Abstract
Few studies have assessed which biomarkers influence mortality risk among those with peripheral arterial disease (PAD). We analyzed data from 556 individuals identified to have PAD (i.e. ankle–brachial index ⩽0.9) with available measurements of C-reactive protein, the neutrophil-to-lymphocyte ratio (NLR), homocysteine, and the urinary albumin-to-creatinine ratio (UACR) in the 1999–2004 National Health and Nutrition Examination Survey. We investigated whether a combination of these biomarkers improved the prediction of all-cause and cardiovascular mortality beyond conventional risk factors. During follow-up (median, 8.1 years), 277 of 556 participants died; 63 deaths were attributed to cardiovascular disease. After adjusting for conventional risk factors, Cox proportional-hazards models showed the following to be most strongly associated with all-cause mortality (each is followed by the adjusted hazard ratio [HR] per 1 standard deviation increment in the log values): homocysteine (1.31), UACR (1.21), and NLR (1.20). UACR alone significantly predicted cardiovascular mortality (1.53). Persons in the highest quintile of multimarker scores derived from regression coefficients of significant biomarkers had elevated risks of all-cause mortality (adjusted HR, 2.45; 95% CI, 1.66–3.62; p for trend, <0.001) and cardiovascular mortality (adjusted HR, 2.20; 95% CI, 1.02–4.71; p for trend, 0.053) compared to those in the lowest two quintiles. The addition of continuous multimarker scores to conventional risk factors improved risk stratification of all-cause mortality (integrated discrimination improvement [IDI], 0.162; p<0.00001) and cardiovascular mortality (IDI, 0.058; p<0.00001). In conclusion, the addition of a continuous multimarker score to conventional risk factors improved mortality prediction among patients with PAD.
Keywords
Introduction
Peripheral arterial disease (PAD) is the third leading cause of atherosclerotic cardiovascular morbidity worldwide. 1 Associated with a marked increase in individuals’ risk of mortality from coronary and cerebrovascular disease,2,3 mortality from PAD continues to increase. 4 Improved risk assessment of these individuals may lead to better risk factor modification and improved outcomes. 5
Multiple physiologic pathways implicated in the pathogenesis of PAD may also contribute to individuals’ mortality risk once PAD has developed. Though limited prior research has evaluated modifiers of risk among those with PAD, such research suggests that inflammation may be critically important. Inflammatory pathways have been linked to the pathogenesis of PAD, 6 and an association between C-reactive protein (CRP), a marker of systemic inflammation, and prevalent and incident PAD is well described.7–14 In the limited existing studies on adverse outcomes among those diagnosed with PAD, CRP has been linked to increases in the combined outcome of death and adverse cardiovascular and peripheral events, 15 and higher rates of reintervention and death following endovascular therapy. 16 An alternative inflammatory marker, the neutrophil-to-lymphocyte ratio (NLR), implicated in the prognosis of other cardiovascular diseases,17–19 was associated with cardiovascular mortality in one study examining patients with critical limb ischemia. 20 PAD has also been linked to homocysteine, 21 a measure of oxidative stress and increased thrombogenicity, 22 and to albuminuria, a marker of endothelial function and atherogenic milieu,23–25 though the prognostic value of these biomarkers among those already with PAD has not been previously considered.
Since it remains undetermined whether multiple biomarkers might be independently associated with mortality in those with PAD, and whether, with simultaneous measurement, one might enhance the ability to risk stratify such patients, we investigated the prognostic value of multiple biomarkers using the 1999 to 2004 National Health and Nutrition Examination Survey (NHANES). Using the multimarker approach advocated elsewhere,26,27 we investigated whether CRP, NLR, homocysteine, and the urinary albumin-to-creatinine ratio (UACR), either alone or in combination, improved prognostication of all-cause and cardiovascular mortality among individuals with PAD.
Methods
Study population
NHANES, a repeated, cross-sectional, stratified, multistage survey of the non-institutionalized US population, has been previously described. 28 In brief, the survey includes questionnaire, physical exam, and laboratory evaluation components. As described ahead, individuals’ mortality status was determined through the National Death Index (NDI). NHANES protocols were approved by the National Center for Health Statistics (NCHS) institutional review board. All participants gave written, informed consent.
Ankle–brachial index
Individuals ⩾40 years of age, weighing ⩽400 pounds (181.4 kg) and without bilateral amputation, were asked to participate in the ankle–brachial index (ABI) examination. Laying supine, systolic pressures were measured preferentially at the right brachial artery, twice among those aged 40–59 years old and once in those ⩾60 years old, and twice in the posterior tibial arteries bilaterally for all age groups. The ABI was calculated automatically (Mini-Lab IV, Model 3100; Parks Medical Electronics, Inc., Aloha, OR, USA) by dividing the mean systolic blood pressure at the ankle by the mean brachial artery systolic blood pressure. PAD was defined as an ABI in either leg ⩽0.90.
Biomarker measurement
Parameters of the complete blood count were determined by a Beckman Coulter MAXM instrument (Brea, CA), with absolute counts estimated by multiplying the white blood cell count by the white cell differential percentages. NLR was derived as the absolute count of neutrophils divided by the absolute count of lymphocytes. High-sensitivity CRP was measured using latex-enhanced nephelometry on a Behring Nephelometer Analyzer System (Somerville, NJ). The lowest reportable CRP level was 0.02 mg/dL. Homocysteine was measured by fully automated fluorescence polarization immunoassay from Abbott Diagnostics that was performed, until 2002, on the Abbott IMx analyzer and, thereafter, on an Abbott AxSYM system (Abbott Park, IL): results were harmonized for comparison across years. UACR was derived as the ratio of urinary albumin, measured by solid-phase fluorescent immunoassay, and urinary creatinine, assessed using the Jaffé rate reaction by a CX3 analyzer (Beckman Coulter, Brea, CA).
Definitions of other variables
Demographic variables included age, sex, race/ethnicity, and smoking status (i.e. never, former, current). Prior recognized cardiovascular disease was based on self-reported personal history; respondents replying affirmatively to any of the questions, ‘has a doctor or other health professional ever told you that you had (coronary heart disease, angina [also called angina pectoris], heart attack [also called myocardial infarction], stroke)?’ were classified as having recognized cardiovascular disease.
Regarding other medical comorbidities, individuals were considered to have diabetes mellitus if they reported a physician diagnosis, reported taking insulin or other oral prescription medication for diabetes, had non-fasting glucose values ⩾200 mg/dL, had fasting glucose values ⩾126 mg/dL, or had a glycosylated hemoglobin (A1c) ⩾6.5%. Patients were considered to have hypertension if they reported a prior diagnosis, were taking anti-hypertensive medications, or had an average measured systolic blood pressure ⩾140 mmHg or diastolic blood pressure ⩾90 mmHg. Chronic kidney disease was defined as an estimated glomerular filtration rate ⩽60 mL/min/m2, as estimated by the Modification of Diet in Renal Disease study equation; 29 associated serum creatinine levels were calculated by use of the Jaffé rate method. Individuals’ body-mass index (BMI) was calculated from their measured weight in kilograms divided by the square of their measured height in meters. While used to define PAD, as discussed above, ABI was not included as a covariate itself. A sensitivity analysis with its inclusion did not materially alter the results presented.
Mortality follow-up
Mortality status was based upon a probabilistic record match with the National Death Index (NDI) using demographic identifiers (e.g. social security number, sex, date of birth) conducted by the NCHS through 2011. 30 In this analysis, we assessed deaths from any cause and from cardiovascular disease (i.e. International Classification of Diseases, 10th Revision codes I00 to I99). The follow-up duration was calculated from the date of examination to either the date of death or to 31 December 2011.
Statistical analysis
Data were summarized as percentages in the case of categorical variables and as means in the case of continuous variables. Of note, we performed unweighted analyses. To maximize the intrinsic validity of our results and to develop the most physiologically valid multimarker scores, we treat participants as any other multi-ethnic cohort despite the survey’s underlying complex selection process. Accordingly, we avoid inappropriately privileging certain individuals’ biomarker values above others in score development but, in so doing, forgo any claim as to the national representativeness of our results. To corroborate the reported results, a sensitivity analysis employing a weighted analysis incorporating the survey’s complex design was conducted. Results did not materially change the conclusions discussed here and are reported in an online Supplemental Appendix. Differences of potential note between the weighted and unweighted analyses are discussed below.
We pursued a multimarker approach designed to limit false positive results and ensure parsimony.26,27 With each outcome of interest, a multivariable Cox proportional-hazards model was fit with all biomarkers entered as a set. Biomarkers were transformed to the logarithmic scale and standardized to improve comparability. Improvement in this model over a model employing conventional risk factors alone was assessed using the likelihood ratio test between the two models. We pursued further analyses only if the multivariable p-value from the likelihood test was <0.05. Parsimony was then achieved using a backward elimination procedure in which the retention threshold was p<0.05. A multimarker score was then constructed based on the identified significant biomarkers. Following others’ approach,26,27 individuals were subsequently categorized by the quintile of that multimarker score. Those in the highest quintile were considered to be at comparatively high risk, those in the second and third were considered to be at intermediate risk, and those in the lower two quintiles were considered to be at comparatively low risk. We then constructed cumulative mortality curves based on these categorizations using the Kaplan-Meier method and, using Cox proportional-hazard regression, calculated hazard ratios (HRs) for these categorizations, accounting for conventional risk factors. Assumptions of proportional hazards were checked with each model by inspection of Schoenfield residuals.
Improvement in risk stratification with the use of the calculated multimarker scores was assessed by receiver-operating-characteristic (ROC) curves and the C statistic, in which the categorical multimarker score was added to a logistic regression model predicting mortality during the follow-up period. Since no standard thresholds of clinical interest are available regarding risk for mortality among those with PAD, an already high-risk group, we calculated the integrated discrimination improvement (IDI) metric using the continuous multimarker scores in order to gauge improvement in risk stratification by inclusion of the score compared to conventional risk factors alone. 31 By comparing appropriate increases in individuals’ estimated risk with inclusion of the multimarker score for those with adverse events alongside appropriate decreases in estimated risk for those without adverse events, the IDI provides a holistic assessment of how the multimarker score affects risk stratification. Data were analyzed using Stata version 11.2 (StataCorp, College Station, TX, USA).
Results
Of the 31,126 participants in the 1999 to 2004 NHANES, 7571 adults ⩾40 years old underwent ABI measurement. Of these, 647 were identified to have PAD, of which 556 had complete measurements of the biomarkers and covariates employed here. Baseline characteristics did not differ between those with and without complete measurements (Supplemental Table 1). Individuals were followed for a median 8.1 years (interquartile range, 4.9–9.8 years; mean, 7.3 years). During that time, 277 of 556 (49.8%) participants died, of whom 164 (59.2%) were male. Of those deaths, 63 were attributed to cardiovascular disease, of whom 39 (61.9%) were male.
Shown in Table 1 are sample characteristics for the analysis. At baseline, individuals were an average 70.9 years old. Approximately half were male and 57.4% were non-Hispanic white individuals. Relatively few had prior recognized cardiovascular disease, though risk factors such as hypertension, chronic kidney disease, and hypercholesterolemia were relatively common.
Baseline characteristics of the study participants.

Data presented as means ± standard deviation and, for biomarker levels, as medians (interquartile range).
Recognized cardiovascular disease includes prior diagnosis of coronary heart disease, angina pectoris, myocardial infarction, or cerebrovascular disease.
Indicates the use of angiotensinogen-converting enzyme/angiotensin II receptor blocker medications.
Table 2 presents age- and sex-adjusted partial correlation coefficients among the biomarkers. After adjustment, NLR was significantly associated with CRP (r = 0.20, p<0.001) and UACR (r = 0.13, p<0.001) but not homocysteine. UACR was, in turn, correlated with CRP(r = 0.09, p=0.04) and homocysteine (r = 0.12, p=0.01). Homocysteine was not, however, associated with either NLR or CRP.
Age- and sex-adjusted partial correlations among biomarkers.
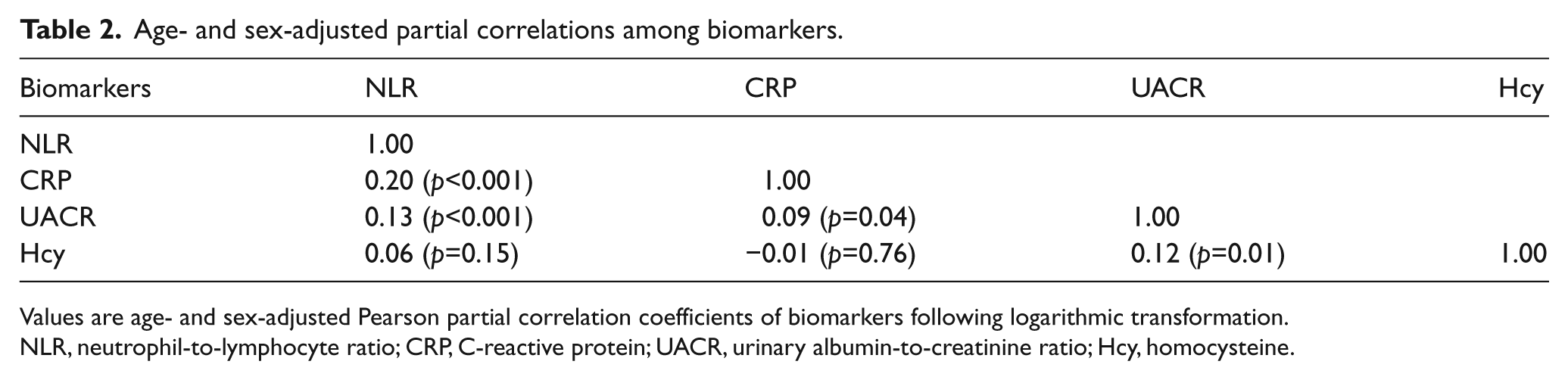
Values are age- and sex-adjusted Pearson partial correlation coefficients of biomarkers following logarithmic transformation.
NLR, neutrophil-to-lymphocyte ratio; CRP, C-reactive protein; UACR, urinary albumin-to-creatinine ratio; Hcy, homocysteine.
The four biomarkers were, in aggregate, significantly associated with all-cause (multivariable p from the likelihood ratio test, p<0.0001) and cardiovascular mortality (multivariable p, 0.012) after adjusting for conventional risk factors. Following backwards elimination, the following biomarkers were retained as predictors of all-cause mortality: homocysteine, UACR, and NLR. Of note, in the weighted analysis, only homocysteine and UACR were retained as significant predictors (Supplemental Table 5). Following backwards elimination, only UACR was found to be a significant predictor of cardiovascular mortality (Supplemental Table 2).
Multimarker scores were then generated using the biomarkers selected by backward elimination, with thresholds of low, medium, and high risk identified (Table 3). Figure 1 displays the cumulative probability of death from all causes and from cardiovascular disease during the follow-up period for individuals classified as low, intermediate, and high risk by the respective multimarker scores. Adjusted HRs for these risk categories are shown in Table 4. Compared to those at low risk, those in the highest risk categories were more likely to die during follow-up (HR, 2.45; 95% CI, 1.66–3.62; p for tend, <0.001) and also more likely to die from a cardiovascular cause (HR, 2.20; 95% CI, 1.02–4.71; p for trend, 0.053). Of note, estimates from the weighted analysis, for both all-cause and cardiovascular mortality, disclosed greater HRs and a statistically significant trend (p=0.001) for cardiovascular death (Supplemental Table 6).
Multimarker scores for the prediction of all-cause and cardiovascular mortality, with cutoff points distinguishing low, intermediate, and high risk. a
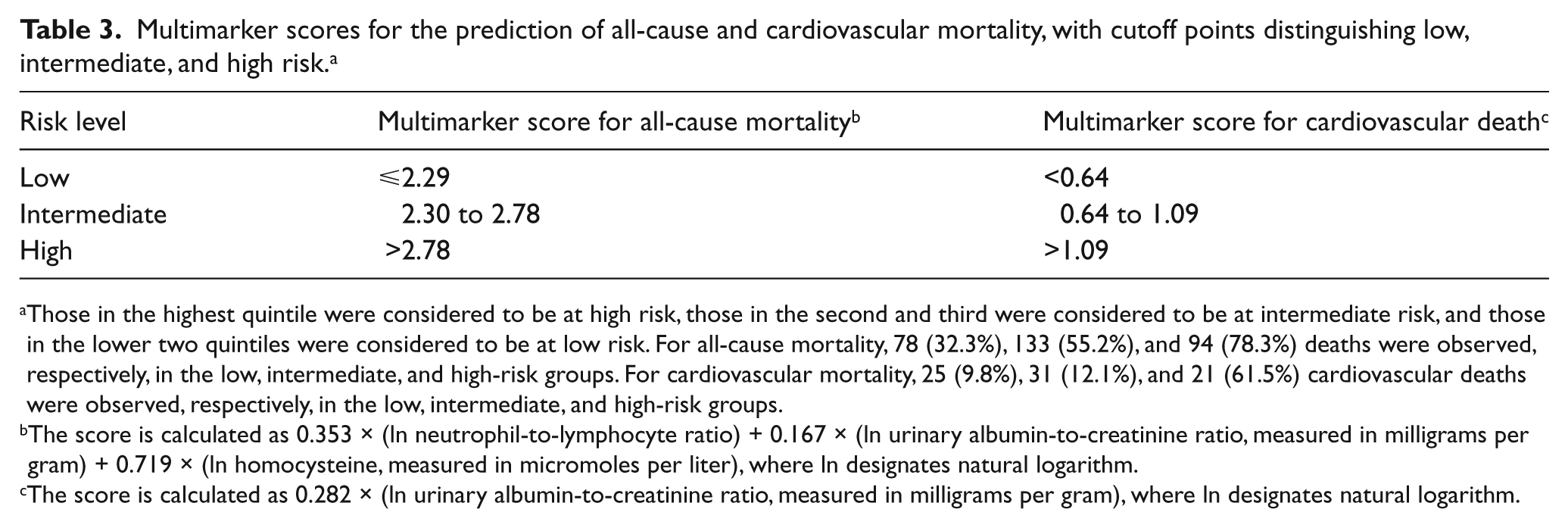
Those in the highest quintile were considered to be at high risk, those in the second and third were considered to be at intermediate risk, and those in the lower two quintiles were considered to be at low risk. For all-cause mortality, 78 (32.3%), 133 (55.2%), and 94 (78.3%) deaths were observed, respectively, in the low, intermediate, and high-risk groups. For cardiovascular mortality, 25 (9.8%), 31 (12.1%), and 21 (61.5%) cardiovascular deaths were observed, respectively, in the low, intermediate, and high-risk groups.
The score is calculated as 0.353 × (ln neutrophil-to-lymphocyte ratio) + 0.167 × (ln urinary albumin-to-creatinine ratio, measured in milligrams per gram) + 0.719 × (ln homocysteine, measured in micromoles per liter), where ln designates natural logarithm.
The score is calculated as 0.282 × (ln urinary albumin-to-creatinine ratio, measured in milligrams per gram), where ln designates natural logarithm.
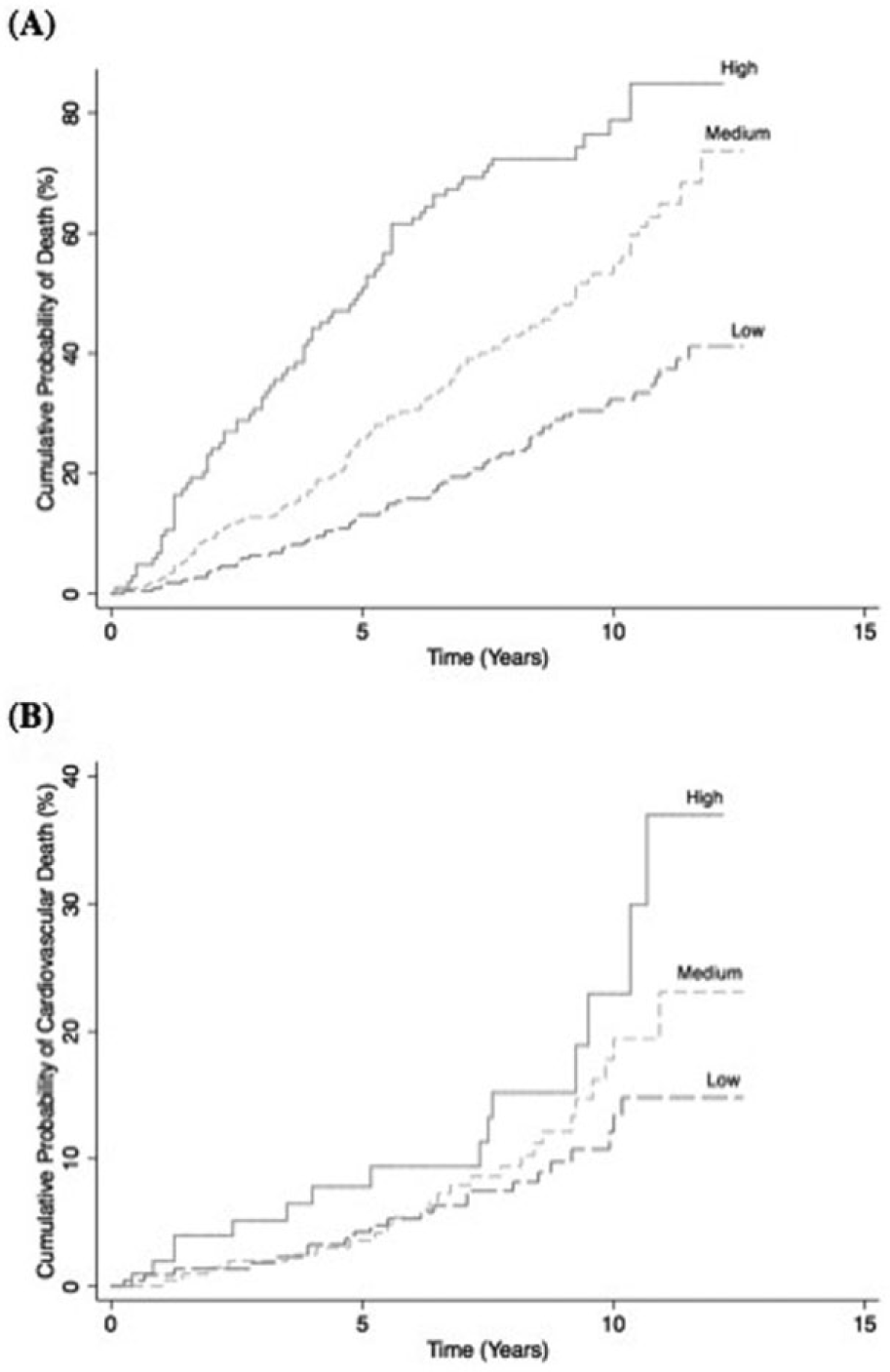
Kaplan-Meier curves of the cumulative probability of death (A) from all causes and (B) from cardiovascular causes, by multimarker score. Those in the highest quintile of the multimarker score were considered to be at high risk, those in the second and third were considered to be at intermediate risk, and those in the lower two quintiles were considered to be at low risk.
Relation of multimarker risk score to outcomes. a
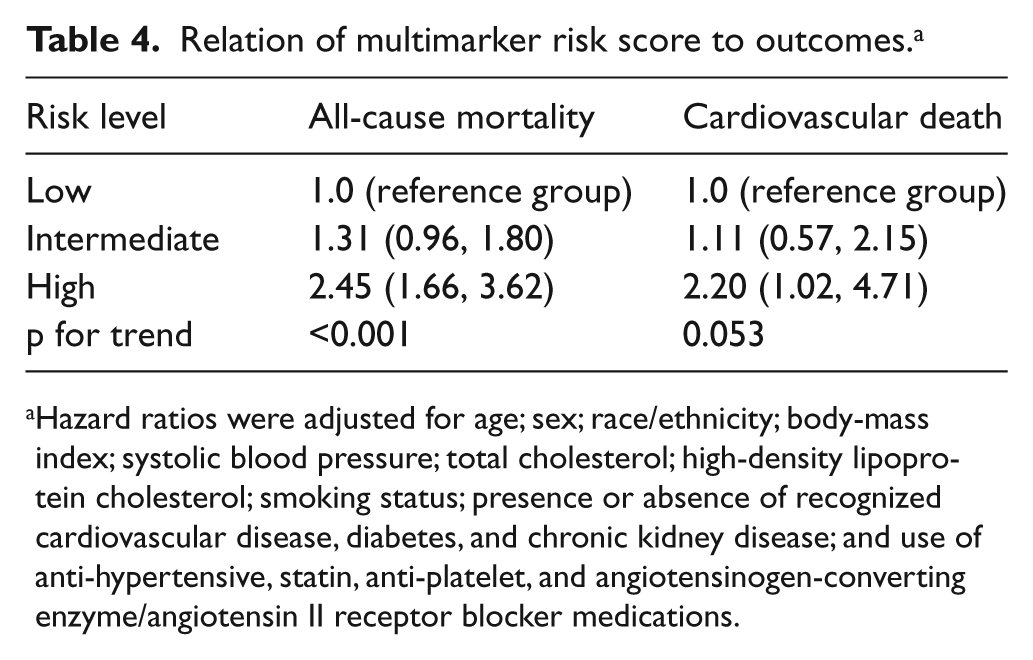
Hazard ratios were adjusted for age; sex; race/ethnicity; body-mass index; systolic blood pressure; total cholesterol; high-density lipoprotein cholesterol; smoking status; presence or absence of recognized cardiovascular disease, diabetes, and chronic kidney disease; and use of anti-hypertensive, statin, anti-platelet, and angiotensinogen-converting enzyme/angiotensin II receptor blocker medications.
For the all-cause mortality analysis, C statistics were 0.746 with age and sex as predictors, 0.805 for age, sex, and all other conventional risk factors, and 0.814 for all conventional risk factors and the multimarker score. In the cardiovascular mortality analysis, C statistics were 0.619 with age and sex as predictors, 0.739 for age, sex, and all other conventional risk factors, and 0.739 for all conventional risk factors and the multimarker score. As shown in Figure 2, the ROC curves overlapped between the conventional risk factors model and the model with the added multimarker score for both all-cause mortality (p=0.17) and cardiovascular mortality (p=0.95). Inclusion of the respective continuous multimarker score, however, improved both classification of all-cause mortality (IDI, 0.162; p<0.00001) and cardiovascular mortality (IDI, 0.058; p<0.00001) compared to conventional risk factors alone.
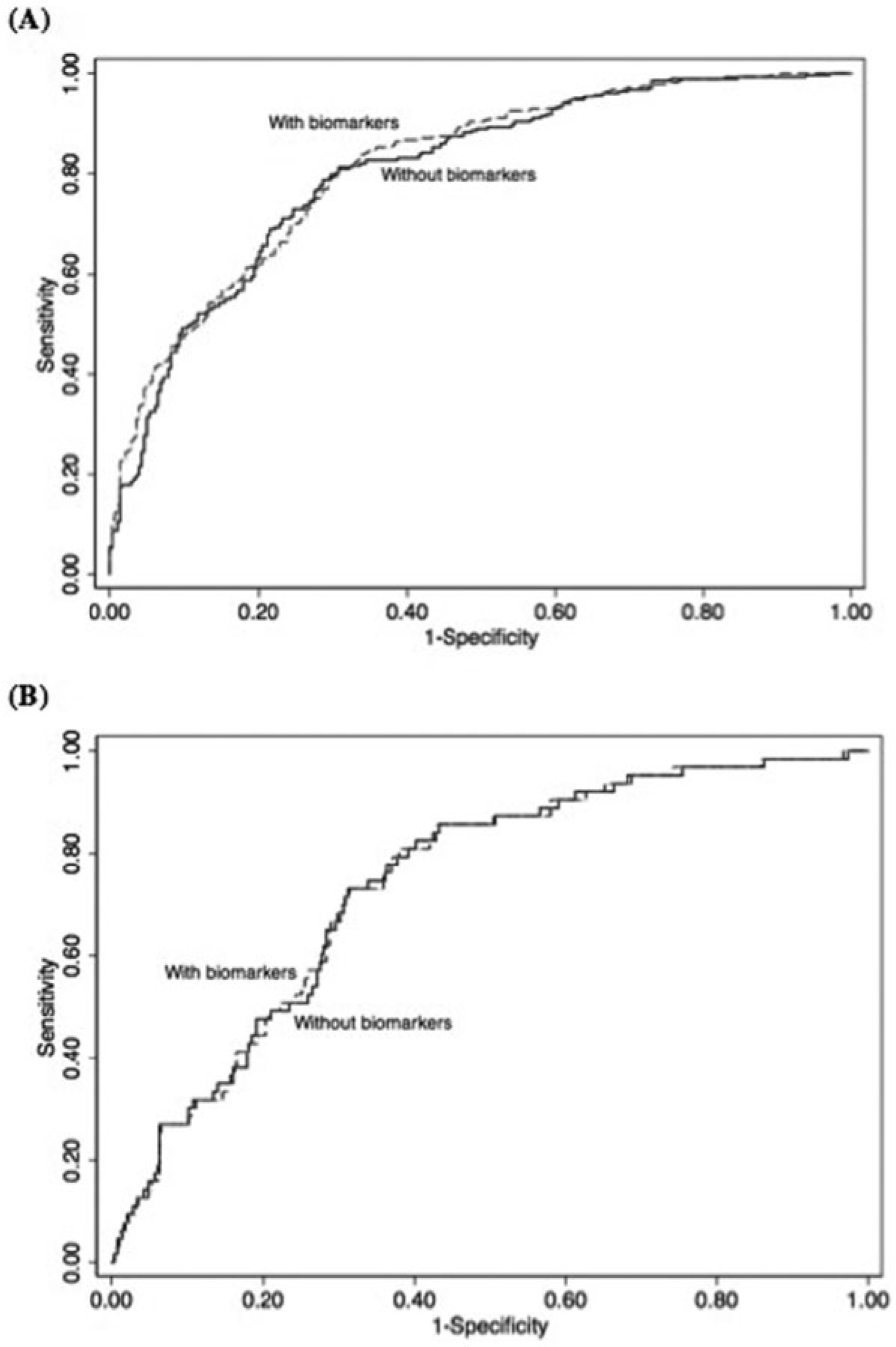
Receiver-operating-characteristic (ROC) curves for death (A) from all causes and (B) from cardiovascular causes during the follow-up period. Compared to conventional risk factors, the C statistic did not improve significantly with the inclusion of the categorical multimarker score for all-cause mortality (C statistic, 0.814 vs 0.805, respectively; p=0.17) or for cardiovascular mortality (C statistic, 0.739 vs 0.739, respectively; p=0.95). As measured by the integrated discrimination improvement, inclusion of the continuous multimarker scores improved classification of all-cause mortality (IDI, 0.162; p<0.00001) and cardiovascular mortality (IDI, 0.058; p<0.00001) compared to conventional risk factors.
Discussion
Improved risk assessment is needed for those with PAD. 5 In this study, we assessed the prognostic value of multiple biomarkers for all-cause and cardiovascular mortality in a multi-ethnic sample with a median 8.1 years of follow-up. We report here that, after adjusting for conventional risk factors, elevated levels of homocysteine, the UACR, and the NLR were significantly associated with increased all-cause mortality. The UACR was also significantly associated with death from cardiovascular causes. Inclusion of these biomarkers as continuous multimarker scores significantly improved risk stratification of those with PAD.
Results from these prospective data should be interpreted in light of prior research. After accounting for other biomarkers, including the alternative inflammatory marker NLR, CRP was not significantly associated with mortality outcomes in our study. This stands in contrast to the limited prior data linking CRP to adverse cardiovascular events in those with PAD.15,16 CRP and NLR have not been previously evaluated simultaneously when considering outcomes among those with PAD. While the significance of these findings is not immediately apparent, a few considerations merit comment. First, these biomarkers were relatively poorly correlated with one another so it is unlikely that these results were driven by statistical reasons alone. Second, at least one prior study has described both NLR and CRP to be associated with an adverse cardiovascular outcome, albeit in a different clinical setting. 32 Third, and relatedly, others have previously suggested that CRP and NLR reflect different underlying pathologic mechanisms. 33 Consequently, the question as to what underlying mechanism, in our sample with patients with PAD, caused NLR but not CRP to be a significant predictor of mortality merits further consideration. Moreover, the potential superior prognostic value of NLR in certain clinical settings 34 might have economic implications since it is a cheap and widely available measure. 35
Homocysteine was also a significant predictor of all-cause mortality in this sample. Previously reviewed as a biologically plausible factor in systemic atherosclerosis, 36 evidence linking homocysteine and PAD has been equivocal,14,21 and homocysteine had not been previously examined as a risk factor for mortality in those already with PAD. Nonetheless, homocysteine has been associated with other adverse cardiovascular outcomes, such as stroke, 37 so our findings are not altogether unsurprising.
Importantly, though, these results raise the possibility of whether hyperhomocysteinemia might be a modifiable risk factor among those with PAD. Randomized control trials targeting homocysteine as a modifiable risk factor in other populations at risk for cardiovascular disease have thus far proved unsuccessful. 38 Yet, while limited in size and number, interventions targeting hyperhomocysteinemia in the high-risk PAD population are slightly more encouraging. The larger of two small trials 39 conducted to address hyperhomocysteinemia found that median ABI improved by 0.07 after 16 weeks with folic acid supplementation, which occurred in the absence of any measurable improvement in patients’ inflammatory biomarkers. 40 Results from our study help support the notion that findings from that trial merit replication. Since lower ABIs predispose individuals to adverse outcomes, 41 such interventions hold promise at altering the clinical course of those with PAD.
UACR, after adjusting for conventional risk factors, including chronic kidney disease, also significantly predicted all-cause and cardiovascular mortality. Albuminuria has previously been linked to carotid plaque initiation and growth, 42 incident hypertension, 43 and PAD prevalence. 44 Results from the Dutch Hoorn study examining both PAD and microalbuminuria found both to be mutually independent risk factors for mortality. 25 Findings here that UACR, among those with PAD, increases mortality risk corroborate and extend those prior results to a multi-ethnic US sample.
Limitations and strengths
An understanding of the study’s limitations is important for interpretation. The mortality analyses presented here are based upon a comparatively small subset of NHANES participants and, consequently, limited our power to observe potential underlying differences. Some covariates relied upon participant self-report; potential misclassification may have biased results in an uncertain direction. Moreover, data are observational and residual confounding may remain. Results may also not be generalizable to those with small-vessel PAD since only ABIs, a marker of large-vessel PAD, were measured, and inflammation appears predictive of large- but not small-vessel disease progression. 41 Classification of individuals with PAD relied upon measurements of the posterior tibial arteries; dorsalis pedis artery pressures were not assessed, and this omission may have misclassified certain individuals with arterial disease present in a dorsalis pedis artery but not that limb’s posterior tibial artery. Additional limitations include the inability to assess time-varying covariates over the follow-up period. Progression of PAD has itself been associated with increased cardiovascular morbidity and mortality and we cannot account for changes in ABI over time. 45 We did, however, account for multiple baseline covariates that correlate with PAD progression; for example, PAD in the young is thought to have a more aggressive clinical course and age was incorporated into our models. 46 Such correlations help to attenuate any potential biases left unaccounted for by the study’s design. In addition, the weighted sensitivity analyses largely, though not perfectly, corroborated our results. In the most significant departure from our unweighted results, weighted sensitivity analysis suggested that, among those with PAD, neither NLR nor CRP was predictive of adverse risk. Unweighted results had indicated that NLR was, in fact, predictive. Such a difference stems from the underlying weighting in the sensitivity analysis, privileging the importance of some deaths above others in order to generate nationally representative results. While representativeness is an admirable goal in studying the population-level effects of biomarkers, our aim to examine and clarify potential improvements in risk prediction among those with PAD leads us to favor, all caveats considered, the unweighted analyses.
The study’s strengths also merit comment. Using a multi-ethnic cohort with validated mortality follow-up data, we provide an assessment of the additional benefit of measuring multiple biomarkers on all-cause and cardiovascular mortality in an increasingly common, high-risk population. We incorporated multiple potential confounders into our models, including baseline medication use, which has often been omitted from prior studies. Our study employed ABI as an objective diagnostic criterion for PAD. ABI has previously been shown to have excellent specificity and very good sensitivity to diagnose PAD. 47 We developed multimarker scores to help risk stratify those with PAD. Use of the multimarker method limits the potential for false positive results and favors identification of the most informative biomarkers. We also assessed the additional benefit in reclassification of those scores compared to conventional risk factors. In so doing, we employed the IDI, a more clinically useful and sensitive metric to risk stratification. 48 We noted that elevated levels of these multimarker scores were independently associated with all-cause and cardiovascular mortality, and contributed further to the risk stratification of individuals with PAD beyond conventional risk factors.
Conclusions
In summary, our findings suggest that, among patients with PAD, homocysteine, the urinary albumin-to-creatinine ratio, and the neutrophil-to-lymphocyte ratio are associated with increased rates of all-cause mortality and that the urinary albumin-to-creatinine ratio is associated with increased rates of cardiovascular mortality. The continuous multimarker scores developed from these measures improved the mortality risk stratification of those with PAD beyond conventional risk factors.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
