Abstract
Propionyl-
Keywords
Introduction
Therapeutic options for peripheral artery disease-associated claudication remain limited despite increasing recognition of the disability associated with the condition.1,2 Supervised exercise rehabilitation is efficacious and considered the medical treatment of choice,2,3 but is available to only a few patients due to cost, a limited number of available facilities, inconvenience and lack of acceptance. Revascularization is beneficial in many patients4,5 but is expensive and its long-term efficacy is unclear. Pharmacotherapies in the United States are limited to pentoxyfylline, which is associated with only mild improvement in patients 6 and cilostazol, whose use has been limited due to vasodilatory adverse effects and residual safety concerns based on its mechanism of action.7,8
Evidence that a secondary myopathy contributes to the exercise limitation in patients with claudication9,10 has suggested novel targets for new drugs to treat the exercise impairment. Propionyl-
PLC has been studied as a treatment for claudication based on these properties. Published studies have supported the hypothesis that PLC is effective but have in general been small and may raise questions of publication bias with respect to the available information. Thus, a comprehensive systematic review of all studies with PLC in patients with claudication performed and for which data were available up to September 2010 was conducted in order to ascertain a better estimate of the effect of PLCs in this population.
Methods
Systematic review of all studies for PLC in claudication
Study identification
A systematic review of all studies of the effect of PLCs on walking performance in patients with claudication was conducted. All studies published in the medical literature in any language, starting from January 1984 and up to September 2010, were retrieved through MEDLINE and EMBASE using the following search strategy: “{[(propionyl AND carnitine) OR propionyl-
In order to minimize publication bias, the proceedings of international congresses of vascular medicine were reviewed to identify abstracts of negative studies. Furthermore, the internal database of Sigma-Tau S.p.A (Pomezia, Italy; the manufacturer and principal developer of PLC for peripheral artery disease) was accessed to identify unpublished studies conducted with PLC in patients with claudication.
Patient-level meta-analysis
Study identification – primary analysis
The complete databases of the six phase III randomized, placebo-controlled studies performed by Sigma-Tau S.p.A. in patients with claudication were made available to the authors, including three studies not previously published. A patient-level meta-analysis of these six studies was prospectively designated as the primary analysis. All studies used 1 g PLC twice daily or matching placebo. For convenience, the studies will be referred to as European, 20 US Dose Ranging, US/Russia, 21 European-Diabetes, US-Diabetes, and US-Exercise, 22 as detailed in Table 1.
Overview of the phase III trials included in the meta-analysis
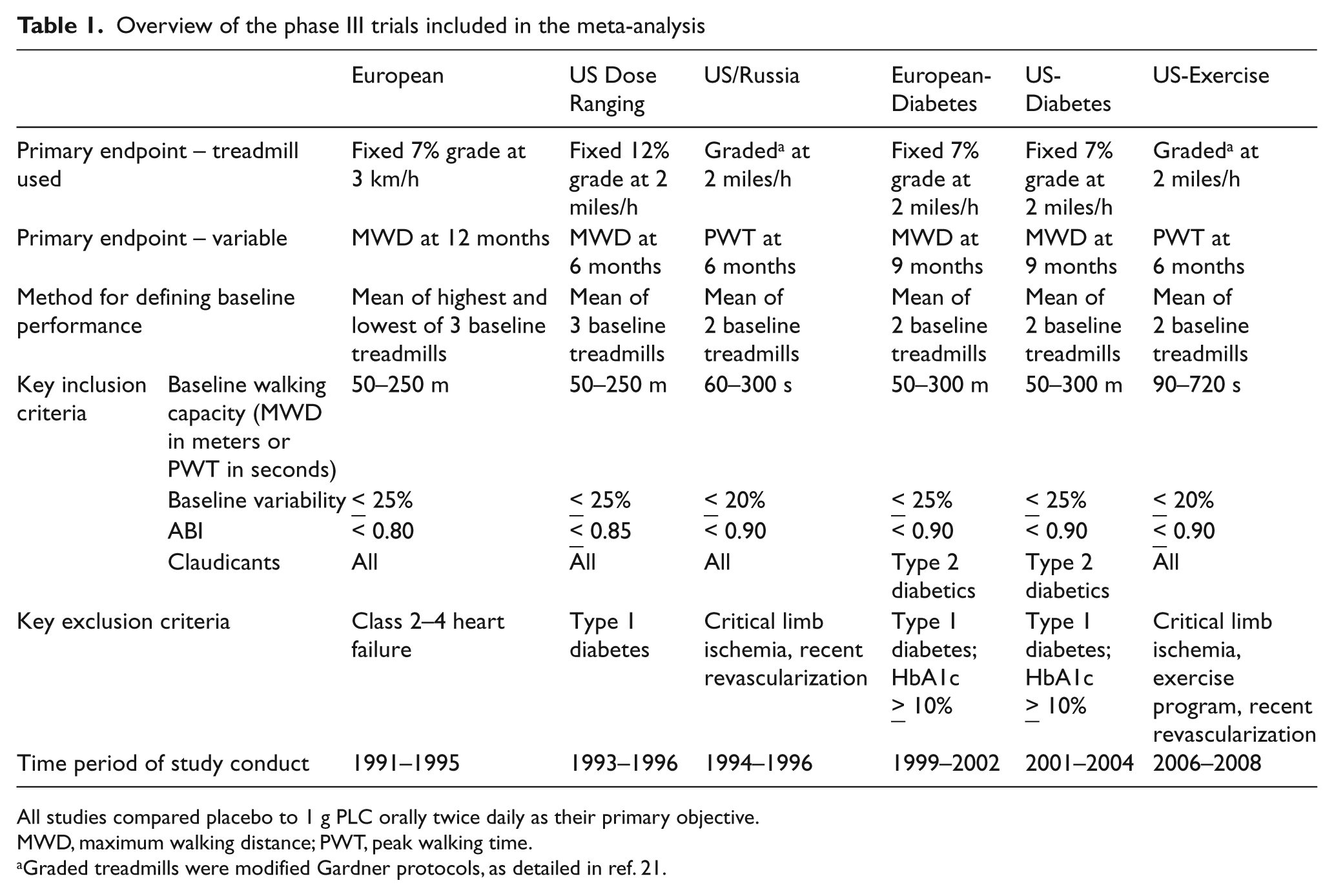
All studies compared placebo to 1 g PLC orally twice daily as their primary objective.
MWD, maximum walking distance; PWT, peak walking time.
Graded treadmills were modified Gardner protocols, as detailed in ref. 21.
Analysis populations – primary analysis
In the patient-level meta-analysis, the intention-to-treat (ITT) population pre-specified by each study’s Statistical Analysis Plan for the primary efficacy analysis was used. In the case of the European study, the primary analysis population was composed of patients with baseline walking distances of 50–250 meters. Similarly, in the US Dose Ranging study, patients treated with 1.0 g PLC twice daily formed the basis for that study’s primary efficacy assessment, and have been used here for the primary analysis. When multiple treadmill assessments were made over the course of therapy, the treadmill time point specified in the study’s protocol as the primary endpoint was utilized in the current analysis. Other protocol-specified treadmill assessments were used for the secondary analysis of time-dependence of PLC effect. Furthermore, in order to identify subgroups of patients with a different response to PLC, a broad array of clinical and demographic parameters was used to check for consistency of PLC effect. In this context, concomitant exercise refers to a patient self-reported, regular exercise regimen or a protocol-specified exercise program during the study period. No information on concomitant exercise was available from the US-Diabetes study.
These patient databases were also used for the safety analysis which was based on incidence of adverse events (AEs), serious AEs, deaths and the number of patient discontinuations.
Harmonization of treadmill peak performance assessments
The six phase III studies identified for the primary meta-analysis evaluated maximum walking capacity (either maximum walking distance (MWD) on a constant-load test or peak walking time (PWT) on a graded test) as the primary efficacy parameter. However, these studies used different treadmill methodologies (Table 1), precluding a direct combination of study results. Therefore, harmonization of the results was performed by converting the results of the different treadmill tests into a common measure for analysis (peak walking distance; PWD). This was accomplished by converting each subject’s peak performance on the treadmill test to the equivalent walking distance on a fixed treadmill of 7% slope and a speed of 3 km/hour. This calculation was done by first converting the peak performance to energy expenditure (joules/kg) using a previously developed methodology.23,24 This energy expenditure was then converted back to the equivalent walking distance at a 7% slope and 3 km/hour speed.
Statistical analysis
Changes in treadmill performance deviated substantially from a normal distribution, and log transformation did not adequately yield an appropriate distribution in most studies. Thus, in accordance with each study’s Statistical Analysis Plan, the meta-analyses of the change in treadmill performance over baseline (PLC vs placebo) were performed using the non-parametric Cochran–Mantel–Haenszel test on the modified ridits, which is equivalent to the van Elteren test. 25 Ridit (Relative to an Identified Distribution) analysis is an empirical cumulative probability transformation used on ordinal data which does not rely on an interval scale. Modified ridits are standardized midrank scores. Similarly, non-parametric approaches were used to calculate the 95% confidence intervals (CIs) and p-values. 26 Of note, the resulting CIs may be asymmetric due to the non-parametric methods needed for the non-normal distribution. The patient-level data were assessed for heterogeneity based on the chi-squared test. If the data were found to not be heterogeneous (i.e. were in fact homogeneous) then the prespecified decision was to use a fixed-effects model for the meta-analysis. The statistical software was SAS Software v.9.2 for Windows (SAS Institute, Inc., Cary, NC, USA).
Study-level meta-analysis of randomized, controlled trials
Study inclusion and exclusion criteria
Studies identified during the systematic review (see before) were included in the study-level meta-analysis if they: included patients with claudication confirmed by history and PAD confirmed on an ankle–brachial index (ABI) or toe–brachial index (TBI); were randomized, double-blinded and had a comparator arm (active or placebo); had complete data on the change (± standard deviation) from baseline for MWD and/or PWT assessed using a treadmill test; used orally administered PLC; were completed according to their protocol; and randomized patients with Leriche-Fontaine stage II disease (i.e. intermittent claudication). Excluded were studies with: no confirmation of claudication; no comparator arm; incomplete or no treadmill data on MWD or PWT; intravenous administration of PLC; incomplete patient data sets due to study discontinuation; and patients with ischemic rest pain or trophic lesions (Leriche-Fontaine stages III–IV). When duplicative studies were identified, only the most recent publication was considered.
Analysis population
In the retrieved articles, the mean change over baseline observed in the ITT population was used. If this endpoint was not available, the result from the randomized patient population was used.
Statistical analyses
Data from each retrieved study were abstracted using a data collection form, and results compared. Quality of the included studies was not graded. For each identified study, the treatment effect size was assessed by computing the mean difference from end of treatment relative to baseline for each group (PLC and placebo) and the difference between these group means was then normalized to their pooled standard deviation. Heterogeneity across studies was assessed by formal statistical testing, using a Q-test for heterogeneity. 27 Trial results were pooled in a statistical meta-analysis using weighted standardized mean differences and the estimator of Hedges and Olkin (Hedges’ g ), as described by Boissel et al. 27 The decision to use a fixed-effects and/or a random-effects model was used based on heterogeneity testing that defined the best model to use. Commercial software was used for all the analyses (MetaWin, statistical software for meta-analysis (v.2.1) by Rosenberg, Adams, and Gurevitch; Sinauer Associates Inc., 2007).
Results
Systematic review of all studies for PLC in claudication
Identified studies
A total of 85 studies including 3394 patients with claudication were identified using the above search strategy. Of these, 72 were excluded from subsequent analysis (n = 1540 patients, 45% of the total) because the reports were incomplete or had no treadmill data (n = 23), were reviews (n = 21), used intravenous PLC (n = 27), or oral PLC was administered only as a single dose (n = 1) (Figure 1). Appendix 1 (online only) provides a complete list of the excluded studies.
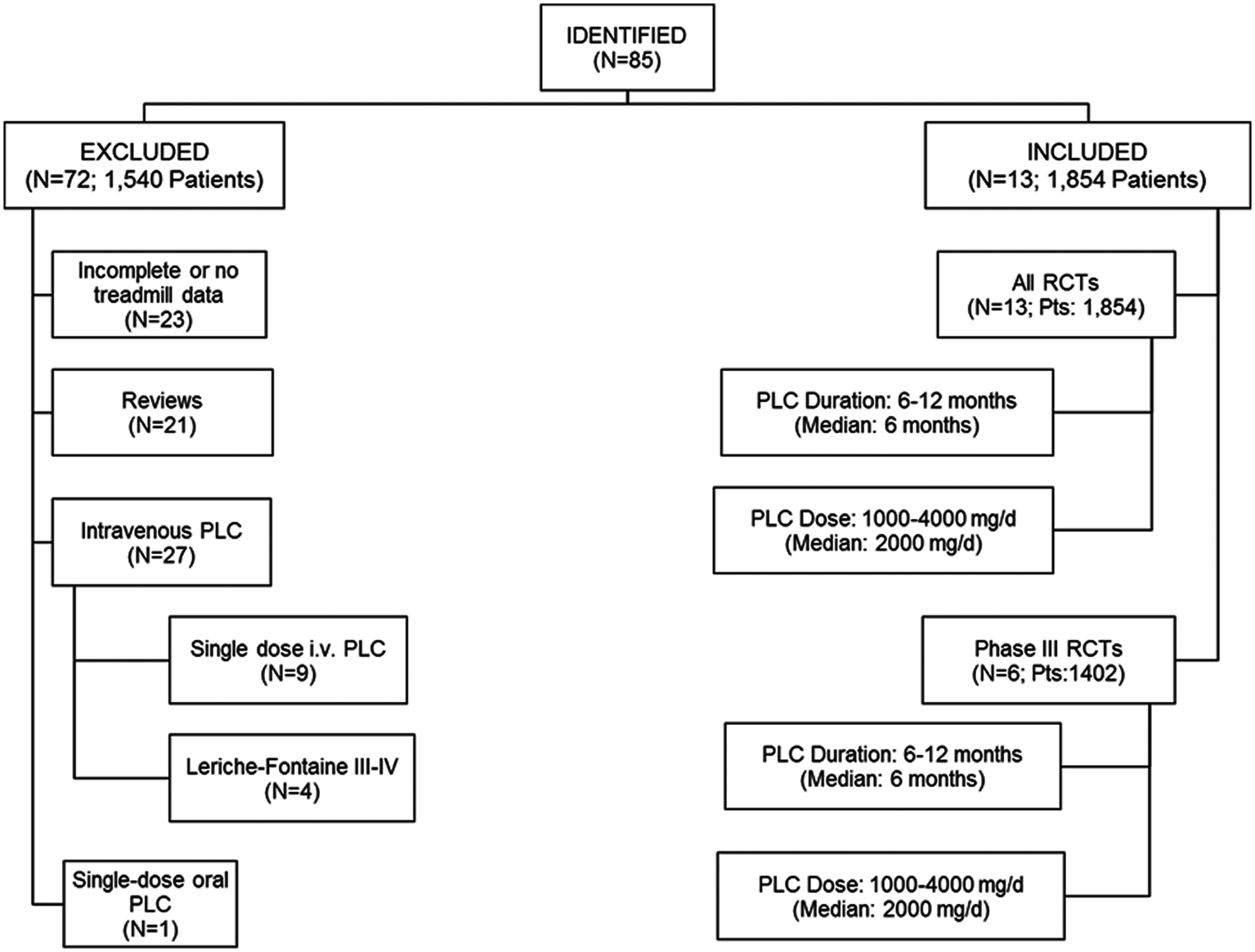
Systematic review: disposition of identified studies using PLC in intermittent claudication (January 1984 – September 2010). N refers to the number of studies and patient numbers are the total number of randomized subjects.
The remaining 13 studies (n = 1854 patients) were included in the subsequent analyses (Figure 1). All 13 studies were randomized controlled trials, exposing 1002 randomized patients with Leriche-Fontaine stage II claudication to a median dose of 2000 mg/day oral PLC (range: 1000–4000 mg/day), mostly in a twice daily schedule, for a median treatment duration of 6 months (range: 6–12). Six out of the 13 studies were phase III trials and contributed substantially to this cohort (n = 1402 randomized patients; Figure 1). An overview of the designs of the six phase III trials is provided in Table 1. The designs of the additional seven studies are summarized in Table 2.
Clinical studies meeting criteria for inclusion in the systematic review
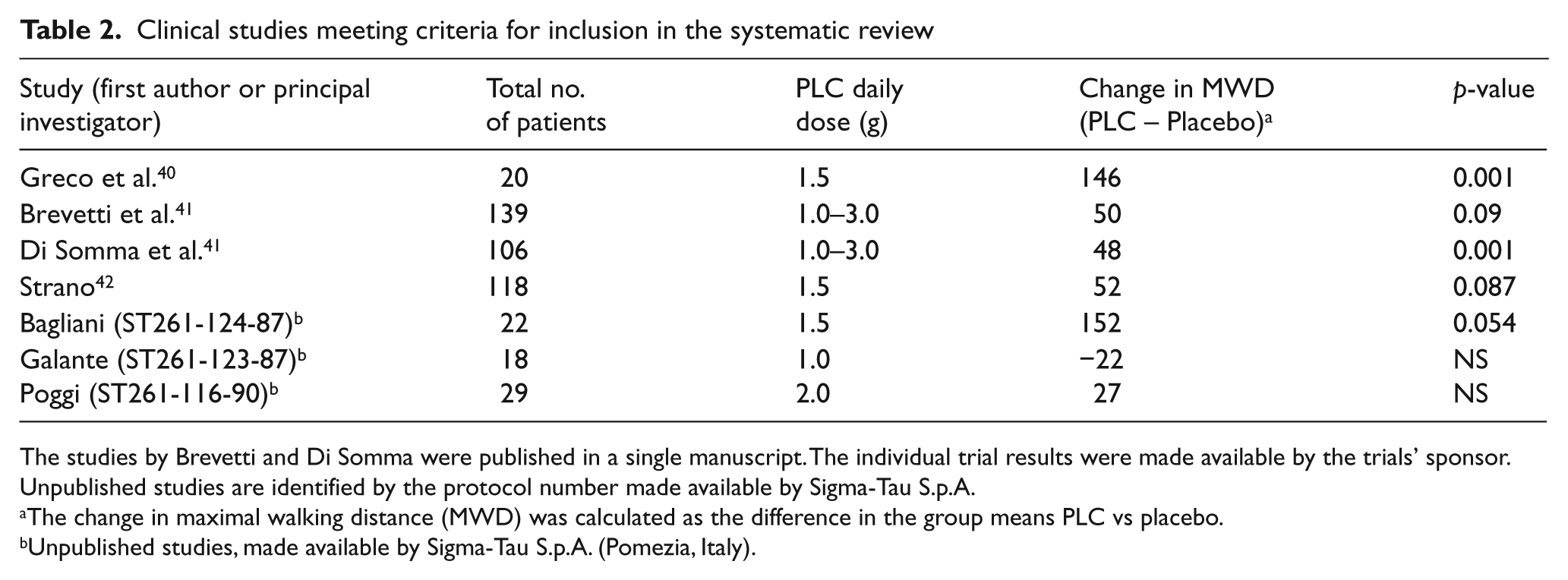
The studies by Brevetti and Di Somma were published in a single manuscript. The individual trial results were made available by the trials’ sponsor. Unpublished studies are identified by the protocol number made available by Sigma-Tau S.p.A.
The change in maximal walking distance (MWD) was calculated as the difference in the group means PLC vs placebo.
Unpublished studies, made available by Sigma-Tau S.p.A. (Pomezia, Italy).
Patient-level meta-analysis
Analysis population
The patient demographics comprising the primary analysis cohorts in the six phase III trials are described in Table 3. Patients were predominately male, were on average in their seventh decade of life, and had a high prevalence of current smoking. Studies differed in their inclusion of diabetics, with two studies (US-Diabetes and European-Diabetes) requiring a diagnosis of diabetes for subjects to be included, and one study (European) tending to under-represent diabetics. Baseline treadmill performance was similar in all studies. The concomitant use of statin and antiplatelet therapies appeared to increase in the more recent versus the earlier studies (Table 3).
Demographics of the ITT populations in the phase III trials included in the primary meta-analysis
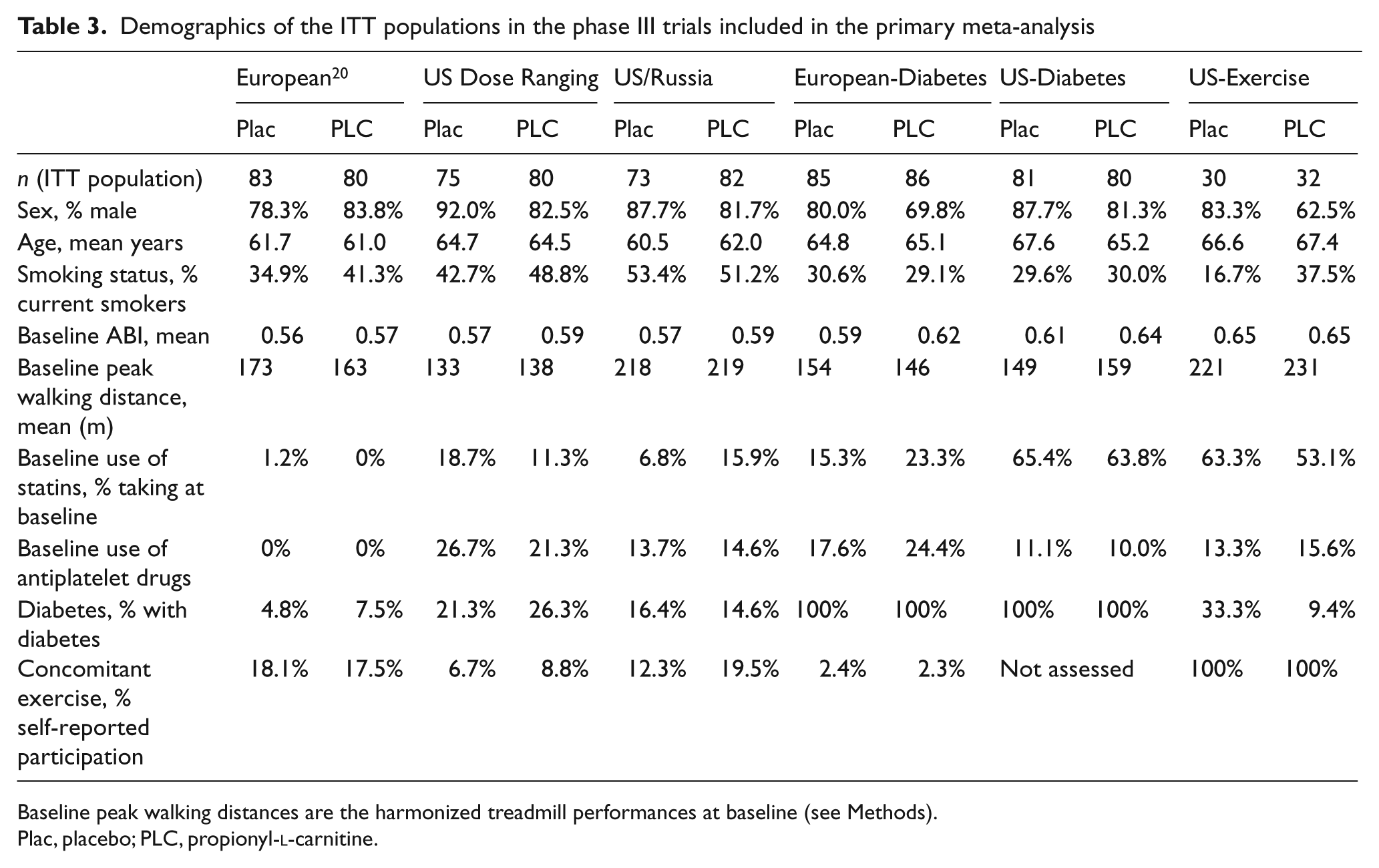
Baseline peak walking distances are the harmonized treadmill performances at baseline (see Methods).
Plac, placebo; PLC, propionyl-
Effect of oral PLC on peak walking distance
The ITT populations included a total of 427 patients randomized to placebo and 440 patients randomized to 1 g PLC orally twice daily. Analysis of the ITT populations demonstrated that PLC was associated with a net improvement of 16 meters (95% CI, 8–20 meters) in PWD as compared with placebo (p = 0.002; Figure 2, standardized effect size 0.11). Examination of the six individual studies demonstrated no heterogeneity in effect size (χ2: 9.36, df: 5; p > 0.05), consistent with the overlapping CIs across studies (Figure 2).
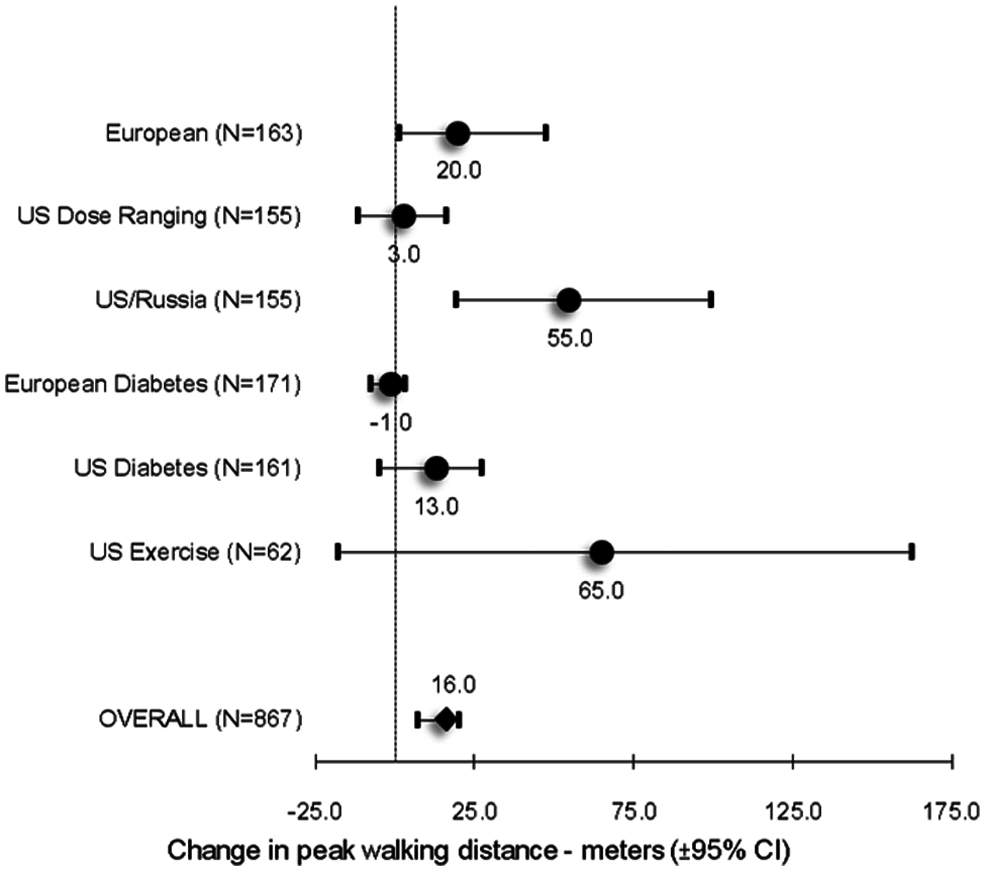
Effect of PLC on harmonized peak walking distance for patient-level meta-analysis. The results from ITT populations in the six phase III studies and the meta-analysis are shown as change over baseline vs placebo (± 95% CIs). The CIs are asymmetric due to the use of non-parametric analyses (see Methods).
The effect of PLC on PWD appeared consistent across a broad array of clinical and demographic parameters, except for suggestions of increased effect in patients who were participating in a concomitant exercise program during the study period, or who were randomized at sites in Russia (Figure 3).
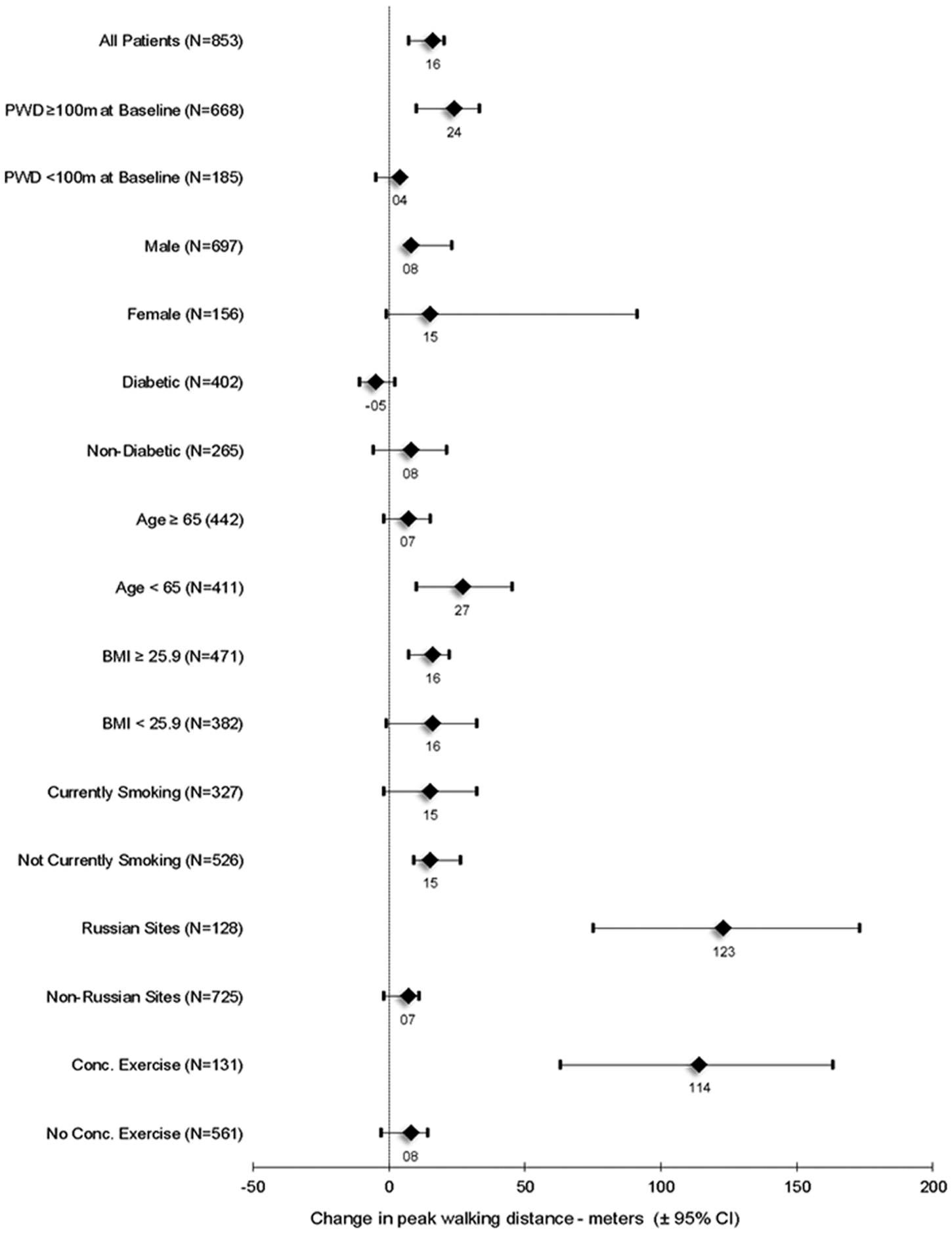
Effect of PLC on harmonized peak walking distance in patient subgroups (ITT populations) for patient-level meta-analysis. The results are from the six phase III studies and are based on patient characteristics at study entry. The data are expressed as change in peak walking distance over baseline vs placebo ± 95% CIs. The CIs are asymmetric, reflecting the use of non-parametric analyses (see Methods). N refers to the number of subjects in each subgroup and the point estimate is presented numerically. Conc, concomitant; PWD, peak walking distance (harmonized).
The six studies varied in duration from 6 to 12 months (Table 1) and included protocol-defined treadmill assessments at earlier time points on a protocol-specific basis. Estimates of improvement in PWD as a function of time suggested that the benefits of PLC were maintained over a treatment interval of 3–12 months (Figure 4).
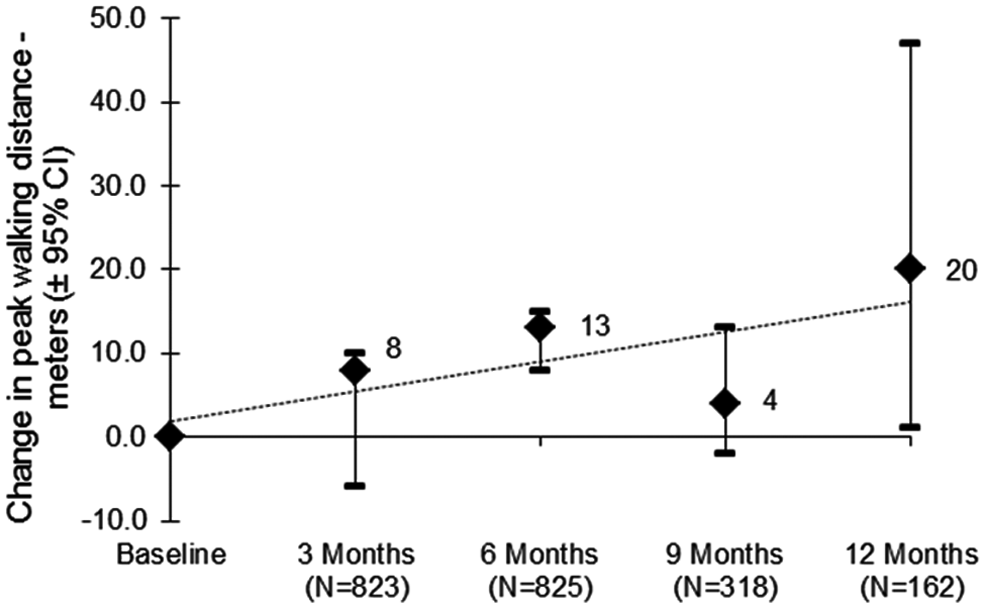
Time dependence of PLC effect on harmonized peak walking distance for patient-level analysis. Protocol-specified treadmill assessments were used. As the time points studied varied by protocol, the number of patients contributing to each time point in the figure also varies and a weighted regression was used. Results are the change over baseline vs placebo ± 95% CIs based on the ITT populations. Numerical values for the point estimate at each time point are provided. The CIs are asymmetric, reflecting the use of non-parametric analyses (see Methods). A trend line is shown for descriptive purposes.
Safety of oral PLC
PLC was generally well tolerated by patients with claudication, with discontinuation rates similar between placebo (n = 626) and PLC groups (n = 797) (Table 4). The incidence of serious AEs, cardiovascular serious AEs and deaths were balanced across the placebo and PLC arms. Across the six studies there were a total of 95 (15.2%) patients with serious AEs in patients randomized to placebo and 133 (16.7%) patients with serious events in the PLC arms. Gastrointestinal adverse events, the most common adverse events associated with PLC, 28 were similar between treatment groups (11.8% placebo, 14.3% PLC).
Safety profile of PLC (1 g orally twice daily) in patients with claudication (Safety population)
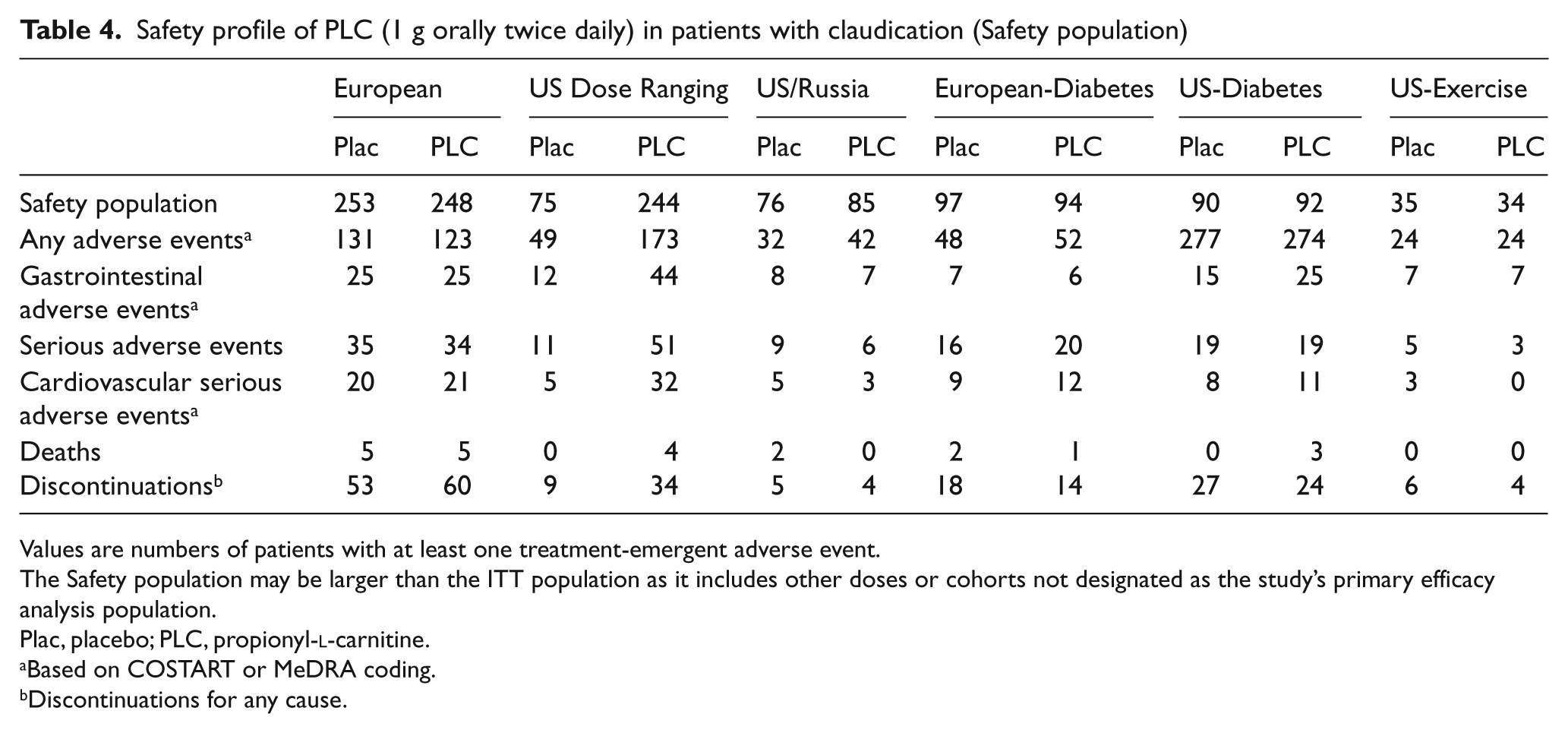
Values are numbers of patients with at least one treatment-emergent adverse event.
The Safety population may be larger than the ITT population as it includes other doses or cohorts not designated as the study’s primary efficacy analysis population.
Plac, placebo; PLC, propionyl-
Based on COSTART or MeDRA coding.
Discontinuations for any cause.
Meta-analyis of all randomized controlled trials
Effect of oral PLC on peak walking performance
An inspection of the funnel plot of the standardized difference in PWD observed in each of the 13 included studies versus the study’s sample size was consistent with an absence of publication bias, as confirmed by a Kendall’s test (tau: –0.29, p = 0.17; Figure 5). Similar results were obtained when the variance in each study effect size was used instead of sample size (data not shown).
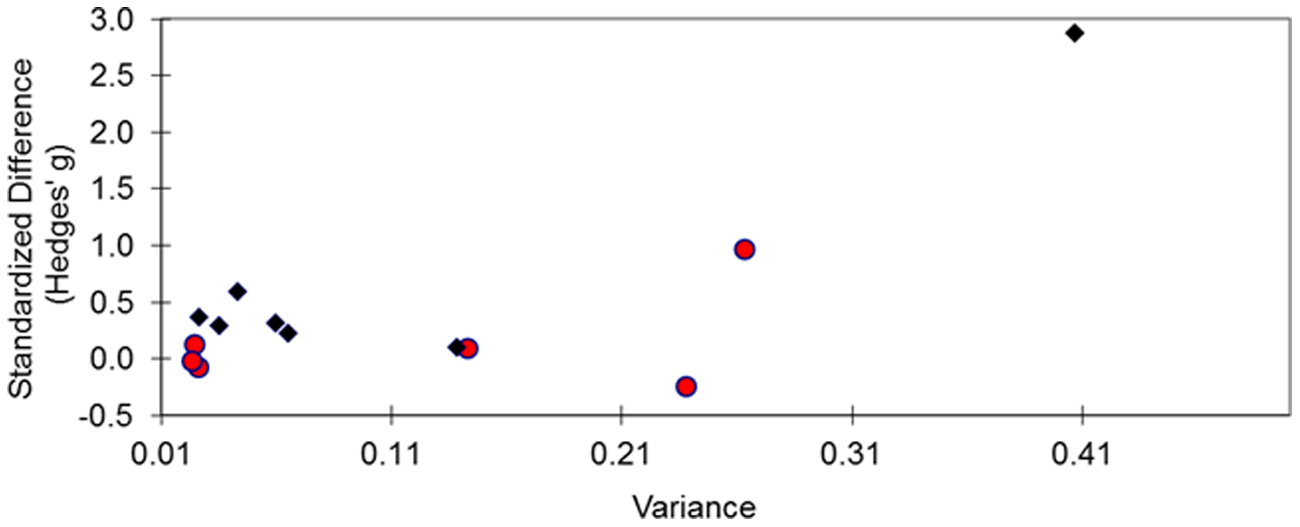
Funnel plot of PLC effect size (standardized difference) vs sample size (total number of randomized patients) in each individual study. Diamonds identify published studies.
The study-level meta-analysis of the 13 randomized controlled trials identified a Q-test for heterogeneity that was not significant at p = 0.08 when using a random-effects model. When the random-effects model was used, the improvement in PWD induced by PLC was 45 meters (95% CI, 7–84 meters, standardized effect size 0.28; Figure 6). In contrast, a fixed-effects model demonstrated the studies were heterogeneous (Q-test for heterogeneity, p < 0.01). With the fixed-effects model there was a statistically significant effect of PLC in patients with claudication, with an improvement in PWD of 35 meters (95% CI, 13–57 meters, standardized effect size 0.22).
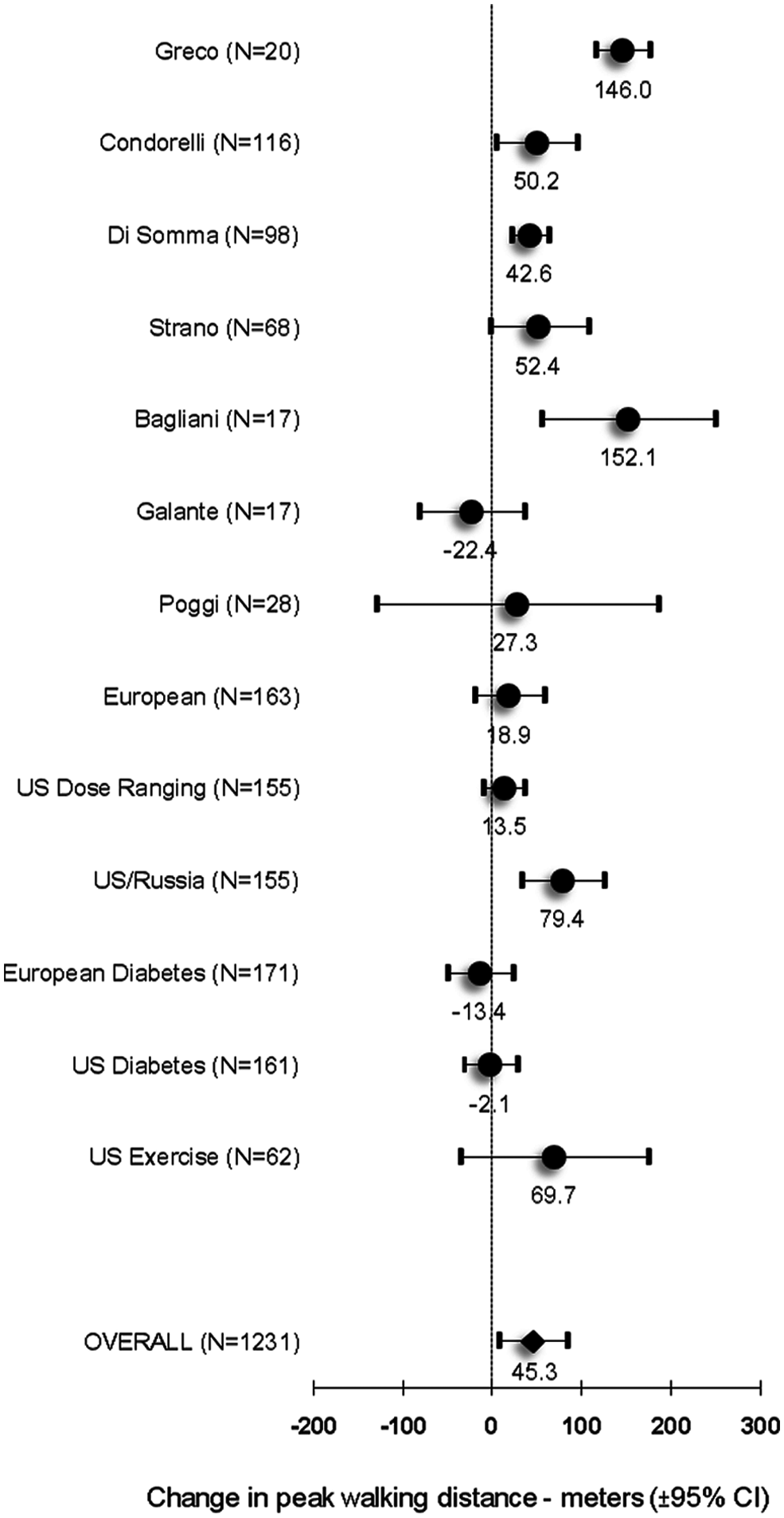
Effect of PLC on maximum walking distance. The results from all included studies and their meta-analysis are shown as change over baseline vs placebo (± 95% CIs, random-effects model).
Discussion
Efficacy of PLC
In PAD patients with claudication, a patient-level meta-analysis of six phase III trials demonstrated that oral PLC (1 g twice daily) increased harmonized treadmill walking distance by 16 meters (95% CI, 8–20 meters), as compared with placebo. This conclusion was confirmed by a systematic review and study-level meta-analysis that included 13 studies overall (including the above six phase III studies), which demonstrated a 45 meter improvement (95% CI, 7–84 meters, random-effects model). These improvements corresponded to standardized effect sizes of 0.11 and 0.28, respectively. The benefit of oral PLC did not appear to be restricted to any subgroup based on demographic or clinical features (Figure 3). Patients were randomized at sites in Russia in two studies (European and US-Russia), and this patient cohort seemed to benefit more from PLC treatment for reasons that may be associated with either the demographics or the standard of care of these patients (Figure 6). However, the specific reasons for this difference are not known, but both studies were conducted to Good Clinical Practice standards and were monitored and audited. Of note, in the US-Russia study, the drug effect was statistically significant even after adjusting for the country effect.
The patient-level meta-analysis was considered primary and the most robust as all individual phase III patient data were included, thus allowing for more accurate estimates. As the total cohort was not truly randomized, but only randomized within study, this approach may have some limitations. Nonetheless, each study was sufficiently large to minimize randomization bias and the effects of PLC were not significantly heterogeneous across trials. The effect estimates from the patient-level meta-analysis differ from those of the study-level meta-analysis because of the different patient groups selected, lack of treadmill data harmonization in the latter, and the specific analysis methods used. Of note, although the study-level meta-analysis included several smaller studies, varied treadmill methods, and PLC administered at different doses, its results overlap with those of the patient-level meta-analysis and thus can be viewed as supportive of its conclusions.
The importance of accessing unpublished data and the potential bias of the published literature are illustrated by the changes in PWD associated with PLC in published versus unpublished studies (Tables 1 and 2). On a sample size-weighted basis, the improvement with PLC in published studies was approximately 44.5 meters versus 8.5 meters in the unpublished trials.
The estimates of the effect of PLCs on maximal walking performance observed in the current analyses are similar to that of other drugs approved for claudication in the United States, including pentoxyphylline (44 meters; 95% CI, 14–74 meters) 29 and cilostazol (42 meters; 95% CI, 21–64 meters). 30 The effect of all drugs, however, appears smaller than that of non-pharmacological interventions, such as supervised exercise training (113 meters; 95% CI, 95–131 meters). 31 Importantly, these apparently modest changes in treadmill walking performance on drug therapy have been associated with patient-perceived increases in ambulatory walking ability. 32
The clinical pharmacology of PLC may limit its efficacy as an oral therapy for claudication. The oral systemic bioavailability of PLC is likely not higher than the approximately 10–15% observed with carnitine, 33 and peak PLC plasma concentrations of only 5 µM are achieved. 34 Further, PLC may have limited access to intracellular sites of action. The skeletal muscle carnitine pool is kinetically isolated from the plasma pool. 35 Uptake of carnitine or acylcarnitines into muscle under normal conditions is slow, and even large doses of intravenous carnitine have minimal acute effects on the muscle carnitine content. 35 Thus, it can be hypothesized that better delivery of PLC to skeletal muscle in patients with PAD might improve its efficacy. Supporting this concept are data showing short-term benefit of intravenous carnitine or PLC in patients with PAD.36,37 In this context it is interesting to note that the full benefit on walking performance of oral PLC appears to require a long duration of treatment (Figure 4), thus suggesting slow accumulation in muscle. Insulin has been shown to enhance muscle uptake of carnitine, 38 but it is unclear whether a similar effect on PLC uptake exists. In this context, a trend towards greater efficacy in non-diabetic patients (i.e. patients without insulin resistance) was observed (Figure 3).
Safety of oral PLC
Published reviews of PLC and other carnitine therapeutics conclude that the drugs have a low incidence of AEs and no serious toxicities.28,39 The data from the six phase III trials support this conclusion in the PAD population. The drug appeared to be well tolerated with discontinuation rates similar between the treatment arms. Gastrointestinal AEs occurred at a low rate, without relevant treatment-related differences observed. The incidence of serious AEs, cardiovascular serious AEs and deaths were balanced between treatment groups. Across the studies, a total of nine (1.4%) deaths were reported in patients randomized to placebo and 13 (1.6%) in patients on PLC.
Limitations of the analysis.
No meta-analysis can likely support conclusions as robust as those obtained from a single, adequately powered, well-conducted clinical trial. While the primary meta-analysis was able to utilize patient-level data, it is important to note that the six phase III studies randomized different patient populations, treatment duration varied by study, and background therapy differed either by study design (e.g. background exercise rehabilitation) or reflecting evolution of standards of care during the 15-year period covered by the six studies (e.g. use of statins). Differences in discontinuation rates across studies (Table 4), while balanced between treatment arms, also suggest potential unrecognized heterogeneity between such studies. Nonetheless, both the overlap of the 95% CIs for the estimated PLC effects (Figure 3) and the result of the chi-squared test for homogeneity suggest that the PLC effect did not vary appreciably across studies. Further, analysis of subpopulations failed to identify dramatic differences in PLC effect, with very few notable exceptions (Figure 3), although female patients were under-represented. It is possible, but unlikely, that additional clinical trial data with more unfavorable results exist but were not included in this systematic review. Finally, while treadmill performance is an accepted surrogate for clinical function based on quality of life and other ambulatory assessments, the current analyses focused exclusively on treadmill performance and did not directly assess clinical impact.
Conclusions
Analyses of patient-level data from six phase III studies, as well as a study-level meta-analysis of clinical trial data from 13 randomized controlled trials, demonstrate that PLC improves walking performance in patents with claudication. The overall standardized effect size was 0.11 in the patient-level analysis of the phase III studies and 0.28 in the publication-level meta-analysis, each of which was statistically significant (p < 0.002) and of a magnitude representing a net benefit of 16–45 meters. PLC was in general well tolerated, with no relevant safety signals identified.
Footnotes
Acknowledgements
The authors thank Sigma-Tau S.p.A. and Joe Testa at Sigma-Tau Research, Inc. for facilitating access to the study databases for analysis.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
EPB and WRH were consultants to Sigma-Tau S.p.A. DK is an active consultant for Sigma-Tau S.p.A. AA is an employee of Sigma-Tau S.p.A.
