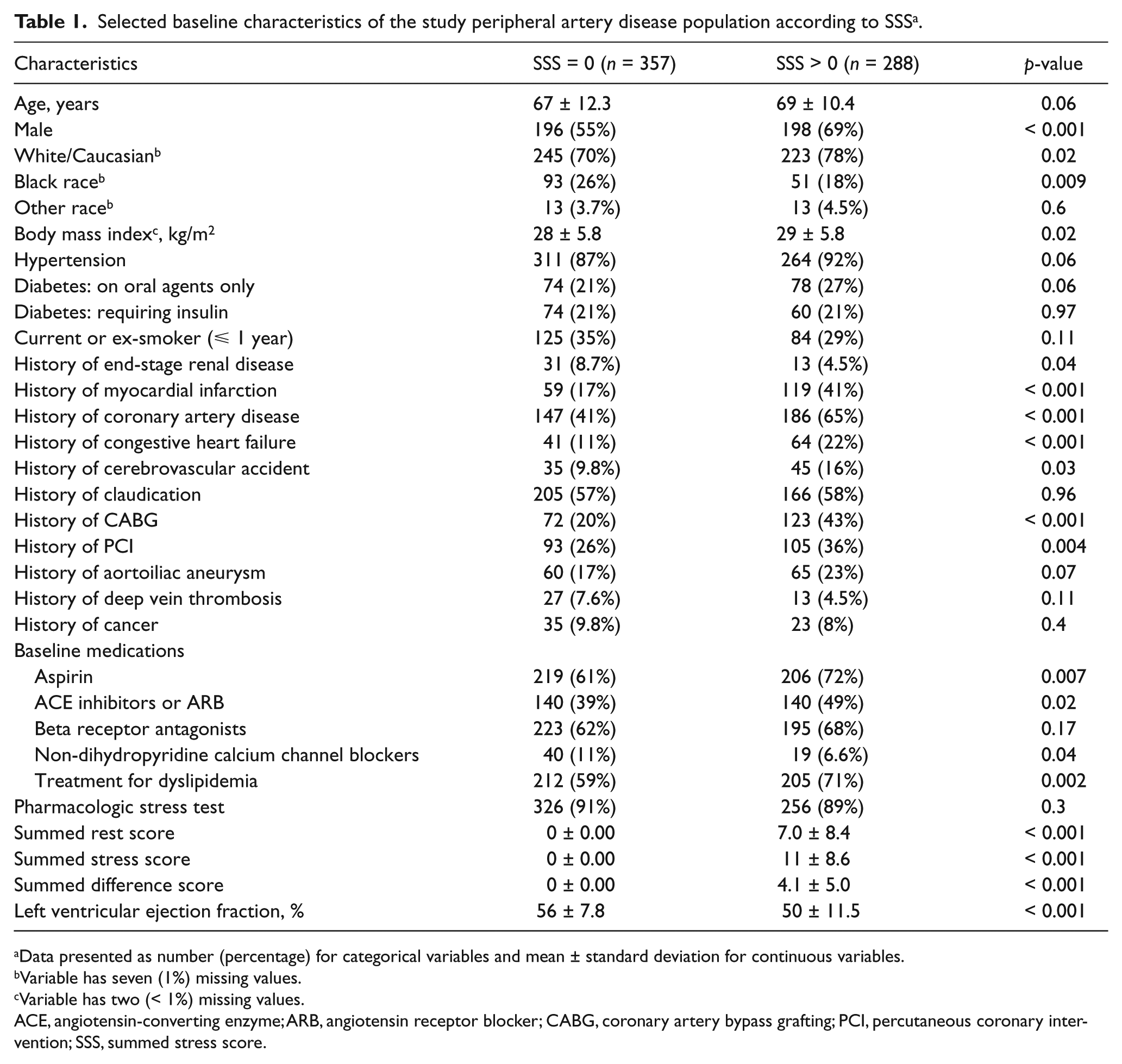
Data presented as number (percentage) for categorical variables and mean ± standard deviation for continuous variables.
Variable has seven (1%) missing values.
Variable has two (< 1%) missing values.
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CABG, coronary artery bypass grafting; PCI, percutaneous coronary intervention; SSS, summed stress score.
Association of SDS > 0 with subsequent coronary revascularization
Fifteen percent of our study cohort underwent coronary revascularization after undergoing stress testing; 30% of those with an SDS > 0. A multivariable Cox proportional hazard regression analysis showed that a positive stress test (SDS > 0) was associated with a significant increase in the occurrence of future coronary revascularization (adjusted HR: 5.46, 95% CI: 1.99–15.00; p = 0.001). This was despite adjustments for pre-stress test coronary intervention in addition to the reliable baseline predictors of post-stress test coronary revascularization as identified by the bootstrapping methods: history of myocardial infarction, family history of CAD, history of pulmonary hypertension, abdominal aortic aneurysm, and shortness of breath.
All-cause mortality in PAD – the impact of SDS and SSS
There were 192 deaths (30%) among our study patients during follow-up. Mortality in the positive stress test group (SDS > 0) was 35% versus 27% in the negative stress test group (SDS = 0), and 35% in the SSS > 0 group versus 25% in the SSS = 0 group. The Kaplan–Meier curves showed no significant mortality difference between positive and negative cardiac stress test cohorts (SDS = 0 vs SDS > 0, p = 0.6) (Figure 2, Panel A) or when comparing SSS > 0 vs SSS = 0 groups, p = 0.1 (Figure 2, Panel B). In unadjusted univariate analysis, only the magnitude of jeopardized myocardium (SSS) was associated with a slight increase in all-cause mortality (HR: 1.02, 95% CI: 1.003–1.04; p = 0.02) (Table 2).
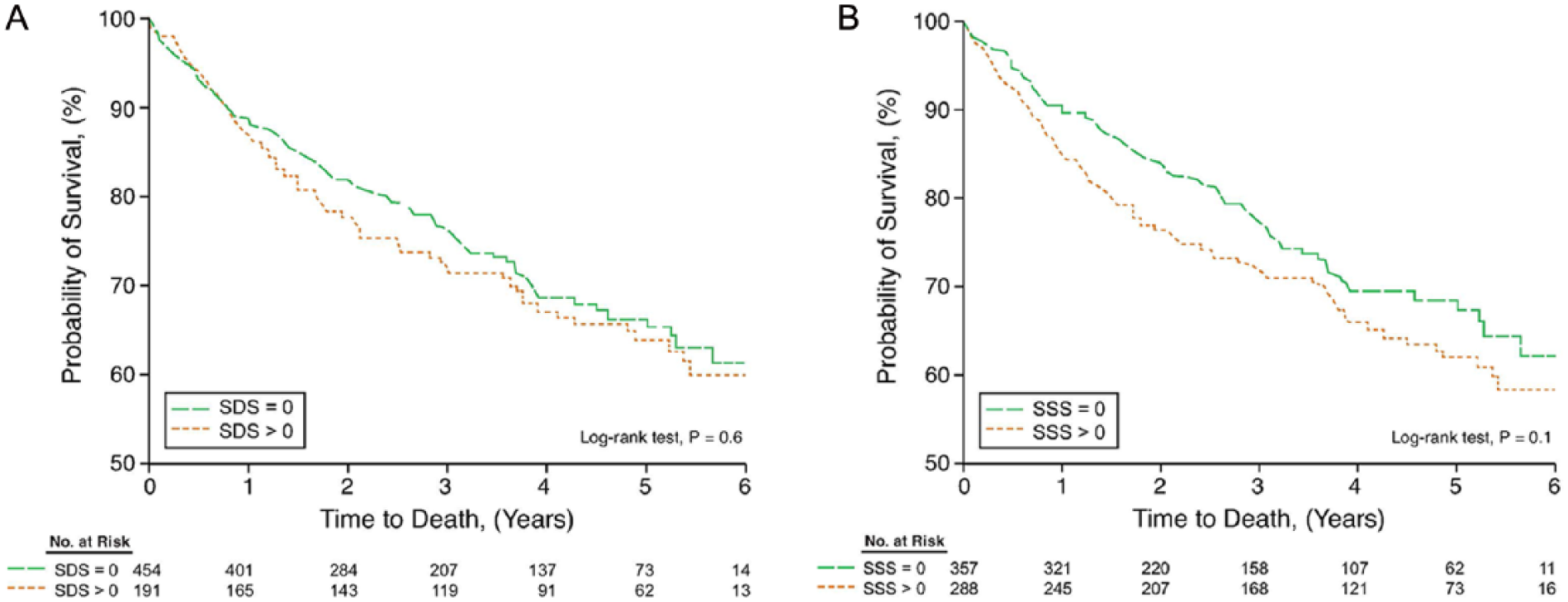
Unadjusted comparison of all-cause mortality among peripheral artery disease (PAD) patients according to SDS (ischemic myocardium) (Panel A) and SSS (jeopardized myocardium) (Panel B). Kaplan–Meier curves for all-cause mortality in PAD patients show no significant differences between SDS = 0 and SDS > 0 groups (Panel A), or between SSS = 0 and SSS > 0 groups (Panel B). (SDS, summed difference score; SSS, summed stress score.)
Univariate and multivariable analyses for all-cause mortality in peripheral artery disease patients.
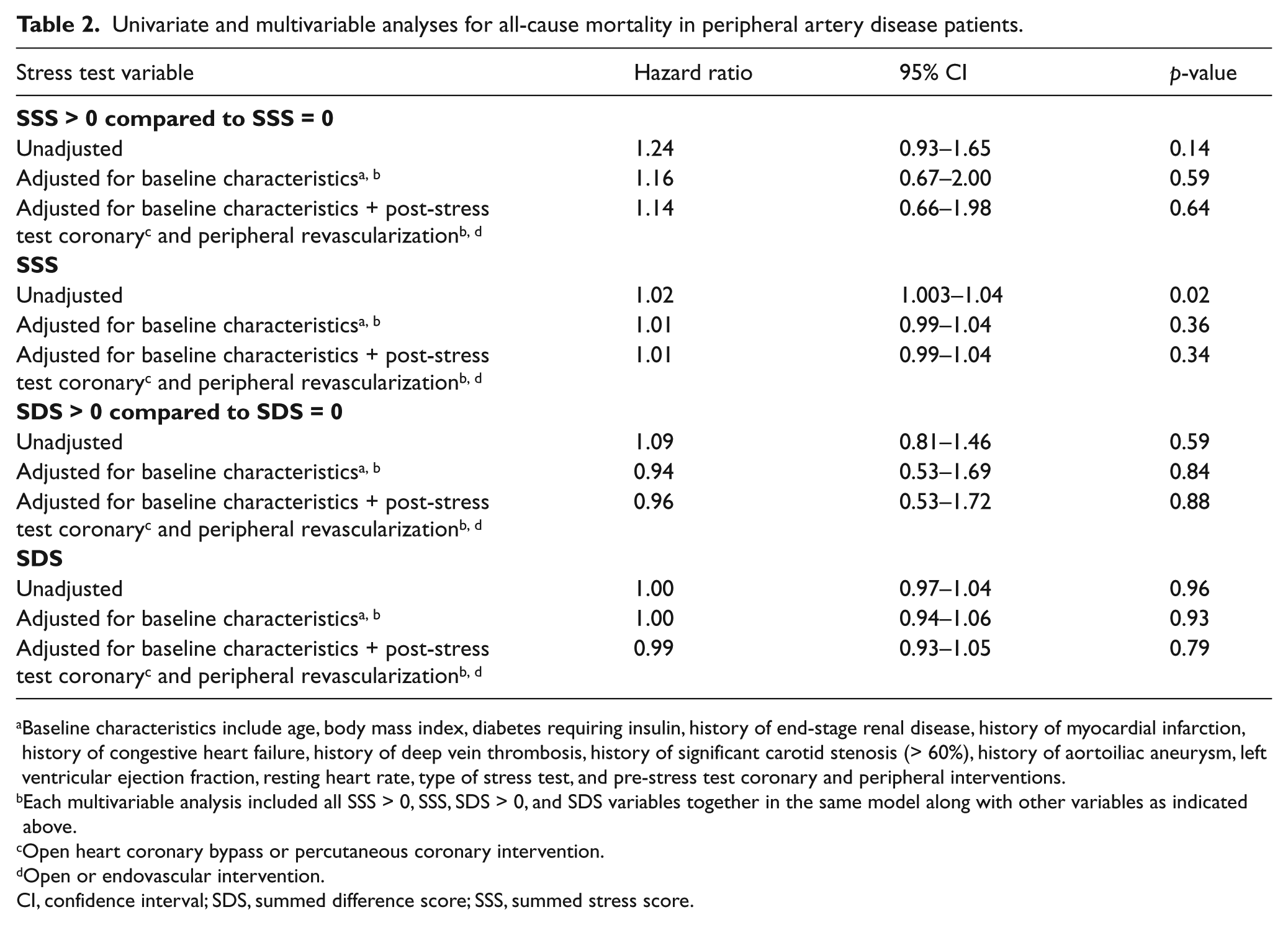
Baseline characteristics include age, body mass index, diabetes requiring insulin, history of end-stage renal disease, history of myocardial infarction, history of congestive heart failure, history of deep vein thrombosis, history of significant carotid stenosis (> 60%), history of aortoiliac aneurysm, left ventricular ejection fraction, resting heart rate, type of stress test, and pre-stress test coronary and peripheral interventions.
Each multivariable analysis included all SSS > 0, SSS, SDS > 0, and SDS variables together in the same model along with other variables as indicated above.
Open heart coronary bypass or percutaneous coronary intervention.
Open or endovascular intervention.
CI, confidence interval; SDS, summed difference score; SSS, summed stress score.
The reliable baseline predictors of mortality identified by bootstrapping methods were age, body mass index, diabetes requiring insulin, end-stage renal disease, history of congestive heart failure, myocardial infarction, abdominal aortic aneurysm, significant carotid stenosis (> 60%), lower extremity arterial intervention, or deep vein thrombosis (marginally significant), LVEF, and resting heart rate. In a multivariable Cox proportional hazard model that adjusted for reliable predictors and pre-stress test coronary and lower extremity arterial revascularizations, positive stress test (SDS > 0) (adjusted HR: 0.94, 95% CI: 0.53–1.69; p = 0.84) or an abnormal stress test (SSS > 0) (adjusted HR: 1.16, 95% CI: 0.67–2.00; p = 0.59) did not have any significant effect on survival. More importantly, in a hierarchical model, further adjustments for post-stress test coronary and lower extremity arterial interventions had no significant impact on all-cause mortality or on the association between the SDS or SSS and all-cause mortality (Table 2).
After excluding ejection fraction from all-cause mortality models, and adjusting for reliable predictors and pre-stress test coronary and lower extremity arterial revascularization, only continuous SSS became statistically significant (adjusted HR: 1.03, 95% CI: 1.002–1.06; p = 0.03). Adjustments for post-stress test coronary and lower extremity arterial interventions in a hierarchical approach did not impact these results (Appendix, Table 2).
In separate analyses including only those with cardiac symptoms (n = 278), SDS and SSS variables still had no significant association with all-cause mortality, similar to our results in the whole study population (Appendix, Table 3).
Discussion
This study found no association between all-cause mortality and functional markers for CAD represented by SDS and SSS in PAD patients, even among those with cardiac symptoms. Therefore, flow-limiting CAD and infarcted scar tissue (SDS and SSS), important precursors for ventricular arrhythmia, do not seem to predict the increased all-cause mortality observed in the PAD population. Our findings are based on adjusted analyses that included multiple baseline characteristics, LVEF, medication use, type of stress test, and any coronary or lower extremity revascularization after obtaining the result of the stress test and ABI. LVEF might be a better marker of myocardial response to ischemic burden; therefore, analyses were repeated without ejection fraction in the models. Of all 12 models tested in Table 2, only continuous SSS became statistically significant (Appendix, Table 2). Ejection fraction is an important prognostic variable; however, from a clinical standpoint, a low LVEF does not preclude ischemic work-up when indicated.
Data have repeatedly shown that patients with PAD have an increased risk of all-cause mortality, 1 with the majority dying from cardiovascular causes. 16 In addition, observational data show a high prevalence of CAD among individuals with PAD,8,9 mostly asymptomatic due to the claudication-limited physical activity in these patients. As a result, there has been an emerging clinical practice trend towards recommending testing for CAD in PAD patients; 8 however, it remains unknown whether uncovering the presence of coexisting CAD would help further risk stratification of these patients beyond what has been recommend by the guidelines. 13 In this study, we used hierarchical multivariable models to understand the role of SDS and SSS (markers of myocardial ischemia and scar) as possible predictors of all-cause mortality in this population. Available evidence shows that SPECT MPI perfusion scores (SDS and SSS), in addition to providing a quantitative assessment of total ischemic burden and total jeopardized myocardium, are independent predictors of all-cause mortality.5–7 Therefore, we believe, SDS and SSS are well suited to address the study objective.
In one study (1992–1995) by Thatipelli et al. (n = 395), no correlation was found between the wall motion score index on dobutamine stress echocardiography and ABI. Moreover, in multivariable analysis, inducible and fixed wall motion abnormalities on dobutamine stress echocardiography did not predict all-cause mortality. 10 This study, however, was limited by small sample size and the heterogeneity of its population (14% had no PAD), though ABI was adjusted for in the multivariable analysis. In another study, Ishihara et al. 17 found that the rate of all-cause mortality in PAD patients was lowest in the no-CAD group and highest in the triple-vessel CAD group. The limitations included small sample size, lack of functional assessment of CAD (coronary angiography was used), and lack of adjusted analysis. Our study has a larger sample size, applies multivariable adjustment using reliable predictors from bootstrapping methods, and also involves a contemporary patient population on appropriate guideline-recommended medications. Moreover, we present for the first time a step-wise hierarchical approach to test the impact of post-stress test coronary and peripheral interventions on all-cause mortality and its association with SDS and SSS (Table 2).
There are several studies that have shown that the SPECT MPI cardiac stress test can predict early18,19 and late20,21 coronary revascularization. Similarly in our study, patients with a positive stress test (SDS > 0) were more likely to have subsequent (post-stress test) coronary revascularization despite adjustment for all reliable predictors identified by bootstrapping methods in addition to history of (pre-stress test) coronary and lower extremity arterial intervention. However, there was still no association between SDS > 0 and all-cause mortality in PAD patients, even after adjusting for post-stress test coronary revascularization. Therefore, it appears that patients with PAD and a positive stress test are more likely to undergo coronary revascularization; however, in this analysis, revascularization did not impact clinical outcomes.
Limitations
While this study proposes a new perspective to the well-known association between PAD and mortality, a few limitations need to be highlighted. First, ABI measurements and cardiac stress testing were, for the most part, symptom-driven in a population with multiple co-morbidities typical for a tertiary care center. Hence, the results may not be easily generalized to the asymptomatic ‘silent’ PAD population. Second, patients with an abnormal stress test (SSS > 0) as a group had a significantly higher prevalence of cardio-protective medication use that included aspirin, statin, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, and beta-blockers that may have modified the association between SSS and mortality. Nevertheless, our results are based on adjustments for cardio-protective medication use. Lastly, despite multiple adjustments and prospectively collected data, this is a retrospective analysis and the results must be verified by well-designed randomized clinical trials.
Conclusions
In this study, there was no association between the presence or extent of functionally significant CAD and all-cause mortality in patients with PAD. The myocardial ischemic burden and total jeopardized myocardium do not appear to predict the high mortality seen in the PAD population. Furthermore, neither coronary nor lower extremity arterial revascularization after cardiac stress and ABI testing had any significant impact on all-cause mortality. Hence, cardiac stress testing is likely of limited additional value for risk stratification in the PAD population beyond the aggressive risk stratification recommended by the guidelines. However, it is often unavoidable in the setting of questionable cardiac symptoms.
Footnotes
Acknowledgements
Suzanne Turner, Department of Graphics, Heart and Vascular Institute, Cleveland Clinic and Kathryn Brock, BA, Editorial Services Manager, Heart and Vascular Institute, Cleveland Clinic.
Dr. Joshua Beckman served as guest editor for this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
