Abstract
Objective:
We conducted a prospective pilot study in patients with diabetes to analyse the interobserver reliability of the ankle–brachial index, toe–brachial index and distal pulse palpation depending on the training of the professional involved.
Materials and Methods:
The ankle–brachial index, toe–brachial index and distal pulses were assessed by three clinicians with different levels of experience on the same day. Measurements were supervised and recorded by a fourth clinician.
Results:
Twenty-one patients (42 ft) were included in this study. We observed moderate agreement between clinicians in the palpation of posterior tibial arteries (K = 0.45, p < 0.001) and low agreement in dorsalis pedis arteries (K = 0.33, p < 0.001). The measurement of ankle–brachial index had moderate agreement between clinicians in patients with medial arterial calcification (K = 0.43, p < 0.001) and low agreement in patients with normal ankle–brachial index (K = 0.4, p < 0.001). The measurement of toe–brachial index had moderate agreement between clinicians in patients with a normal toe–brachial index (K = 0.4, p < 0.001) and in patients with medial arterial calcification (K = 0.60, p < 0.001).
Conclusion:
Palpation of distal pulses, ankle–brachial index and toe–brachial index determination in patients with diabetes are not highly reproducible and reliable between clinicians with different levels of experience under routine conditions.
Keywords
Introduction
Diabetes mellitus is an important risk factor for the development of peripheral arterial disease (PAD). 1 The prevalence of PAD in patients with diabetes among different studies varies between 8% and 33%. 2 Patients with diabetes and PAD have a greater risk of adverse outcomes including progression to critical limb ischaemia, amputation and death. 3 Despite the correlation between PAD and diabetes, PAD is underdiagnosed in primary care practice and physician awareness of the PAD diagnosis is relatively low. 4 The assessment of peripheral vascular status in a clinical setting includes a physical examination which includes the palpation of distal pulses combined with non-invasive tests such as the ankle–brachial index (ABI) and toe–brachial index (TBI). 5
It is not clear whether the palpation of distal pulses, ABI and TBI in patients with diabetes can be reliably determined by non-expert clinicians, or whether their use should be restricted to specialist clinicians. In patients with diabetes, we observed some situations that could affect the interpretation of these tests such as calcification in the pedal arteries or swelling around the ankle, especially in patients with neuropathy, where anatomical considerations are important and can affect distal pulse palpation. 6 To date, methodological research on this topic is limited and it is important to know if acquired skill or practice can favourably influence the amount of observer variability or error among these tests. Recording a pulse as present when it is absent (underdiagnosis) can result in a dangerous delay in diagnosis with a subsequent influence on treatment in these patients. 7 Our study aim was to analyse the interobserver reliability of ABI, TBI and distal pulse palpation in patients with diabetes depending on the training of the professional involved.
Material and methods
We conducted a cross-sectional pilot study which included patients with diabetes attending our Diabetic Foot Unit as outpatients. Participants were over 18 years of age with type 1 or type 2 diabetes mellitus. Patients with diabetic foot ulcers, patients without a big toe where it had not been possible to measure the TBI, patients who had a vasomotor condition such as Raynaud’s disease were excluded. The ABI, TBI and distal pulses were assessed by three clinicians on the same day using a manual 8 MHz Doppler (Doppler II, Huntleigh Healthcare Ltd, South Glamorgan, UK), and the toe systolic pressure was taken with a digital plethysmograph (SysToe, Atys Medical, Quermed, Madrid, Spain).
The ABI and TBI were calculated, separately for each leg, by dividing the higher of the two systolic pressures (from posterior tibial and dorsalis pedis arteries) at the ankle or toe pressure readings by the highest brachial reading between the right and left arms. Each observer first palpated distal pulses and recorded each as present or absent. Subjects were placed in a supine position for at least 10 min before starting the measurements in a heated room (20°C–23°C).
We classified the clinicians into three levels of experience in the management of patients with diabetes: inexperienced (no experience), medium experience (6 months but less than 1 year) and experienced (more than 5 years). Measurements were supervised and recorded by a fourth clinician.
We considered PAD to have a TBI value less than 0.7; normal TBI values were between 0.7 and 0.99, and TBI ⩾ 1 was considered as distal arterial calcification. We considered PAD to have an ABI value less than 0.9; normal ABI values were between 0.9 and 1.39, and an ABI value of ⩾1.4 was considered to represent poorly compressible vessels related to medial arterial calcification (MAC). 8 To analyse the correlation between qualitative variables, we applied the kappa coefficient, using the Landis and Koch criteria to study the strength of association. 9
Results
A total of 21 patients (42 ft) were included in this study, of whom 15 (71.4%) were men and 6 (28.6%) were women; the mean age of patients was 67 ± 8.7 years, with a mean diabetes duration of 16.7 ± 9.8 years. All patients had type 2 diabetes. Mean glycated haemoglobin (HbA1c) was 52 ± 8 mmol/mol (6.9 ± 0.9%) and the mean glucose level was 7.3 ± 1.1 mmol/L. In our study, 18 patients (85.7%) had neuropathy, 19 patients had hypertension (90.5%), 1 patient had nephropathy (4.8%), 7 patients had retinopathy (33.3%), 17 patients had dyslipidaemia (81%) and 6 patients were smokers (28.6%). Figure 1 illustrates the kappa coefficient between clinicians in the interpretation of distal pulses palpation, ABI and TBI.
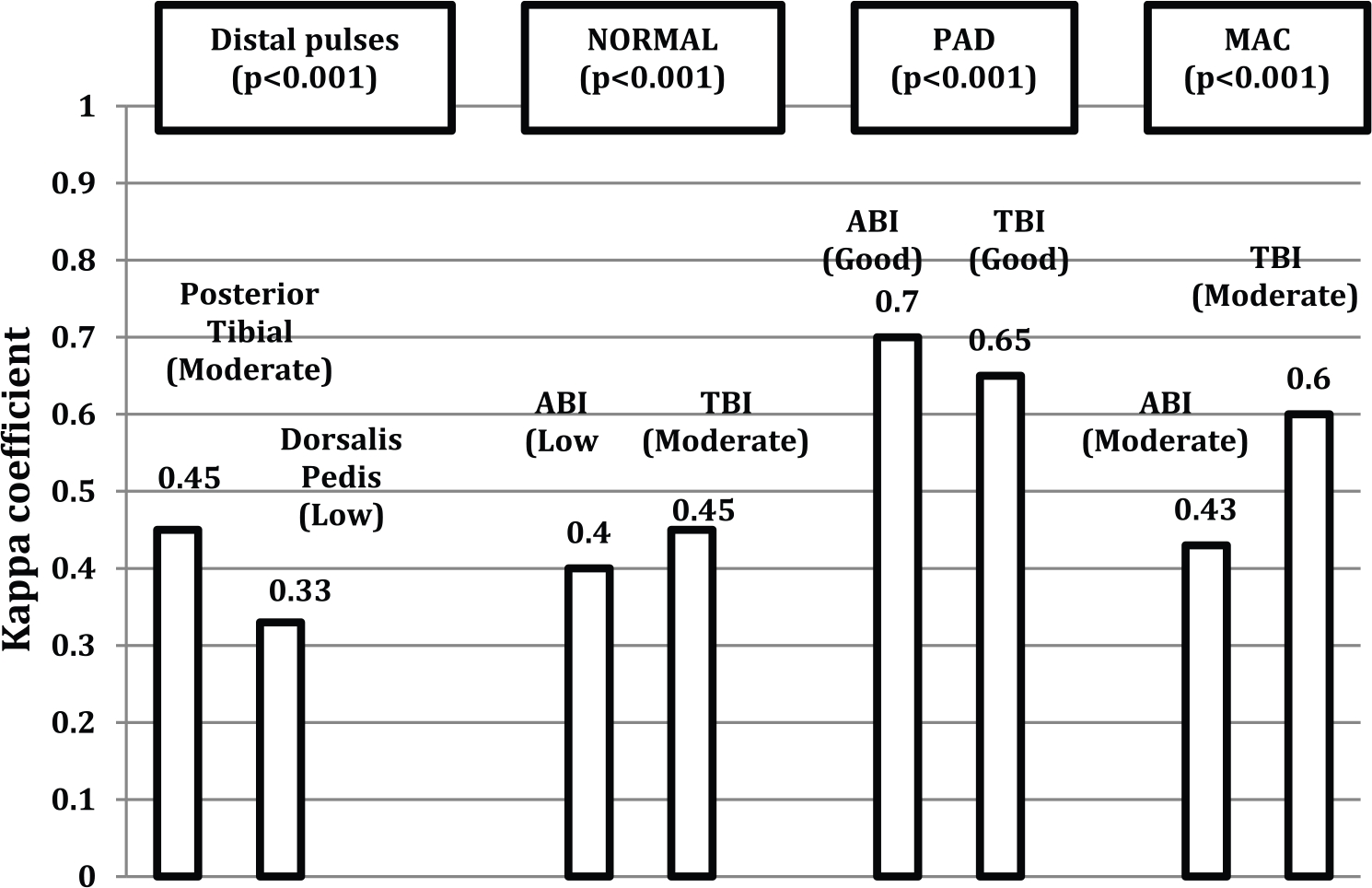
Kappa coefficient between clinicians in the interpretation of distal pulses palpation, the ankle–brachial index (ABI) and the toe–brachial index (TBI).
Related to palpation of distal pulses, 29 (69%), 18 (42.9%) and 25 (59.5%) posterior tibial pulses and 34 (81%), 23 (54.8%) and 26 (61.9%) dorsalis pedis pulses were present for the very experienced, medium-experienced and inexperienced clinicians, respectively. Of these 29 (69%) posterior tibial pulses present for the very experienced clinician, 11 (37.9%) and 8 (27.6%) were absent for the medium-experienced and inexperienced clinicians, respectively. Of the 13 posterior tibial pulses absent for the very experienced clinician, all were absent for the medium-experienced and 4 (30.8%) were present for the inexperienced clinician.
Of the 34 (81%) dorsalis pedis pulses present for the very experienced clinician, 12 (35.3%) and 11 (32.4%) were absent for the medium-experienced and inexperienced clinicians, respectively. Of the eight dorsalis pedis pulses absent for the very experienced clinician, one (12.5%) and three (37.5%) were present for the medium-experienced and inexperienced clinicians, respectively.
Conclusion
In our pilot study, we observed that the palpation of distal pulses in patients with diabetes is operator dependent among clinicians with different levels of experience. We observed moderate agreement between clinicians in the palpation of posterior tibial arteries (K = 0.45, p < 0.001) and low agreement in dorsalis pedis arteries (K = 0.33, p < 0.001). Four posterior tibial (30.8%) and three dorsalis pedis (37.5%) pulses, respectively, were absent for the very experienced clinician but these were present for the inexperienced clinician (probably underdiagnosis). Nevertheless all posterior tibial pulses absent for the very experienced clinician were absent for the medium-experienced clinician and only one dorsalis pedis (12.5%) absent for the very experienced clinician was present for the medium-experienced clinician. These results suggest the same as a previous study of Meade et al., 10 that when palpating foot pulses ‘acquired skill or practice may favourably influence the amount of observer variability or error’. Based on our results, distal pulse palpation by an inexperienced clinician has to be critically interpreted. The presence of distal pulses examined by an inexperienced clinician does not rule out the presence of PAD, and for this situation, it is necessary to combine the palpation of distal pulses with non-invasive screening like ABI and TBI.
We observed that the measurement of ABI and TBI is operator dependent among our clinicians, especially in the diagnosis of normal patients and patients with MAC. Nevertheless, we observed good agreement between clinicians in the diagnosis of PAD with the ABI and TBI.
Holland-Letz et al. 11 observed that reproducibility of the ABI measurement was high, in a cohort of normal elderly patients, among observers with different training. In this study, no elevated ABI values of >1.40 related with MAC were recorded. It is important to know that calcification in the pedal arteries is common in patients with diabetes, especially those with neuropathy. 6 In our study population, 85.7% of patients had neuropathy and 50% of legs explored (n = 21) had an ABI related to MAC by the experienced clinician. We observed a moderate agreement between clinicians in patients with MAC (K = 0.43, p < 0.001). On the other hand, TBI is the method of choice in the presence of MAC and we observed that 47.5% of legs explored (n = 19) had a TBI related to MAC, and agreement between clinicians was moderate (K = 0.60, p < 0.005). Holland-Letz et al. 11 reported that reproducibility of ABI may be easier for ‘non-experts’ in normal arteries, compared with arteries that are hard to find or if the pressure is lower, but we observed low agreement between clinicians in our patients with a normal ABI (K = 0.40, p < 0.001).
Based on our results, we think that ABI and TBI determinations are not highly reproducible and reliable under routine conditions in patients with diabetes, especially in patients with normal ABI or TBI and in patients with MAC. We have not studied the reproducibility of ABI and TBI measurements among experienced clinicians but we think that the measurement of both tests in patients with diabetes should not be done with little training, as Holland-Letz et al. 11 suggested based on their results with normal elderly patients. A previous pilot study suggested that a significant lack of knowledge exists among internal medicine residents regarding the ABI test. 12 We fully agree, as in this study, that training should include practical instruction on performing an ABI and TBI as well as didactic teaching on both test calculation and interpretation guidelines.
Our study has several limitations. First, the characteristics of our study population, in a specialised diabetic foot unit, are probably different than in a basic health-care unit where the prevalence of PAD is lower. Second, this reliability study can only comment on the interobserver reliability associated with the tests, but not definitively on which observer is correct in their findings, because we did not compare these tests with a gold standard and three is the small sample size. Further studies are required to provide more specific recommendations.
Key messages
Palpation of distal pulses and ABI and TBI determination in patients with diabetes are not highly reproducible.
The presence of distal pulses examined by an inexperienced clinician does not rule out the presence of PAD.
The measurement of ABI and TBI is operator dependent especially in the diagnosis of normal patients and patients with MAC or DAC.
Footnotes
Acknowledgements
The authors thank the study participants and staff for contributing to this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
