Abstract
Both catheter-directed thrombolysis (CDT) and ultrasound-assisted thrombolysis (USAT) are novel treatment modalities for patients presenting with acute pulmonary embolism (PE). The objective of this study was to compare clinical and quality-of-life (QOL) outcomes for patients undergoing either treatment modality. We retrospectively studied 70 consecutive patients treated with either CDT or USAT over 3 years at a multicenter health system. The primary clinical efficacy endpoint was right ventricular systolic pressure (RVSP) reduction post-procedurally. Safety endpoints were mortality and bleeding incidents based on Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Arteries (GUSTO) criteria. Long-term QOL was assessed using the 36-Item Short-Form Health Survey (SF-36) via phone interview. Thirty-seven patients (53%) in our study underwent USAT and 33 (47%) patients were treated with conventional CDT. Among all patients studied, 96% had echocardiographic evidence for right ventricular strain on admission. Mean RVSP decreased by 18 ± 13 mmHg in the USAT group post-procedurally as compared to 14 ± 16 mmHg in the CDT group, without significant difference between groups (p = 0.31). Rates of moderate and severe bleeding were largely identical between USAT and CDT groups (USAT: 3%; CDT: 0%; p = 0.09). There was no death in either group during admission. At long-term follow-up, there was no significant difference in QOL between both treatment modalities in all eight functional domains of SF-36. Our retrospective study demonstrated using USAT over conventional CDT for acute submassive or massive PE did not yield additional clinical, safety, or long-term QOL benefit.
Keywords
Introduction
Pulmonary embolism (PE) causes an estimated 50,000–100,000 deaths annually in the USA.1,2 Many of those who survive have persistent symptoms that negatively impact quality of life (QOL).3–5 The US Food and Drug Administration’s (FDA) approved treatments for PE include oral anticoagulants, systemic thrombolysis, surgical thrombectomy, and catheter-directed thrombolysis (CDT). 6 PE severity typically guides the utilization of these therapies. Categorization of PE severity, defined as massive or high-risk, submassive or intermediate-risk, or low-risk PE, is based upon hemodynamic status and markers of right ventricular strain, as demonstrated by clinical exam, imaging, and laboratory cardiac enzymes. 7 While the majority of PE patients present with minimal symptoms and are safely treated with anticoagulation alone, patients exhibiting right ventricular (RV) compromise or hemodynamic instability may benefit from acute intervention to decrease clot burden, in an effort to reduce RV afterload.7,8
Catheter-directed thrombolytic therapy has in recent years emerged as a tool for the treatment of acute submassive and massive PE patients. 9 The American Heart Association endorsed CDT as a viable alternative to systemic thrombolysis, particularly in patients with high bleeding risks.6,7 The EkoSonic Endovascular System (EKOS Corp., Bothell, WA, USA) is an ultrasound-assisted CDT (USAT) system that is purported to accelerate the fibrinolytic process via the application of ultrasound energy.10–12 Several observational series and single-arm prospective analyses have shown USAT to acutely decrease clot burden and improve RV size and function.13,14 However, whether USAT is superior to conventional CDT in PE patients is still unknown. We therefore sought to compare the efficacy and safety for USAT versus conventional CDT in patients admitted with massive and submassive PE. In addition, as post-PE QOL has never been systemically studied in patients treated with catheter-directed therapies, our study also aimed to compare functional impairment between patients in both cohorts at long-term follow-up.
Methods
Study design and population
We retrospectively reviewed the records of patients with acute PE who underwent CDT or USAT from January 2014 to January 2017 at four institutions (North Shore University Hospital, Long Island Jewish Medical Center, Lenox Hill Hospital, Southside Hospital) within a single health system. Institutional Review Board approval was obtained at all study centers and written informed consent was obtained prior to every procedure. CDT was implemented for massive PE in patients with a high bleeding risk and for submassive PE in patients with high-risk features at the discretion of the treating clinicians. These high-risk features were defined as fulfilling one or any combination of the following criteria: (a) persistent dyspnea or hypoxia (SaO2 < 88%) on exam requiring oxygen supplementation; (b) persistent tachycardia (heart rate > 110 bpm) despite therapeutic anticoagulation; (c) worsening symptoms related to PE (e.g., uncontrolled pleuritic chest pain); and (d) PE progression signs (e.g., worsening RV strain, defined by RV dilatation or dysfunction) demonstrated by any imaging modality after full anticoagulation within 24 hours of admission. No patients were excluded from the study.
Medical records were reviewed and clinical data including symptoms, risk factors for venous thromboembolism, treatment details, and hospital length-of-stay (LOS) were recorded for 70 consecutive patients. Additionally, patients were contacted via telephone 1 year after discharge to complete a QOL questionnaire, which was obtained after informed verbal consent.
Device and interventional procedures
The choice of either conventional CDT or USAT and the procedural technique used for the delivery of the chosen catheter system were based on individual physician preference and discretion. CDT was performed using Cragg-McNamara (Boston Scientific, Marlborough, MA, USA) catheters and USAT was performed using the EkoSonic (EKOS) endovascular system.
Prior to the procedure, all except two patients received systemic anticoagulation with an initial intravenous bolus of unfractionated heparin of 80 units/kg followed by a weight-based infusion to target a goal activated partial thromboplastin time (aPTT) of 60–90 seconds. Information was not available in one patient and the other had previously confirmed history of heparin-induced thrombocytopenia. All procedures were performed in a standard angiography suite using fluoroscopic imaging guidance. Venous access was obtained from a femoral approach with ultrasound guidance. Areas of heavy thrombus burden were identified via pre-procedural CT angiography and intra-procedural pulmonary angiography. These areas were established as ideal targets for catheter placement. Two infusion catheters were placed when bilateral emboli were present. Prior to thrombolytic initiation, baseline pulmonary arterial pressures were measured via the inserted catheter.
A loading dose of 2–5 mg tissue plasminogen activator (tPA) through each catheter was administered at the operators’ discretion following catheter placement. Subsequently, tPA was infused at a rate of 0.5–1 mg/hour through each catheter for a duration of 12–24 hours, unless bleeding risk or complications mandated a shorter infusion period. During tPA infusion, intravenous unfractionated heparin was continued at a reduced rate ranging from 250 to 400 units/hour. The ultrasound system, when applicable, was activated prior to departure from the catheterization suite. All patients were admitted to an intensive care unit for close monitoring throughout the infusion period. Fibrinogen levels and aPTT were recorded at established intervals throughout the treatment period. Low fibrinogen levels (< 150 mg/dL) prompted a decrease in the thrombolytic infusion rate. The low-dose heparin infusion was titrated in increments or decrements of 50 units/hour to target a reduced goal aPTT of 30–40 seconds. After completion of thrombolytic administration, the infusion catheters were removed, and standard full-dose heparin infusion was resumed. All patients were then transitioned to oral anticoagulation therapy prior to discharge.
Outcome measures
The primary efficacy endpoint was change in right ventricular systolic pressure (RVSP), which was estimated by pre- and post-procedural echocardiography performed at diagnosis and at 24–48 hours after thrombolytic cessation, respectively. Secondary efficacy endpoints included change in heart rate, oxygen supplementation requirements, and RV strain, as well as overall inpatient LOS. Additional procedural-related characteristics including thrombolytic dose and duration of infusion were also captured.
The primary safety endpoint was the incidence of moderate and severe bleeding as defined by GUSTO (Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries) criteria. Severe bleeding was defined as intracranial hemorrhage or bleeding resulting in hemodynamic compromise requiring intervention. Moderate bleeding was defined as bleeding requiring transfusion but not associated with hemodynamic compromise. 15
QOL among patients treated by either intervention was also assessed. Post-discharge follow-up was accomplished via telephone interview between February and March 2018 by trained research personnel. QOL was assessed using the Short-Form Health Survey (SF-36). The SF-36 is a generic measure of QOL that has been widely validated for use across a range of clinical settings and patient populations, including the post-discharge assessment of PE patients treated with a variety of PE therapies. 16 The survey measures QOL across eight emotional and physical domains: physical functioning, role limitations due to physical health, role limitations due to emotional problems, energy/fatigue, emotional well-being, social functioning, pain, and general health. The score in each domain ranges from 0 to 100, with a higher score indicating a better QOL. 17
Statistical analysis
Statistical analysis was performed by means of Pearson chi-squared test or Fisher’s exact test in categorical variables, pertaining mostly to patient demographics and clinical outcomes. Within-group comparisons of continuous variables (i.e., pre- and post-procedural clinical data) were performed with the paired-samples t-test. Between-group continuous variables were compared with the independent-sample t-test or Wilcoxon rank sum test. Continuous data are presented as mean ± SD and sample proportions are reported as percentages. The p-values were reported as two-sided, and a p-value < 0.05 was considered statistically significant. All statistical analyses were performed with the use of SPSS software, Version 23.0 (IBM Corp., Armonk, NY, USA).
Results
Baseline demographics and clinical characteristics
A total of 70 patients (age = 61.8 ± 13.2 years, 52.9% male) were included during the study period; 33 (47.1%) were treated with CDT and 37 (52.9%) with USAT. There were no significant differences in baseline demographics between two groups (Table 1). The duration of symptoms prior to diagnosis was 6 days (IQR 1–7) in the CDT group and 4 days (IQR 1–7) in the USAT group (p = 0.35). PE severity on presentation was comparable, with a similar percentage of submassive (75.8% vs 91.9%, p = 0.69) and massive PE (18.2% vs 13.5%, p = 0.46) patients noted in the CDT and USAT groups, respectively. Additional clinical characteristics including cardiac biomarker levels and initial echocardiography findings (Table 2) did not differ between the two groups. All patients had bilateral PE.
Demographics and clinical history of patients who underwent CDT and USAT.
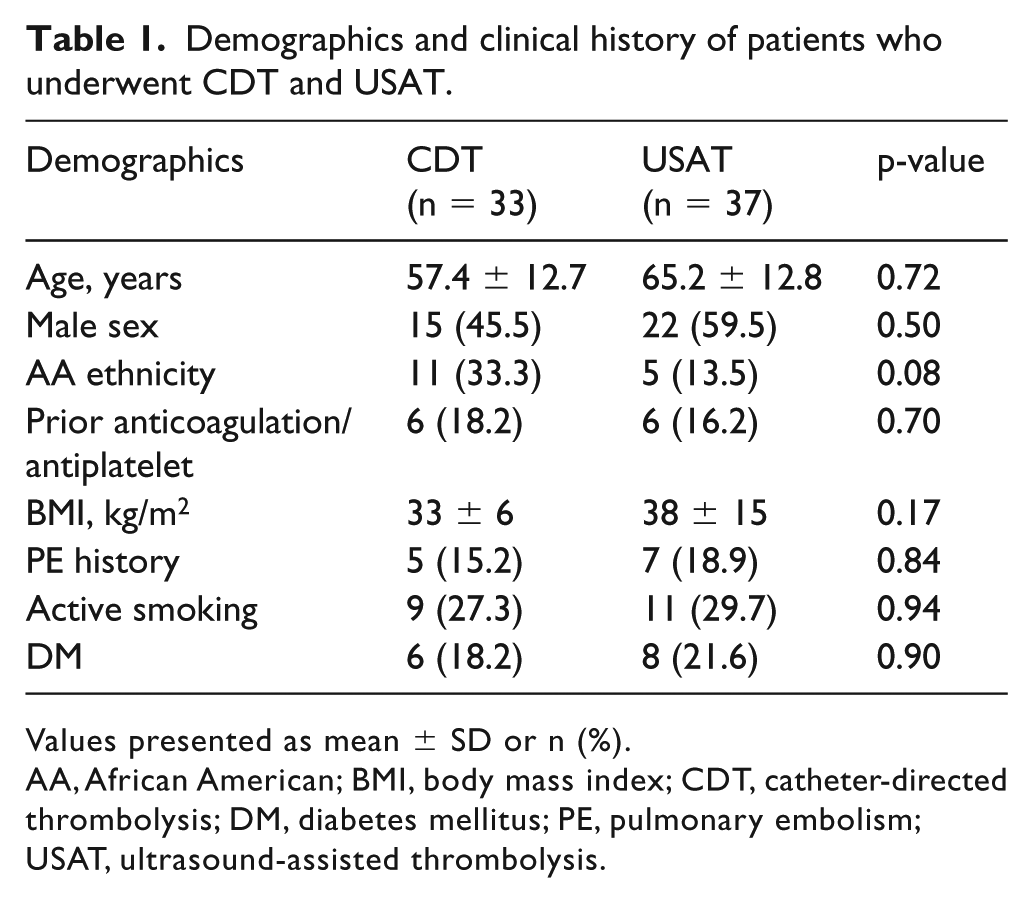
Values presented as mean ± SD or n (%).
AA, African American; BMI, body mass index; CDT, catheter-directed thrombolysis; DM, diabetes mellitus; PE, pulmonary embolism; USAT, ultrasound-assisted thrombolysis.
Clinical presentation of patients who underwent CDT and USAT.
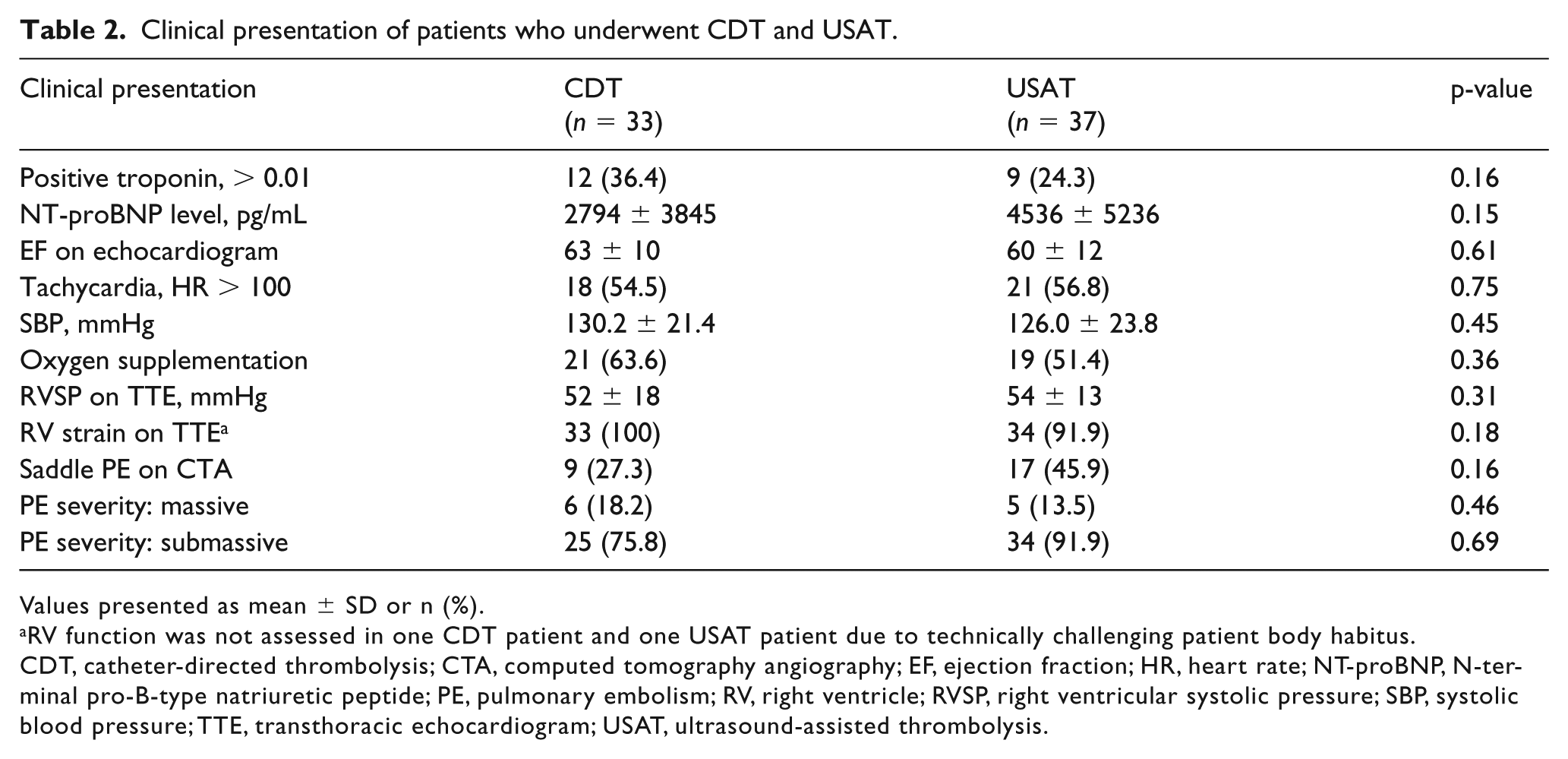
Values presented as mean ± SD or n (%).
RV function was not assessed in one CDT patient and one USAT patient due to technically challenging patient body habitus.
CDT, catheter-directed thrombolysis; CTA, computed tomography angiography; EF, ejection fraction; HR, heart rate; NT-proBNP, N-terminal pro-B-type natriuretic peptide; PE, pulmonary embolism; RV, right ventricle; RVSP, right ventricular systolic pressure; SBP, systolic blood pressure; TTE, transthoracic echocardiogram; USAT, ultrasound-assisted thrombolysis.
Procedural details
Pre-procedural unfractionated heparin was initiated in 32 (97.0%) and 36 (97.3%) patients in the CDT and USAT groups, respectively (p = 0.31). Successful placement of the infusion catheters was achieved in all patients. There were no catheter-related complications. All patients received two infusion catheters (one in each side). The intra-procedural, catheter-derived, pulmonary arterial systolic pressures were similar in both groups (CDT 48 ± 15 vs 52 ± 13 mmHg, p = 0.45) (Table 3). Overall doses of tPA (24.7 ± 12.2 vs 25.1 ± 6.3 mg, p = 0.09) and infusion duration (13.3 ± 3.9 vs 13.3 ± 3.2 hours, p = 0.31) were similar between the CDT and USAT groups, respectively.
Comparison of procedural details in patients who underwent CDT and USAT.
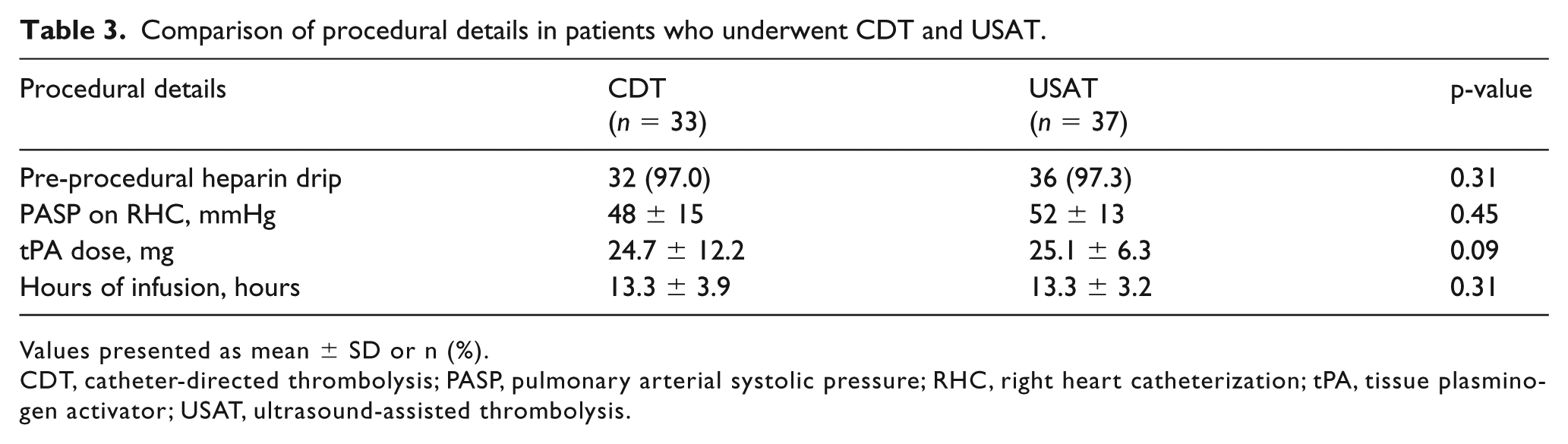
Values presented as mean ± SD or n (%).
CDT, catheter-directed thrombolysis; PASP, pulmonary arterial systolic pressure; RHC, right heart catheterization; tPA, tissue plasminogen activator; USAT, ultrasound-assisted thrombolysis.
Efficacy outcomes
The primary efficacy endpoint was similar in both groups: ΔRVSPs are −14 ± 16 mmHg and −18 ± 13 mmHg in the CDT and USAT groups, respectively (p = 0.31; Figure 1). Among all patients studied, 97% had echocardiographic evidence for RV strain on admission (CDT 100% vs USAT 94.4%, p = 0.18). At 24–48 hours post-procedure, repeat echocardiography demonstrated that 64.3% in the CDT group and 59.3% of patients in the USAT group continued to exhibit signs of persistent RV strain (p = 0.70). The percentage of patients who remained tachycardic (CDT 9.1% vs USAT 5.9%, p = 0.62) and required oxygen supplementation (CDT 27.3% vs USAT 17.6%, p = 0.35), despite undergoing catheterization-based therapies, were similar in both groups. Lastly, the overall LOS was comparable in the two groups (CDT 7.7 days, IQR 3–6 vs USAT 6.5 days, IQR 3–7; p = 0.54).
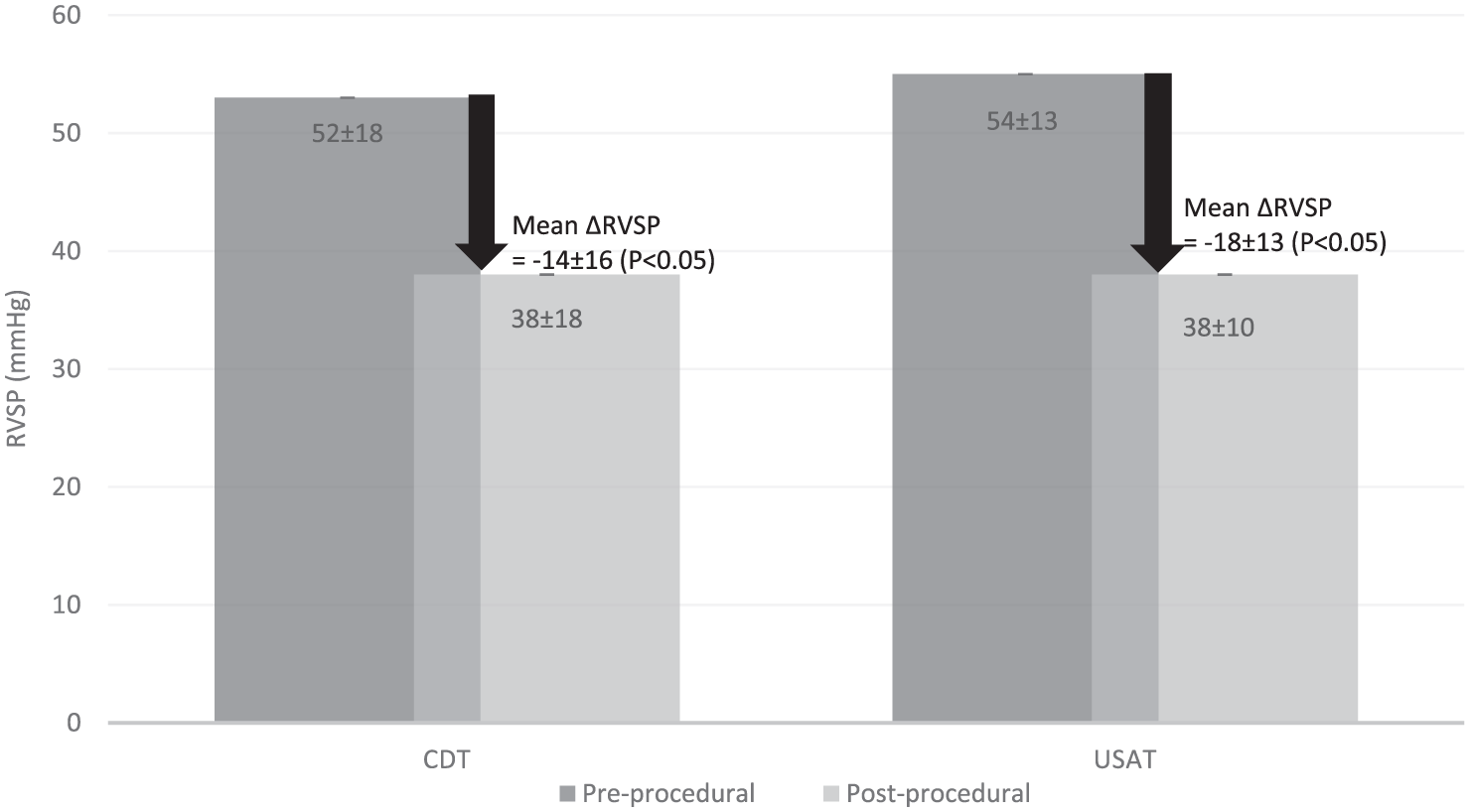
Comparison of ΔRVSP between catheter groups.
Safety outcomes
There were no deaths noted until hospital discharge in both groups. No significant bleeding events were reported in the CDT group (0%). However, one GUSTO moderate bleeding event was noted in the USAT group (3%, p = 0.09) in the form of a retroperitoneal bleed requiring blood transfusion.
Quality of life
Overall, 47 (67.1%) patients completed the SF-36 questionnaire (age 62.6 ± 12.9 years, 15.1% African American, 48.9% male). All QOL-related post-discharge follow-up was accomplished via telephone between February and March 2018. Baseline characteristics, disease presentation, and therapeutic and safety outcomes conferred by catheter-based therapies did not differ between subjects who did and those who did not complete the questionnaire (online Supplementary Table 1S). Of patients who responded to the survey, the average times to follow-up were 16.6 ± 7.1 months in the CDT group (n = 22) and 17.9 ± 11.4 months in the USAT group (n = 25) (p = 0.58). Subsequent analysis demonstrated no difference in the scores across all studied QOL domains, including physical function, role limitations due to physical health, role limitations due to emotional health, energy/fatigue, emotional well-being, social role functioning, bodily pain, and general health. Individual scores and their inter-group correlation are illustrated in Figure 2.
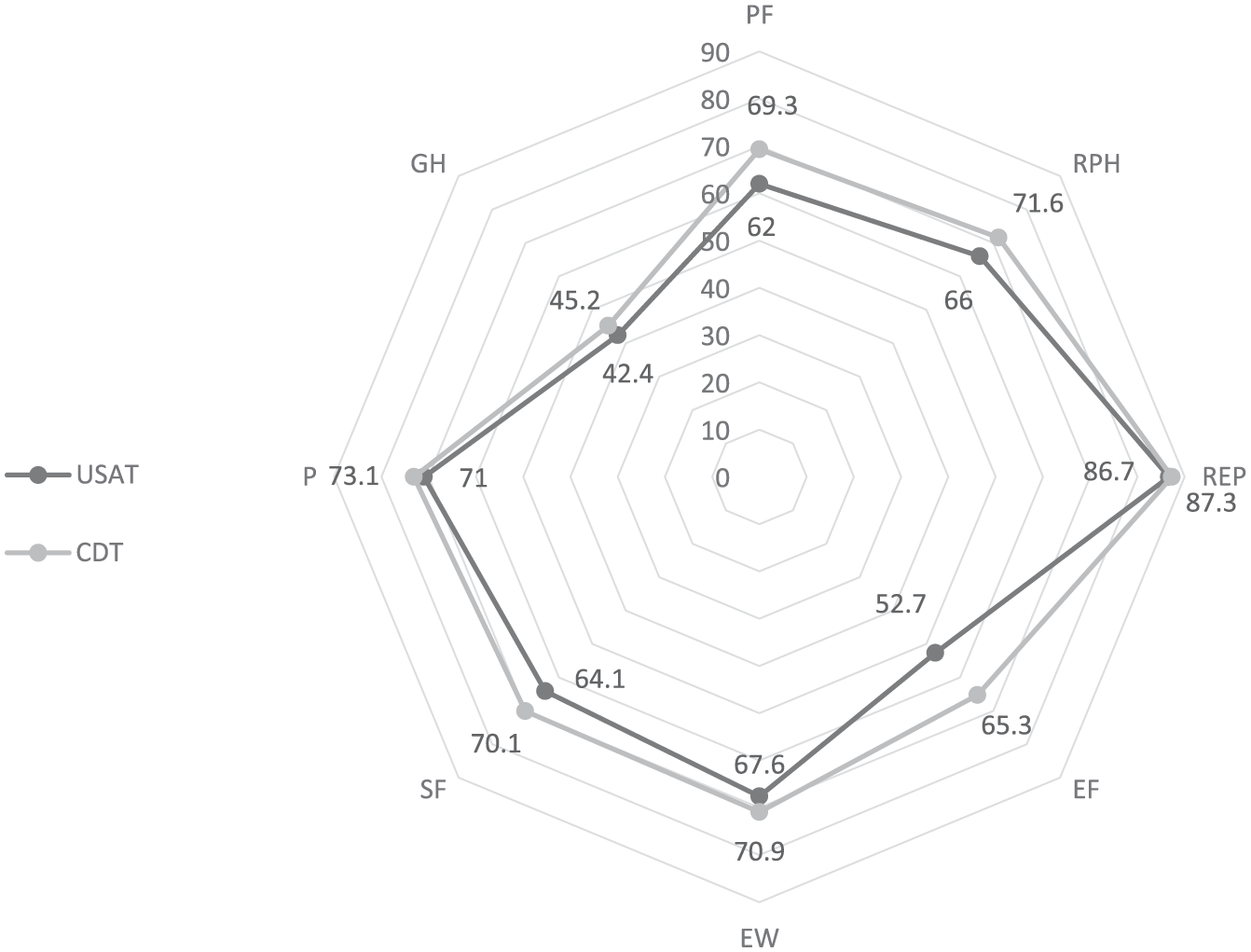
USAT (n = 25) versus CDT (n = 22) QOL comparison.
Discussion
In this study, comparing CDT and USAT in the treatment of acute, intermediate and high-risk PE, we demonstrated no difference between the two treatment groups in terms of clinical efficacy, safety and QOL outcomes. CDT and USAT achieved similar early reductions in RVSP, RV strain, tachycardia and oxygenation requirements. Moreover, the safety profile in both therapies is comparable, with very few adverse bleeding incidents. No deaths were noted in either group at the time of discharge. Thus, this study failed to identify any hemodynamic, physiologic, mortality or safety advantage potentially attributable to the ultrasound component of USAT.
Very few comparisons between USAT and CDT have been performed to date. In a study of 33 patients with massive PE reported by Lin et al. in 2009, USAT with the EKOS platform was compared with CDT delivered via a straight UniFuse catheter (AngioDynamics, Latham, NY, USA). 18 Similar mortality rates and similar reductions in Miller PE scores were noted in the CDT and USAT groups (14.2% vs 9.1%, p > 0.05). Bleeding rates, however, were significantly lower in the CDT group (CDT: 0%, USAT 21.4%, p = 0.02). 18 The impact of catheter-directed therapies on RVSP was not described. More recently, the Pulmonary Embolism Response to Fragmentation, Embolectomy, and Catheter Thrombolysis (PERFECT) registry examined 101 patients undergoing CDT for acute PE. 19 Subgroup analysis demonstrated no significant difference in thrombolytic infusion dosage (CDT 25.63 vs USAT 30.27 mg, p > 0.05), change in RVSP (CDT −14.02 vs USAT −13.76 mmHg, p > 0.05), or major bleeding events. 19 These findings were further corroborated by Liang et al. in a retrospective study of 63 PE patients. Similar complication rates were noted in CDT and USAT cohorts (CDT 11% vs USAT 13.9%, p = 0.8). 20 Lastly, Graif et al., in 2017, compared CDT and USAT in a single-center retrospective study of 60 patients with either massive or submassive PE. Again, similar reductions in RVSP (CDT −11.1 vs USAT −10.1 mmHg, p = 0.69) and comparable complication rates (CDT 5.6% vs USAT 20.7%, p = 0.07) were demonstrated in the two groups. 21 It is worth noting that the USAT group in Graif et al. underwent an overall lower dose of tPA (30.4 mg) compared with the CDT group (27.1 mg). However, this difference was deemed not significant statistically (p = 0.075). In summary, four published studies to date have failed to demonstrate any significant outcome difference between CDT and USAT. The Standard vs Ultrasound-assisted Catheter Thrombolysis For Submassive Pulmonary Embolism (SUNSET sPE) trial is an ongoing, randomized, head-to-head clinical trial that should establish whether USAT confers any additional benefit compared with USAT alone. 22
Consistent with results summarized above in the current published literature, our study re-demonstrated that CDT and USAT are, from a clinical standpoint, highly comparable modalities. Specifically, the physiologic benefits, procedural requirements for thrombolytic dosage, improvements in clinical status, bleeding rates, and mortality rates of CDT and USAT were not statistically different.
Strengths and limitations
The strength of this study is the retrospective collection of comprehensive data from real-world practice in multiple centers within a busy regional health-care system. Additionally, to our knowledge, this study is the first to systematically evaluate QOL in patients undergoing catheter-directed therapies for PE. We performed a functional impairment assessment to identify any difference in QOL between CDT and USAT at long-term follow-up. Similar to the other outcomes described in this study, there was no significant difference in all eight domains assessed by the standardized SF-36 health survey matched between both cohorts up to 4 years after their initial presentation.
The study is subject to several limitations. First, the retrospective, non-randomized nature of this study subjects it to the inherent weakness of such analyses. Specifically, unmeasured bias could have affected the choice of one therapy over the other. However, it is notable that we found no baseline clinical differences among patients treated with either therapy. Furthermore, although our study is first in assessing long-term QOL among PE patients treated with catheter-directed therapies, the retrospective design unavoidably conferred sizable variation in the follow-up period. In theory, this could have diminished the accuracy of our long-term outcome assessment. Second, despite this current study being large relative to previous publications, the sample size remains small. Finally, this study did not assess cardiopulmonary dysfunction, change in RVSP, or RV function at long-term follow-up. Thus, the QOL measure could not be correlated with any objective measure of physiological features. While such data would have been interesting, recent study has suggested that the post-PE syndrome and post-PE QOL are independent of RV systolic function, RVSP, or other measures of cardiopulmonary fitness. 23
Conclusion
CDT with ultrasound-assisted energy appeared to not confer any additional clinical, safety, or QOL advantages for intermediate and high-risk PE patients when compared to catheter-directed lysis without ultrasound assistance. Given our limitations, our study is hypothesis-generating and a larger, randomized study would be the gold standard to definitively compare these modalities. However, until the time when such a study becomes available, our results, in combination with the additional equipment costs of USAT, create a compelling argument for performing CDT without ultrasound assistance.
Supplemental Material
10.1177_1358863X19838334_Supplementary_table – Supplemental material for Ultrasound-assisted versus conventional catheter-directed thrombolysis for acute pulmonary embolism: A multicenter comparison of patient-centered outcomes
Supplemental material, 10.1177_1358863X19838334_Supplementary_table for Ultrasound-assisted versus conventional catheter-directed thrombolysis for acute pulmonary embolism: A multicenter comparison of patient-centered outcomes by Gaurav Rao, Hai Xu, Jason J Wang, Andrew Galmer, Jay Giri, Michael R Jaff, Raghu Kholluri, Joe F Lau, Samy Selim, Ido Weinberg and Mitchell D Weinberg in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jay Giri: PERT Consortium – directors board; AstraZeneca – advisory board; St Jude and ReCor Medical – research funds to institution. Michael R Jaff: Boston Scientific – non-compensated advisor; Embolitech – equity investor; BTG, Medtronic, Philips/Volcano, Sanofi – compensated advisor. Ido Weinberg: Novate Medical – scientific advisory board; BTG – non-compensated advisor. Mitchell D Weinberg: INARI Medical – grant funding; Medtronic – advisory board and consultant; Cardiovascular Systems Inc. – consultant.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
