Abstract
Endothelial dysfunction, evaluated by flow-mediated dilatation (FMD), predicts adverse cardiovascular events in patients with intermittent claudication (IC). IC is an example of repeated ischemia/reperfusion injury that may contribute to the progression of vascular disease by worsening endothelial function, a trigger for acute cardiovascular events. The predictive value of effort-induced endothelial dysfunction for cardiovascular events in patients with IC has not been studied previously. The objective of this study was to assess whether exercise-induced endothelial dysfunction is predictive of adverse cardiovascular outcome in IC. In 44 patients with IC, we measured brachial artery FMD by B-mode ultrasonography at rest and 10 minutes after a maximal treadmill exercise. Treadmill exercise halved the FMD (from 3.5 ± 0.6% to 1.45 ± 0.46%, p < 0.05). After a follow-up period of 85 (72–98) months, a total of 20 major cardiovascular events occurred. In a multivariate analysis, a post-exercise reduction of brachial FMD > 1.3% was predictive for cardiovascular events. Maximal exercise-induced endothelial dysfunction is predictive of cardiovascular events in patients with IC.
Keywords
Introduction
Intermittent claudication (IC), one of the symptomatic stages of peripheral artery disease (PAD), is associated with enhanced incidence of ischemic cardiovascular events and mortality.1–4 Endothelial dysfunction plays a crucial role in the development, progression, and clinical manifestations of PAD.5,6 Indeed, reduced flow-mediated vasodilation (FMD), a marker of endothelial dysfunction, 7 correlates with disease stage, degree of impairment of the pain-free walking distance, and disability. 8 Moreover, in patients with PAD, endothelial dysfunction is predictive of long-term ischemic cardiovascular events9–12 and has an incremental predictive value over the measurement of the ankle–brachial index (ABI), 10 a marker of disease severity and a crucial prognostic index.13,14
Lower limb muscle ischemia triggered by leg exercise, the cause of walking-elicited pain in patients with IC, is associated with a chain of pathologic cellular events known as ischemia/reperfusion injury, which lead to the generation of several mediators, like oxygen radicals and cytokines, noxious to the endothelium. 15 In fact, FMD decreases drastically and endothelial-activation markers increase significantly early after maximal leg exercise in patients with IC.16,17 Not only does endothelial dysfunction inversely correlate with pain-free walking distance in patients with IC, 8 but it is improved by active pharmacologic treatments and by supervised physical exercise, which also ameliorate functional ability.17–19
It is thus conceivable that the degree of the walking-induced drop of endothelial function in patients with IC may correlate with the impairment of cardiovascular homeostasis and that it may identify patients at higher risk of subsequent ischemic cardiovascular events.
The aim of our study was to assess the prognostic value for major adverse cardiovascular events (MACE) of exercise-triggered endothelial dysfunction, measured by the acute drop of brachial FMD and the increase of circulating endothelial activation biomarkers, in a cohort of patients with IC.
Materials and methods
Study population
Forty-four patients (mean age 66 ± 9 years; five females) with IC enrolled in a randomized, placebo-controlled study on the effect of a nitric oxide (NO)-donating agent (NCX 4016) compared with aspirin 20 on the acute endothelial dysfunction induced by exercise in patients with IC, 17 were clinically reassessed at follow-up for up to 11 years. Inclusion criteria were: IC Fontaine stage II stable for over 6 months, a pain-free walking distance (PFWD) of between 50 and 500 m, a resting ABI < 0.85 in the symptomatic leg, and age 18–75 years. Exclusion criteria were: intolerance to aspirin, peptic ulcer, kidney or liver failure, thrombocytopenia (< 100 × 109/L), acute coronary syndrome within the previous 6 months, illnesses interfering with the treadmill test performance, including COPD, and concomitant use of drugs potentially interfering with the end point, such as non-steroidal anti-inflammatory agents, COX-2 inhibitors, anticoagulant or antiplatelet agents, nitrates, pentoxifylline, and PDE-5 inhibitors.
All patients gave their informed, written consent and the study was approved by the local Ethics Committee (CEAS Umbria).
PFWD and maximal walking distance (MWD) were measured at enrollment prior to randomization to the two intervention groups (aspirin, 100 mg/d or NCX 4016, 800 mg bid) by a standardized treadmill test at 3 km/h and a fixed incline of 10%.17,21 MWD was the distance walked until strong/heavy pain (5–6 perceived exertion-triggered leg pain – Borg category ratio-10 scale) occurred. 22 Endothelial function was measured at baseline and 10 minutes after the end of maximal treadmill exercise, while blood sampling was made before (baseline) and immediately after the end of maximal treadmill exercise, in the morning in patients under fasting conditions who were abstaining from smoking and from consuming caffeine-containing food or beverages for at least 12 hours before the study. Subjects were advised to continue their daily activities in the 24 hours before testing. FMD variations occur transiently after acute exercise and a 10-minute time after the end of exercise was therefore chosen for repeated FMD.16,17
After enrollment, all patients were advised to stop smoking and to exercise regularly, and optimal control of risk factors were prescribed.
Follow-up was carried out by direct interview with the patients at routine clinical follow-up visits or by phone interview, or, when not possible, by interview with family members or the family doctor. For patients impossible to reach, the regional health system database of disease-related groups (DRG) for hospitalization or for causes of death was consulted.
Major cardiovascular events (peripheral artery surgical revascularization, lower limb amputation, ischemic stroke, myocardial infarction, cardiovascular death) were defined as previously described.20,23
Endothelial function
B-mode ultrasonography with a 10 MHz linear-array transducer and an Acuson 128 XP/10 System (Acuson, Mountain View, CA, USA) were used to assess FMD according to current guidelines, 7 as previously described.24,25
Ultrasound scan data of an average of four consecutive cardiac cycles were recorded on super VHS video for later analysis. Before FMD assessment, systolic and diastolic blood pressure was measured (sphygmomanometer) and heart rate was obtained from continuously recorded ECG.
Circulating soluble biomarkers
Soluble parameters were measured in serum or in plasma obtained from EDTA-anticoagulated blood. Soluble VCAM-1 (sVCAM-1), soluble ICAM (sICAM), soluble E-selectin (sE-sel), and interleukin-8 (IL-8) were measured by ELISA (Cell Com, Beckman Coulter, Inc. Fullerton, CA, USA). The lower detection limit was 0.74 ng/mL, 100 pg/mL, 50 pg/mL, and 30 pg/mL, respectively, and intra-assay and inter-assay coefficient of variation (CV) were below 10% for all the assays. Soluble P-selectin (sP-sel) was measured by ELISA (R&D Systems, Minneapolis, MN, USA). The lower detection limit was 0.5 ng/mL, intra-assay CV was 4.9–5.6%, and inter-assay CV was 7.9–9.9%. 26
β2-Microglobulin was measured by ELISA (Immundiagnostik, Bensheim, Germany), as described. 27 The detection limit was 0.1 mg/L, intra-assay CV was 1.1–11%, and inter-assay 1–12%.
High-sensitivity C-reactive protein (hs-CRP) was measured in serum using a commercially available kit based on nephelometry (N-Latex CRP II; Dade Behring Marburg Gmbh, Marburg, Germany) as described. 28 The detection limit was 0.17 mg/L, intra-assay CV was 4.7–6.8%, and inter-assay < 20%.
Serum TxB2 levels, the stable metabolite of TxA2, were measured using an enzyme immunoassay (EIA) (Cayman Chemical, Ann Arbor, MI, USA) as described. 29 The detection limit was 5 pg/L, intra-assay CV was 8.2–15.3%, and inter-assay 9.9–12.9%.
Urinary biomarkers
Urinary excretion of 8-iso-prostaglandin F2α (8-iso-PGF2α), a validated index of in vivo lipid peroxidation and oxidative stress, was measured from overnight urine collections. To prevent the formation of 8-iso-PGF2α in vitro, 1 mmol/L of the antioxidant 4-hydroxy-TEMPO (2,2,6,6-tetramethylpiperidine-N-oxyl) was added at collection and urine samples were stored at −80°C for later assay. Samples were analyzed by immunoassay (Cayman Chemical) after chromatographic purification, as previously described. 30 The sensitivity was 3 pg/mL and the measuring range was 7–20 pg/mL. Intra-assay CV was 6.4–20% and inter-assay CV was 9.9–24% and results were expressed as the ratio of urinary metabolite concentration to urinary creatinine concentration. Urinary creatinine was assayed with the Jaffè method using an automatic system (Roche/Hitachi MOD P800; Roche Diagnostics Corporation, Indianapolis, IN, USA).
The NO degradation end-products nitrites and nitrates (NOx) were measured spectrophotometrically using a colorimetric non-enzymatic assay (Oxford Biomedical Research, Oxford, MI, USA) 31 ; sensitivity was 1 pmol/mL.
Urinary cyclic GMP (cGMP) levels were measured by competitive immunoassay (R&D Systems). The detection limit was 3.06 pmol/mL and the measuring range was 2.1–500 pmol/mL. Intra-assay CV was 5.3–8.1% and inter-assay CV was 8.2–9.9%.
Statistical analysis
All data were tested for normal distribution with the D’Agostino Omnibus normality test and are presented as means and SD or median and IQR, as appropriate. Categorical data were analyzed using the χ2 test. Univariate analysis was performed using the paired t-test for the comparison before versus after treadmill exercise, or the unpaired t-test, with Welch correction, as appropriate. A stepwise logistic regression analysis for the evaluation of the odds ratios for the parameter effort-induced levels of variation in relation to occurrence of events at follow-up was performed, setting the insertion probability at p < 0.05 and the removing probability at p > 0.1. Moreover, cumulative event rates were estimated with Kaplan–Meier survival curves, and probability values of free from events survival time were calculated with the log-rank test. Brachial artery FMD variations were divided into values below and above the median decrease of FMD induced by exercise in the overall population. Multivariate Cox proportional hazard regression was used to evaluate the association of FMD drop greater than the median and the risk factor variables with a p-value less than 0.05 in univariate analysis (namely: current smoking, baseline sVCAM-1 levels, baseline sICAM, and soluble P-selectin with MACE). 32 All analyses were performed using MedCalc, version 19.0.6 (MedCalc Software bvba) software.
Results
Clinical characteristics of the enrolled patients are reported in Table 1. Patients were followed-up for a median of 90 months (95% CI: 62 to 120). A total of 20 MACE occurred in 20 patients (45.4%) (four ischemic strokes, three myocardial infarctions, six lower limb artery surgical revascularizations, one lower limb amputation, and six cardiovascular deaths: five from myocardial infarction and one from abdominal aneurism rupture).
Baseline characteristics of the study population.
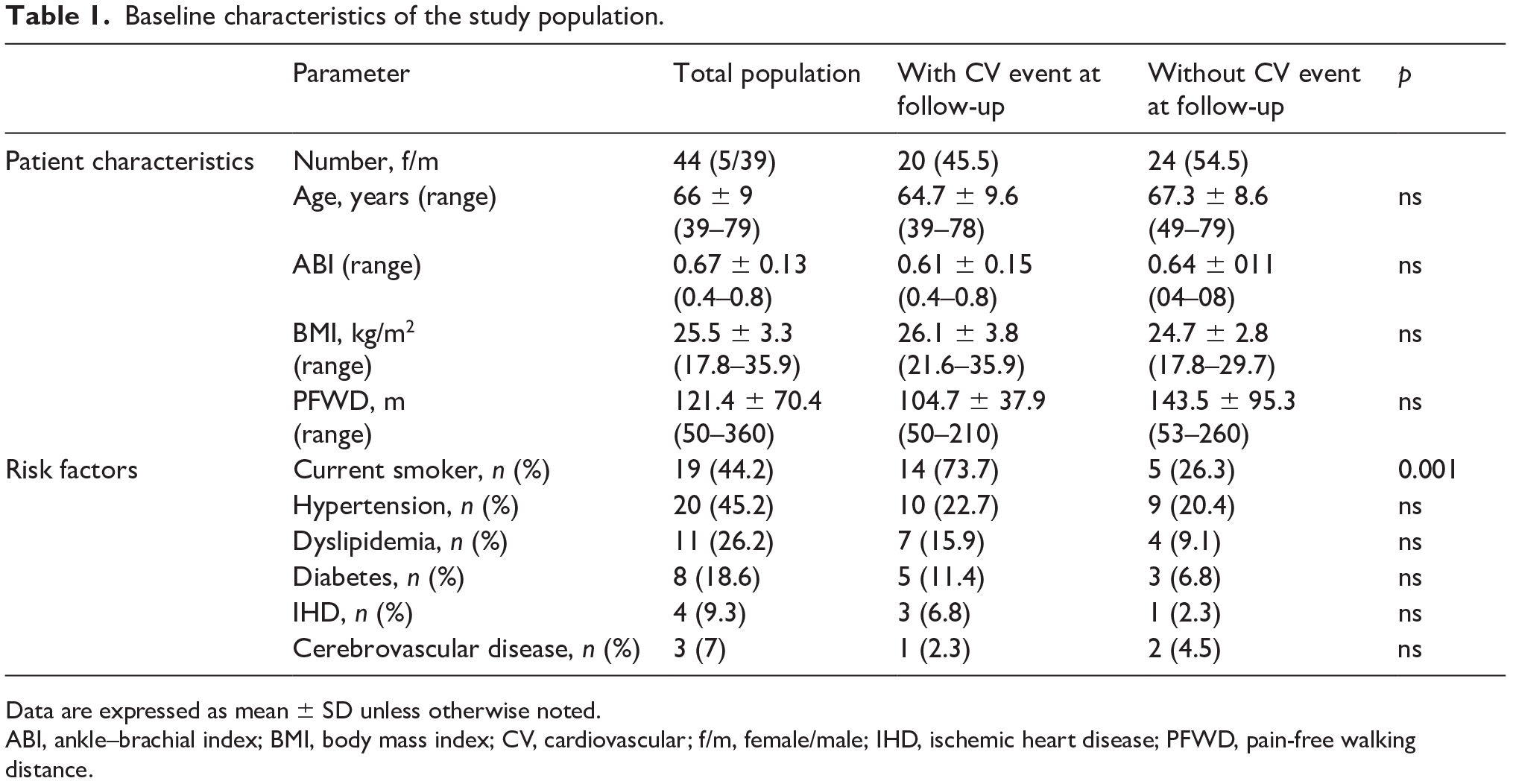
Data are expressed as mean ± SD unless otherwise noted.
ABI, ankle–brachial index; BMI, body mass index; CV, cardiovascular; f/m, female/male; IHD, ischemic heart disease; PFWD, pain-free walking distance.
Endothelial function
The amount of exercise was evaluated by measuring the maximal walking time (min) and the pain-free walking time (min). We did not find any correlation between walking maximal time and the difference in FMD% measured before and after exercise (slope = 0.192, 95% CI: −0.069 to 0.453, correlation coefficient = 0.238, r2 = 0.057, p = 0.144), as well as pain-free walking time, suggesting that the generation of ischemia, independently from the amount of exercise performed, was responsible for the drop in FMD. Baseline brachial artery diameter before and after exercise were similar (4.5 ± 0.6 mm vs 4.4 ± 0.5 mm, p = 0.765, by paired t-test). FMD at rest in patients with IC was 3.5 ± 0.6% (i.e. considerably lower than values found in healthy subjects),24,25 and maximal treadmill test leg exercise further reduced it, strikingly and significantly (Figure 1).
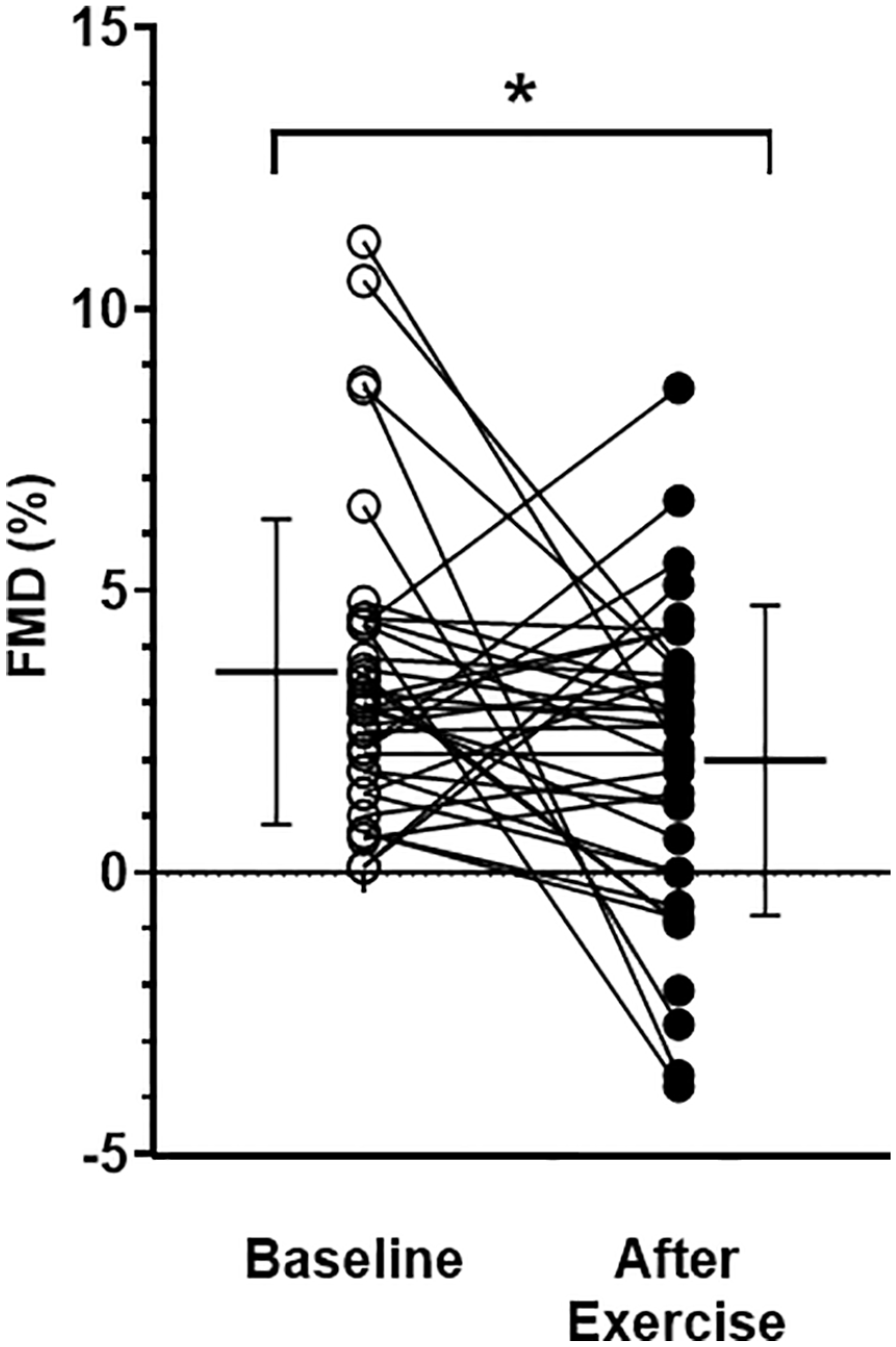
FMD at rest and after exercise.
Baseline FMD or ABI at rest did not predict the occurrence of subsequent ischemic events. On the contrary, an exercise-induced drop of FMD greater than the median decrease (i.e. > 1.3%) was strongly predictive of subsequent MACE (HR: 3.9, 95% CI: 1.2 to 12.2), with an event-free survival time strikingly and significantly shorter than that of patients with a FMD drop lower than the median (i.e. < 1.3%) (Figure 2).
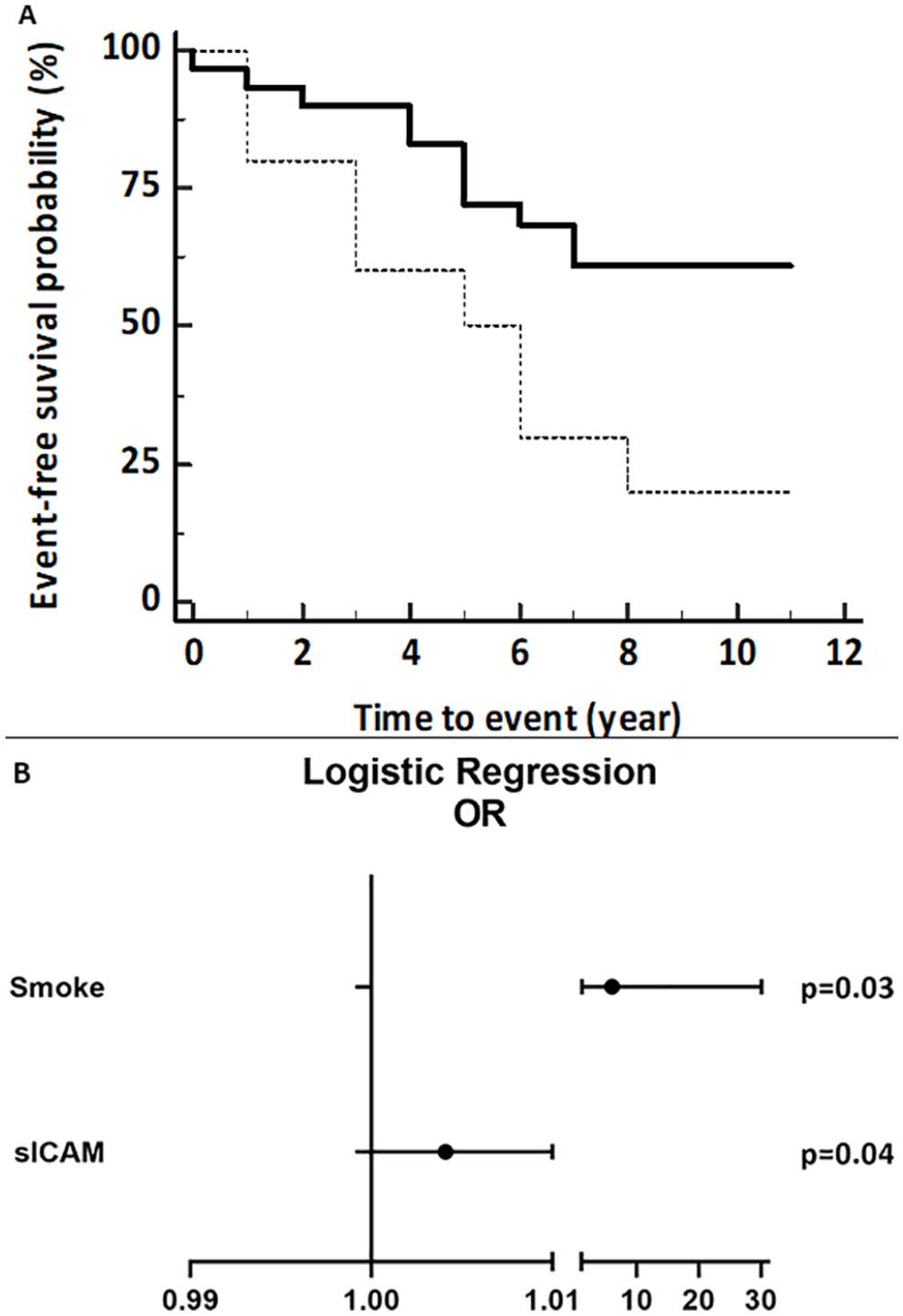
Survival probability and odd ratios. (
Of the soluble and urinary biomarkers measured (online supplementary Table 1), baseline sP-Sel, sICAM-1, and 8-iso-PGF2α levels were significantly higher in patients with subsequent ischemic events than in subjects without events at univariate analysis (Figure 3). As a marker of oxidative stress generated by ischemia/reperfusion injury, we measured 8-iso-PGF2α urinary levels in samples collected during the 24 hours prior to exercise and in samples collected 24 hours after. We indeed did not observe an increase of 8-iso-PGF2α after exercise, but the timing of sample collection (24 hours) was probably too long to highlight the variations induced by effort diluting the short-lasting oxidative stress generated by exercise. However, we found that urinary levels of 8-iso-PGF2α in samples collected in the 24 hours after exercise were significantly higher in subjects with MACE at follow-up than in PAD patients without MACE at follow-up, suggesting that higher levels of oxidative stress in patients with PAD are associated with higher MACE incidence and logistic regression analysis showed that higher baseline sP-sel levels were predictive of subsequent ischemic events (OR: 1.082, 95% CI: 1.01 to 1.16, p = 0.03). Instead, none of the exercise-induced changes of circulating or urinary biomarkers was predictive of ischemic events (online supplementary Table 2).
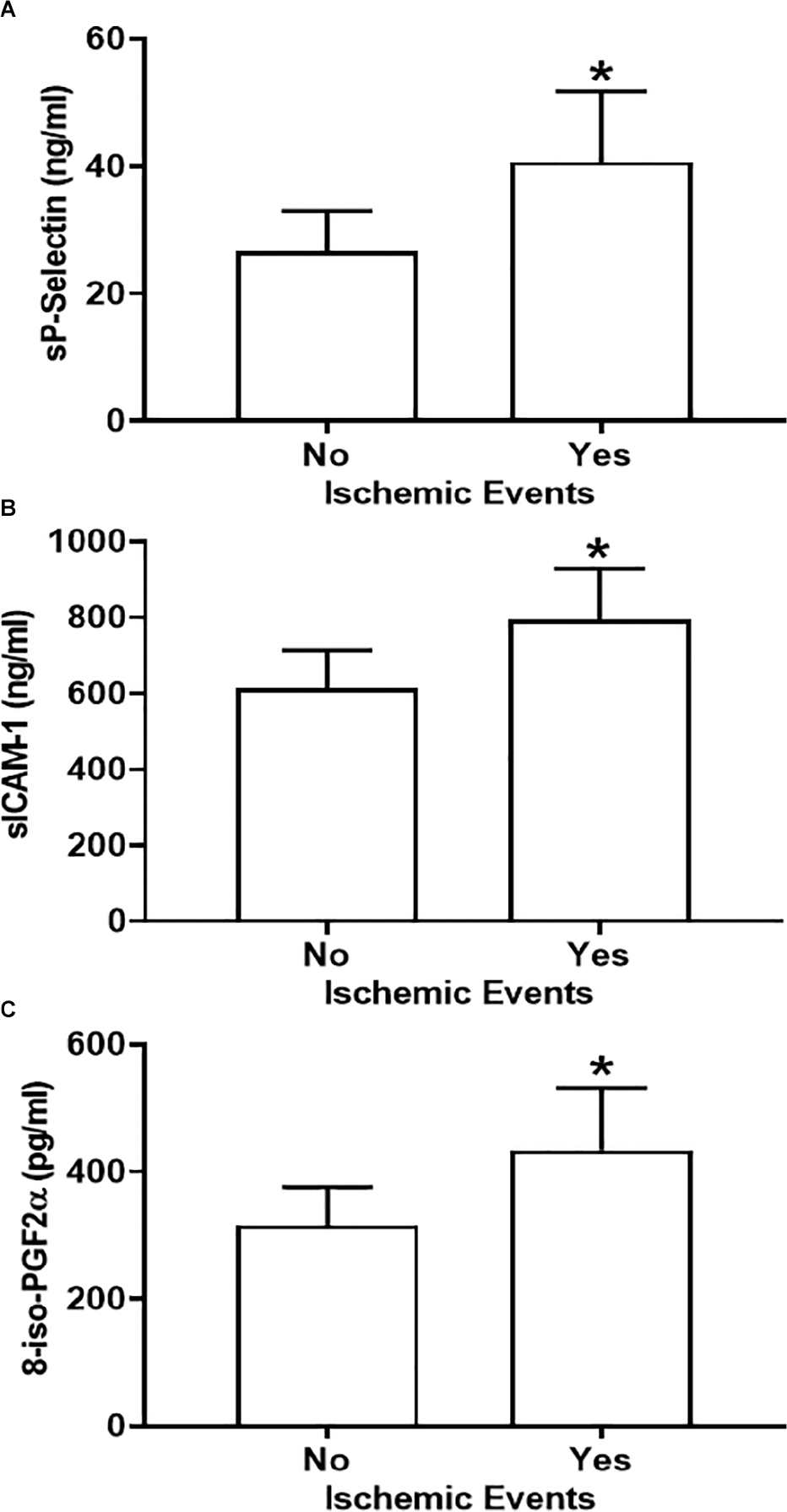
Soluble P-selectin (A), soluble ICAM-1 (B), and 8-iso-PGF2α (C) baseline levels in patients with an ischemic event compared to those without events at follow-up.
Among risk factors, smoking (OR: 6.05, 95% CI: 1.22 to 30.1, p = 0.04) and baseline sICAM levels (OR: 1.004, 95% CI: 1.001 to 1.1, p = 0.03) showed a predictive value for MACE. Unfortunately, the information about current smoking at follow-up was not available and separate analysis by current smoking was not possible. However, the exercise-induced decrease in FMD at enrollment was similar in current smokers and non-smokers (FMD decrease = −1.96%, 95% CI: −4.69 to 0.78 and −1.26%, 95% CI: −2.92 to 0.39, respectively, p = 0.64), Moreover, 55.6% of smokers had an exercise-induced FMD drop > 1.3% compared to 44% of non-smokers (p = 0.52).
In multivariable Cox regression analysis, an exercise-induced FMD drop greater than 1.3% was independently associated with MACE (HR: 2.7, 95% CI: 1.1 to 6.9, p = 0.03).
Discussion
Our study shows for the first time that systemic endothelial dysfunction induced by maximal leg exercise, assessed by the acute drop of brachial artery FMD, is strongly predictive of subsequent adverse ischemic cardiovascular events in patients with IC. Moreover, baseline sP-sel also predicted MACE, while several other biomarkers did not.
Many studies have previously shown that impaired endothelial function, measured as reduced FMD, is a prognostic marker of ischemic events in patients with IC,9,10,12 similar to that observed in other patient groups at high risk of cardiovascular events. 33 A predictive value of endothelial dysfunction for cardiovascular events and restenosis was also shown in patients with PAD undergoing surgical revascularization.34,35 However, no previous studies have analyzed the predictive value of an exercise-induced drop of FMD for cardiovascular events. Indeed, while in healthy subjects undergoing a 6-minute walk performed prior to FMD measurement the endothelial function was improved, 36 in both healthy subjects and patients with abdominal aortic aneurysms FMD was decreased after maximal exercise. 37 In patients with IC, maximal exercise significantly and drastically reduced FMD, a phenomenon prevented by administration of vitamin C but not of placebo, suggesting a role of ischemia/reperfusion injury in the reduction of FMD. 16
Ischemia/reperfusion injury is a crucial trigger of endothelial dysfunction.38–40 It is thus conceivable that a particularly striking worsening of endothelial function induced by maximal leg exercise, a test eliciting systemic ischemia/reperfusion injury,15,41–43 may identify a subgroup of patients with PAD at higher risk of subsequent ischemic cardiovascular events. For instance, it has previously been shown that the degree of myocardial ischemia/reperfusion injury induced by coronary revascularization in patients with coronary artery disease (CAD) has a predictive value for subsequent major ischemic cardiovascular events, 44 but no studies to our knowledge have assessed the prognostic value of exercise-induced endothelial dysfunction in patients with PAD.
Recent advances in the pathophysiology of PAD have identified mitochondrial dysfunction as a key element in the deleterious processes associated with lower limb exercise-induced ischemia/reperfusion injury, with several observations suggesting that skeletal muscle mitochondrial function may be considered as a prognostic factor in PAD. 45 Indeed, changes in walking performance induced by exercise correlated with mitochondrial function in patients with IC, with patients displaying a worsening of the peak walking time after exercise showing decreased mitochondrial respiration. 46 Therefore, acute exercise-induced ischemia/reperfusion injury may exert more deleterious effects in some patients with IC than in others, and this may associate with adverse long-term cardiovascular events.
Study limitations
Our study has some limitations. First, it has a relatively low sample size; however, this is partly compensated for by the relatively long follow-up and the high cardiovascular risk of the study population. Indeed, 45.4% of patients suffered a MACE during a median follow-up of 5 years, with a yearly incidence of 9.2%. Moreover, some landmark studies showing the prognostic value of a low ABI for subsequent cardiovascular events in PAD were of a similar size. 47 Secondarily, there was an imbalance in the baseline characteristics of patients with or without subsequent events, with a higher number of smokers among patients with events. Smoking is associated with impaired endothelium-dependent arterial dilation47–51 and with increased cardiovascular risk. 50 However, chronic smoking does not appear to enhance the detrimental effect of vasoconstrictor stimuli or of NO inhibitors on endothelial function,52,53 thus it is unlikely that this imbalance may have influenced the effects of exercise on FMD. Moreover, smoking is expected to approximately double the risk of a new cardiovascular event, 54 while the risk ratio in our PAD subpopulation with greater impaired exercise-induced FMD was 3.9 (95% CI: 1.2 to 12.2). Finally, multivariable Cox regression analysis confirmed that impaired exercise-induced FMD was an independent predictor of subsequent MACE. 54
Conclusions
In conclusion, our data show that the transient, acute worsening of systemic endothelial function induced by maximal leg exercise in patients with IC is predictive of subsequent ischemic cardiovascular events with apparently a higher sensitivity than FMD at rest. Patients with symptomatic PAD are at high cardiovascular risk. The identification by exercise-induced endothelial dysfunction of a subgroup of patients at even higher risk may have an incremental value for risk assessment, permitting recognition of those patients who require stricter monitoring of adherence to therapeutic and lifestyle modification prescriptions. Moreover, our findings imply that the prevention of ischemia/reperfusion damage might represent a therapeutic target in patients with IC. Unfortunately, despite evidence for a pathogenic role in several cardiovascular disorders, so far no intervention or substance able to protect from ischemia/reperfusion injury has emerged. Further research on this is highly warranted. 55
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X211001927 – Supplemental material for Walking-induced endothelial dysfunction predicts ischemic cardiovascular events in patients with intermittent claudication
Supplemental material, sj-docx-1-vmj-10.1177_1358863X211001927 for Walking-induced endothelial dysfunction predicts ischemic cardiovascular events in patients with intermittent claudication by Rino Migliacci, Giuseppe Guglielmini, Chiara Busti, Emanuela Falcinelli, Pietro Minuz and Paolo Gresele in Vascular Medicine
Footnotes
Acknowledgements
Collaboration with A.M. Mezzasoma (University of Perugia, Perugia, Italy) for biomarkers measurement and the skilled editorial assistance of S. Spaccini (University of Perugia) are gratefully acknowledged.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by a grant to PG from MIUR-FIRB (Protocol # RBFR12W5V5_004) and by Regione Umbria (Progetto Ricerca Finalizzata Regione Umbria 2013).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
