Abstract
Hypoxia inducible factor (HIF) stabilization by HIF-prolyl hydroxylase (PHD) inhibitors may improve ischemic conditions such as peripheral artery disease (PAD). This multicenter, randomized, placebo-controlled study evaluated the safety and efficacy of GSK1278863 (an oral PHD inhibitor) in subjects with PAD. The study assessed two active treatment paradigms: single dosing and subchronic daily dosing (300 mg single dose and 15 mg daily for 14 days, respectively). Neither regimen improved exercise performance compared with placebo (change from baseline in the 6-minute walk test (6MWT; feet), (GSK1278863, placebo): single dose (–46, –44), p=0.96; repeat dose (9, 8), p=0.99; change in number of contractions to onset of claudication (goniometry): single dose (4, –1), p=0.053; repeat dose (–2, 1), p=0.08). A calf-muscle biopsy substudy showed no increases in mRNA or protein levels of HIF target genes. More subjects receiving GSK1278863 than placebo experienced adverse events, particularly following the 300 mg single dose. Thus, assessing the safety of GSK1278863 in this setting would require a larger population exposed to the agent for a longer duration. These data do not support a benefit of GSK1278863 in PAD using the regimens tested.
Keywords
Introduction
Peripheral artery disease (PAD) has an estimated worldwide prevalence of 4–10% in adults, increasing to 15-20% in people over 70 years old, and affects around 27 million people in Europe and North America. 1 Supervised exercise therapy improves symptoms of claudication and increases exercise capacity by various mechanisms, including ischemic preconditioning (IPC). 2 Nonetheless, there is a need for new therapeutic options, as many patients remain symptomatic and functionally limited despite available treatment strategies.
Hypoxia-inducible factor (HIF) is a key regulator in the induction of genes facilitating cellular adaptation and survival during hypoxia (erythropoietin (EPO), vascular endothelial growth factor (VEGF), glucose transporter-1 (GLUT1), pyruvate dehydrogenase kinase 1 and 4 (PDK1 and PDK4), and cyclooxygenase-4 (COX4-1 and COX4-2))3–12; therefore, HIF stabilization by prolyl hydroxylase (PHD) inhibition may represent a therapeutic target in the treatment of ischemic conditions such as PAD.
GSK1278863 is a potent, selective, orally available inhibitor of HIF-PHDs-1, 2, and 3.8,9 Pharmacologic inhibitors of PHD, including GSK1278863, that mimic the HIF-stabilizing effects of ischemia are being developed as therapies for the treatment of anemia of chronic disease because of their ability to stimulate erythropoiesis by inducing EPO production and improving iron availability. 13 The activity of the compound may have unique application in other ischemic-related disease states such as PAD in which modulating oxygen delivery and utilization could be critical factors in the treatment of the condition.
In subjects with chronic ischemia and in pre-clinical models of aging, evidence suggests that there is a decrease in HIF activity resulting in the inability to adequately compensate for decreased oxygen availability. 14 Further, Van Weel et al. have reported down-regulation of the HIF targets, VEGF and stromal cell-derived factor-1 (SDF-1), in the muscle tissue from the limbs of subjects with chronic ischemia associated with PAD. 15 In the preclinical setting, PHD inhibition by siRNA-mediated knockdown improved vascularization following hindlimb ischemia in mice, and this was associated with HIFα accumulation and upregulation of VEGF and eNOS expression in skeletal muscle. 16
In preclinical work utilizing a PHD inhibitor similar to GSK1278863, oral administration of the inhibitor for 4 weeks in a mouse hindlimb ischemia model demonstrated engagement of the PHD/HIF axis in the soleus muscle as evidenced by elevation of HIF gene targets (e.g. VEGF, PDK1, GLUT1), and small increases in both the phosphocreatine/Adenosine triphosphate (PCr/ATP) ratio and the wheel-running distance (unpublished results). These results and supporting scientific evidence from other laboratories supported investigation of GSK256883 in the clinical setting in an exploratory effort to reveal evidence for muscle effects in patients with PAD.
We hypothesized that subjects with PAD experiencing repeated muscular ischemia may benefit from acute or subchronic pharmacologic augmentation of HIF activity in muscle, thereby delaying the onset and increasing the maximal tolerance of ischemic symptoms during exercise.
The primary objectives of this exploratory clinical study were to assess the safety, tolerability, and pharmacokinetics (PK) of GSK1278863 administered as a single dose and as subchronic dosing in subjects with claudication-limited PAD. In addition, the study assessed the effect of GSK1278863 on several performance measures, using functional measures of calf muscle endurance, fatigability, and timed walking distance, including the bilateral heel rise (goniometry) and 6-minute walk tests (6MWTs). The effect of GSK1278863 on HIF stabilization and activation of HIF-dependent target genes was evaluated based on the hematologic profile and level of circulating EPO. A substudy was conducted to evaluate changes in the mRNA and protein levels of HIF target genes in skeletal muscle biopsies.
Methods
Study design
This multicenter, randomized, single-blind, parallel-arm, placebo-controlled study evaluated a single dose of GSK1278863 (300 mg) followed by a 14-day subchronic treatment (15 mg) in subjects with claudication-limited PAD. Twelve investigative centers in the United States participated in this study from 15 October 2010 to 1 November 2011. The study was conducted in compliance with the International Conference on Harmonisation – Good Clinical Practice (ICH-GCP) guidelines and the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board at all centers, and written informed consent was obtained from each subject (NCT01673555).
Patients were randomized to receive a single dose of GSK1278863 300 mg or placebo within 21 days of screening. Pharmacokinetics/pharmacodynamics (PK/PD) evaluations were performed at baseline and post-dosing. After a 14-day washout period, the randomized cohort was treated with GSK1278863 15 mg or placebo daily for 14 days. PK/PD evaluations were performed at baseline, after the first dose, and at the end of the 14-day treatment period. A follow-up evaluation was scheduled approximately 30 days after the end of treatment (Figure 1).
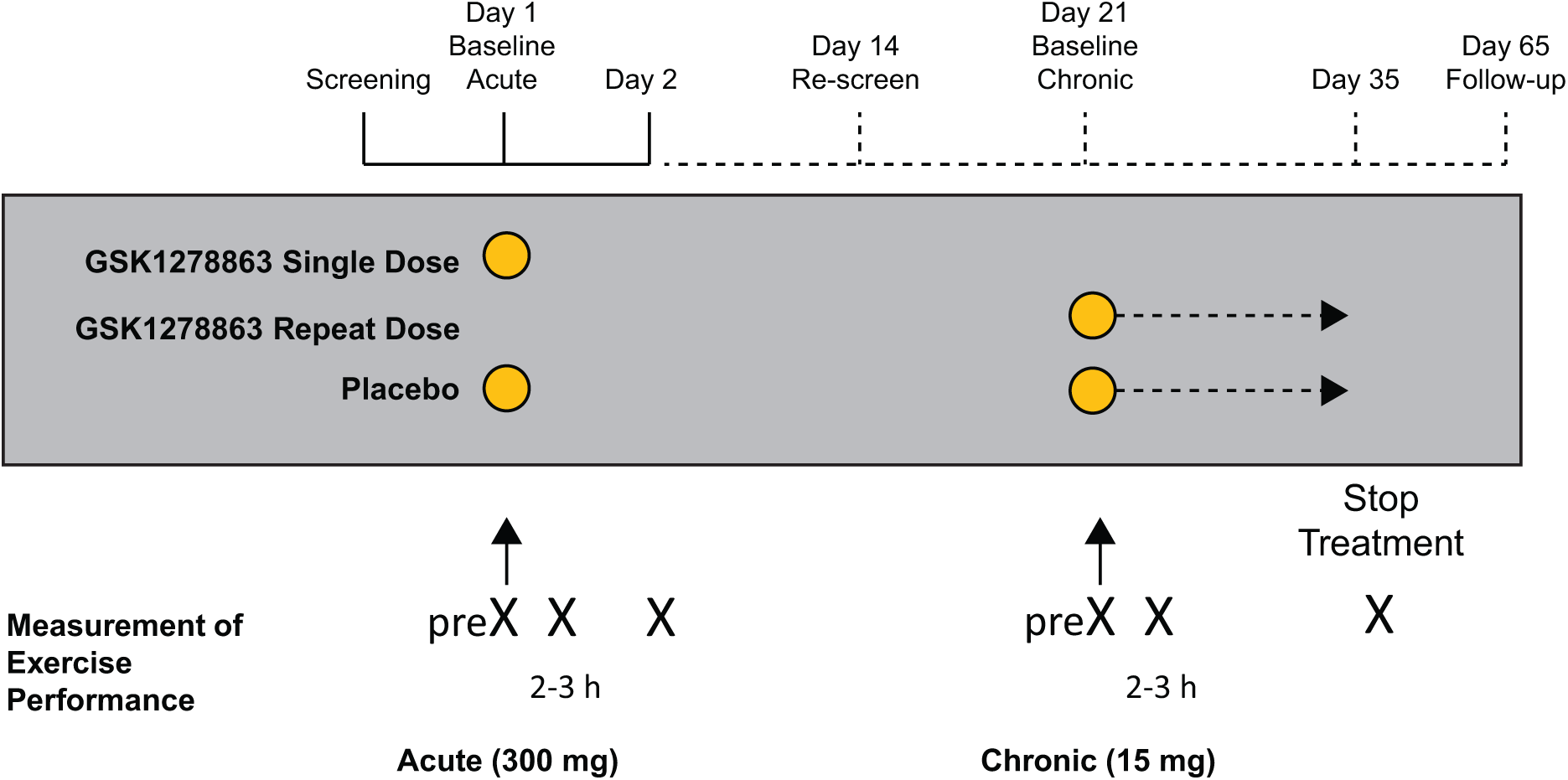
Study design.
Early termination was considered any time between administration of the single dose and end of the repeat dose on day 14. The investigators and subjects were blinded; GlaxoSmithKline internal personnel were not blinded. A muscle-biopsy substudy was conducted at selected sites to determine if GSK1278863 induced detectable increases in HIF target gene expression in muscle.
Dose rationale
GSK1278863 dose selection was based on PK/PD data on erythropoiesis from healthy subjects and extrapolation of rat quantitative whole-body autoradiography (QWBA) for estimated drug levels in human muscle. The acute single dose and subchronic dosing regimens were selected to maximize muscle exposure to GSK1278863 and to avoid increases in erythropoiesis-related blood volume and viscosity. Specifically, 300 mg was the highest single dose demonstrated to be safe in healthy volunteers that produced a serum EPO level increase 3–4 hours post-dose (without an increase in hemoglobin up to day 14), suggesting transcriptional changes could also occur in skeletal muscle simultaneously. The 15 mg dose for 14 days in the repeat-dose regimen was the highest dose at which neither the hemoglobin level nor elevation rate exceeded 15.0 and 1.0 g/dL/week, respectively, in healthy volunteers. In that study, a significant increase in serum EPO level suggested the potential for activation of HIF target genes in skeletal muscle.
Furthermore, data from tissue exposure in rats and in vitro enzyme inhibition assays were used in a physiologically based PK model (PBPK) to simulate potential levels of PHD inhibition at the site of action (i.e. human skeletal muscle), based on predicted levels of local drug concentrations. The model’s prediction of PHD inhibition up to 80% in human skeletal muscle was considered sufficient to meet the study objectives.
Subjects
Men and women ≥40 years of age with PAD, defined as an ankle–brachial index (ABI) ≤0.90 at rest in at least one leg (index leg), which had claudication for at least 3 months before screening, were enrolled. The ABI calculation utilized the higher (among the two arms) systolic blood pressure measurements and the highest ankle vessel pressure (the dorsalis pedis or posterior tibial arteries in each ankle). No additional evaluations were performed to assess for non-atherosclerotic forms of PAD.
Key exclusion criteria included: recent transient ischemic attack (TIA), cerebrovascular accident (CVA), unstable angina, or acute myocardial infarction (MI) within 6 months prior to randomization; recent peripheral revascularization (within 6 months prior to screening) or possible requirement of revascularization during the course of the trial; critical leg ischemia, defined as pain at rest or ischemic ulceration (Fontaine stages 3–4); elevated baseline hemoglobin level (men or postmenopausal women, >15.5 g/dL; premenopausal women, >14.5 g/dL); retinopathy; poorly controlled hypertension (defined as seated blood pressure >160/95 mmHg); comorbidities limiting exercise tolerance; creatinine >2.5 mg/dL or undergoing hemodialysis; venous thrombosis; and gastrointestinal bleeding.
Subjects experiencing new-onset gastrointestinal bleeding, significant abdominal pain, change in blood pressure, and hemoglobin level >17 g/dL were to be withdrawn.
Safety and efficacy endpoints
The primary study endpoint was the clinical safety and tolerability profile of GSK1278863, including spontaneous adverse event (AE) reporting, electrocardiograms (ECGs), vital signs, nursing/physician observation and clinical laboratory values, including hematologic parameters. The secondary efficacy PD/biomarker endpoints included evaluation of the bilateral heel rise test, maximal distance covered during the 6-MWT, and changes in EPO, hemoglobin, and lipid levels. The exploratory endpoints included: changes in high-sensitivity C-reactive protein (hsCRP) and in the expression of HIF target gene mRNA (COX4, GLUT1, hemoxygenase-1 (HO1), PDK1, PDK4, and VEGF) and proteins (GLUT1, HIF-1, HO1, VEGF, and inducible nitric oxide synthase (iNOS)) in muscle biopsy samples.
Pharmacokinetic and pharmacodynamic assessments
Single-dose period
EPO levels were assessed pre-dose and 2.5, 3, and 24 hours post-dose, and samples for PK were assessed pre-dose and 1, 2, 3.5, and 24 hours post-dose. Hemoglobin and hsCRP levels were assessed pre-dose and 24 hours post-dose. The 6-MWT and bilateral heel rise test were performed pre-dose and 24 hours post-dose.
Repeat-dose period
EPO levels were assessed pre-dose, 2.5 and 3 hours after the first dose, and at the end of the 14-day treatment. Samples for PK were assessed pre-dose, at 1, 2 and 3.5 hours, and at the end of the 14-day treatment. Hemoglobin, hsCRP, lipid levels, and the 6-MWT and bilateral heel rise test were performed pre-dose and at the end of the 14-day treatment.
Muscle biopsy substudy
To assess HIF target genes and proteins, biopsy of the gastrocnemius muscle was done using a method similar to that of Vigano et al. 17 and Bao et al., 18 both at baseline and on day 14 prior to the study drug administration and the 6MWT.17,18
Bilateral heel rise test
After familiarization with the test at the screening visit, calf muscle endurance in a standardized bilateral heel rise test was assessed using the number of heel rises at the onset of claudication, total number of heel rises, time to onset of claudication, time to intolerable claudication pain or maximum calf muscle fatigue, and Heel Rise Work Index (HRWI) – and adjudicated by a central core laboratory. 19
Six-minute walk test
The test involved the subject walking at a self-selected pace for 6 minutes through a predefined walking course of 50 feet with cones marking the boundaries; there was a rest for a minimum of 1 hour between the heel rise test and 6MWT. 20
Blood sample assessment
Blood samples for PK were evaluated by GSK Worldwide Bioanalysis (Upper Merion, PA, USA). Plasma samples were analyzed for GSK1278863 and six of its metabolites using a validated analytical method based on protein precipitation, followed by high-performance liquid chromatographic tandem mass spectrometric (HPLC/MS/MS) analysis. Biochemical and biomarker analyses were performed by Quest Diagnostics (Valencia, CA, USA).
Statistical analysis
A sample size of 44 (22 subjects/group) was specified to achieve a final sample size of 18 subjects per group, providing 88% power to detect an increase of eight heel rises (SD, 7.34) and 25% power to detect a 39-meter increase in the 6-minute walking distance (SD, 88.77).21,22 Planned analyses were performed after study completion and database freeze or unblinding. PD endpoints, after loge transformation, were analyzed using a mixed-effects model including a term for regimen, visit, interaction of regimen and visit, and subject as random effects. All statistical analyses were the responsibility of Clinical Statistics, GlaxoSmithKline, Philadelphia, PA, USA and done with SAS 9.2 (Cary, NC, USA). No alpha adjustments for multiple comparisons of efficacy endpoints were specified in this early-stage exploratory study.
Results
Subject disposition and baseline characteristics
In total, 46 subjects (GSK1278863, 26; placebo, 20) were randomized and included in the safety population; 42 (91%) subjects completed the study: two (8%) subjects from the GSK1278863 treatment group were withdrawn due to AEs and one subject each from the GSK1278863 and placebo groups withdrew consent (Figure 2). Demographic characteristics were similar between the GSK1278863 and placebo groups (Table 1).
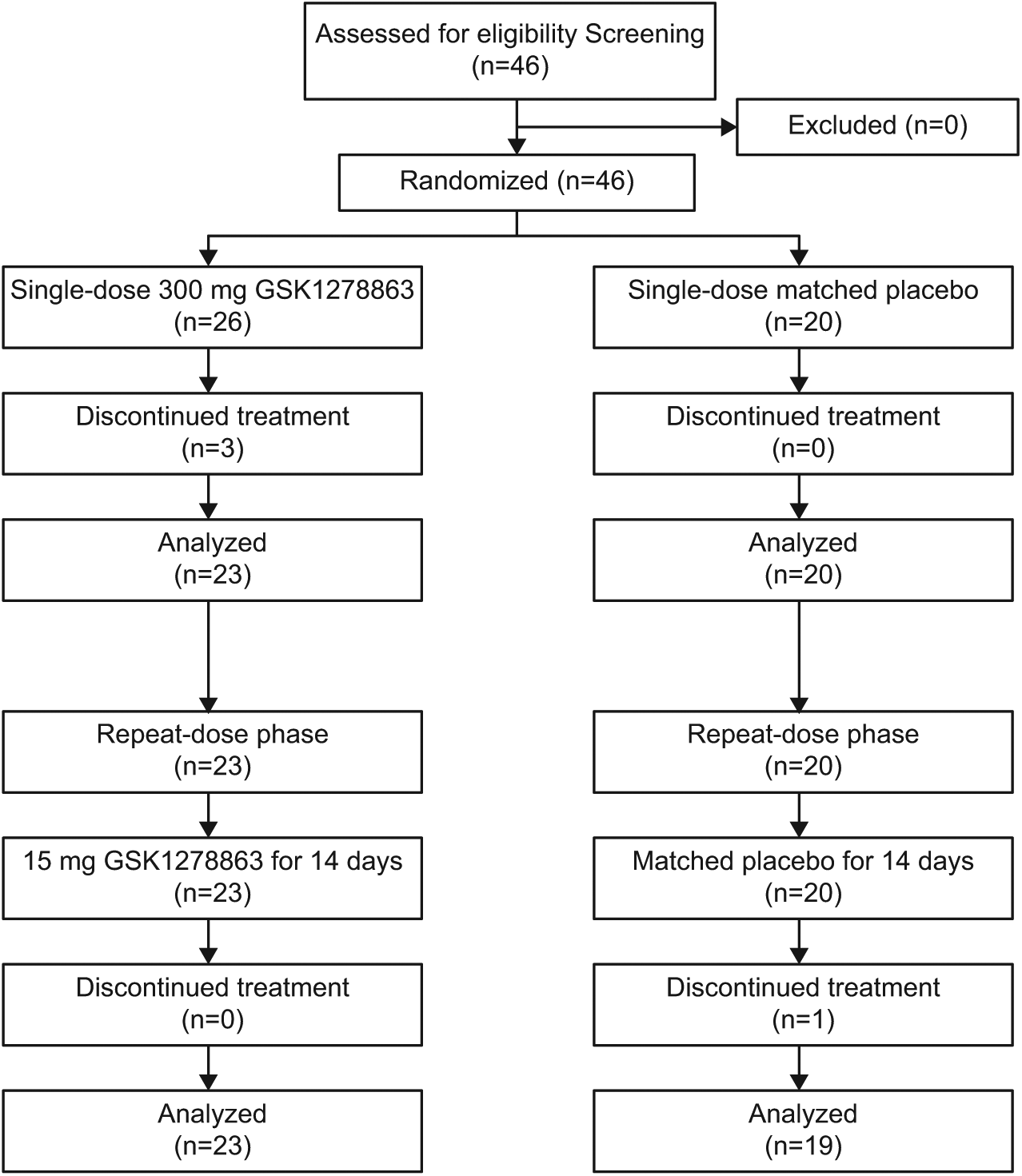
CONSORT flow diagram.
Baseline demographics.
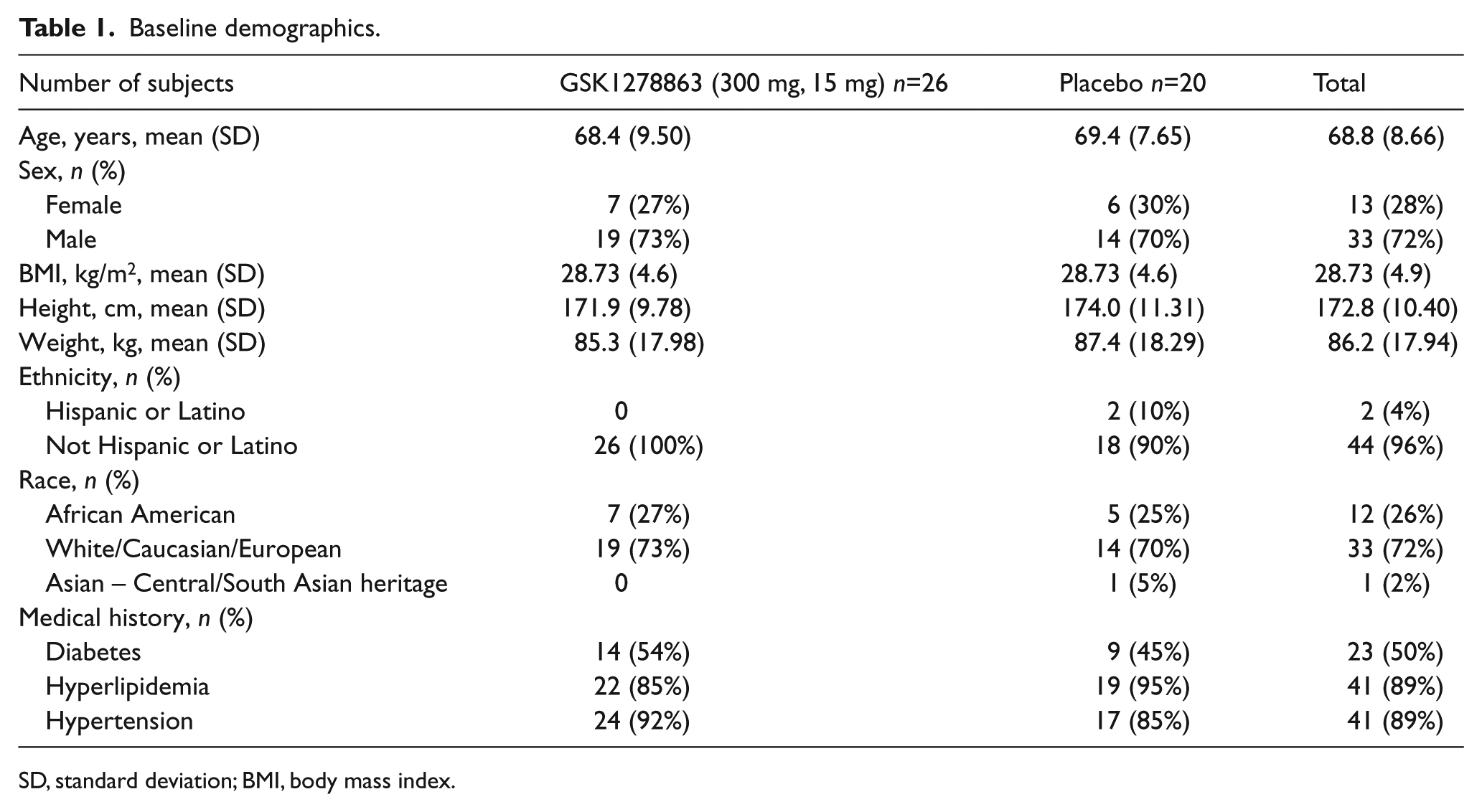
SD, standard deviation; BMI, body mass index.
Safety results
Adverse events occurred in two subjects (4%) during screening, 14 subjects (54%) during single-dose GSK1278863 treatment, six subjects (30%) during single-dose placebo treatment, nine subjects (39%) during repeat-dose GSK1278863 treatment and three subjects (16%) during repeat-dose placebo treatment. Three subjects (two on single-dose GSK1278863 and one on placebo) experienced serious adverse events (SAEs); no deaths were reported during the study.
Table 2 displays all the AEs that occurred in >1 subject (all on GSK1278863: pain in extremity, dyspepsia, diarrhea, upper respiratory tract infection, and fatigue), according to treatment and study period, as well as a description of the SAEs.
All the AEs that occurred in >1 subject.
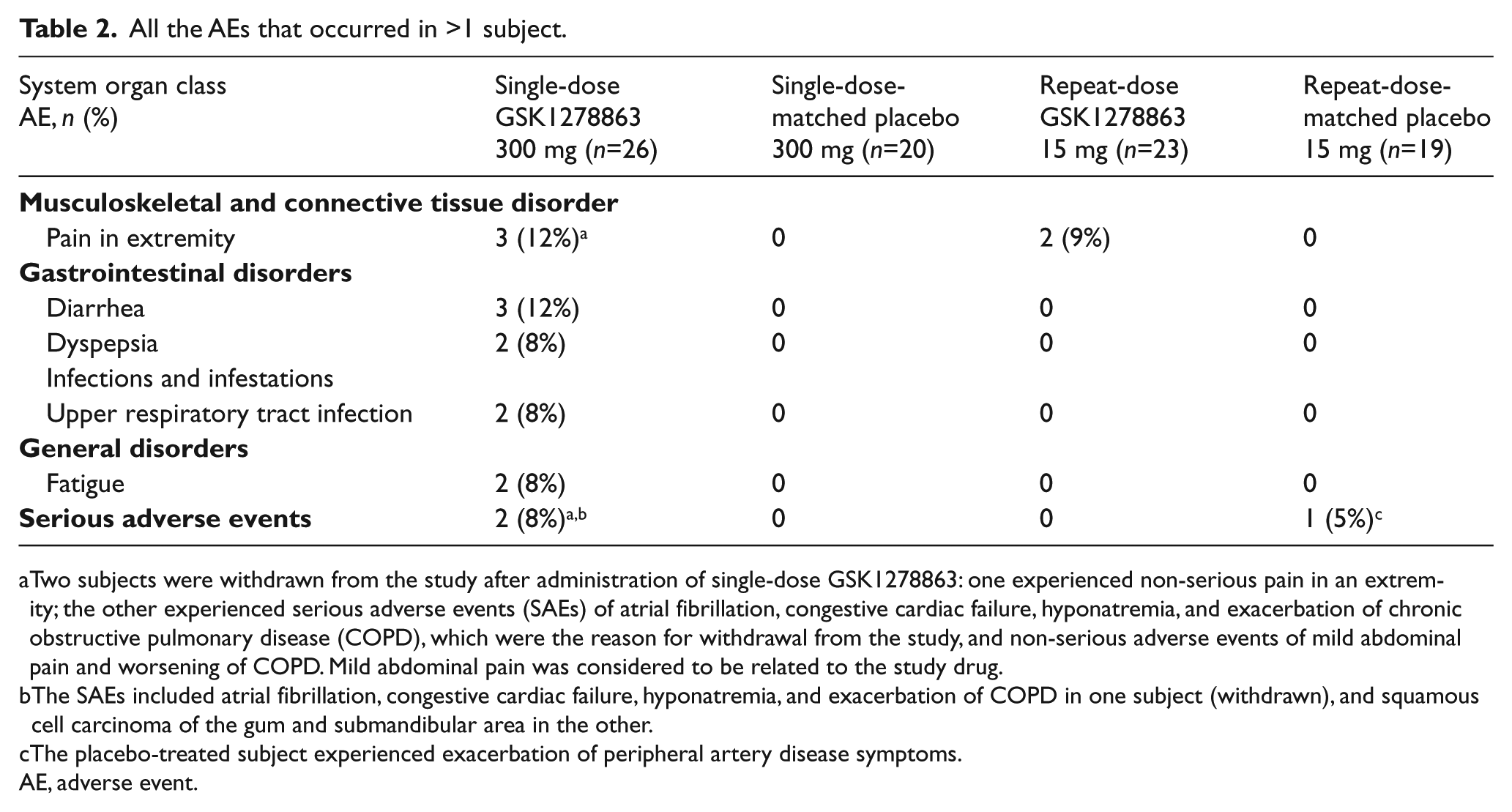
Two subjects were withdrawn from the study after administration of single-dose GSK1278863: one experienced non-serious pain in an extremity; the other experienced serious adverse events (SAEs) of atrial fibrillation, congestive cardiac failure, hyponatremia, and exacerbation of chronic obstructive pulmonary disease (COPD), which were the reason for withdrawal from the study, and non-serious adverse events of mild abdominal pain and worsening of COPD. Mild abdominal pain was considered to be related to the study drug.
The SAEs included atrial fibrillation, congestive cardiac failure, hyponatremia, and exacerbation of COPD in one subject (withdrawn), and squamous cell carcinoma of the gum and submandibular area in the other.
The placebo-treated subject experienced exacerbation of peripheral artery disease symptoms.
AE, adverse event.
Of note, during the GSK1278863 repeat-dose treatment period, the majority of AEs were mild or moderate in intensity, and none required drug discontinuation.
Pharmacokinetic results
GSK1278863 was absorbed readily following a single oral dose of 300 mg or 15 mg, with median time to maximum concentration (Tmax) in the range of 0.9–3.7 and 0.8–3.6 hours post-dose, respectively. For a 20-fold increase in dose (between 15 mg and 300 mg), there was an approximate 17-fold increase in the Cmax of GSK1278863. The metabolite:plasma ratios ranged from 2% to 11%, similar to those observed in previous studies with healthy volunteers. Concentrations of GSK1278863 and six of its metabolites at the end of the dosing interval (Ctau) for the 15 mg dose group were generally below the assay quantitation limit, and the variability in PK parameters was higher (coefficient of variance (CV) >70%) than that observed in previous phase I studies (CV <42%). Furthermore, the PK parameters for both dose groups were lower than those in previous dose-escalation studies in healthy subjects (Supplementary Tables S1 and S2).
Functional endpoints
Bilateral heel rise test
Compared with placebo, neither single- nor repeat-dose GSK1278863, respectively, significantly increased the total number of contractions to onset of claudication (p=0.053; p=0.08), total work performed to onset of claudication (p=0.18; p=0.12), total number of contractions (p=0.45; p=0.18), or total work performed (p=0.86; p=0.0.32). Although total exercise time prior to the onset of claudication was statistically increased for the single-dose period (p=0.025; increase of 11 seconds), no significant increase with repeat dosing (p=0.15) was noted (Table 3; Figure 3).
Comparison of bilateral heel rise test variables and 6-minute walk test between the GSK1278863 and placebo groups.
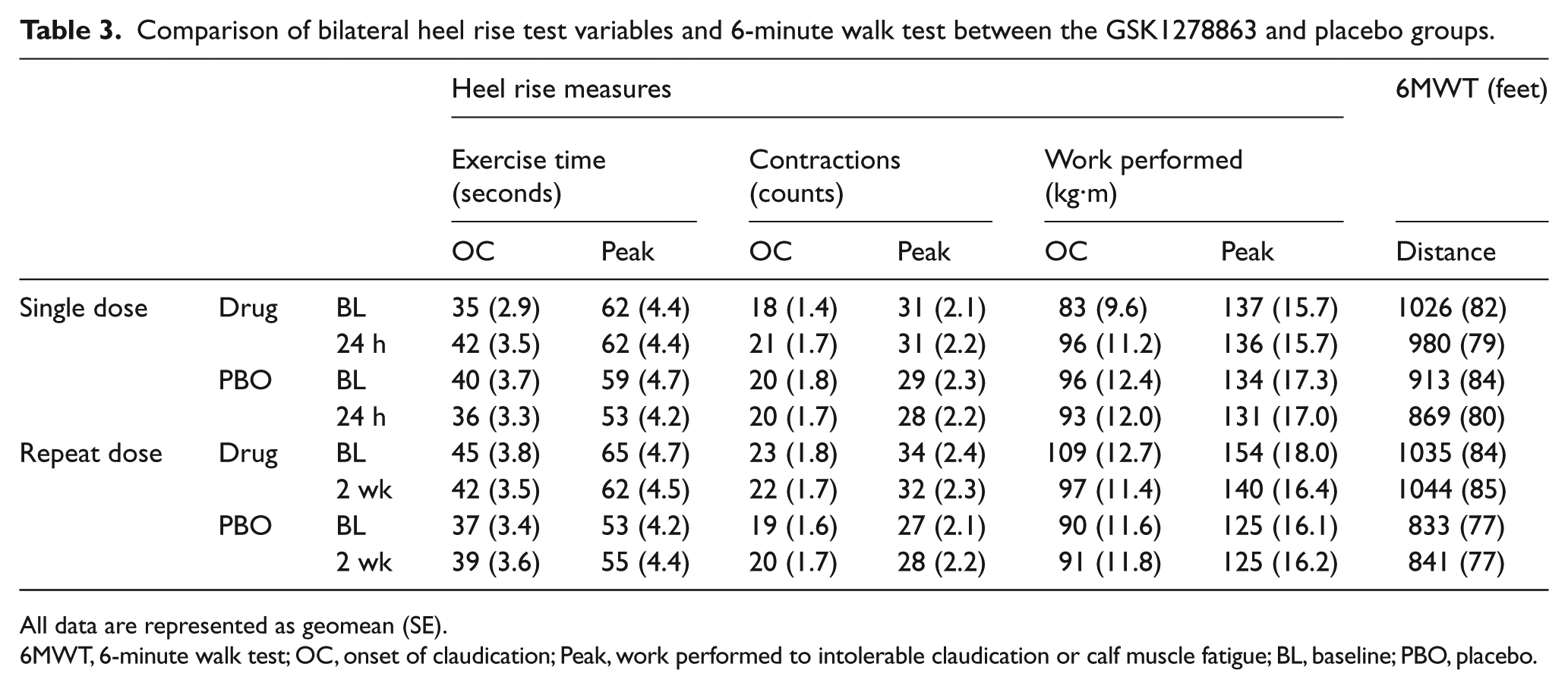
All data are represented as geomean (SE).
6MWT, 6-minute walk test; OC, onset of claudication; Peak, work performed to intolerable claudication or calf muscle fatigue; BL, baseline; PBO, placebo.
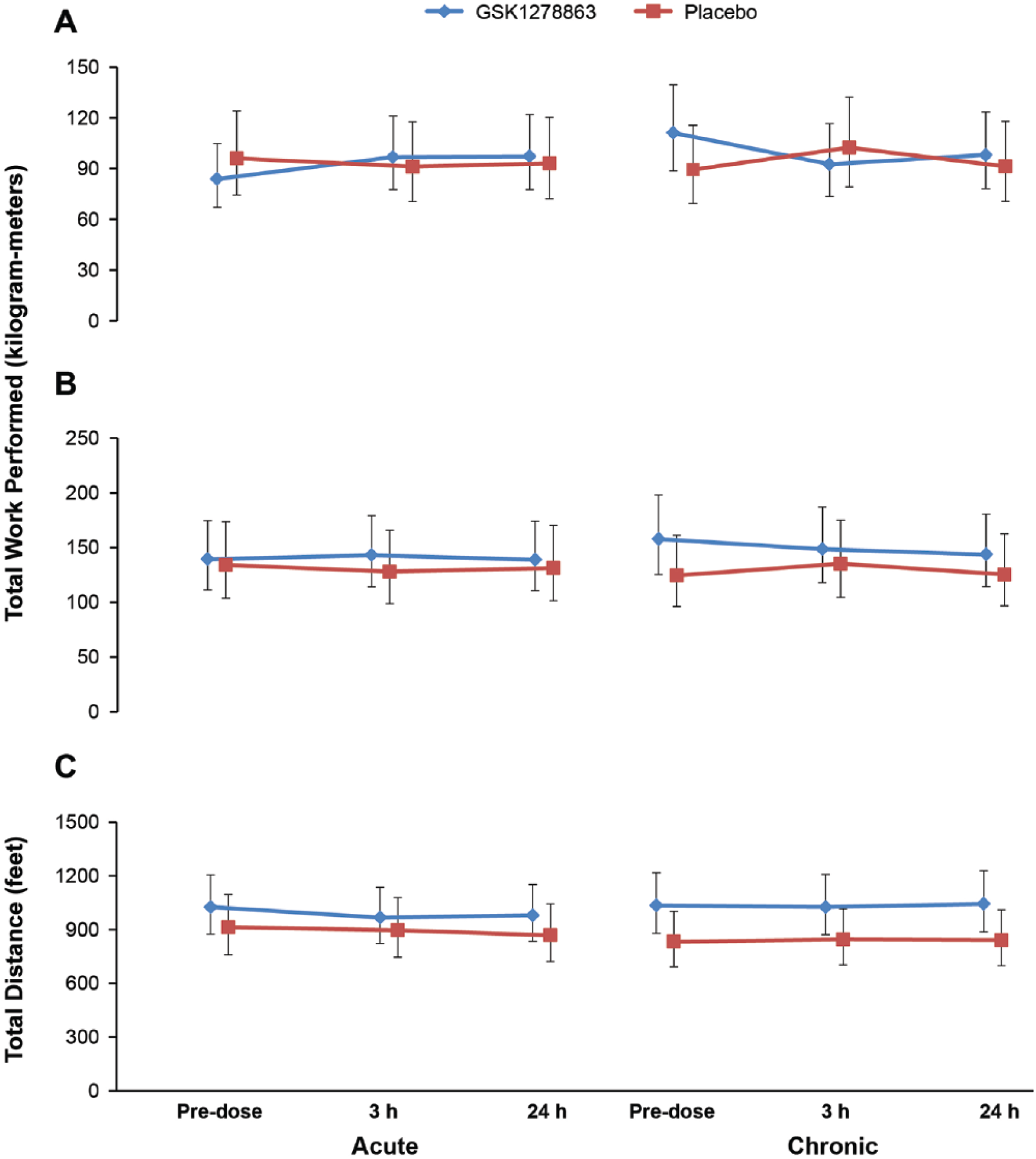
Bilateral heel rise test. Plot of least squares (LS) mean and 95% confidence intervals for (A) total work performed to onset of claudication (kilogram-meters) and (B) total work performed to claudication-limited maximal muscle performance (kilogram-meters) by visit and treatment. (C) Six-minute walk test (total distance covered). Plot of LS mean and 95% confidence intervals for the 6-minute walk test (feet) by visit and treatment.
Six-minute walk test
No significant differences were observed in the distance covered in the 6MWT between the two active treatment and placebo groups (single dose, p=0.96; repeat dose, p=0.15) (Table 3).
Clinical laboratory evaluations
Erythropoietin and hemoglobin
There was a significant increase in the EPO level from baseline after single-dose GSK1278863, but not with repeat-dose GSK1278863 and placebo (Supplementary Table S3) at all measured time points (2.5 hours (p=0.011), 3 hours (p<0.001), and 24 hours (p<0.001) post-dose).
No significant changes from baseline hemoglobin were detected after single-dose GSK1278863, although it increased significantly with repeat-dose GSK1278863 (13.4–14.3 g/dL, p<0.001; Supplementary Table S3).
Lipids
The majority of subjects were on antihyperlipidemic treatment, and repeat-dose GSK1278863 significantly decreased total cholesterol (TC; p<0.001), low-density lipoprotein (LDL; p=0.003), and high-density lipoprotein (HDL; p=0.016) by the end of treatment, compared with placebo; no statistically significant changes in triglycerides were observed (Supplementary Table S3).
Biomarkers
The mean hsCRP level increased significantly (from 1.96 to 4.31 mg/L) after single-dose GSK1278863 (least squares (LS) mean ratio with 95% CI: 2.20 (1.60, 3.02); p≤0.001) but not after repeat-dose GSK1278863 (2.31 and 2.36 mg/L pre- and post-GSK1278863 15 mg; LS mean ratio (95% CI): 1.02 (0.73, 1.43); p=NS) (Supplementary Table S3).
In 13 patients who underwent muscle biopsy, no increases from baseline mRNA or proteins of HIF target genes were detected in the GSK1278863 and placebo groups at the end of treatment (Supplementary Tables S4 and S5).
Discussion
This is the first clinical study examining exercise performance measures in patients with PAD and claudication treated with an orally administered PHD inhibitor. The key findings were that numerically more subjects receiving single- and repeat-dose regimens of GSK1278863 experienced AEs than those receiving placebo, and that neither regimen improved exercise performance despite plasma biomarker evidence of HIF-dependent pharmacology activation.
Despite therapeutic advances, a medical need for symptomatic relief in patients with claudication due to PAD exists, and recent attempts to develop novel therapeutics have been unsuccessful.23–25 Impairment in muscle function and exercise performance in PAD cannot be entirely ascribed to limitation in blood flow. 26 Changes in mitochondrial respiration, microcirculatory function and structure, skeletal muscle organization, nerve-muscle interface, and muscle metabolic function also significantly contribute to the disease pathophysiology.26–32
Accordingly, the hypothesis tested in the present study was predicated on evidence, demonstrating the ability of GSK1278863 to induce HIF-responsive mechanisms, such as EPO and HIF target gene mRNA induction within hours of administration, and suggesting that acute exercise and IPC can induce HIF-responsiveness.33,34 Although both regimens of GSK1278863 studied were adequate to engage the PHD pathway, no symptomatic or performance benefit ensued. Without chronic exposure, the single high dose (300 mg) increased EPO but was insufficient to increase hemoglobin levels. In contrast, the much lower repeat dose had an impact on hemoglobin, indicating both regimens had the expected effect on hematopoiesis. Poor tissue penetration might have limited target engagement in skeletal muscles; however, given the increase in hemoglobin following short-term therapy, poor tissue penetration cannot be overcome by increasing exposure without the risk of potential complications due to excess erythropoiesis.
Creager et al. studied intramuscular administration of an engineered type-2 adenovirus vector encoding a truncated form of HIF-1α in subjects with claudication. 25 Subject demographic and disease-burden profiles were similar to those in the present study. No significant improvements in performance measures were noted at 6 months, inducing speculation that adjunctive tissue ischemia may be necessary to realize the potential benefit of HIF stabilization, and that limited muscle distribution of the virus and its expression level may have influenced the outcome. 25
This finding was contrary to animal experiments in PHD2 haplo-sufficient mice (Phd2+/–) 35 and Phd1–/– mice resistant to tissue ischemia.35,36 Furthermore, administration of naked DNA encoding HIF-1α into the ischemic rabbit hindlimb caused a relatively rapid and robust development of collateral vessels that were visualized easily by angiography. 37 In addition, intramuscular injection of naked plasmid DNA encoding VEGF induced therapeutic angiogenesis in selected patients with critical limb ischemia, 38 but was not associated with improved exercise performance or quality of life. 24
The regulation of HIF in muscle during exercise and in response to hypoxia is complex and not well characterized. Chronic exercise therapy is well known to increase exercise performance in claudication-limited patients with PAD with blunting of muscle HIF response.39–43 Similarly, experiments involving subjects exposed to acute and chronic environmental hypoxia (4559 m altitude equivalent), showed unchanged HIF protein levels despite evidence for increased erythropoiesis—a known target effect of HIF elevation. 17 In contrast, acute exercise has been shown to elevate HIF levels. 33 These data suggest that the chronicity of acute bouts of ischemia together with the potential for tissue-specific HIF response may be more relevant to HIF regulation than hypoxia alone. This apparent paradox emphasizes the uncertain relationship among hypoxia, HIF stabilization, and exercise performance, suggesting that therapeutics to improve muscle performance and angiogenesis in PAD should evolve to counter the lack of efficacy in the majority of human trials utilizing molecular, genetic, and cellular modalities. 23
Administration of repeat-dose GSK1278863 15 mg was associated with a decrease from baseline lipid levels. These effects were unexpected and further investigation is warranted. The possible mechanisms may involve metabolic shifts associated with HIF activation toward more glycolytic mechanisms for adenosine triphosphate (ATP) generation, or the effects of EPO elevation that are associated with LDL reduction in the context of recombinant EPO administration.44,45
This study has several important limitations. Only acute and short-term treatment regimens were evaluated based on the hypothesis that immediate benefit could result from HIF stabilization and IPC effects. In contrast, the well-demonstrated benefits of chronic exercise therapy alleviate symptoms only after several months21,39,41,46,47; therefore, long-term therapy with GSK1278863 could possibly confer benefit. Furthermore, as this was the first study of this compound in patients with PAD, and primarily aimed to assess initial safety and PKs, it was not adequately powered to be a robust test for pharmacodynamic endpoints; this therefore limits the strength of interpretation of the results. From preclinical models and physicochemical properties of the drug, we can infer that skeletal muscle penetration may have been insufficient to elicit a robust HIF response and a true test of the hypothesis may require more efficient delivery to the site of action to be adequately tested.
In conclusion, more subjects receiving GSK1278863 single-dose (300 mg) and repeat-dose (15 mg for 14 days) regimens experienced AEs than those receiving placebo. Additionally, neither regimen improved functional measures of exercise performance or increased expression of HIF target genes in patients with PAD. HIF stabilization may still be an efficacious approach to the treatment of claudication-limited PAD if there is improved target engagement in skeletal muscle, longer therapy duration, or if administered in combination with other interventions such as revascularization or exercise training. Careful safety monitoring will be required to evaluate the AE profile, with a focus on limitations in hemoglobin elevation. Given the prevalence and symptom burden of this disease, additional exploration of this potential therapeutic approach is warranted.
Footnotes
Acknowledgements
All listed authors met the criteria for authorship set forth by the International Committee for Medical Journal Editors. The authors wish to acknowledge the following individuals for their assistance with study management and critical review during the development of the manuscript: Emile Mohler, MD, for scientific input and Douglas L Wicks ISMPP CMPP™, an employee of GlaxoSmithKline, for management of manuscript development. Editorial support, in the form of outline development, assembling tables and figures, collating author comments, copyediting, fact checking, and referencing, was offered by Cactus Communications Inc., and funded by GlaxoSmithKline. CPC Clinical Research, Aurora, Colorado, managed the study.
Declaration of conflicting interest
Funding
Funding for this study was provided by GlaxoSmithKline (NCT01673555).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
