Abstract
Tirasemtiv (CK-2017357), a novel small-molecule activator of the fast skeletal muscle troponin complex, slows the rate of calcium release from troponin, thus sensitizing fast skeletal muscle fibers to calcium. In preclinical studies, tirasemtiv increased muscle force and delayed the onset and reduced the extent of muscle fatigue during hypoxia in vitro and muscle ischemia in situ. This study evaluated the effect of single doses of tirasemtiv on measures of skeletal muscle function and fatigability in patients with stable calf claudication due to peripheral artery disease (PAD). Sixty-one patients with an ankle–brachial index ≤0.90 in the leg with claudication received single double-blind doses of tirasemtiv 375 mg and 750 mg and matching placebo in random order about 1 week apart. After 33 patients were treated, the 750 mg dose was decreased to 500 mg due to adverse events and these dose groups were combined for analysis. On each study day, bilateral heel-raise testing was performed before and at 3 and 6 hours after dosing; a 6-minute walk test was performed at 4 hours after dosing. Claudicating calf muscle performance was increased at the highest dose and plasma concentration of tirasemtiv; however, the 6-minute walk distance decreased with both the dose and plasma concentration of tirasemtiv, possibly due to dose-related adverse events, particularly dizziness, that could impede walking ability. In conclusion, the mechanism of fast skeletal muscle troponin activation improved muscle function but not 6-minute walking distance in patients with claudication due to PAD.
Introduction
Peripheral artery disease (PAD) represents a major systemic atherosclerotic syndrome which affects an estimated 202 million individuals in the world. 1 Individuals with PAD suffer a high risk of fatal and non-fatal cardiovascular ischemic events; however, affected individuals also are impacted in their daily lives by reductions in exercise performance, community walking ability and health-associated quality of life. 2 The exercise impairment is attributable to an insufficient increase in blood flow and oxygen delivery relative to the increase in metabolic demand of the exercising skeletal muscle. Additional mechanisms may further contribute, including alterations in muscle strength and muscle oxidative metabolism. 3 One effective claudication treatment, supervised exercise, works in part by improving skeletal muscle function. 4 Thus, new pharmacological treatments targeting the affected skeletal muscle are under consideration.
Tirasemtiv (formerly CK-2017357), a novel and selective small-molecule activator of the fast skeletal muscle troponin complex, has been developed to improve skeletal muscle function in disease states associated with insufficient neuromuscular input, muscular weakness or fatigue. Tirasemtiv slows the rate of calcium release from the troponin regulatory complex of fast skeletal muscle fibers, thus sensitizing the sarcomere to calcium. It is highly selective for fast skeletal muscle troponin, and much less effective in activating slow skeletal muscle with no activity in cardiac muscle. 5 In a preclinical femoral artery ligation rat model of vascular insufficiency, tirasemtiv both delayed the onset and reduced the magnitude of muscle fatigue during repeated trains of electrical muscle stimulation delivered every 3 seconds over 10 minutes. 6 In a study of healthy male volunteers, tirasemtiv produced dose- and concentration-dependent increases in the isometric force developed by the anterior tibialis muscle during transcutaneous stimulation of the common peroneal nerve. 7 More recently, increases in measures of skeletal muscle strength and endurance, including pulmonary function parameters, have been observed during the administration of tirasemtiv to patients with amyotrophic lateral sclerosis 8 and myasthenia gravis. 9 The most frequently reported dose-related adverse event (AE) has been dizziness, which is usually mild and typically resolves within 2 weeks of continued treatment.8,9
These data suggest that tirasemtiv may delay the onset of skeletal muscle dysfunction and fatigue during exercise in patients with claudication due to PAD. The current study was designed to evaluate the effect of single doses of tirasemtiv on skeletal muscle function and 6-minute walking ability in patients with calf claudication due to PAD.
Methods
Patients
Men and women were enrolled who were ≥40 years of age, had a body mass index of 18–30 kg/m2, and stable calf claudication due to PAD in at least one leg. The ankle–brachial index (ABI), defined as the ratio between the higher of the two pedal systolic blood pressures (dorsalis pedis or posterior tibialis) and the greatest systolic brachial pressures, was ≤0.90 in the leg with claudication. Patients were familiarized with a modified bilateral heel-raise test and standard 6-minute walking protocol. Patients were excluded if they had critical leg ischemia, hypotension or uncontrolled hypertension, life-threatening ventricular arrhythmias, unstable angina, stroke and/or myocardial infarction within the past 3 months, moderate/severe heart failure, Type 1 or uncontrolled Type 2 diabetes, or the need for supplemental oxygen. Patients could not have hepatic or renal insufficiency or anemia. Previous investigational treatments with gene therapy or other vascular endothelial growth factor (VEGF)-related therapy were not permitted. The study was approved by the investigational review boards at each site and all patients provided written informed consent prior to enrollment in the study.
Study design
This was a Phase II, double-blind, randomized, placebo-controlled, three-period crossover study of single oral doses of placebo, and a low dose (375 mg) and high dose (500 or 750 mg) of tirasemtiv (Figure 1). Eligible patients were randomized to one of six different treatment sequences, each consisting of three dosing periods (Table 1). On Day 1 of each dosing period, patients received a single dose of one of the three treatments: placebo, 375 mg tirasemtiv, or 750 mg tirasemtiv (which was subsequently reduced to 500 mg by a protocol amendment), administered in a double-blind fashion and in random order. Each single dose was followed by a 6–10-day washout period. Study assessments were performed over a 24-hour period following each of the three single doses. Five to 10 days after the final study treatment, the patient returned for follow-up assessments.
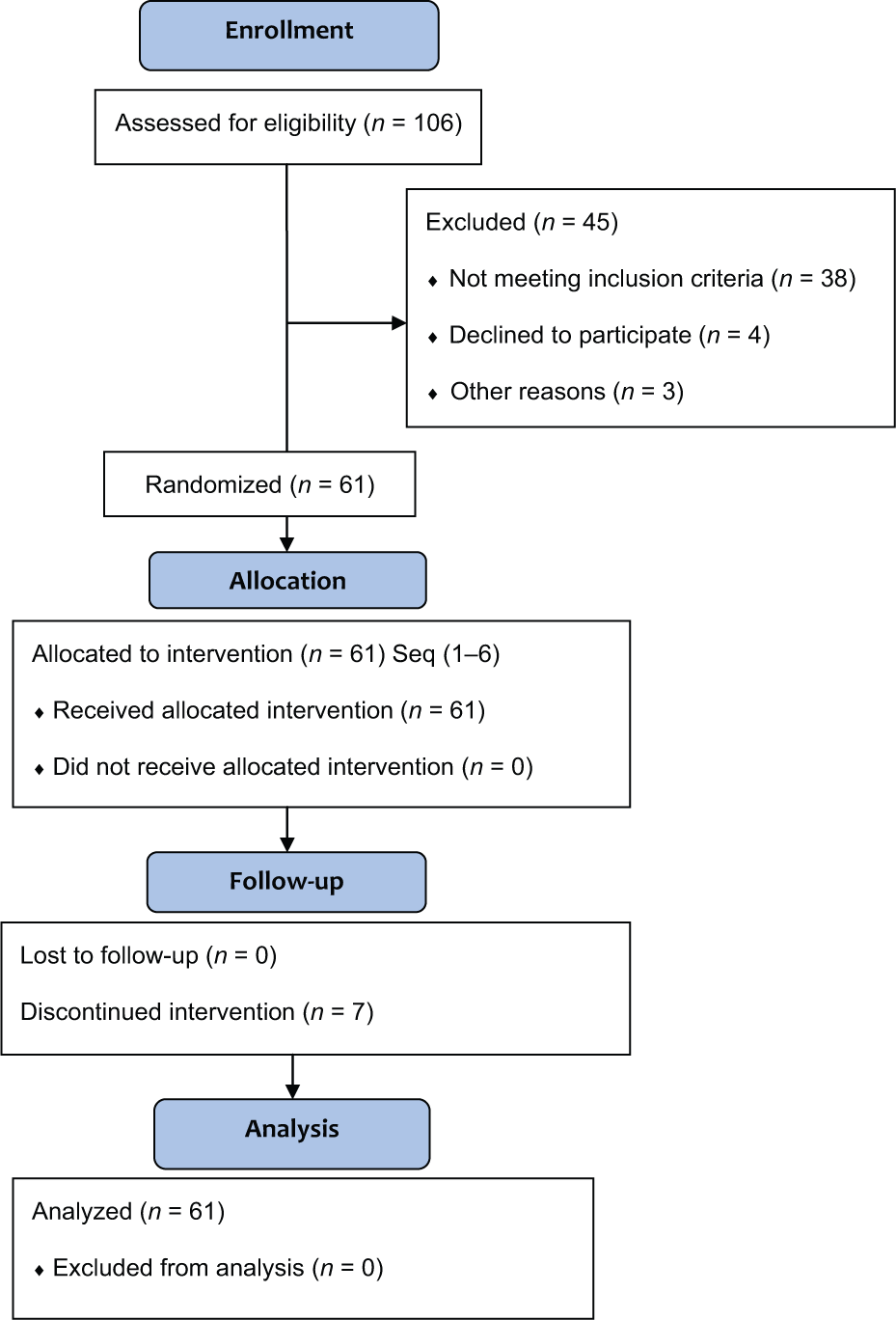
CONSORT diagram of trial design and enrollment.
Distribution of treatment assignments.
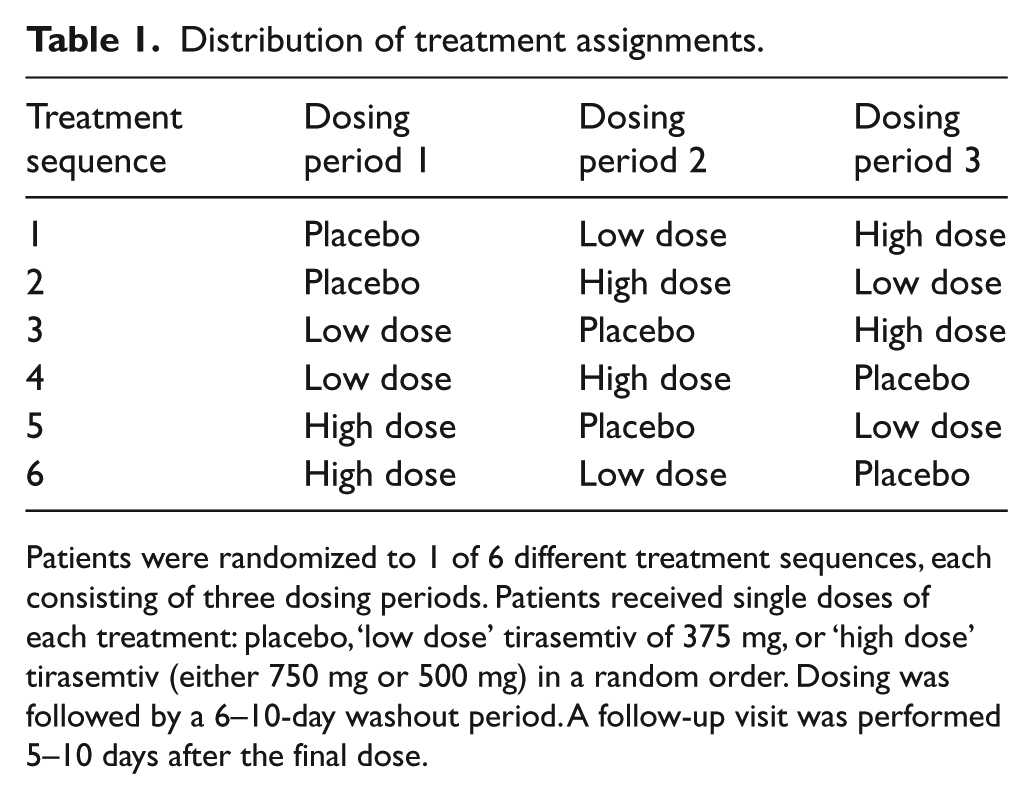
Patients were randomized to 1 of 6 different treatment sequences, each consisting of three dosing periods. Patients received single doses of each treatment: placebo, ‘low dose’ tirasemtiv of 375 mg, or ‘high dose’ tirasemtiv (either 750 mg or 500 mg) in a random order. Dosing was followed by a 6–10-day washout period. A follow-up visit was performed 5–10 days after the final dose.
Interim safety reviews of blinded data were pre-specified and conducted by the trial steering committee. The first review occurred after the first eight subjects completed dosing and again after the first 18 consecutive patients completed the study. The requirement for an overnight stay after each dose of study drug was discontinued following the initial safety review. The high dose of tirasemtiv (750 mg) was reduced to 500 mg following the second interim safety review because of concerns about tolerability of the 750 mg dose.
Pharmacodynamic assessments
Bilateral heal-raise test
As a direct measure of calf muscle performance, bilateral heel raises were performed to maximal claudication symptoms or muscle fatigue at 3 and 6 hours post-dose on Day 1 of each dosing period. The maximal claudication endpoint was defined as the inability to continue the exercise, similar to criteria for defining peak walking time during an incremental treadmill test. Bilateral heel raises were monitored by an electrogoniometer (Noraxon, Scotsdale, AZ, USA) placed on the ankle of the index leg; heel raises were performed once every other second to an audible cue until the patient was limited by intolerable claudication pain or muscle fatigue. Heel raises were only counted if a minimum angular displacement of 20 degrees was achieved from the subject’s native ankle position (0 degrees). The measurement range of the goniometer is ± 160 degrees, with a sensitivity of 25 mV/degree (range of measurement ± 5 V) and linearity of the goniometer is reported as ± 1%.
During each heel-raise test, the number of heel raises and the time to both onset of claudication and intolerable claudication pain or maximum calf muscle fatigue were measured. In addition, a cumulative heel-raise work index was assessed for the onset of claudication symptoms and for maximal claudication pain. The heel-raise work index was calculated as the product of peak heel-raise height and body mass for each heel raise assessed as the sum of the contractions over the entire duration of the test. The tests were performed at the Screening Visit (for familiarization) and on Day 1 of each dosing period – pre-dose, and 3 and 6 hours post-dose. All heel-raise test endpoints were adjudicated by a central core lab.
The 6-minute walk test
Patients were instructed to walk as far as possible (in a straight, measured hallway – reversing direction every 50 feet (15.24 m)) for 6 minutes, with rest as needed. 10 The test was administered 4 hours post-dose on Day 1 of each dosing period.
Pharmacokinetic assessments
Blood samples for pharmacokinetic analysis were taken on Day 1 of each dosing period at pre-dose, and 3, 4, 6, 9, 12, and 24 hours post-dose.
Analytical methods
Plasma concentrations of tirasemtiv were determined using a validated HPLC/MS/MS method at Alturas Analytics, Inc. (Moscow, ID, USA). An aliquot was injected onto an HPLC/MS/MS triple quadrupole mass spectrometer (Sciex API4000), and a Discovery HS C18, 3 µm HPLC column (2.1 × 50 mm) (Supelco, Bellefonte, PA, USA) was used to separate tirasemtiv and the internal standard, CK-2066398, from interfering compounds. The peak area of the study drug and internal standard were measured.
Safety assessments
AEs were monitored throughout the study. A physical examination was performed at the Screening and Follow-up Visits. Vital signs, clinical laboratory measurements, and 12-lead electrocardiogram (ECG) results were recorded at Screening and Day 1 of each period at pre-dose and at pre-specified times post-dose within each period, and at the Follow-up Visit. Cardiac enzymes troponin I and creatinine phosphokinase (CPK) were evaluated on a weekly basis by the study steering committee.
Safety population
The safety population consisted of all patients who were treated with at least 1 dose of the study drug. An AE was assigned to the corresponding tirasemtiv dose level and dosing period if it occurred after taking the study drug but prior to taking the study drug in the next period. An AE was assigned to the last treatment if the onset date was within 30 days from the date of the last dose. Safety data were summarized using descriptive methods for continuous measures, and counts and percent of patients for categorical parameters. All AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA), Version 12.0.
Statistical analysis
Demographic and baseline parameters were summarized by descriptive statistics for continuous variables and frequency distributions for categorical variables.
Pharmacodynamics (PDs)
The baseline for the heel-raise test in each period was defined as the same-day pre-dose assessment; the baseline for the 6-minute walk test was the Screening Visit assessment. The pharmacodynamic assessments and changes from baseline were summarized descriptively by treatment and time point. Because the heel-raise data were not normally distributed, a non-parametric analysis was used to compare heel-raise parameters at each time point following active treatment and placebo. A Cochran–Mantel–Haenszel (CMH) test on standardized mid-rank scores adjusting for treatment period and sequence was used to calculate p-values for treatment differences. Median and interquartile ranges (Q1–Q3) for the changes from baseline (pre-dose) by treatment and p-values for treatment differences were computed. The 6-minute walk test distance changes from baseline were analyzed using a repeated-measures analysis of covariance (ANCOVA) model with treatment, sequence, and period as fixed categorical effects, baseline measurement as a continuous covariate, and patient as a random effect. At each time point, p-values and 95% confidence intervals (CIs) were calculated for differences between each dose level and placebo in terms of placebo-corrected change from baseline. A partial correlation coefficient between AEs of interest and the change from baseline in the 6-minute walk distance was calculated, controlling for the baseline value and tirasemtiv dose. Multiplicity was not addressed in this Phase II study.
Pharmacokinetics (PKs)
Pharmacokinetic parameters were calculated using Phoenix WinNonlin 6.1 (Pharsight, Mountain View, CA, USA). Descriptive statistics (arithmetic mean, standard deviation, and geometric mean) were determined for maximum plasma concentration (Cmax), time to maximum plasma concentration (Tmax), and area under the plasma concentration–time curve (AUC) using non-compartmental PK methods. All analyses of AUC were conducted using AUC12hr. Dose proportionality of tirasemtiv on AUC and Cmax was assessed by a power model with period and sequence as fixed effects, and patient as a random effect. Point estimate of the slope with 90% CI and p-value were determined.
PK-PD correlation analysis
Plasma concentrations were divided into quartiles (>0 to 7, >7 to 10, >10 to 14, and >14 µg/mL). ANCOVA was used to compare each PD parameter per concentration range compared with placebo. The placebo-corrected least square (LS) mean change from baseline for the PD parameter was plotted at the mid-point of each concentration range. A correlation analysis between PK concentrations and the change from baseline in PD parameters was performed using an ANCOVA model, pooling all time points among all periods at which both PD and PK data were available; PK concentration and baseline measurements were continuous covariates; period and sequence were fixed categorical effects; and patient as a random effect. The slope and corresponding p-value were determined.
Results
Dosing and baseline characteristics
A total of 61 eligible patients were randomized into the study. Most patients (88.5%) completed the study and received all three treatments; therefore, baseline characteristics are shown for the overall population (Table 2). The study population was predominantly male and Caucasian with a mean age of 67 years; 39.3% were current smokers. The mean baseline ABI of 0.7 was typical for this population. Initially, 33 patients were randomized to one of six crossover sequences including placebo, tirasemtiv 375 mg, and tirasemtiv 750 mg. Following the second safety review the 750 mg dose of tirasemtiv was replaced with a 500 mg dose of tirasemtiv. Subsequently, 28 patients were randomized to crossover sequences including placebo, tirasemtiv 375 mg, and tirasemtiv 500 mg. The number of patients receiving each treatment included: placebo (n=57), tirasemtiv 375 mg (n=56), tirasemtiv 500 mg (n=27), and tirasemtiv 750 mg (n=33).
Demographics and baseline characteristics.
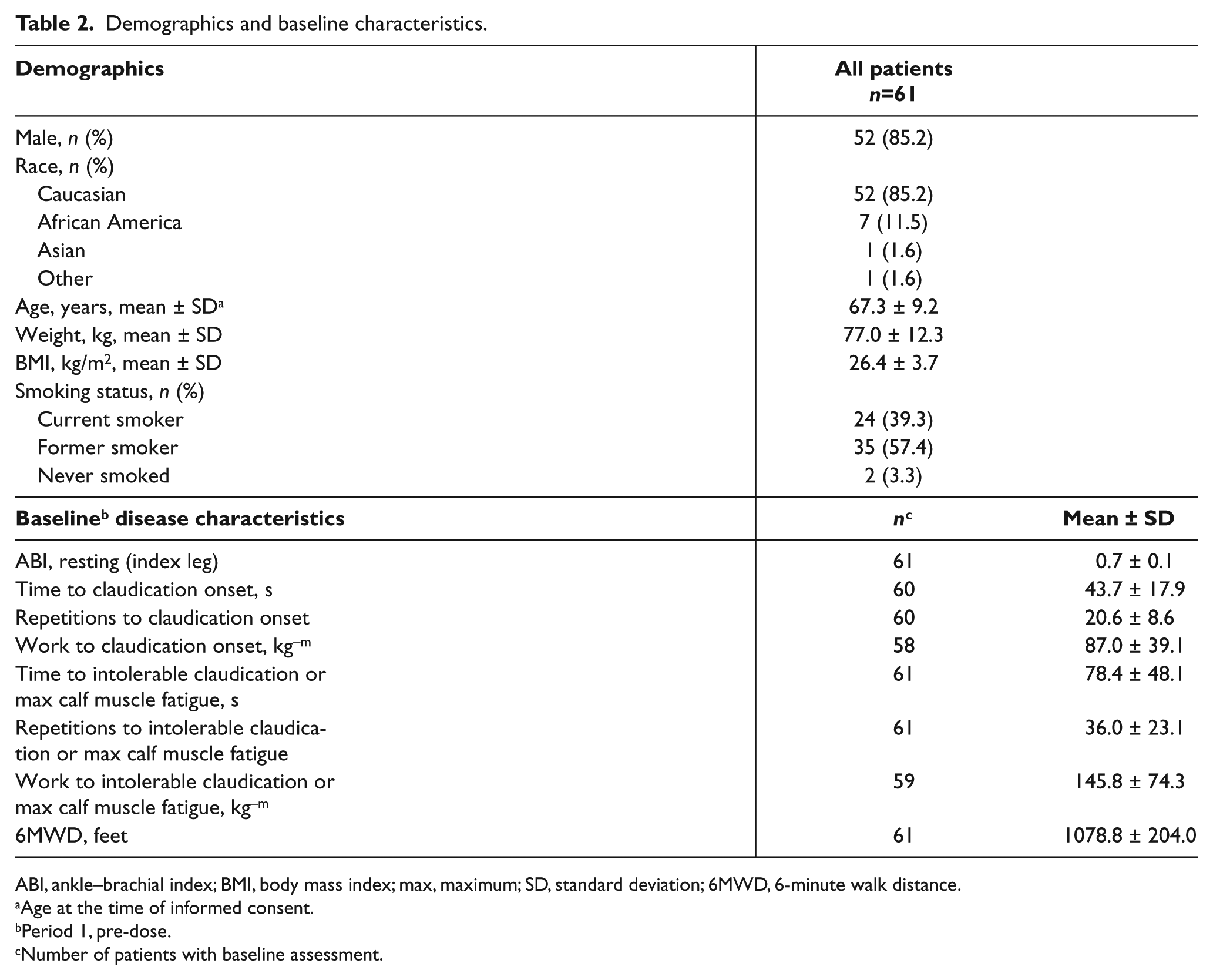
ABI, ankle–brachial index; BMI, body mass index; max, maximum; SD, standard deviation; 6MWD, 6-minute walk distance.
Age at the time of informed consent.
Period 1, pre-dose.
Number of patients with baseline assessment.
Pharmacodynamics
Heel raise to intolerable claudication or maximum calf-muscle fatigue
Statistically significant or borderline significant (p <0.10) increases in changes from baseline versus placebo were observed for time to intolerable claudication pain 3 hours after the 375 mg tirasemtiv dose and 6 hours after the 500 mg and 750 mg tirasemtiv doses (Table 3). Results for the number of heel-raise repetitions to intolerable claudication were similar to the time to intolerable claudication results, with both endpoints demonstrating statistically significant or borderline significant increases from baseline versus placebo observed 3 hours after the 375 mg tirasemtiv dose and 6 hours after the 500 mg tirasemtiv dose (Table 3).
Pharmacodynamic parameters: change from same-day baseline.
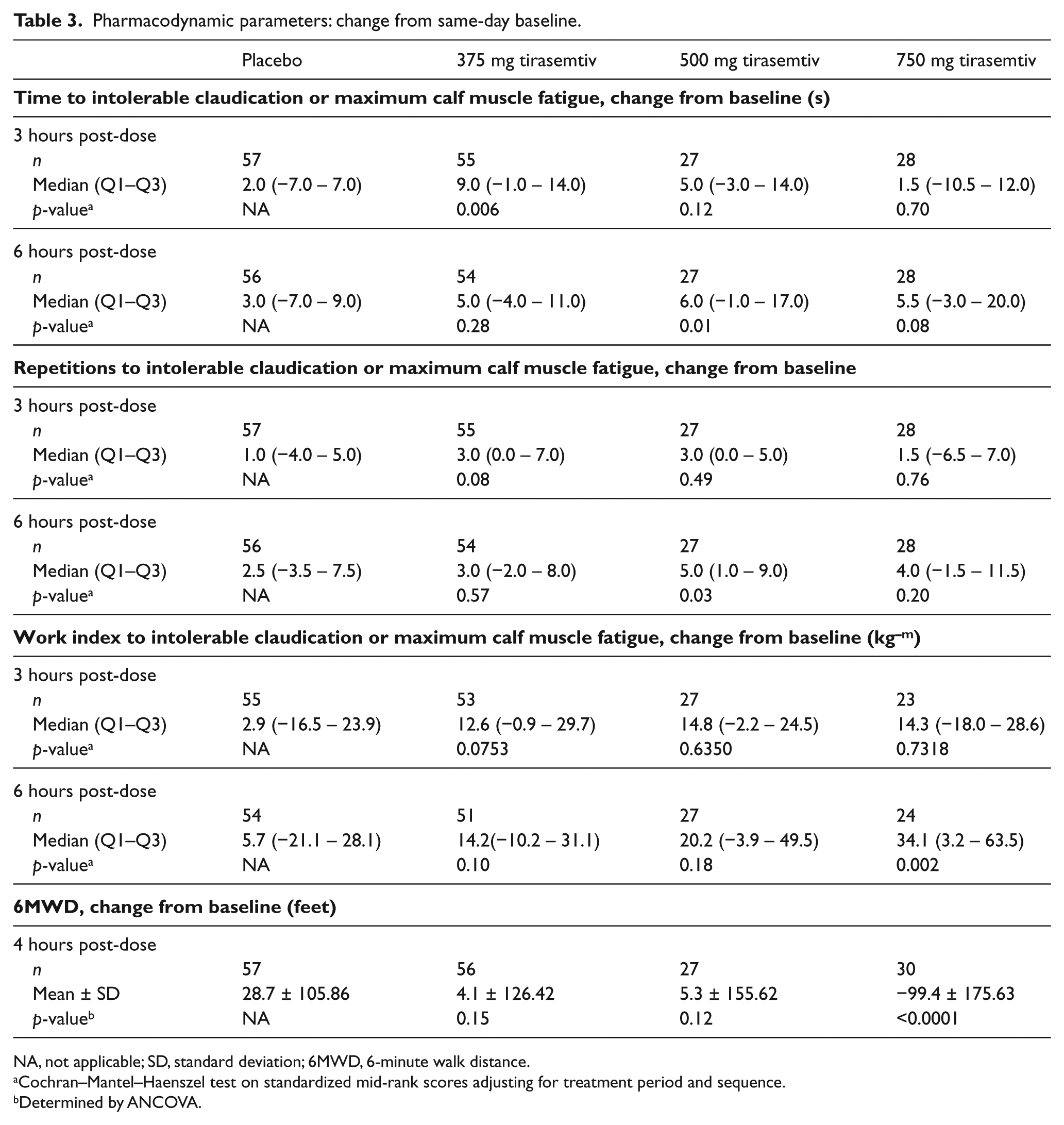
NA, not applicable; SD, standard deviation; 6MWD, 6-minute walk distance.
Cochran–Mantel–Haenszel test on standardized mid-rank scores adjusting for treatment period and sequence.
Determined by ANCOVA.
Heel raise to onset of claudication
The changes from baseline for time, number of repetitions, and work index to onset of claudication for each of the three tirasemtiv doses versus placebo showed no statistically significant differences at either the 3- or 6-hour time point (data not shown).
Figure 2 shows the change from baseline in the median work index to intolerable claudication pain 6 hours after dosing. Each tirasemtiv dose produced a numeric increase from baseline and compared to placebo. Although the 750 mg tirasemtiv dose was the only treatment to produce a statistically significant improvement in work index over placebo at the 6-hour time point (p=0.0019), a statistically significant overall dose–response was evident (see PK-PD correlation across all doses) (p=0.0067).
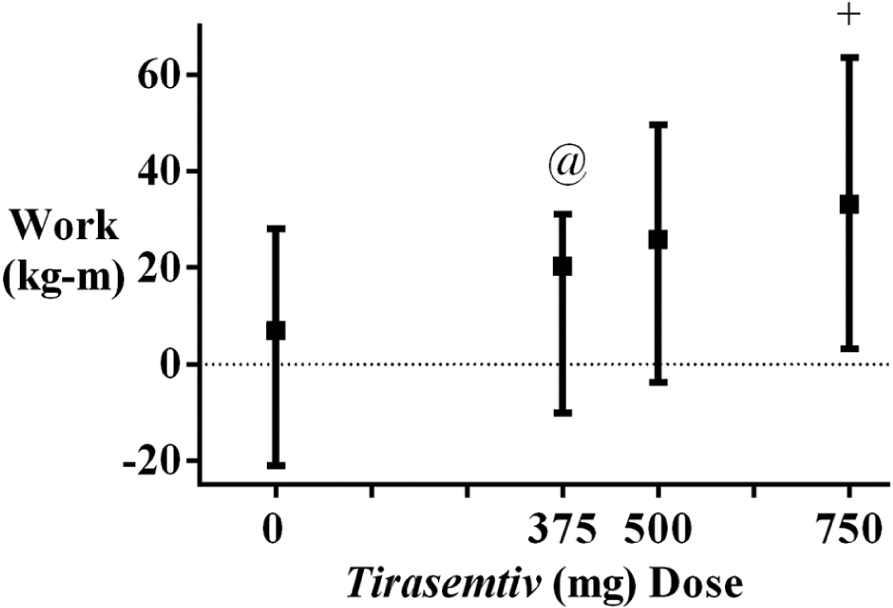
Work index to intolerable claudication or muscle fatigue. The median (± interquartile range) change from baseline for work index at 6 hours post-dose was plotted per treatment, where 0 mg tirasemtiv = placebo. Baseline was the same day, pre-dose assessment. (@p <0.10 versus placebo; +p <0.002 versus placebo; overall dose–response, p = 0.0067.)
6-Minute walk test distance
There was a dose-dependent decrease in the placebo-corrected mean change from baseline for the 6-minute walk distance (Table 3, Figure 3A). The placebo-corrected changes observed were modest relative to the mean baseline distance (1079 (328.88 m); Table 2); however, the decrease in the 6-minute walk distance was most notable and significantly different from placebo after the 750 mg tirasemtiv dose (mean change: −99.4 feet (–30.30 m); p <0.0001). AEs, particularly at the high dose, associated with tirasemtiv treatment may have negatively affected the performance of the walk test (see ‘Safety’).
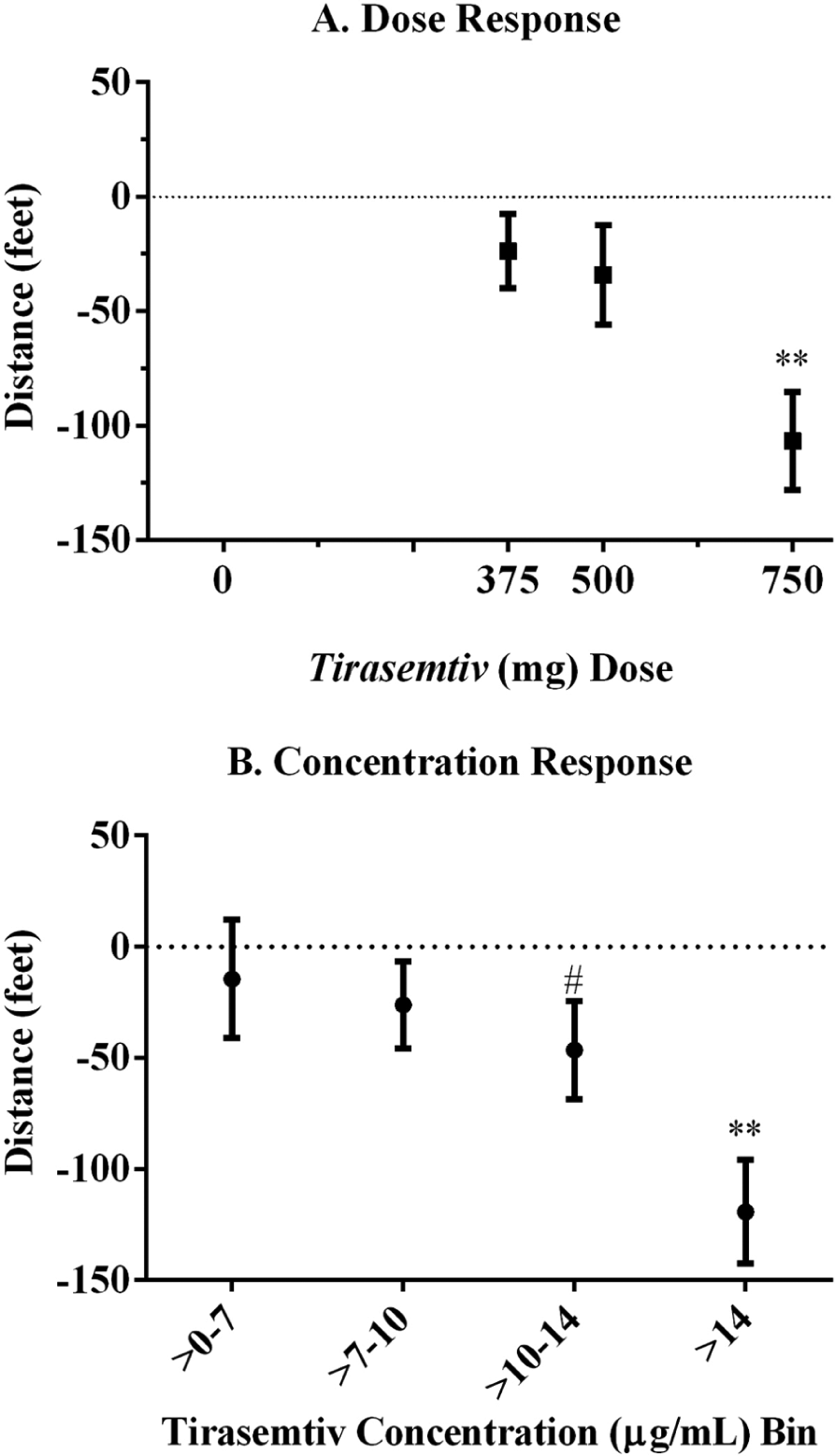
Impact of drug dose on the 6-minute walk distance. Least square mean (± standard error) placebo-corrected changes from baseline were plotted per tirasemtiv dose administered (A) and by measured plasma concentration (B). For panel A, 0 mg = placebo. Baseline was the Screening Visit assessment and the 6-minute walk distance was assessed again 4 hours post-dose. There was a statistically significant relationship between both dose (panel A, p <0.0001) and plasma concentration (panel B, p <0.0001). (#p <0.05 versus placebo; **p <0.0001 versus placebo.)
Pharmacokinetics and PK-PD correlation
Maximum plasma concentrations were reached at approximately 6 hours after all doses of tirasemtiv (median Tmax = 6 hours for each tirasemtiv dose) (Table 4). Cmax increased with increasing dose, ranging from 9.6 ± 2.2 µg/mL after the 375 mg dose up to 18.3 ± 3.4 µg/mL after the 750 mg dose. These PK results are consistent with those previously reported. 7
Pharmacokinetics of tirasemtiv by dose.
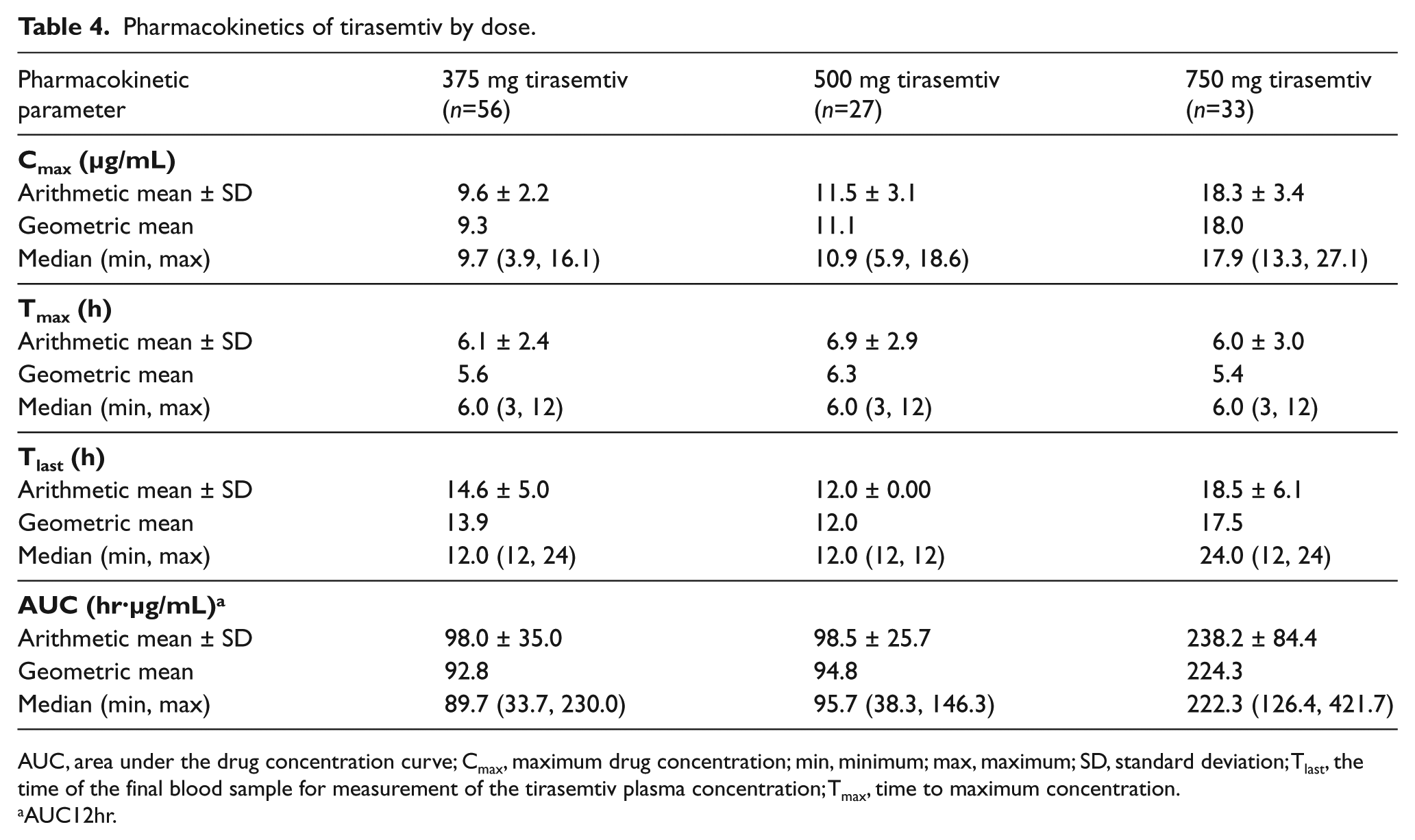
AUC, area under the drug concentration curve; Cmax, maximum drug concentration; min, minimum; max, maximum; SD, standard deviation; Tlast, the time of the final blood sample for measurement of the tirasemtiv plasma concentration; Tmax, time to maximum concentration.
AUC12hr.
The onset to claudication pharmacodynamic parameters showed minimal treatment effect over placebo when analyzed by plasma concentration range, except for the highest concentration range (>14 μg/mL), in which exercise time increased by 3.9 ± 1.9 s [SE] (p = 0.04), the number of repetitions by 2.3 ± 1.0 repetitions (p = 0.03), and work index by 9.5 ± 5.4 kg−m (p = 0.08). Both time and number of repetitions to onset of claudication showed a positive, statistically significant concentration–response (p = 0.02 and p = 0.04, respectively). The treatment effects on the assessments of intolerable claudication pain were more evident, with the highest tirasemtiv concentration range showing a statistically significant increase over placebo observed for each parameter (Table 5). A positive, statistically significant concentration–response relationship was observed for each parameter to intolerable claudication (Table 5).
Change from baseline for pharmacodynamic parameters by tirasemtiv concentration range.
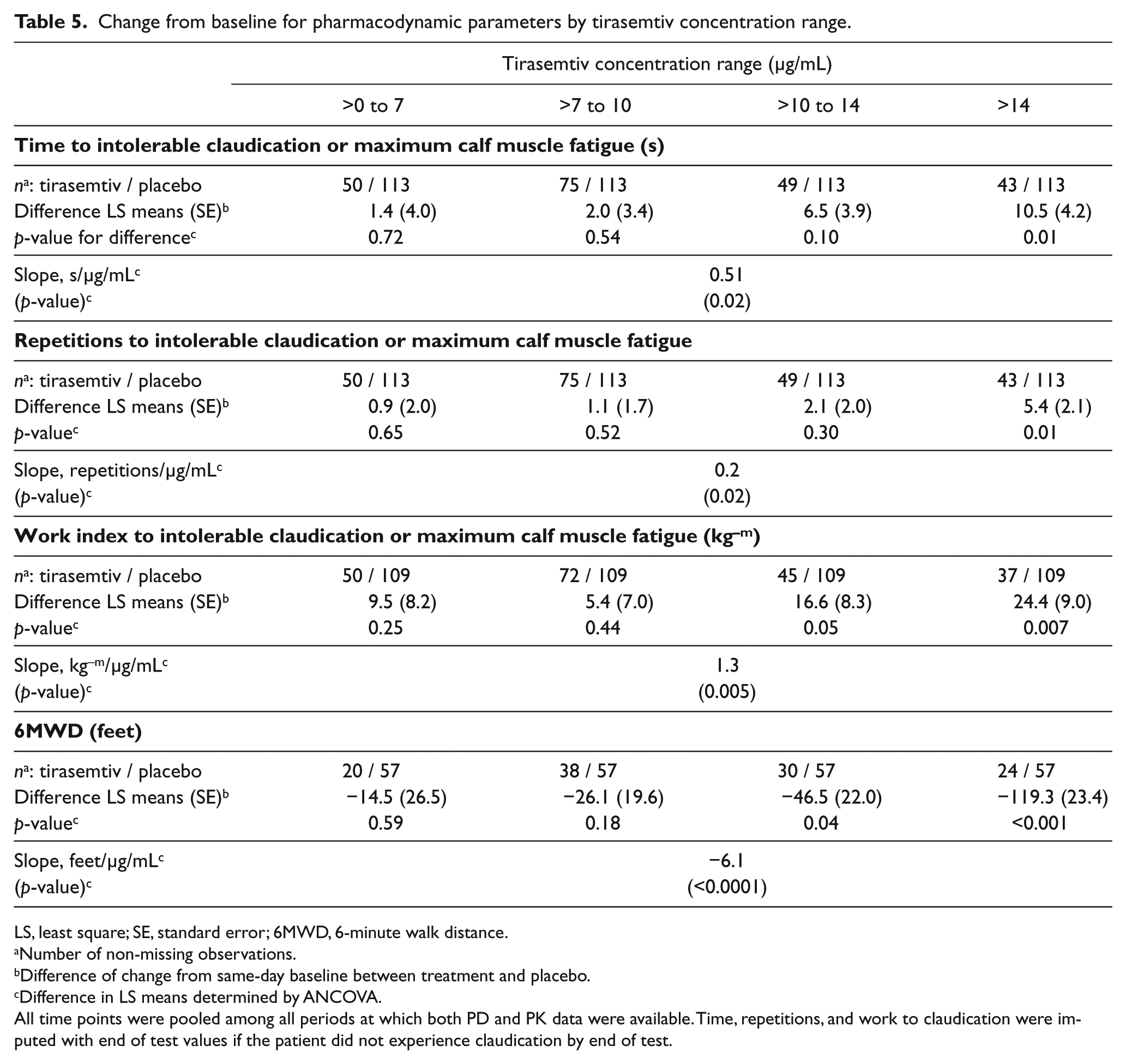
LS, least square; SE, standard error; 6MWD, 6-minute walk distance.
Number of non-missing observations.
Difference of change from same-day baseline between treatment and placebo.
Difference in LS means determined by ANCOVA.
All time points were pooled among all periods at which both PD and PK data were available. Time, repetitions, and work to claudication were imputed with end of test values if the patient did not experience claudication by end of test.
Figure 3B presents the inverse relationship between placebo-corrected change from baseline in 6-minute walk distance and concentration range. There were significant decreases from baseline for the two highest concentration ranges, with a statistically significant negative slope for the concentration–response relationship (Table 5).
Safety and tolerability
Of the 61 patients receiving at least one dose of study treatment, most (93.7%) had at least one AE during the study, and AEs were reported more frequently with tirasemtiv treatment compared to placebo. The AE profile appeared to be dose-dependent. Overall, the most commonly reported AEs were dizziness, euphoric mood, fatigue, and gait disturbance (Table 6), consistent with previous reports.8,9 Most AEs were mild to moderate in severity; however, severe AEs that occurred in more than one patient included dizziness (n=4), gait disturbance (n=2), and somnolence (n=2), of which, all occurred during the 750 mg tirasemtiv dosing period. Five patients prematurely discontinued the study due to AEs related to the central nervous system. There were three serious AEs experienced by three patients. One patient had worsening cholecystitis following 500 mg tirasemtiv which resolved without study discontinuation. Two patients were hospitalized with severe dizziness following dosing with 750 mg tirasemtiv; these events resolved and both patients were discontinued from the study. Laboratory and ECG abnormalities were infrequent, and there was no indication of a relationship to tirasemtiv. Changes in cardiac troponin I levels were rare, and although there were several increases in CPK above the normal range, none appeared to be dose-dependent or associated with a cardiac event or any other AE.
Most common (≥5%) on-treatment adverse events.
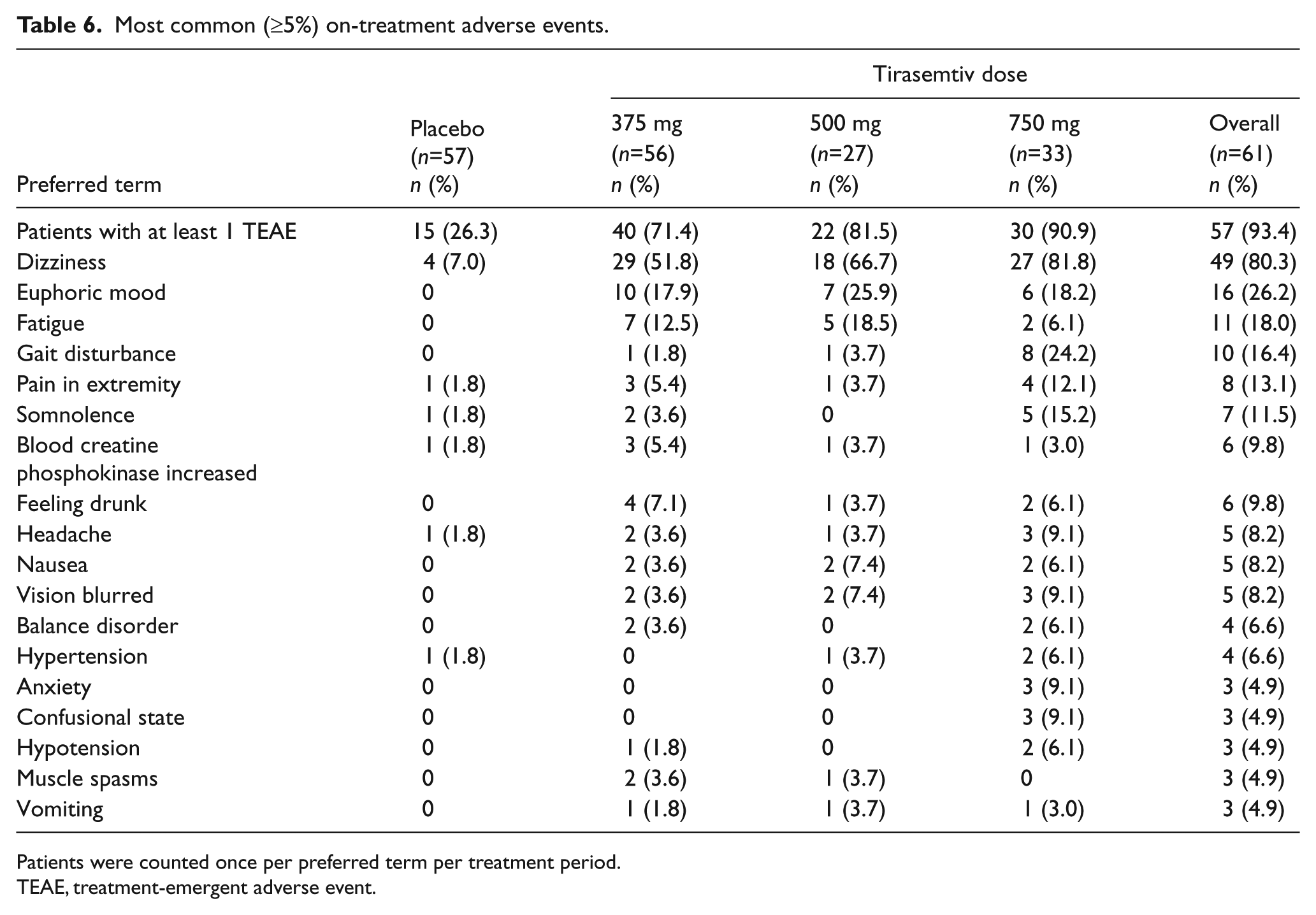
Patients were counted once per preferred term per treatment period.
TEAE, treatment-emergent adverse event.
A substantial percentage of patients experienced dose-dependent dizziness, and/or gait disturbance during the 6-minute walk (1.8% for placebo, 37.5% for 375 mg, 48.1% for 500 mg, and 72.7% for 750 mg). These AEs were associated with a decrease in the 6-minute walk distance from baseline with a correlation coefficient of r = −0.18 (p = 0.02).
Discussion
This study provides new evidence that a novel small-molecule activator of the fast skeletal muscle troponin complex can improve calf muscle exercise performance as measured by sequential heel-raise muscle contractions and total muscular work performed. This pharmacodynamic effect appeared to be both dose and plasma drug concentration dependent. This direct muscle effect was associated with an improvement in claudication-limited maximal calf muscle performance as detected by an increase in the number and duration of calf muscle contractions, and in the amount of muscular work performed at the highest dose and plasma concentration of the drug. While this novel activator of the fast skeletal muscle troponin complex improved skeletal muscle performance, the AE profile and reduced 6-minute walk distance at the highest dose would limit interest in this particular small molecule for clinical development in PAD.
This study evaluated the pharmacodynamic impact of this new molecule using two contrasting outcome modalities, designed to directly test calf muscle endurance (the heel-raise test) and overall ambulatory function (the 6-minute walk test). While calf muscle exercise performance showed signals of improvement with increasing doses of tirasemtiv, the 6-minute walk distance was adversely impacted by tirasemtiv in a dose-dependent manner. There was an association between the AEs of dizziness and gait disturbance and loss of 6-minute walk distance. Thus, while local calf muscle performance appeared to be improved at the highest dose of tirasemtiv, the integrated walking performance of the patient was adversely impacted. These results provide evidence that use of more than one method of functional exercise assessment may be particularly useful in the evaluation of new claudication drug efficacy, as traditional endpoints of peak walking time on a graded treadmill may have missed the potential therapeutic effect of this candidate small molecule.
PAD is known to be associated with a complex pathophysiology that is initiated by an insufficient increase in skeletal muscle blood flow to exercising skeletal muscle. Over time, repeated bouts of ischemia and reperfusion lead to muscle damage as indicated by muscle fiber denervation, mitochondrial DNA injury and reductions in several components of the electron transport chain.11–14 These changes result in altered skeletal muscle oxidative metabolism as indicated by accumulations of skeletal muscle acylcarnitines and slowing of muscle respiration during exercise.15,16 In this context, depletion of high-energy substrates and impaired oxidative energy metabolism likely lead to impaired excitation-contraction coupling and subsequent muscle contractile dysfunction in PAD. 17 Tirasemtiv, an activator of the fast skeletal muscle troponin complex, is an attractive agent for this disease as it slows the rate of calcium release from the regulatory troponin complex of fast skeletal muscle resulting in a sensitization of the contractile apparatus to calcium. In intact rat skeletal muscle in situ, the drug sensitizes muscle to nerve stimulation, increasing force at sub-maximal levels of nerve stimulation. 6 Thus, in PAD, where both denervation and high-energy phosphate depletion are components of the muscle impairment, this agent might provide a strategy to improve muscle function. In the current study, a testing environment that isolated the gastrocnemius and soleus muscles appeared to demonstrate a positive effect on muscle function with single doses of drug.
The positive effect on muscle performance in PAD must also be weighed against its safety profile. Tirasemtiv has been previously studied in healthy volunteers and patients with amyotrophic lateral sclerosis (ALS), and myasthenia gravis.8,9 The side effect profile appears similar in all cases, with dizziness – described most often as a lightheadedness – being most commonly reported. The incidence of dizziness is clearly dose related but appears to abate with continued dosing in the majority of those individuals treated. 8 An ongoing trial of tirasemtiv in ALS is employing a dose titration strategy to implement therapy. In this study, the drug was associated with the systemic side effects of dizziness, euphoria and gait disturbance. As a consequence, these observed AEs may have had untoward effects on the PD endpoints, including an observed decrease in functional walking performance and the potential for subject unblinding. It is difficult to quantify to what extent the AE profile may have influenced subject performance on any of the PD endpoints. However, a plausible consequence of these AEs is consistent with the reduced 6-minute walk performance. In this regard, the 6-minute walk test appeared to be a sensitive barometer of the ability of patients to integrate their walking ability under conditions where gait and balance contribute to the distance walked as well as muscle performance. Whether the drug would have improved treadmill exercise performance is unknown as that endpoint would also be influenced by the positive effects of the drug on muscle performance coupled with the potential adverse effects of dizziness and gait disturbances. A drug of this class with a similar mechanism of action but without a central nervous system component would be desirable.
Limitations
This was an early stage trial with three endpoints of the heel-raise test at the point of maximal claudication: number of contractions, time to maximal claudication pain, and calculated work. As noted in the statistical analysis section, there was no adjustment of the p-value for multiplicity and the investigators did not declare a primary endpoint in an attempt to identify a signal of benefit.
Conclusion
Tirasemtiv, a selective small-molecule activator of the fast skeletal muscle troponin complex, was associated with a positive effect on calf muscle performance at the highest dose and plasma concentrations in individuals with PAD and claudication. The drug was associated with significant side effects and a loss of 6-minute walk performance. The adverse functional effects of the drug demonstrate that the 6-minute walk test may serve as a sensitive barometer of the ability of patients to integrate their walking ability under conditions where gait and balance contribute to the distance walked. Similar drugs of this class, which may be able to improve skeletal muscle performance without central nervous system effects, would provide a potentially desirable method to improve functional capacity and quality of life in patients with PAD.
Footnotes
Acknowledgements
The authors acknowledge Jacqueline Lee for management of the study and Michael M Chen for statistical support.
Declaration of conflicting interest
A. Wolff, F. Malik, and L. Meng are current or former employees of Cytokinetics. T. Bauer and W. Hiatt received grant support from Cytokinetics through CPC Clinical Research. A. Hirsch received funding from Cytokinetics for participation on the steering committee. K. Rogers had no reported conflicts.
Funding
This study was sponsored by Cytokinetics, Inc.
