Abstract
The relationship between ankle–brachial index (ABI) remains uncertain relative to clinical and angiographic features of critical limb ischemia (CLI). From July 2011 to February 2013, 89 consecutive patients with CLI had non-invasive testing for indications of rest pain (n=23, 26%), as well as minor (n=29, 33%) and major (n=37, 42%) ischemic tissue loss. All patients subsequently underwent ABI testing and lower extremity angiography with visualization of the infragenicular arteries. Toe–brachial index (TBI) testing was available among 31 of these patients. Among patients with any ischemic tissue loss, 29% had an ABI between 0.7 and 1.4. Patients with rest pain alone had reduced odds of abnormal arterial runoff in univariate (OR 0.75, 95% CI 0.63–0.90; p=0.002) but not multivariate (p=0.50) analysis. Advanced age, increased ABI, reduced creatinine clearance, hyperlipidemia, and prior coronary artery disease were predictive of abnormal infragenicular runoff. Despite limitations in statistical power, median TBI, compared to ABI, tended to increase when infragenicular arterial runoff was preserved. Overall, the association of TBI with abnormal runoff was not significant (p=0.38). In conclusion, in the evaluation of CLI, nearly one-third of patients with any ischemic tissue loss had a normal or mildly reduced ABI. Assessment of TBI may augment the diagnostic accuracy of ABI in the evaluation of CLI.
Introduction
For patients with resting leg pain or wounds associated with critical limb ischemia (CLI), prompt diagnosis provides opportunity to improve tissue perfusion, relieve pain, and promote wound healing. 1 The ankle–brachial index (ABI) offers a quick, convenient, office-based comparison of systolic blood pressure between the ankle and arm, whereby the indexed limb pressures are stratified, from borderline to severely reduced, to characterize the severity of peripheral artery disease (PAD). However, a consistent relationship may not be observed between severely reduced ABI results that are congruent with the degree of arterial obstruction. 2 To our knowledge, there are currently no published studies in the literature addressing the angiographic validation of non-invasive indices such as the ABI and TBI (toe–brachial index) among patients with CLI. This study highlights the shortcomings of these modalities in the evaluation of patients with CLI, who should undergo angiography to adequately characterize the burden of disease, and to determine the best revascularization strategy.
The present study describes the relationship between ABI, TBI, Rutherford–Baker classification, and angiographic findings among consecutive CLI patients. Based on our clinical experience, we hypothesize that the clinical severity of CLI may be more closely associated with the number of patent arteries below the knee (i.e. infragenicular arterial outflow) than stratified ABI results. Among patients with both ABI and TBI results, we also explore the relationship of TBI and infragenicular arterial outflow relative to that of ABI. Finally, this report characterizes the clinical variables most commonly associated with abnormal infragenicular arterial runoff.
Methods
Patient population
Between July 2011 and February 2013, 89 consecutive patients with CLI were retrospectively identified after presenting to the Department of Vascular Medicine. All patients subsequently underwent lower extremity angiography in the catheterization lab at Cleveland Clinic. After Institutional Review Board approval, patient characteristics and details of non-invasive and invasive arterial assessments were collected.
Definitions and study variables
The Rutherford–Baker classification (RC) system provides an objective scale to describe a clinical continuum of PAD. 3 The scale ranges from 1 to 6, including patients with mild, moderate, and severe ambulatory limb claudication (i.e. RC 1, RC 2, and RC 3). CLI is represented by resting limb pain (i.e. RC 4) as well as those with minor and major ischemic tissue loss (i.e. RC 5 and RC 6, respectively); therefore, this study population includes only those patients classified as RC 4–6. Major tissue loss was defined as gangrene and/or tissue loss proximal to the mid-foot that threatened a functional foot. Minor tissue loss was any non-gangrenous ulceration, including tissue loss distal to the mid-foot.
Several clinical variables were retrospectively collected from the medical record. Age, sex, race, body mass index, creatinine clearance, and RC were recorded at the time of diagnosis. Historical variables were also collected at the time of ABI, including presence of hypertension, hyperlipidemia, diabetes mellitus, current or former tobacco abuse, coronary artery disease, including prior myocardial infarction and/or prior coronary revascularization, prior cerebrovascular accident (CVA) or transient ischemic attack (TIA), prior lower extremity revascularization for PAD, and any prior pedal, below-knee, or above-knee amputation for ischemic tissue loss. The baseline characteristics for the cohort were summarized for the overall cohort and by number of infragenicular runoff arteries (Table 1).
Univariate predictors according to the number of patent infragenicular arteries.
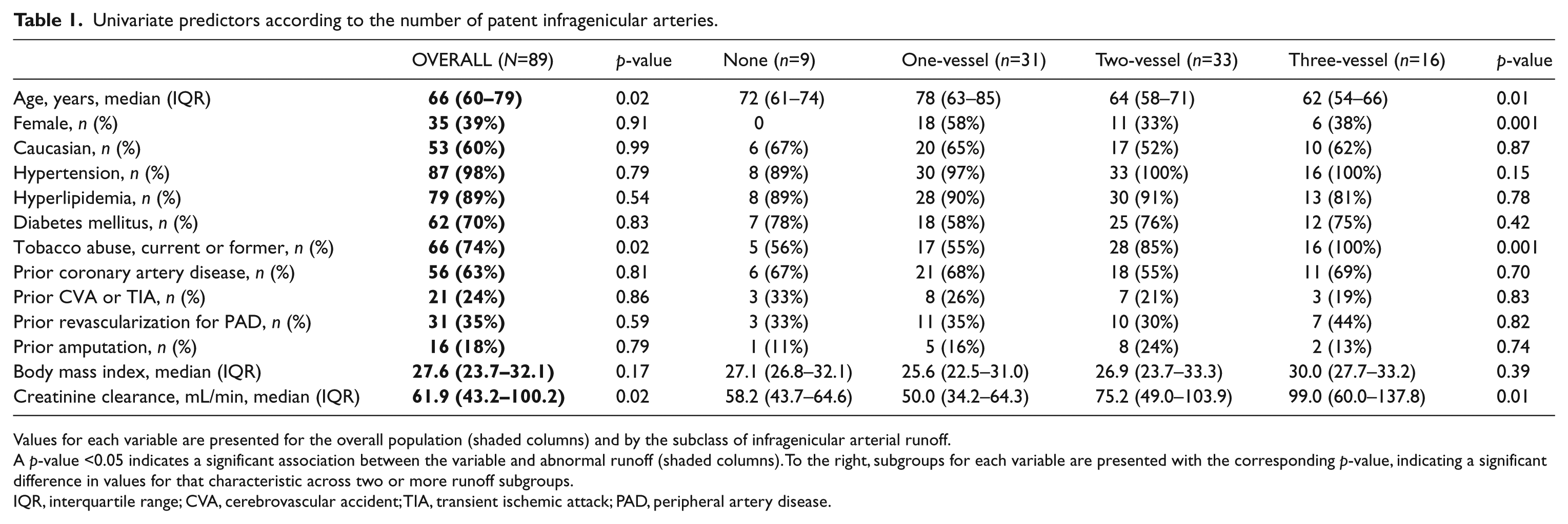
Values for each variable are presented for the overall population (shaded columns) and by the subclass of infragenicular arterial runoff.
A p-value <0.05 indicates a significant association between the variable and abnormal runoff (shaded columns). To the right, subgroups for each variable are presented with the corresponding p-value, indicating a significant difference in values for that characteristic across two or more runoff subgroups.
IQR, interquartile range; CVA, cerebrovascular accident; TIA, transient ischemic attack; PAD, peripheral artery disease.
Procedures
Non-invasive hemodynamic assessment
The ABI is the ratio of systolic pressure in the ankle to the systolic pressure in the arm (i.e. brachial pressure). The resting ABI was performed with patients supine for 5–10 minutes prior to checking the brachial pressure in the arms. Next, the ankle pressures were performed over the dorsalis pedis and posterior tibial arteries on each leg. The ABI was calculated by indexing the higher of the two ipsilateral ankle pressures divided by the higher of the arm pressures. In a similar fashion, the TBI was calculated using a single systolic pressure at the great toe compared to the higher of the two brachial systolic pressures. All measurements were performed using photoplethysmography (PPG) and in accordance with Intersocietal Accreditation Commission – Vascular Testing standards. 4 Peak systolic ankle pressure and pulse volume recordings were not included in this analysis. Exercise ABI testing was not performed among this cohort given the presence of resting limb pain or ulceration.
ABI assessment was attempted in all patients prior to invasive angiography. A non-diagnostic ABI was observed among 14 (16%) patients due to non-compressible arteries. The remaining 75 patients were subgrouped by ABI as follows: borderline or normal ABI, 0.9–1.4 (n=12); mildly reduced ABI, 0.7–0.89 (n=10); moderately reduced ABI, 0.4–0.69 (n=35); and severely reduced ABI, <0.4 (n=18). Owing to the limited number of patients in the borderline (0.9–0.99) group, these patients were added to the normal (1.0–1.4) ABI group to enhance statistical power between arterial patency subgroups.
Ipsilateral toe blood pressure was assessed by PPG at the time of ABI in 31 patients. A TBI ≥0.70 was considered normal as reported elsewhere.5–7 For a comparison to ABI subgroups, TBI was subgrouped as follows: normal TBI, ≥0.70 (n=3); mildly reduced TBI, 0.40–0.69 (n=5); moderately reduced TBI, 0.20–0.39 (n=15); severely reduced TBI, <0.20 (n=8).
Angiographic assessment
In this study, all patients received lower extremity angiography. Upon confirming patency of the aorto-iliac and femoro-popliteal arteries, detailed characterization of the infragenicular arteries was performed. Normally, the anterior tibial artery, posterior tibial artery, and peroneal artery originate from the popliteal artery, thereby providing a parallel network of arterial perfusion to the foot. Because occlusion of one or more of these major arteries below the knee (i.e. infragenicular arteries) often contributes to the development of CLI, angiographic results from these three arteries were analyzed. Angiographic results from each infragenicular artery were recorded as patent (i.e. ≤50% arterial narrowing), stenotic (i.e. >50% arterial narrowing), or occluded. A point was given toward a total runoff score, ranging from 0 to 3, for each non-occluded artery that reached the ankle. For example, if the anterior tibial, posterior tibial, and peroneal artery had continuous flow to the ankle, a score of 3 would be given. For outcomes analysis, abnormal runoff was defined as a runoff score <3. Patients were subgrouped by the number of infragenicular arteries providing lower leg runoff, as follows: none (n=9); one-artery (n=31); two-artery (n=33); and three-artery (n=16). Angiographic findings were summarized relative to demographic and historical information, vital statistics, laboratory values, and non-invasive hemodynamic assessment, including ABI and TBI.
Statistical analysis
Continuous variables were summarized as median with corresponding interquartile range (IQR). Categorical variables are summarized as percentages. Comparisons of continuous variables were performed with the Wilcoxon rank-sum test with continuity correction. Comparisons of categorical variables were performed with Fisher’s exact test. The Kruskal–Wallis test was used when comparing means among more than two continuous variables. To test the significance of patient characteristics between infragenicular arterial runoff subgroups, results were reported as p-values of multiple comparisons. A two-sided alpha level of 0.05 was used for all superiority testing. All data were analyzed and Figure 1 was generated using R software (Version 2.13.1; The R Foundation for Statistical Computing, 2011).
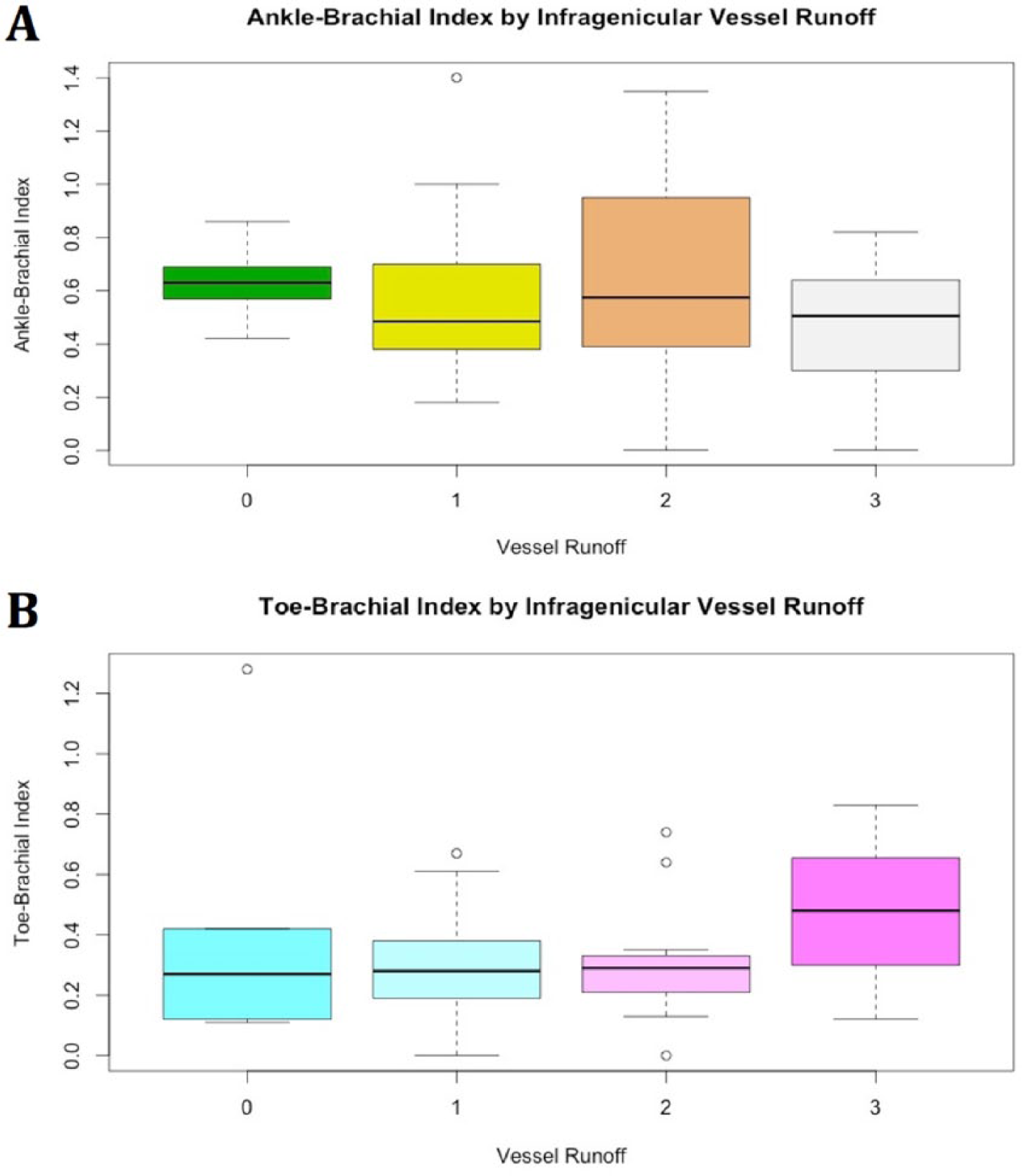
ABI and TBI by number of patent infragenicular runoff arteries. Panel A: Distribution of ABI by number of patent infragenicular runoff arteries among 75 patients with compressible ABI results. Panel B: Distribution of TBI by number of patent infragenicular runoff arteries among 31 patients with available results. In each plot, the dark horizontal line represents the result median contained within a colored box indicating the IQR. Outliers are represented by open circles.
Two-way analysis of variance (ANOVA) was used to compare the mean ABI based on various clinical factors, then assessed with Q-Q plots and residual plots for relevant model assumptions. Logistic regression was used to model associations between clinical factors and having less than three-artery infragenicular runoff. To avoid over-fitting of data, a stepwise model path with an analysis of deviance function (stepAIC function) was used for variable selection (MASS package) to simplify an initial model of 18 variables (Akaike Information Criterion = 71.4) to one of nine variables (Akaike Information Criterion = 57.0).
Results
Baseline characteristics
Among 89 patients presenting with CLI the median age was 66 years (IQR, 60–79), 39% were female, and 60% were Caucasian (Table 1). The population had several comorbidities, including diabetes mellitus (70%), current or former tobacco abuse (74%), coronary artery disease (63%), prior CVA/TIA (24%), and prior revascularization (35%) or amputation (18%) for advanced PAD. Based on presenting the severity of CLI, 23 (26%) patients presented at RC 4, 29 (33%) had RC 5, and 37 (42%) had RC 6 PAD (Table 2).
Baseline characteristics according to the number of patent infragenicular arteries.
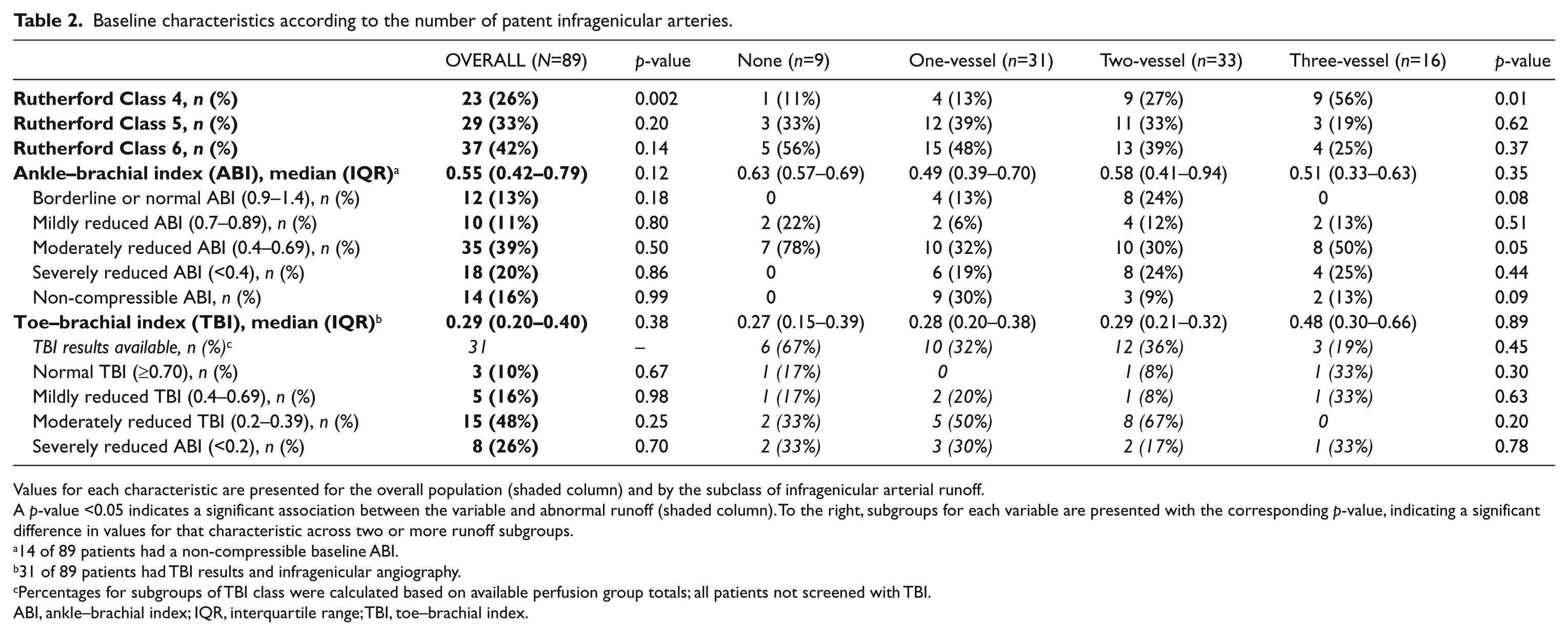
Values for each characteristic are presented for the overall population (shaded column) and by the subclass of infragenicular arterial runoff.
A p-value <0.05 indicates a significant association between the variable and abnormal runoff (shaded column). To the right, subgroups for each variable are presented with the corresponding p-value, indicating a significant difference in values for that characteristic across two or more runoff subgroups.
14 of 89 patients had a non-compressible baseline ABI.
31 of 89 patients had TBI results and infragenicular angiography.
Percentages for subgroups of TBI class were calculated based on available perfusion group totals; all patients not screened with TBI.
ABI, ankle–brachial index; IQR, interquartile range; TBI, toe–brachial index.
In univariate analysis, clinical factors associated with abnormal runoff included advanced age (p=0.02), lack of tobacco abuse (p=0.02), and reduced creatinine clearance (p=0.02) (Table 1). Patients with resting limb pain without tissue loss were less likely to have abnormal runoff (p=0.002) (Table 2). In univariable analysis, there was no difference in ABI (p=0.12) or TBI (p=0.38) results based on the presence of abnormal runoff (Table 2). In multivariable analysis, the odds of abnormal runoff were increased with a higher ABI at baseline (p=0.01), reduced creatinine clearance (p=0.02), and history of hyperlipidemia (p=0.048) (Table 3).
Association between ABI and abnormal lower extremity runoff.
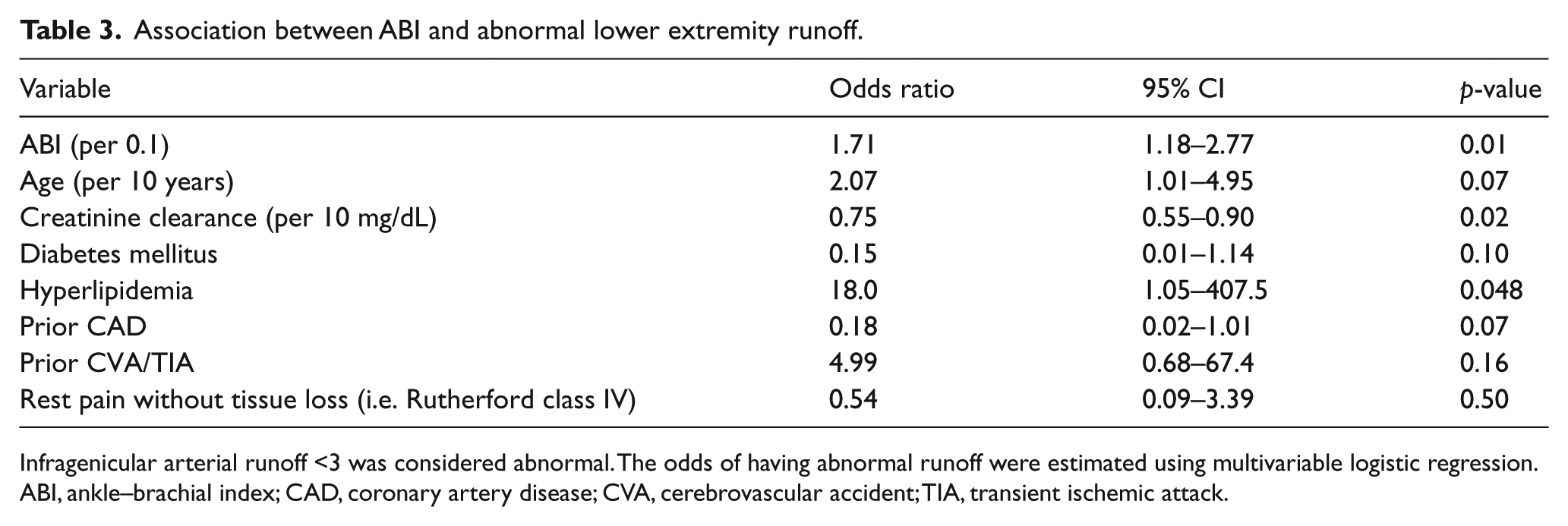
Infragenicular arterial runoff <3 was considered abnormal. The odds of having abnormal runoff were estimated using multivariable logistic regression.
ABI, ankle–brachial index; CAD, coronary artery disease; CVA, cerebrovascular accident; TIA, transient ischemic attack.
Non-invasive perfusion assessment
Among patients with a measureable result, the median ABI was 0.55 (IQR, 0.42–0.79) (Table 2). Overall, 70.8% had an ABI <0.9, whereas 13% had a normal ABI (0.9–1.4). Among our CLI population, a moderately reduced (0.40–0.69) ABI was most commonly observed (39%). The median TBI was 0.29 (IQR, 0.20–0.40) (Table 2). Among patients with TBI results, 97% had a TBI <0.7.
Overall, ABI did not have a significant linear correlation with TBI (p=0.20). Among 31 CLI patients with available ABI and TBI results, 19 (61%) had a TBI <0.7 and a non-compressible or resting ABI <0.9. Conversely, no patients with a borderline or normal ABI (0.9–1.4) had a normal TBI (≥0.7).
Non-invasive perfusion assessment and arterial runoff
Lower extremity angiography of the infragenicular arteries was performed at a median 10.5 days (IQR, 3–21) following office-based perfusion assessment. After excluding 14 patients with non-compressible ABI, the remaining ABI results from 75 patients were stratified by infragenicular runoff (Figure 1 and Table 2). When evaluated across all runoff subgroups, the highest median ABI was noted in the 0-artery runoff group (0.63; IQR, 0.57–0.69) (Figure 1 and Table 2). ABI was not significantly different across arterial runoff subgroups (p=0.35). Fewer patients had available TBI results (n=31). Across all runoff subgroups, TBI values trended higher as arterial supply to the distal extremity improved, particularly for patients with three-artery infragenicular arterial runoff who had the highest median TBI (0.48; IQR, 0.30–0.66) (Figure 1 and Table 2). Across infragenicular arterial runoff subgroups, there was no significant difference in TBI results (p=0.38).
Rutherford classification and infragenicular arterial runoff
Abnormal infragenicular arterial runoff was present among 14 of 23 (61%) without tissue loss (i.e. RC 4 CLI) and 59 of 63 (94%) with tissue loss (i.e. RC 5 and 6 CLI). In univariable analysis, RC 4 CLI was associated with reduced odds of abnormal runoff (OR 0.75, 95% CI 0.63–0.90; p=0.002). However, in multivariable analysis the presence of rest pain alone was no longer associated with abnormal runoff (p=0.50) (Table 3).
Discussion
While the ABI remains a key test in assessing PAD, important limitations exist when applying the test to CLI. Among a contemporary, real-world CLI population, 29% had near-normal or normal ABI, despite having significant infragenicular arterial disease. Additionally, those with the lowest number of patent infragenicular arteries tended to have the highest median ABI results. However, among patients with borderline to normal ABI results, concurrent TBI was abnormal in all. Furthermore, discrimination for abnormal runoff was improved somewhat by considering the clinical severity of CLI. In univariable analysis, rest pain alone tended to have reduced odds of having abnormal runoff although this association was not preserved in multivariable regression analysis. Overall, these results reinforce the need for a comprehensive assessment of CLI, particularly if a treatment strategy incorporating revascularization is considered.
The exact mechanism for near-normal to normal ABI in patients with CLI and isolated infragenicular arterial disease is unknown and likely multifactorial. Vascular calcification is associated with an abnormally elevated ABI (>1.4), and both are strongly associated with occlusive PAD. In the presence of heavily calcified, non-compressible infragenicular arteries characteristic of CLI and often associated with renal failure or diabetes, the ABI often delivers non-diagnostic results.8,9 In this study, worsening renal function was associated with reduced arterial runoff.
Aberrant ABI results may be inherent to how the test is performed. Variations in technique and expertise in interpreting test results limit the accuracy of the ABI. 10 The reproducibility of the test worsens with advanced arterial disease, with higher interobserver variability during ABI interpretation and may be a particularly relevant point for the CLI population. 10 When calculating the ABI, the difference in systolic blood pressures at the ankle may be large, with preserved pressure in one artery (e.g. anterior tibial/dorsalis pedis artery) relative to the other (e.g. posterior tibial artery). As a consequence, using the higher of the two ankle pressures may provide ostensibly normal or mildly abnormal ABI results that are, in fact, falsely negative. This consideration is particularly relevant when considering angiosome-directed revascularization whereby the goal includes restoration of in-line arterial flow to the wound bed. 11 Without clinical suspicion for arterial insufficiency and careful scrutiny of results, ABI could offer a somewhat distorted representation of arterial patency below the knee (Figure 1).
Collateral circulation may also produce a normal or nearly normal ABI (0.9–1.4) at rest, even among those with complete occlusion of all three infragenicular arteries. Diffuse distal collateralization, particularly from the peroneal artery, may maintain sufficient systolic pressure to the ankle but not necessarily sustain sufficient flow to the distal foot. Our study’s finding that all patients with borderline or normal ABI (0.9–1.4) had abnormal TBI (<0.7) supports this consideration. Therefore, suspicion for CLI should not be simply dismissed by the presence of a near-normal, borderline, or normal ABI; these suspicions should be confirmed with additional testing, such as TBI. The use of TBI in these patients may be helpful in confirming clinical suspicions of severe arterial insufficiency, particularly when arterial patency below the knee is incomplete.
Diabetes has been reported as a dominant factor associated with non-compressible, calcified arteries often represented by an elevated ABI. 10 In our cohort, 71% of those with non-compressible arteries were diabetic. Among our cohort, a history of diabetes was evenly distributed across perfusion subgroups. Although diabetes was not independently associated with abnormal runoff, many of our patients with CLI and two- or three-artery runoff were diabetic, highlighting the role of microvascular arterial disease in the development of CLI despite preserved macrovascular flow (Table 1).
ABI provides perfusion assessment to the ankle, and not the foot, where many ulcers occur. Thereby, ABI may underestimate the extent of microvascular occlusive disease, 2 particularly among those with diabetes or chronic kidney disease. In such circumstances, TBI may supplement the evaluation of distal limb perfusion. The supportive role of TBI in evaluating PAD, particularly in the presence of a normal ABI, has been confirmed elsewhere.9,10 Studies among patients with suspected PAD have used lower normal limits ranging from <0.6 to <0.75, with reported sensitivity ranging from 90% to 100% and specificity of 65% to 100%. 12 Among healthy populations not suspected to have PAD, normal lower limits for TBI range from 0.49 to 0.74. 12 In our cohort of CLI patients, 13% of subjects had a borderline or normal ABI (0.9–1.4), 10% had a normal TBI, but none had both results simultaneously. TBI did not predict abnormal runoff in multivariable analysis, likely due to limited statistical power. However, patients with three-artery runoff tended to have a distinctly higher median TBI result compared to those with abnormal runoff. Qualitatively this threshold phenomenon could potentially aid in identifying abnormal runoff and should be tested prospectively in a larger population.
Limitations
Variations of brachial–ankle blood pressure relationships have been suggested to be more sensitive than the current standard for ABI assessment,2,13 although were not evaluated in this analysis. This analysis focused on the ABI and not ankle systolic pressure, which is a recommended non-invasive physiologic confirmation of CLI. 6 Furthermore, the addition of pulse volume recordings and Doppler waveforms to the assessment of CLI can help in localizing arterial disease. ABI had no association with abnormal runoff in univariable analysis, although it had a significant association in multivariable analysis. An interaction effect is suspected between ABI and other covariates within the multivariable model. Given the retrospective nature of data collection and subjective interpretation of disease severity, opportunity for bias exists. Our analysis was limited by relatively few patients with both TBI and ABI results. Prospectively collected data on ABI and TBI at the time of presentation for all patients would offer improved statistical power. Efforts to improve the grading of CLI and limit biases from retrospective analyses, such as this one, are fortunately underway in the form of prospective registries and trials sure to enhance the understanding of non-invasive and angiographic assessment in CLI.14,15
Conclusion
A significant portion of patients with CLI and below-knee disease present with a normal or borderline ABI. In fact, nearly one-third of patients with ischemic tissue loss had a normal or borderline ABI. Assessment of clinical severity and use of TBI likely enhance the diagnostic evaluation of CLI over ABI alone, especially among those individuals with tissue loss. These findings may have further relevance among the elderly and those with diabetes or chronic kidney disease. Future prospective studies are needed to evaluate the complimentary role of non-invasive testing and angiography in this population. Such studies may offer insights that benefit the evaluation of CLI patients, leading to improved therapies and outcomes. Until these results are available, a high index of suspicion for CLI must be maintained in the face of normal or near-normal ABI results.
Footnotes
Declaration of conflicting interest
Dr. Shishehbor is a consultant for Abbott Vascular, Medtronic, and BARD, however, he does not receive any personal honoraria from this work. Drs. Bunte and Jacob as well as Mr. Nudelman have no conflicts of interest to report.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
