Abstract
The aim of this study was to develop and pilot a group education programme for promoting walking in people with intermittent claudication. Patient focus groups (n=24) and literature reviews were conducted to inform the development of the education programme, which involves a three-hour group-based education workshop and follow-up telephone support. A pilot study was subsequently conducted in which 23 new patients (Rutherford category 1–3) were randomly assigned to usual care (control) or usual care plus the education programme. Outcomes were assessed at baseline and six weeks including daily steps (tri-axial accelerometer), walking capacity (six-minute walk test and Gardner treadmill test), and quality of life (Intermittent Claudication Questionnaire [ICQ]). Exit interviews were conducted to assess the acceptability and usefulness of the programme. Compared with controls, the intervention group had superior walking capacity and quality of life at six weeks. Mean differences in six-minute walk distance, treadmill maximum walking distance and ICQ score were 44.9 m (95% confidence interval [CI], 6.9 to 82.9), 173 m (95% CI, 23 to 322), and −10.6 (95% CI, −18.9 to −2.3), respectively. The daily step count did not differ between groups. The exit interviews indicated that participants valued attending the programme, that it gave them a greater understanding of their condition, and that they had been walking more for exercise since attending. The results suggest that the education programme is feasible, acceptable, and potentially useful for improving walking capacity and quality of life. A fully-powered trial exploring clinical and cost effectiveness is needed.
Introduction
Supervised exercise training can evoke clinically-meaningful improvements in walking distances and quality of life in people with intermittent claudication due to peripheral arterial disease (PAD). 1 As such, the United Kingdom’s (UK’s) National Institute for Health and Care Excellence (NICE) recommends a three-month programme of supervised exercise as a first-line therapy. 2 However, few patients participate in supervised exercise because of limited availability of programmes and the requirement to travel regularly to an exercise centre. 3 The common alternative of providing basic advice to walk more tends not to result in successful behaviour change.4,5
Structured, home-based exercise programmes that promote self-managed walking offer a promising alternative. Such programmes have the potential to improve exercise behaviour at a much lower cost compared to medically-supervised programmes, and may be more acceptable to patients because the requirement to travel is removed. However, the evidence for the effectiveness of home-based exercise programmes is weak and mixed. 6 Many of the studies have methodological flaws, and some of the more rigorous contemporary trials have used interventions that may not be easily implemented in the UK’s tax-funded healthcare system.7,8 For example, the recent Group Oriented Arterial Leg Study (GOALS) investigated a home-exercise programme that involved a 90-minute group session each week for six months, with each session comprising 45 minutes of facilitator-led discussions about exercise for people with PAD and 45 minutes of walking around an indoor track. 7
This paper describes the development and preliminary testing of the SEDRIC (Structured EDucation for Rehabilitation in Intermittent Claudication) programme, which is a group structured education programme that promotes self-managed walking in people with intermittent claudication. The programme has been designed to be sustainable, affordable and suitable for delivery throughout the National Health Service of the UK. The intervention was modelled on the structured education self-management programmes used in diabetes care.9,10
Methods
This section describes the development of the SEDRIC programme and then a pilot study of the intervention. The study was approved by the Research Ethics Committee for Yorkshire and the Humber − Sheffield. Written informed consent was obtained from participants prior to enrolment.
Study population
Participants in this project all had clinically-diagnosed intermittent claudication due to peripheral arterial disease (Rutherford category 1−3).
Development of SEDRIC
The process followed in developing the SEDRIC programme is outlined in Figure 1.
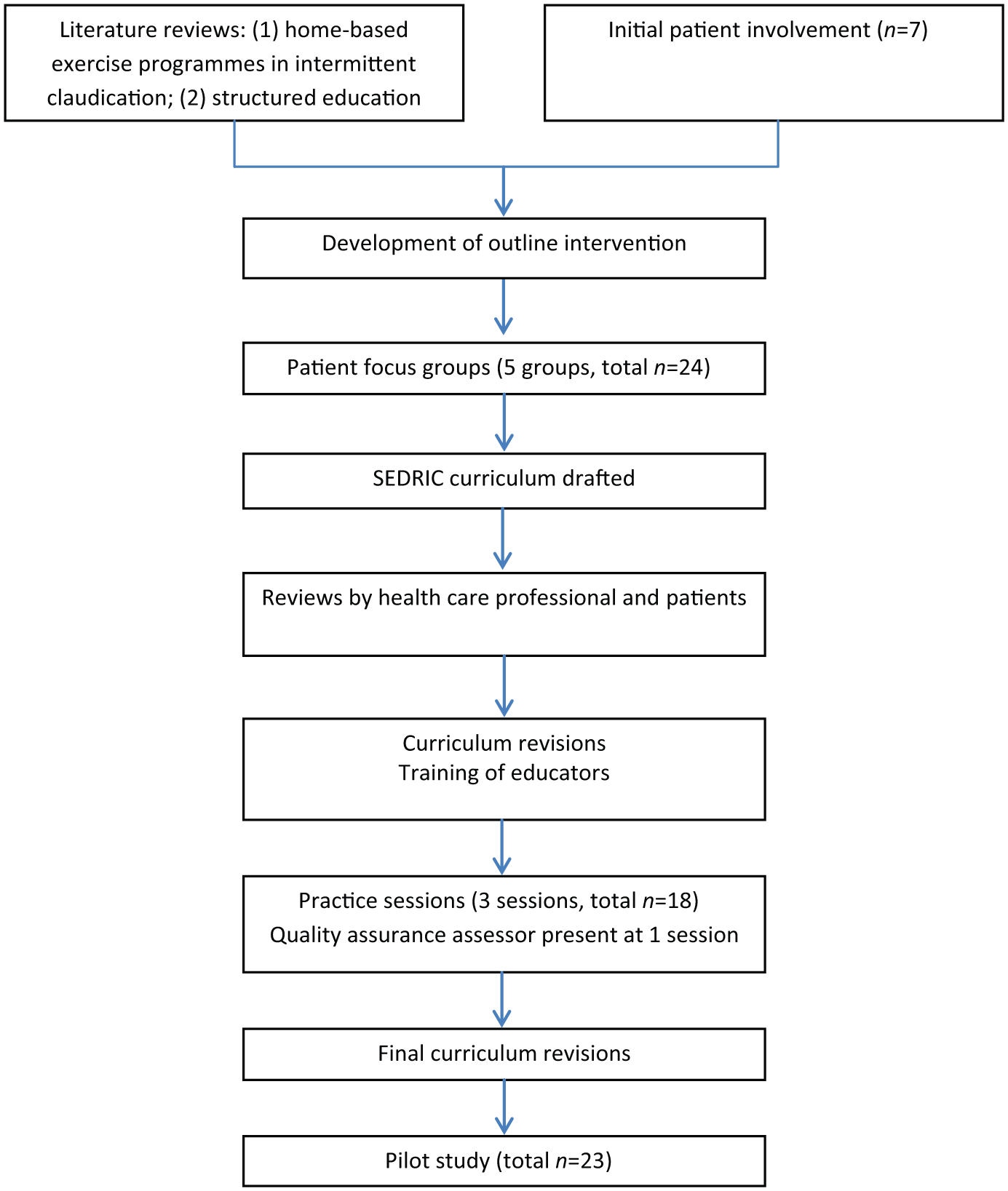
Process of developing and testing the SEDRIC programme.
Patient involvement
We initially conducted two patient involvement sessions with seven patients to obtain their views on and perceptions of PAD, their opinions about possible intervention strategies, target behaviours for intervention, research design, recruitment strategies, and suitable outcome measures.
Literature reviews
The literature on structured, home-based exercise programmes (HEPs) for individuals with intermittent claudication was systematically reviewed; the full results of which are published elsewhere. 6 The review highlighted the potential of HEPs; however, there was no clear indication about which combination of behaviour change strategies was optimal and some used resource-intensive approaches (i.e. interventions that require high levels of staff time) that may be incompatible with the UK healthcare system. Therefore, we opted to base the SEDRIC programme on the content and behaviour change techniques of the successful Prediabetes Risk Education and Physical Activity Recommendation and Encouragement (PREPARE) programme. 9 A non-systematic review of the literature was performed to help familiarise the research team with the structured education approach on which PREPARE and the related Diabetes Education and Self-Management for Ongoing and Newly Diagnosed (DESMOND) programme are based. 10
Focus groups
To further inform the development of the SEDRIC programme, focus group work was conducted, a full description of which is presented elsewhere. 11 In brief, 24 individuals with intermittent claudication (71% male, age 71±8 years; same eligibility criteria as for the subsequent pilot study – described below) participated in one of five focus group sessions. Each session lasted 60−75 minutes and was facilitated by two qualitative researchers. Topics for discussion included experiences of living with intermittent claudication, illness and treatment beliefs, attitudes and beliefs about physical activity, and thoughts about a new structured education programme to promote walking (an overview of which was presented to the participants). The sessions were audio-recorded and transcribed verbatim. A thematic analysis was employed to analyse the data. Consensus and agreement was reached on emerging themes. After five focus groups, there was consensus agreement between the interviewers that no new information was emerging and therefore further groups were not required.
The focus groups provided evidence that strategies are needed to help people with intermittent claudication understand their condition better, and to support them in effective self-management. Two over-riding themes emerged: uncertainty, and lack of support and empathy from medical professionals. Participants expressed uncertainty about PAD and intermittent claudication, what the risk factors are and how they work, treatment options and whether lifestyle change, particularly walking, would help.
Structured education as a possible intervention strategy was described to the participants. The vast majority expressed that this would be useful to them and that they would be interested in attending. They liked the idea of a group setting, and also expressed a preference for some level of direct follow up either by phone or face to face after the workshop. Knowledge and empathy were identified as the key skills for the workshop educators.
Programme development
The content of the PREPARE structured education programme was adapted to make it relevant to the specific focus of the SEDRIC programme. An 89-page curriculum was written by a behaviour change specialist (TG) and an exercise scientist (GT), and this was subsequently reviewed by other exercise scientists, patients, vascular surgeons, a vascular nurse specialist and a general practitioner.
Table 1 provides an overview of the content and structure of the SEDRIC programme. The programme is designed to be delivered to six to ten individuals by two trained educators during one three-hour session. The primary aim is to promote self-managed walking by targeting perceptions and knowledge of the condition as well as promoting self-regulatory skills such as goal setting and self-monitoring. Self-regulation is based around pedometer use, which was demonstrated to be crucial to the success of the PREPARE programme. 9 Specifically, individuals are encouraged to gradually build up their total daily steps to more than 7500, 12 and to perform at least 2500 of these steps as ‘exercise steps’, which should approximate 30 minutes of walking assuming a mean step cadence of 90 steps/minute, 13 consistent with clinical practice guidelines for supervised exercise and the UK physical activity guidelines for adults.2,14 For their ‘exercise steps’, participants are advised to walk at a speed that evokes a strong claudication pain and to persevere walking as long as is tolerable. To support this, sections of the education programme were devoted to helping individuals understand what claudication pain is, and how walking for exercise with strong claudication pain can result in improved symptoms. Participants are supported in developing short and long-term goals for walking, with reference to their baseline daily steps count recorded by wearing an accelerometer (ActiGraph GT3X+, ActiGraph, Pensacola, FL) for seven days before attending the workshop. They are also supported in developing an action plan detailing where, when and how their first initial goal will be reached and they are encouraged to repeat this process for each new goal. Participants are encouraged to wear their pedometer (Yamax SW-200 Digi-Walker) on a daily basis and to self-monitor their ambulatory activity and intensity of claudication during walking using a specifically-designed exercise diary. Two weeks after the educational workshop, participants are contacted by telephone to review progress and discuss goal setting and barriers, with the aim of supporting maintenance of behaviour change.
Outline of the SEDRIC programme.
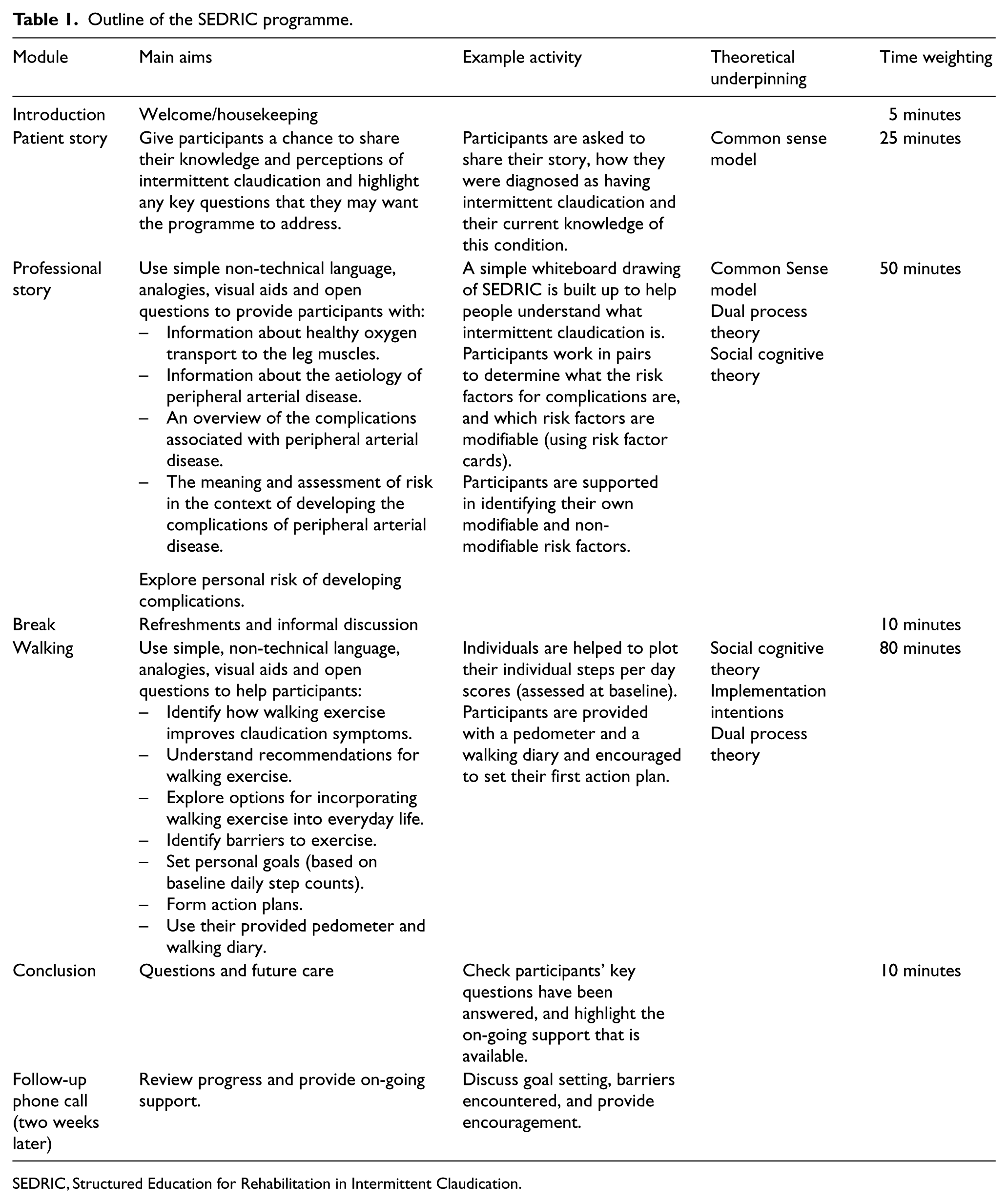
SEDRIC, Structured Education for Rehabilitation in Intermittent Claudication.
The programme is underpinned by a framework of mutually complementary health behaviour theories, including Bandura’s social cognitive theory, 15 Gollwitzer’s implementation intentions, 16 Leventhal’s common sense model, 17 and Chaiken’s dual process theory, 18 and was informed by and modelled on the person-centred empowerment philosophy and learning techniques developed for the DESMOND programme; 10 a national self-management programme for individuals with type 2 diabetes. The programme was developed in line with the Medical Research Council’s Framework for Complex Interventions to Improve Health, 19 and the NICE guidelines for the management of diabetes, 20 which provide key criteria for high-quality structured education programmes.
Refinements to the structure and content of the SEDRIC programme were undertaken using a process that involved training two educators (GT, HT) in the core principles of structured education (conducted by the DESMOND collaborative) and then piloting the programme to a group of exercise scientists (n=6) whereupon participant, educator and observer-level feedback were collected and the programme revised accordingly. This process was repeated twice with two groups of representative patients (both n=6), and the last of these was independently assessed by an experienced Quality Assurance Assessor.
Pilot study
After the SEDRIC programme was developed, a pilot randomised controlled trial was conducted to provide a preliminary assessment of the effectiveness and acceptability of the approach. This was a pragmatic, single-centre, parallel-group study, conducted between August 2013 and May 2014, in which participants were randomised (ratio 2:3) to receive usual care (control) or usual care plus the SEDRIC programme. Usual care included provision of a brief information leaflet on PAD. Randomisation was achieved using a block randomisation sequence (block size 10) generated prior to recruitment (nQuery Advisor 6.01, nQuery Statistical Solutions, Boston, MA, USA), and was implemented by an individual who was not involved in the recruitment or data gathering processes. Group allocation was concealed from participants while baseline information was gathered.
Patient identification and selection
Potentially eligible patients were identified through screening vascular clinic letters at a hospital in the north of England. Recruitment packs were posted to patients, and individuals who responded as ‘interested’ were invited to an initial visit during which written informed consent was obtained and eligibility was confirmed by a vascular surgeon (SN, WA). Demographic data were collected, and resting ankle-brachial indices were calculated by dividing the higher of the posterior tibial and dorsalis pedis pressures by the higher of the right and left arm pressures. 21
Inclusion criteria were age ⩾18 years and stable intermittent claudication for ⩾3 months. Exclusion criteria were critical limb ischaemia, previous or planned lower-limb revascularisation, presence of contraindications to exercise (e.g. unstable angina) or co-morbidities that limited walking to a greater extent than the intermittent claudication (e.g. severe arthritis), and major surgery, myocardial infarction or stroke in the previous six months.
Outcome measures
Outcome measures were assessed at baseline (before randomisation) and after six weeks. Mean daily step count was assessed using a tri-axial accelerometer (ActiGraph GT3X+) over seven days. Pain-free and maximum walking distances were assessed using the Gardner incremental treadmill test. 22 Walking capacity was also assessed using the six-minute corridor walking test. 23 Self-reported ambulatory ability was assessed using the Walking Impairment Questionnaire (WIQ) 24 and the Walking Estimated-Limitation Calculated by History (WELCH) questionnaire. 25 Disease-specific quality of life was assessed using the Intermittent Claudication Questionnaire, 26 and generic health status using the EuroQol EQ-5D-5L. 27 We also assessed various psychological outcome measures that were targeted by the intervention, including self-efficacy related to walking different distances at a brisk pace along with the desire to be able to complete these walking challenges, 28 acceptance of PAD-related pain/discomfort, 28 and personal control and understanding of one’s illness. 29 Outcome measurement was conducted by a researcher blinded to group assignment. One-to-one exit interviews were conducted with intervention group participants after the six-week assessment. These interviews lasted 15 to 20 minutes and explored the acceptability of and satisfaction with the SEDRIC programme, components that were useful or not useful, and suggestions for changes.
Statistical analysis
Analyses were conducted on an intention-to-treat basis using Stata 13. Outcome data were analysed using linear regression models adjusting for the baseline measure. 30 The difference in outcomes between groups at six weeks and corresponding 95% confidence intervals (CI) from the model are presented. The normality of the standardised residuals was checked using a q-q plot, and homoscedasticity assessed by a scatter plot of the standardised residuals against fitted values. For those variables where the model assumptions were in doubt, the outcome data were transformed prior to analysis.
Results
Recruitment and participant flow
Between August 2013 and April 2014, invitation letters were sent to 262 potentially eligible patients. Of the 105 patients who responded (40%), 32 expressed an interest in participating. Non-responders were not sent reminder letters because the target recruitment for the pilot study had been reached. Five patients were deemed ineligible on telephone screening, whereas the remaining 27 attended a familiarisation and screening visit and provided written informed consent. The eligibility status changed for four patients prior to completing the baseline assessment visit, hence only 23 participants were randomised, 14 to the intervention group and 9 to control (Figure 2). Table 2 demonstrates the baseline characteristics of participants. Compared to control, the intervention group had slightly higher levels of physical activity (daily steps), walking capacity and quality of life.
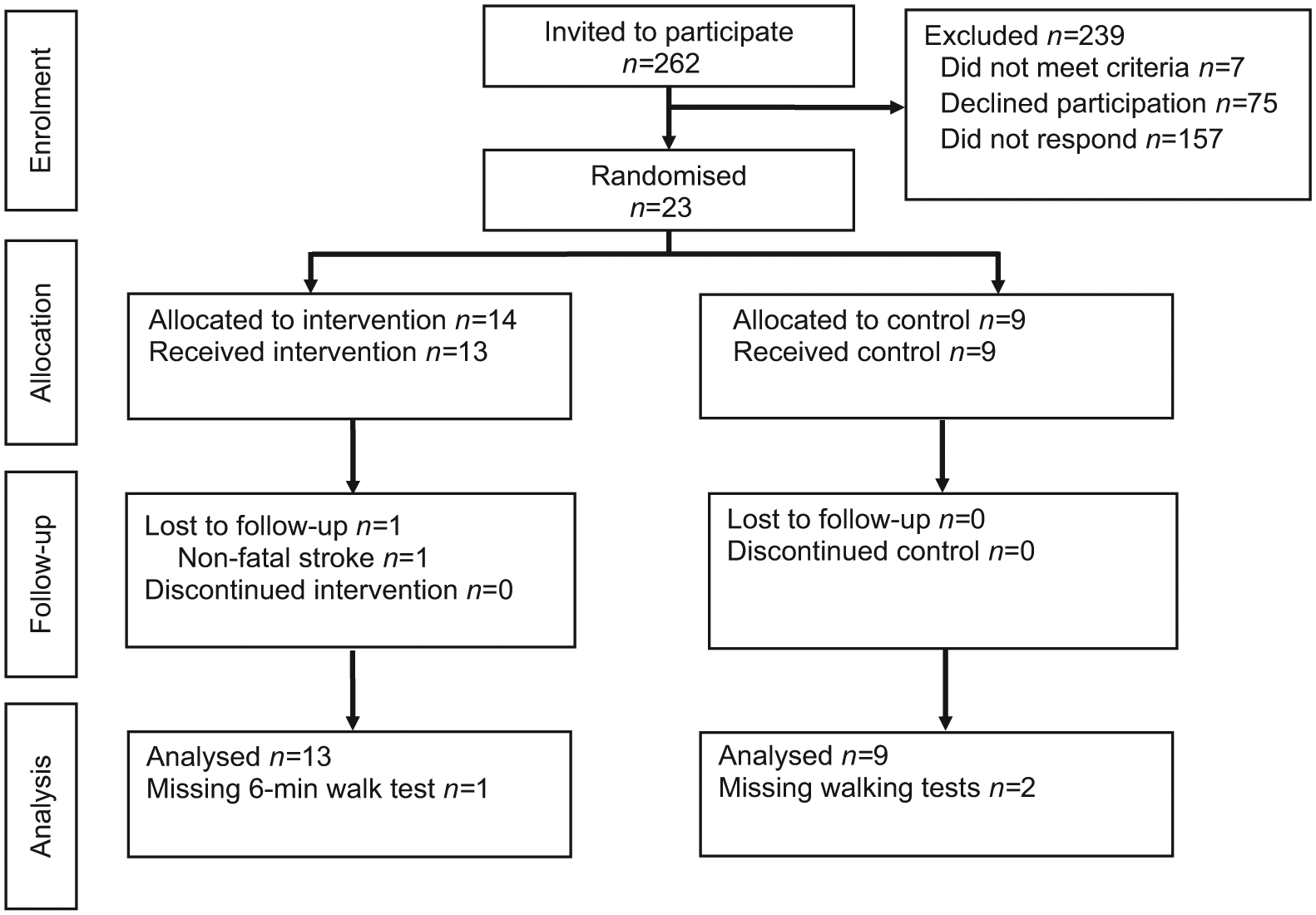
Consolidated Standards of Reporting Trials (CONSORT) diagram.
Participant characteristics at baseline.
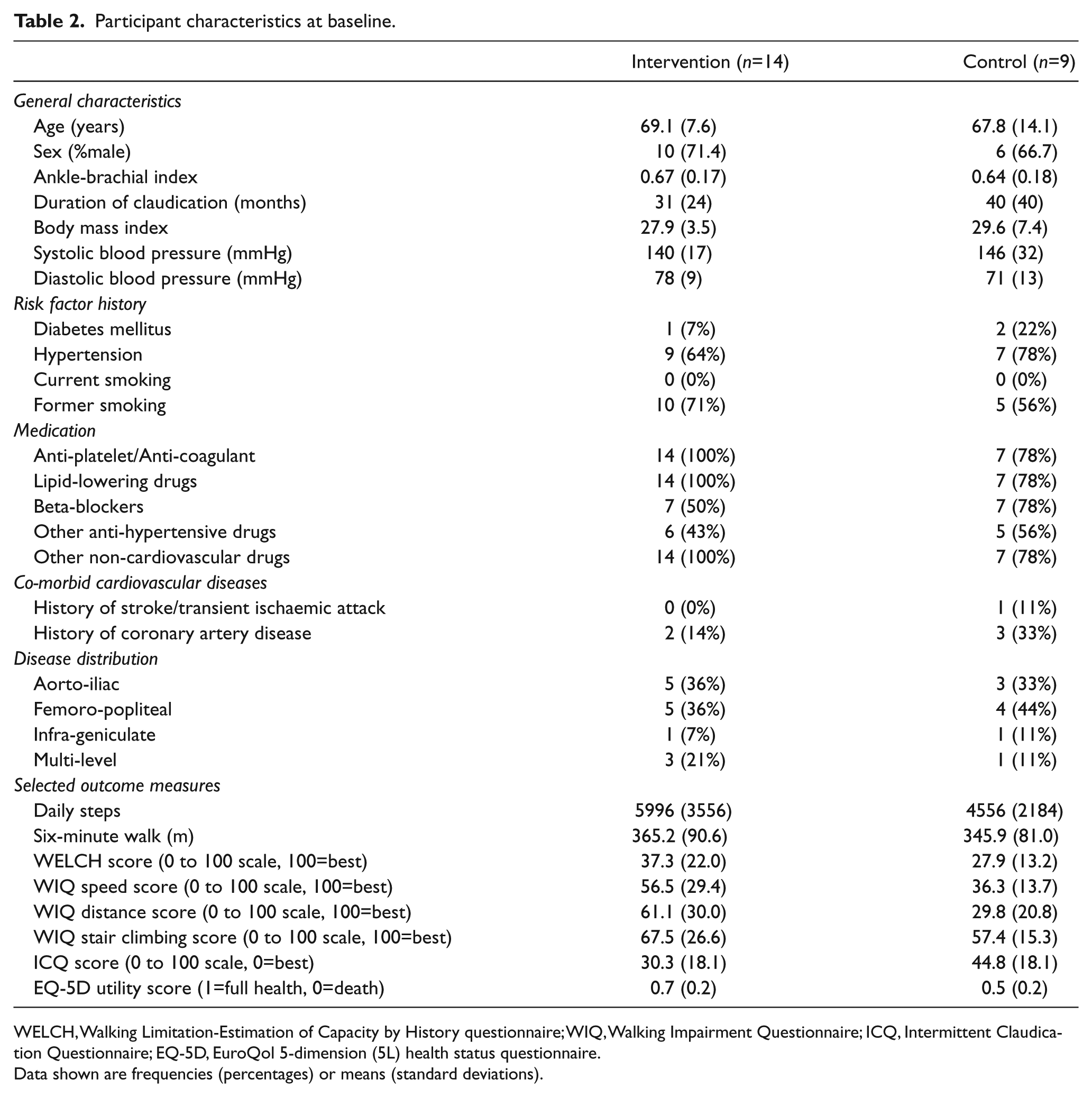
WELCH, Walking Limitation-Estimation of Capacity by History questionnaire; WIQ, Walking Impairment Questionnaire; ICQ, Intermittent Claudication Questionnaire; EQ-5D, EuroQol 5-dimension (5L) health status questionnaire.
Data shown are frequencies (percentages) or means (standard deviations).
One participant in the intervention group was unable to attend the education workshop, but completed the follow-up assessments. Another participant in the intervention group was withdrawn from the study one week before completing follow-up assessments because they suffered a non-fatal stroke, which was judged to be unrelated to the research. Two control participants were unable to attend their follow-up walking assessments. However, they completed the follow-up questionnaires by post.
Of the 13 participants who attended the SEDRIC workshop, seven returned the exercise diary that they had completed during the six-week period between baseline and follow up. The total number of steps walked each day (self-recorded using a pedometer) increased from 7714±2233 (mean±standard deviation [SD]) in the first week following the workshop to 8877±2154 in the sixth week. The number of steps walked specifically for exercise each day increased from 2969±1723 in the first week to 3798±2697 in week six. All seven participants reported performing between five and seven sessions of purposeful walking exercise each week.
Outcome measures
Data for the main outcome measures are presented in Table 3. Compared with controls, the intervention group had superior walking capacity across six measures (six-minute walk distance, treadmill maximum walking distance, WELCH score, and WIQ speed, distance and stair-climbing scores). However, the confidence intervals spanned zero for daily steps and treadmill pain-free walking distance. The intervention group also demonstrated superior disease-specific quality of life at six weeks but there was no difference in EQ-5D utility score.
Outcome measures.
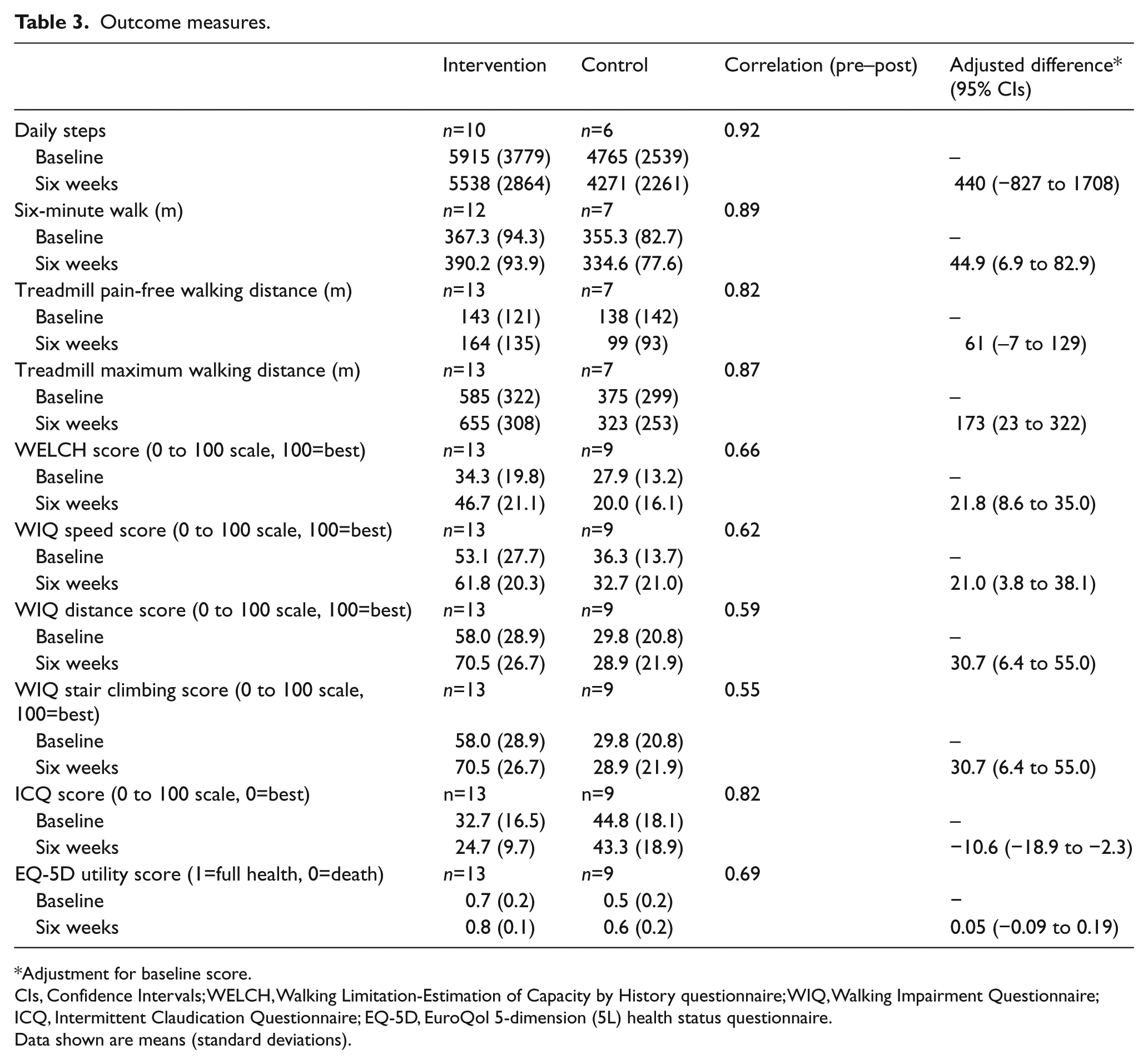
Adjustment for baseline score.
CIs, Confidence Intervals; WELCH, Walking Limitation-Estimation of Capacity by History questionnaire; WIQ, Walking Impairment Questionnaire; ICQ, Intermittent Claudication Questionnaire; EQ-5D, EuroQol 5-dimension (5L) health status questionnaire.
Data shown are means (standard deviations).
Psychological outcome measures
Data for the psychological outcome measures are presented in Supplementary Table 1. At six weeks, the intervention group had superior scores for walking performance self-efficacy and perceived control over illness.
Acceptability data
The exit interviews of the pilot study indicated that participants valued participating in SEDRIC, that it provided them with a greater understanding of their condition, and that they had been walking more for exercise since attending. The pedometer was valued, and seen as a useful tool for motivation, self-monitoring and goal setting. No changes were suggested, perhaps because of the rigorous development work that preceded the pilot study.
Discussion
This report describes the development and preliminary testing of a pragmatic group-based structured education programme for promoting self-managed walking in people with intermittent claudication. A comprehensive approach to developing the SEDRIC programme was used, including literature reviews, focus groups with patients, consultation with other patients and relevant healthcare professionals and academics, development of programme resources, training and practice in intervention delivery, and an independent quality assurance assessment. The subsequent pilot study met key objectives by helping us identify useful information to assist the design of a full-scale trial. The required recruitment target was easily achieved, retention and programme attendance was good, participants provided positive feedback about the programme, and improvements in walking capacity and disease-specific quality of life were observed at six weeks.
Although the pilot study was not powered to detect significant changes in outcome, potentially important differences in favour of the intervention group were observed for walking capacity and disease-specific quality of life at six weeks. Although no differences were evident for accelerometer-measured daily steps, interview data indicated that intervention group participants were walking more for exercise after attending the SEDRIC workshop, and this was supported by the exercise diaries of seven participants, which showed that they were walking regularly (⩾5 sessions per week) and achieving their exercise goals. The reasons for the discrepancy between the accelerometer and patient-reported physical activity data are unclear. However, accelerometer wear time was relatively low in some participants (e.g. three days of valid wear instead of the ideal seven), so we are exploring the use of alternative pedometers/accelerometers, which do not need to be removed intermittently, to improve wearing compliance and data completeness in future studies. 31 In addition, it is possible that while the total number of steps was unchanged, participants changed their walking patterns so that they were walking further before having to stop or walking faster; this has been observed elsewhere. 32 In summary, the preliminary findings are positive and reinforce the potential value of conducting a full-scale study. However, given the small sample size, the large number of statistical tests performed, and the fact this this was a single-centre study, the results of the pilot study should be viewed with caution.
Structured education programmes are recognised by NICE and have been widely adopted in the UK healthcare system, at least in the management of diabetes mellitus. 20 This pilot study suggests that they may also have a role in the treatment of PAD. By specifically addressing knowledge gaps and uncertainty around the disease process and the role that walking may play in its management, the SEDRIC programme appears to provide impetus for at least short-term behaviour change. The group-based approach with knowledgeable and empathetic facilitators appears to address a palpable need within this patient group for recognition and support.
Although our systematic review concluded that HEPs appear superior to basic advice to walk more, 6 recent larger trials examining HEPs in PAD have reported varying effects on walking capacity and quality of life.7,8,32,33 One recent report from the UK that has some similarities with the current study involved a psychologist holding two, one-hour, face-to-face meetings with individual patients in their homes. 34 The intervention, which was also based on Leventhal’s common sense model, 17 aimed to promote self-managed walking by modifying participants’ beliefs about their illness and walking, and supporting goal setting, action planning and self-monitoring. Positive results were observed for daily steps at both four-month and two-year follow up. Although the contact time with a healthcare professional in this programme is broadly similar to that in SEDRIC, the breadth of information covered in SEDRIC is greater, and the group-based approach delivered in a community setting will likely make it a more feasible and cost-effective approach. Another noteworthy recent study is GOALS, 7 which investigated a group-mediated cognitive behavioural intervention for patients with PAD (including patients with and without intermittent claudication). The intervention required weekly visits to a medical centre and consisted of a complex intervention requiring group support and a facilitator. At six months, six-minute walk distance had significantly increased in the intervention group compared with a comparison group that had received a matched volume of attention in the form of general health education meetings. The group mean difference (54 m) was not markedly different to that observed in the present study (45 m) despite the fact that the GOALS intervention involved much greater levels of healthcare contact. Importantly, the intensive approach of the GOALS intervention is not compatible with the infrastructure and resources available in the UK healthcare system, whereas the SEDRIC programme potentially could be. Finally, Gardner et al. recently reported similar improvements in six-minute walk distance (+45 m from baseline) in patients with intermittent claudication after they had completed the NEXT Step Home-Exercise Rehabilitation Programme for a period of three months. 32 This HEP involved encouragement to exercise three times each week, with each session comprising 20–45 minutes of intermittent walking at mild-to-moderate claudication pain. Participants were asked to wear a step activity monitor during each exercise session, which did not provide immediate feedback to the user (StepWatch3TM; Orthoinnovations, Inc, Oklahoma City, OK), and participants met briefly with a researcher four times during the three-month period to obtain feedback on exercise data recorded on the monitor and in an exercise logbook. This study provides a good indication that the walking ability of people with intermittent claudication can be improved with a relatively simple HEP. Again, the scope of the SEDRIC programme is greater in that it aims to educate patients about their condition, as well as to promote self-managed walking exercise.
Conclusions
This paper describes the development and preliminary testing of the SEDRIC programme, which is a pragmatic, group-based structured education programme for promoting self-managed walking in people with intermittent claudication. The pilot study results suggest that the programme is feasible, acceptable, and potentially useful for improving walking capacity and disease-specific quality of life. Further work is needed to assess the clinical and cost effectiveness of the SEDRIC programme in relation to both basic advice to walk more (which is what is typically offered in the UK) and supervised exercise (which is what is recommended in NICE guidelines). The SEDRIC programme should not be considered a replacement for supervised exercise. However, it has the potential to be less expensive to implement and maintain and more acceptable to patients and carers, and to provide more lasting benefits through establishing long-term behavioural change.
Footnotes
Acknowledgements
Dr Trish Gorely acknowledges the support provided by The Carnegie Trust for the Universities of Scotland which facilitated travel for this project. Sheffield Hallam University was the sponsor. Sheffield Clinical Research Facility conducted the initial screening of patients. We thank Anna Myers for her assistance in conducting the initial focus group sessions, Carolin Taylor for independently assessing the SEDRIC programme for quality assurance, Graham Thompson for acting as a patient representative, and Dr Oliver Hart for acting as a primary care/General Practitioner representative.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This project was funded by The Bupa Foundation (Ref: PPW12–033F).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
