Abstract
Objective
High-sensitivity C-reactive protein, a key inflammatory biomarker, can indicate the risk of cardiovascular diseases. The cardiometabolic index has been associated with metabolic and cardiovascular outcomes; however, its direct relationship with high-sensitivity C-reactive protein remains unclear. This study aimed to explore this association and identify influencing factors in adult population.
Methods
This retrospective cross-sectional study used data from the 2015–2018 National Health and Nutrition Examination Survey. A multivariate logistic regression model was applied to assess the association between cardiometabolic index and high-sensitivity C-reactive protein levels (>3.0 mg/L) in 3970 adults, with adjustment for demographic characteristics, comorbidities, and lifestyle factors.
Results
Cardiometabolic index was independently associated with high-risk high-sensitivity C-reactive protein (adjusted odds ratio = 1.11, 95% confidence interval: 1.03–1.19, p = 0.007). Subgroup analyses indicated that the association was more pronounced in females and non-Hispanic Black participants (p values for interaction were <0.001 and 0.004, respectively). Restricted cubic spline curves confirmed a nonlinear relationship between cardiometabolic index and high-risk high-sensitivity C-reactive protein (nonlinearity p < 0.001).
Conclusion
Cardiometabolic index demonstrated a significant positive association with high-risk high-sensitivity C-reactive protein in US adults, serving as a marker linking cardiometabolic status to chronic inflammation. Further studies are needed to validate causality and explore cardiometabolic index–guided prevention.
Keywords
Introduction
Cardiovascular diseases (CVDs) remain the leading cause of death in older individuals. Inflammation is widely recognized as a key contributor to the onset and progression of CVDs. C-reactive protein (CRP), an acute-phase reactant and a nonspecific indicator of inflammation, is broadly accepted as a sensitive inflammatory biomarker. 1 CRP is primarily synthesized in hepatocytes of the liver and plays a vital role in inflammatory responses. 2 Evidence from existing literature suggests that high-sensitivity C-reactive protein (hs-CRP) is associated with unstable atherosclerotic plaques, 3 ischemic stroke, 4 atrial fibrillation,5–7 and impaired infarct healing following revascularization in patients with ST-elevation myocardial infarction.8–10 A CRP level >3.0 mg/L is associated with an increased risk of CVDs, whereas hs-CRP >10.0 mg/L is typically caused by infectious diseases, trauma, and autoimmune diseases. 11
Obesity, defined by abnormal lipid overaccumulation, is significantly associated with an increased risk of all-cause mortality. 12 This condition is frequently linked to metabolic disturbances that can trigger the onset of insulin resistance and related complications, including hypertension, cardiovascular disorders, type 2 diabetes mellitus (T2DM), and dyslipidemia. Traditional indicators such as body mass index (BMI) are used to assess obesity-related cardiovascular risks. However, BMI cannot effectively reflect the actual distribution of body fat. 13 The cardiometabolic index (CMI) is a readily obtainable metric for quantitatively assessing cardiometabolic risk. Initially designed as a marker for predicting the risk of diabetes mellitus (DM), 14 it has demonstrated a strong association with metabolically obese status compared with traditional anthropometric indicators. 15 With the advancement of clinical applications, CMI has emerged as a promising biomarker in cardiovascular risk stratification. Growing evidence indicates that CMI is a valuable prognostic marker for CVDs in the general population, and elevated CMI may be related to all-cause mortality, new-onset cerebral infarction, and heart failure.16–19
Although both CMI and hs-CRP significantly contribute to the occurrence and progression of CVDs, their direct association has not been fully elucidated. Clarifying this relationship is important to understand the intrinsic connection between cardiometabolic abnormalities and chronic inflammation as well as for optimizing risk assessment and prevention strategies for CVDs. Therefore, this study aimed to systematically investigate the association between CMI and serum hs-CRP levels and to determine whether CMI is independently associated with elevated hs-CRP after adjustment for potential confounders.
Materials and methods
Data source and study population
Beginning in 1999, National Health and Nutrition Examination Survey (NHANES) has functioned as a large, nationally representative cross-sectional study, surveying participants every 2 years. It aims to collect data on the health and nutritional status of US citizens, using a stratified multistage random sampling method to obtain representative samples. A comprehensive dataset is collected, including demographic data, questionnaire responses from structured household interviews, physical examination results, and laboratory measurements obtained at analytical facilities. 20 Data from NHANES are publicly available at https://www.cdc.gov/nchs/nhanes/. We used the NHANES database for the 2015–2018 cycles, as these cycles include data on hs-CRP, a key variable in our analysis. This study was conducted in accordance with the Declaration of Helsinki 1975, as revised in 2024. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 21
For the analysis, data from two consecutive NHANES cycles (2015–2018) were combined. Figure 1 presents a flowchart of the study. A total of 19,225 participants were initially included. The following predefined exclusion criteria were applied: (a) participants aged <18 years (N = 7377); (b) participants with missing hs-CRP values (N = 1332); (c) participants with hs-CRP values >10 mg/L (N = 933); (d) participants with missing CMI values (N = 5368); and (e) participants with missing key covariates, including history of hypertension (N = 5), diabetes (N = 4), heart failure (N = 195), coronary heart disease (N = 19), chronic bronchitis (N = 12), cancer or malignancy (N = 4), and smoking history (N = 3). The final analytic sample included 3970 participants.

Flowchart of study participants.
Determination of hs-CRP
Serum samples from participants were stored at −30°C before being transported to Collaborative Laboratory Services for analysis. In the 2015–2016 cycle, the Beckman Coulter UniCel DxC 800 Synchron and Beckman Coulter UniCel DxC 660i Synchron Access chemistry analyzer (DxC 660i) were used, whereas in the 2017–2018 cycle, the Roche Cobas 6000 chemistry analyzer (Cobas 6000) was used. Detailed sample processing and laboratory testing procedures are available on the official NHANES website. A serum hs-CRP threshold of 0.02 mg/mL was used. Consistent with previous studies, hs-CRP ≤3.0 mg/L was categorized as low risk, whereas hs-CRP in the range of 3.1–10.0 mg/L was categorized as high risk. 11
Definition of CMI
CMI was calculated using the following formula.
CMI was derived from waist circumference, height, and blood measurements. Trained health technicians collected anthropometric data, including waist circumference, height, and weight, at the mobile examination center (MEC). After an overnight fast (≥8 h), blood samples were collected, stored at −30°C, and transported to the laboratory for analysis.
Covariates
In our study, covariates included sex, age, marital status (married/other), educational level (below high school, high school, and above high school), race/ethnicity (Mexican Americans, non-Hispanic Blacks, non-Hispanic Whites, Hispanics, and other races), family poverty income ratio (PIR), smoking, hypertension, diabetes, chronic bronchitis, and CVDs. Among these, smoking, hypertension, diabetes, chronic bronchitis, and CVDs were ascertained by self-report.
Statistical analysis
Data processing was performed using R software (version 4.3.2). NHANES uses a complex sampling design that includes features such as stratification, clustering, and weighting; therefore, direct use of traditional statistical methods may introduce bias. We incorporated clustering (SDMVPSU), stratification (SDMVSTRA), and weighting (WTMEC2YR) variables from NHANES to construct an object (NHANES_design) that reflects the complex sampling design. When analysis functions from packages such as ‘survey’ and ‘rms’ were applied, passing this object automatically accounts for the sampling design and ensures valid statistical inference. For categorical variables in the complex survey sample, the Wilcoxon rank-sum test coupled with χ2 test were used. Continuous variables were presented as mean with 95% confidence intervals (CIs), whereas categorical variables were presented as percentages and 95% CIs. Multivariable logistic regression analyses were performed to evaluate the association between CMI and high-risk hs-CRP, with results reported as odds ratios (ORs) and 95% CIs. Three models were constructed: (a) an unadjusted model; (b) a model adjusted for sex, educational level, and hypertension; and (c) a model adjusted for age, sex, income level, educational level, total cholesterol, hypertension, diabetes, and chronic bronchitis. Subgroup analyses were conducted by sex, age, race/ethnicity, and hypertension status. Restricted cubic spline (RCS) curves were used to flexibly characterize and graphically represent the nonlinear association between CMI and hs-CRP. We used the rcsplot function for RCS analysis, with node positions set by default according to quantiles as 0.05, 0.35, 0.65, and 0.95.
Results
Baseline characteristics
According to Table 1, among the 3970 participants, 1973 (49%) were female, the mean age was 48.0 years, and the majority were non-Hispanic Whites (65%). A total of 2046 participants (54%) were married, and 3129 had an educational level of high school or above. Additionally, 1220 participants (30.7%) had high-risk hs-CRP levels (>3.0 mg/L). Compared with participants with higher CMI scores, those with lower CMI scores were younger, more likely to be female, had higher educational levels and income, and were less likely to smoke. In terms of chronic diseases, participants with lower CMI scores had a lower probability of hypertension, diabetes, and heart failure.
Baseline characteristics of the study participants grouped according to CMI (weighted).

Data are presented as median (IQR) or n (%).
CMI: cardiometabolic index; HDL-C: high-density lipoprotein cholesterol; hs-CRP: high-sensitivity C-reactive protein; LDL-C: low-density lipoprotein cholesterol; PIR: family poverty income ratio; TC: total cholesterol; BMI: body mass index; TG: triglycerides; IQR: interquartile range.
Association between CMI and hs-CRP
The CMI score demonstrated a positive association with high-risk hs-CRP. To evaluate this association, we used three weighted multivariable logistic regression models (Table 2). The covariates adjusted for in each model are detailed in the Materials and methods section. When CMI was modeled as a continuous variable, all three models confirmed a positive association. In Models 1, 2, and 3, each one-unit increase in CMI was associated with a 9%, 10%, and 11% higher risk of high-risk hs-CRP, respectively. In addition, a sensitivity analysis assessed the association between categorical CMI and high-risk hs-CRP. When CMI was analyzed as a categorical variable, it remained positively associated with high-risk hs-CRP. In Model 2, compared with patients in the T1 CMI group, those in the high CMI group were more likely to have high-risk hs-CRP (OR = 1.34, 95% CI: 1.27–1.40; p < 0.001).
Association between CMI and high-risk hs-CRP (weighted).

Model 1: unadjusted model; Model 2: model adjusted for sex, educational level, and hypertension; and Model 3: model adjusted for age, sex, income level, educational level, total cholesterol, hypertension, diabetes, and chronic bronchitis.
CMI: cardiometabolic index; CI: confidence interval; hs-CRP: high-sensitivity C-reactive protein; OR: odds ratio.
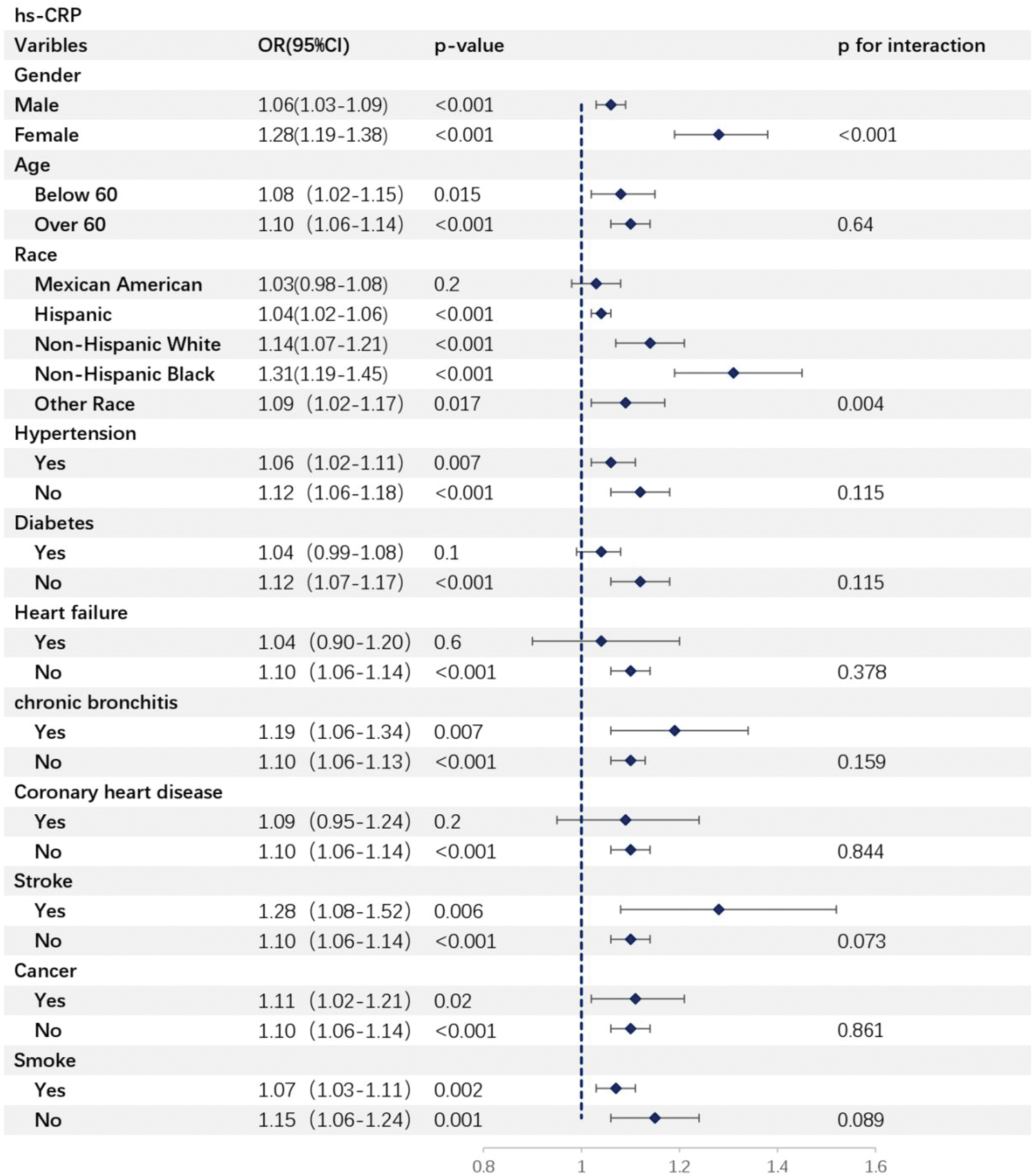
Subgroup analysis
Subgroup analyses were conducted to assess the relationship between CMI and high-risk hs-CRP, as shown in Table 3. In most subgroups, higher CMI scores were positively associated with high-risk hs-CRP. Significant interaction effects were observed for sex (p < 0.001) and race/ethnicity (p = 0.004), with the largest ORs observed among females and non-Hispanic Black participants, respectively.
Subgroups analysis for the association between CMI and high-risk hs-CRP.

CMI: cardiometabolic index; CI: confidence interval; hs-CRP: high-sensitivity C-reactive protein; OR: odds ratio.
RCS analysis
RCS regression analysis was performed to evaluate the nonlinear association between CMI and high-risk hs-CRP. As shown in Figure 2, a clear nonlinear relationship persisted (nonlinearity p < 0.001) even after adjustment for covariates. In the RCS curve, high-risk hs-CRP appeared to plateau when CMI reached 1.21. This phenomenon may be related to the threshold for high-risk hs-CRP being set at 3.0; if a higher threshold was used, the association between the two variables might become more pronounced.

RCS regression analyses of the association between CMI and high-risk hs-CRP. Models were adjusted for sex, educational level, and hypertension. CMI: cardiometabolic index; hs-CRP: high-sensitivity C-reactive protein; RCS: restricted cubic spline.
Discussion
This cross-sectional study, using data from the NHANES 2015–2018 cycles, confirmed an independent association between CMI and high-risk hs-CRP among US adults, which remained robust after multivariable adjustment. Moreover, this association was stable across subgroups stratified by age, sex, ethnicity, and hypertension status.
CMI is a novel indicator obtained by integrating the triglyceride–glucose index and BMI. Originally designed as a marker for predicting the risk of DM, 14 CMI has been shown to be significantly associated with hyperglycemia 14 and to have a stronger association with metabolically obese status than the traditional anthropometric indicators. 15 Subsequent investigations have further examined the relationship between CMI and diabetes. Multiple studies have confirmed the association of CMI with insulin resistance, prediabetes, and diabetes 22 as well as extended its association to CVD and other diseases. Satapathy et al. 23 conducted a meta-analysis demonstrating a significant association between elevated CMI and the risk of stroke, with a pooled relative risk (RR) of 1.66 (95% CI: 1.25–2.20). Yan et al. 16 reported that CMI is significantly and positively correlated with nonalcoholic fatty liver disease (NAFLD) (adjusted OR = 1.44, 95% CI: 1.44–1.45) and liver fibrosis (adjusted OR = 1.84, 95% CI 1.84–1.85). Liu et al. 24 found that CMI was positively correlated with all-cause mortality (hazard ratio (HR) = 1.03, 95% CI: 1.02–1.04), cardiovascular mortality (HR = 1.04, 95% CI: 1.03–1.05), and cancer mortality (HR = 1.03, 95% CI: 1.02–1.05) among the general population. Xu et al. 25 demonstrated that elevated CMI was positively associated with all-cause mortality among older individuals (HR = 1.11, 95% CI: 1.01–1.21), and this relationship may be partially mediated through inflammatory pathways. According to multiple studies, CMI is linked to various systemic diseases, highlighting its association with poorer prognosis.
Traditional BMI has limitations in assessing the risk of obesity-related diseases because it cannot accurately distinguish fat distribution, particularly between visceral and subcutaneous fat. In contrast, CMI can identify obesity in young people with normal BMI, which may contribute to the development of new-onset chronic diseases.15,26 Visceral fat accumulation exhibits a strong association with cardiometabolic diseases. Visceral adipose tissue is more likely to induce inflammatory responses than subcutaneous adipose tissue. 27 Chronic low-grade inflammation commonly coexists with obesity and metabolic disorders. Excessive expansion of visceral adipose tissue leads to adipocyte hypertrophy. Increased activity of lipolysis-related enzymes in these hypertrophied adipocytes enhances lipolysis, resulting in elevated concentrations of free fatty acids (FFA). High concentrations of FFA can activate the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway in adipocytes and adipose tissue macrophages, thereby promoting the secretion of pro-inflammatory cytokines such as interleukin (IL) 6 (IL-6), tumor necrosis factor-α (TNF-α), and monocyte chemoattractant protein-1 (MCP-1) and initiating the inflammatory response. 28 Inflammatory processes are activated in the early stages of fat expansion and during chronic obesity, biasing the immune system toward a pro-inflammatory phenotype, which exerts a substantial influence on overall mortality and the risk of myocardial infarction.29,30
CRP is an important biomarker in cardiovascular risk assessment and one of the therapeutic targets for CVDs. Studies have shown that, in patients receiving statin therapy, inflammation assessed with hs-CRP can better predict the risk of future major adverse cardiovascular events (MACE) and death than low-density lipoprotein cholesterol (LDL-C). 9 Furthermore, studies suggest that statins can reduce the rate of MACE in healthy individuals with elevated hs-CRP but without hyperlipidemia. 31 In patients with a history of myocardial infarction and hs-CRP >2 mg/L, Canakinumab, an IL-1β–targeting human monoclonal antibody, has been shown to markedly reduce circulating IL-6 and hs-CRP levels. Moreover, it is associated with a significant reduction in stroke, nonfatal myocardial infarction, cardiovascular death, and other MACE. 32 Colchicine is a compound with potent anti-inflammatory properties. A once-daily 0.5 mg dose of colchicine is known to reduce ischemic atherosclerotic cardiovascular disease (ASCVD) events by 23%, lower lifetime patient costs by 69%, and increase quality-adjusted life-years from 8.82 to 11.68.33,34 Glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs) were initially used for the treatment of patients with T2DM. They have now been approved by the Food and Drug administration (FDA) for weight management and have gained an increasingly strong position in the antiobesity drug market. 35 Additionally, GLP-1RAs exhibit cardiovascular and neuroprotective effects and are promising agents for limiting inflammation in human visceral fat cells.30,36 Incretin-based drugs dominate the current antiobesity pharmaceutical market; however, 1 year after discontinuation, approximately two-thirds of the weight lost by many people is regained. 37 Novel antiobesity therapies, including dual- and triple-incretin–based co-agonists, have achieved highly promising results in terms of weight loss in patients with obesity, often comparable to those following bariatric surgical procedures. 38
CMI offers the advantages of simple operation and low detection cost, making it a promising tool for early screening of metabolic risk in clinical practice. Clinical research data indicate that regular physical activity (with a cumulative duration of ≥330 min per week) can significantly reduce CMI levels. The reduction in CMI in the “long-term regular activity group” is significantly greater than that in the occasional activity group, highlighting the sensitivity of CMI to lifestyle interventions. 39 Based on this, CMI can serve as an objective evaluation indicator before and after the formulation of exercise prescriptions and the implementation of dietary interventions, helping clinicians dynamically monitor intervention effects and providing patients with visualized feedback on health improvement.
Incorporating CMI into routine health check-up items and chronic metabolic disease management processes can enable the “early detection and early intervention” of metabolically related diseases. By formulating precise prevention plans in combination with individual risk characteristics, this approach has important clinical value for reducing the overall incidence of cardiovascular and metabolic diseases in the population.
Limitations
This study has several limitations. First, our study is observational in nature; therefore, causal inferences between CMI and hs-CRP cannot be made. Further clinical trials are needed to verify these findings. Second, due to the lack of data on other cardiovascular biomarkers in the NHANES 2015–2018 dataset, additional analyses could not be performed. Third, diagnoses of hypertension, diabetes, heart failure, coronary heart disease, chronic bronchitis, stroke, and cancer were based on participant questionnaires, which may introduce recall bias and affect diagnostic accuracy. Finally, due to the lack of potential confounding factors such as dietary patterns and physical activity intensity, residual confounding may be present.
Conclusion
This study confirms an independent association between CMI and high-risk hs-CRP in US adults, which persists after multivariable adjustment. These findings support CMI as a useful marker linking cardiometabolic status to chronic inflammation, enhancing cardiovascular risk assessment beyond traditional indices. These findings warrant further studies to validate causal relationships and to explore CMI-guided preventive strategies.
Footnotes
Acknowledgments
We appreciate the personnel who contributed to the NHANES data used in this study.
Author contributions
Yuhao Yan designed the study, analyzed the data, and drafted the manuscript; Yong Tang critically revised the manuscript and approved the final version.
Declaration of conflicting interests
The authors report no conflicts of interest related to this work.
Ethical approval and consent to participate
The NHANES research protocols were approved by the NCHS Research Ethics Review Board, and all participants provided written informed consent.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.