Abstract
Background:
Ozanimod, a selective, oral sphingosine 1-phosphate receptor 1 and 5 modulator, is approved in multiple countries for adults with relapsing forms of multiple sclerosis (RMS).
Objective:
To characterize the long-term safety and efficacy of ozanimod.
Methods:
Participants were eligible for an open-label extension study of ozanimod 0.92 mg/d (DAYBREAK) if they completed a phase 1–3 RMS ozanimod ‘parent’ trial. DAYBREAK began 16 October 2015, and ended 5 January 2023.
Results:
DAYBREAK included 2494 participants with a mean of 60.9 (range 0.03‒81.5) months of ozanimod exposure. During DAYBREAK, 2219 participants (89.0%) had treatment-emergent adverse events (TEAEs), 381 (15.3%) had a serious TEAE and 98 (3.9%) discontinued treatment due to TEAEs. Serious infections (4.3%), herpes zoster infections (2.0%), confirmed macular oedema cases (0.2%), and cardiac TEAEs (4.1 %) were infrequent. Adjusted annualized relapse rate was 0.098 (95% confidence interval, 0.082‒0.117). In total, 69.1% of participants remained relapse-free and 84.8% were free from 6-month confirmed disability progression at DAYBREAK completion. Adjusted mean numbers of new/enlarging T2 lesions/scan and gadolinium-enhancing lesions were low and remained relatively stable.
Conclusions:
Long-term ozanimod treatment had a favourable safety and tolerability profile and provided sustained control of clinical and MRI disease activity in participants with RMS.
Registries:
ClinicalTrials.gov ID: NCT02576717; EudraCT: 2015-002500-91.
Keywords
Introduction
Ozanimod, a selective, oral sphingosine 1-phosphate (S1P) receptor 1 and 5 modulator, is approved in multiple countries for the treatment of adults with relapsing forms of multiple sclerosis (RMS) or moderately to severely active ulcerative colitis.1–3 The safety and efficacy of ozanimod were studied in phase 1–3 RMS ‘parent’ trials—a phase 1 pharmacokinetic/pharmacodynamic trial, a phase 2 trial with a dose-blinded extension (RADIANCE part A and part A extension), and two phase 3 trials (SUNBEAM and RADIANCE part B).4–7 In the phase 3 RMS trials, ozanimod 0.92 mg/d demonstrated superior efficacy to intramuscular interferon (IFN) β-1a 30 µg/wk in clinical and magnetic resonance imaging (MRI) measures of RMS disease activity.6,7 Ozanimod treatment was well tolerated, with low rates of discontinuation and lower incidences of adverse events (AEs) than IFN β-1a.6,7
DAYBREAK was an open-label extension (OLE) trial of oral ozanimod 0.92 mg/d that aimed to determine the long-term safety (primary objective) and efficacy (secondary objective) of ozanimod in participants with RMS who completed a phase 1–3 parent trial.8,9 An interim analysis of DAYBREAK confirmed the favourable safety/tolerability profile of ozanimod and captured sustained benefits on clinical and MRI measures of disease activity. 8 The data presented here describe up to 6.8 years of follow-up in DAYBREAK and constitute the final trial results.
Methods
Detailed methods are described in the Supplemental Material.
DAYBREAK study design
Upon completion of a parent trial, participants were eligible to enter the single-arm, open-label ozanimod 0.92 mg/d, phase 3 DAYBREAK trial (ClinicalTrials.gov ID: NCT02576717; EudraCT: 2015-002500-91) (Figure 1). DAYBREAK began October 16, 2015, and was conducted in 25 countries in Europe and North America, plus South Africa and New Zealand. The last participant assessment was January 5, 2023, with a database lock on April 7, 2023.
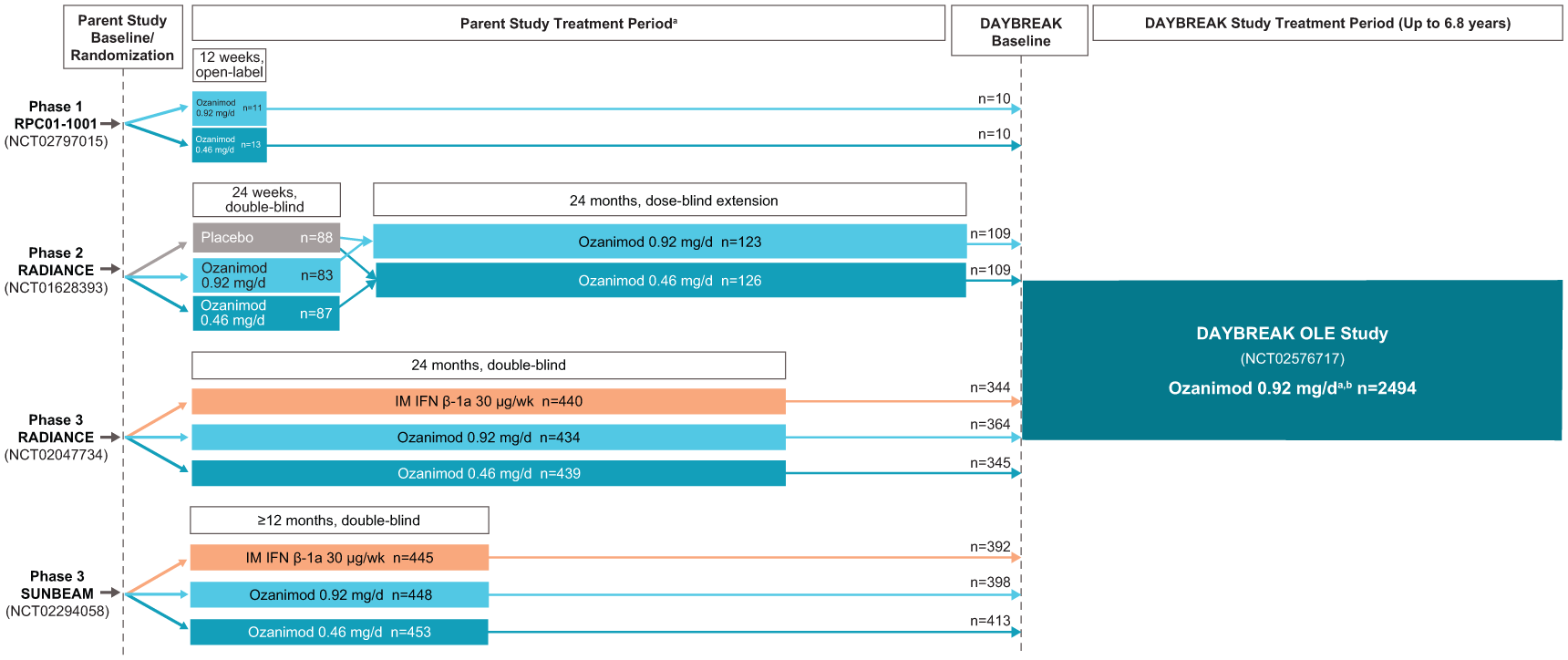
DAYBREAK design.
Oversight and ethical principles
The institutional review board or ethics committee at each site approved the study protocol (Supplemental Table 1). This study was conducted in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki principles. All DAYBREAK participants or their legal representatives provided written consent before participation.
Outcomes
Safety
Pre-specified outcomes included overall treatment-emergent adverse events (TEAEs) and those potentially associated with S1P receptor modulators (infections, malignancies, macular oedema, cardiac and vascular events, and pulmonary and hepatic laboratory abnormalities). Absolute lymphocyte count (ALC) changes are reported as an expected pharmacodynamic effect of ozanimod. Confirmed and suspected COVID-19 cases are also reported.
Efficacy
Pre-specified outcomes included annualized relapse rate (ARR), time to onset of 3- and 6-month confirmed disability progression (CDP-3 and CDP-6; ⩾1-point increase in Expanded Disability Status Scale (EDSS) from DAYBREAK baseline, maintained for 3 or 6 months, respectively), numbers of new/enlarging T2 lesions per scan and gadolinium-enhancing (GdE) lesions on brain MRI, and the proportion of participants free of new/enlarging T2 lesions per scan and GdE lesions. Symbol Digit Modalities Test (SDMT) scores were assessed post hoc to determine long-term impacts on cognitive processing speed in participants from SUNBEAM (SDMT was assessed as part of the MS functional composite score in SUNBEAM but not phase 3 RADIANCE).
Statistical analyses
Safety
TEAEs were reported from DAYBREAK baseline through the end of the trial; laboratory abnormalities were reported from DAYBREAK baseline through months 60 or 72 (data thereafter were limited to a small number of participants). To assess safety by yearly interval, the number of participants with a TEAE during each interval was divided by the total number of participants who were either ongoing or had discontinued treatment in that interval, with participants counted in multiple intervals based on TEAE timing, onset date and treatment duration, but only counted once per interval if the same TEAE occurred more than once. Confirmed and suspected COVID-19 cases were collected using COVID-19 preferred terms (Medical Dictionary for Regulatory Activities 24.1) and assessed for seriousness, action taken with ozanimod and outcome. AE severity was determined by investigators as mild (transient without interfering with daily activities), moderate (low level of inconvenience or concern possibly interfering with daily activities) or severe (incapacitating and interrupting daily activities). AEs were considered serious if they resulted in death, were considered life-threatening, required inpatient hospitalization or prolongation of existing hospitalization, or resulted in persistent or significant disability. Malignancies are reported from the date of first ozanimod exposure to the end of DAYBREAK.
Efficacy
ARR, new/enlarging T2 lesions and number of GdE lesions were calculated using adjusted negative binomial regression models. Time to onset of 3- and 6-month CDP was analysed via Kaplan–Meier analysis. Unless otherwise specified, ARR and CDP were reported from DAYBREAK baseline through the end of the trial or discontinuation; other efficacy outcomes, including ARR by yearly interval and SDMT, were reported from DAYBREAK baseline through months 60 or 72 because data thereafter were limited to a small number of participants.
Results
Disposition, baseline demographics and disease characteristics, and ozanimod exposure
Of 2639 participants who completed a parent trial, 2494 (94.5%) enrolled in DAYBREAK and received ⩾1 dose of ozanimod 0.92 mg (Figure 2). In DAYBREAK, most participants were women (66.9%), White (99.2%) and from Central/Eastern Europe (90.1%) (Table 1). Baseline demographics and disease characteristics were generally similar across parent trial treatment groups (Supplemental Table 2). In DAYBREAK, the overall mean duration of ozanimod 0.92 mg exposure was 60.9 months (median exposure: 67.6 months; maximum exposure: 81.5 months); when including treatment duration in the parent trials, the overall mean duration of exposure to any dose of ozanimod was 74.8 months (median exposure: 81.2 months; maximum exposure: 117.2 months) (Table 2; Supplemental Table 3). Most participants (1989/2494; 79.8%) received ozanimod for ⩾60 months in DAYBREAK (Table 2; Supplemental Figure 1), and most (1950/2494; 78.2%) completed the study, with 544 of 2494 (21.8%) discontinuing (Figure 2). Of the 544 participants who discontinued, 116 (21.3%) discontinued due to family planning or pregnancy.
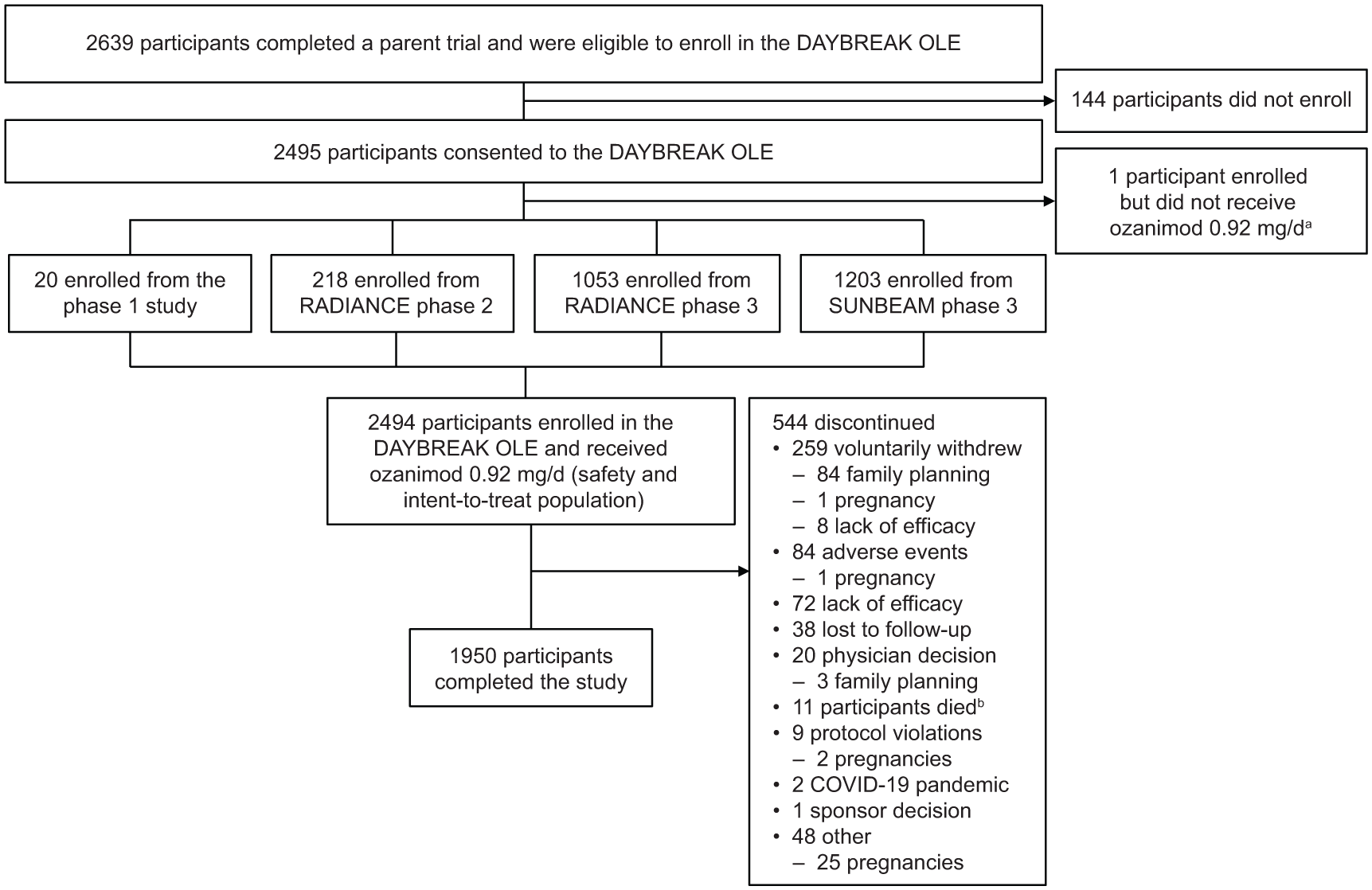
DAYBREAK flow diagram.
Demographics and baseline disease characteristics.

Source: Adapted with permission from Cree et al. 8
Safety population. Baseline characteristics are from DAYBREAK baseline unless otherwise specified. EDSS: Expanded Disability Status Scale; SD: standard deviation.
Rest of the world includes New Zealand and South Africa.
At parent trial baseline.
n = 2474.
Extent of exposure to ozanimod.

Safety population. The extent of exposure to ozanimod during DAYBREAK was defined as days from the first dose date (study day 1) to the last dose date. PY of exposure was the cumulative duration of exposure summed for all participants within a treatment group in days divided by 365.25; the extent of exposure in months was calculated as days divided by 30.44. max: maximum; min: minimum; OLE: open-label extension; PY: person-years; SD: standard deviation.
Safety
Overall
One or more TEAEs occurred in 2219 of 2494 (89.0%) participants, with an incidence rate (IR) of 650.8/1000 person-years (PY) (95% CI: 624.3‒678.5), the most common being nasopharyngitis (531/2494 (21.3%)), headache (426/2494 (17.1%)), COVID-19 (412/2494 (16.5%)), upper respiratory tract infection (310/2494 (12.4%)), and lymphopenia (257/2494 (10.3%)) (Table 3). Serious TEAEs occurred in 381 of 2494 (15.3%) participants, severe TEAEs in 240 of 2494 (9.6%), and TEAEs leading to permanent treatment discontinuation in 98 of 2494 (3.9%) (Table 3). Of the serious TEAEs, three events (pyelonephritis, sensory disturbance and visual impairment) reported in two participants were considered treatment-related. The percentages of participants with TEAEs generally decreased over time (Supplemental Figure 2A), while the percentages of participants with severe or serious TEAEs remained low and stable (Supplemental Figure 2BC). The percentages of participants with any, severe, or serious TEAEs were similar across parent trial treatment groups (Supplemental Table 4). The most common TEAEs leading to treatment discontinuation were increased alanine aminotransferase (7/2494 (0.3%), increased aspartate aminotransferase (5/2494 (0.2%), decreased ALC (4/2494 (0.2%); 0.1–0.2 × 109/L), breast cancer (4/2494 (0.2%) and macular oedema (4/2494 (0.2%); 3 of these cases were confirmed).
Overall safety during DAYBREAK.
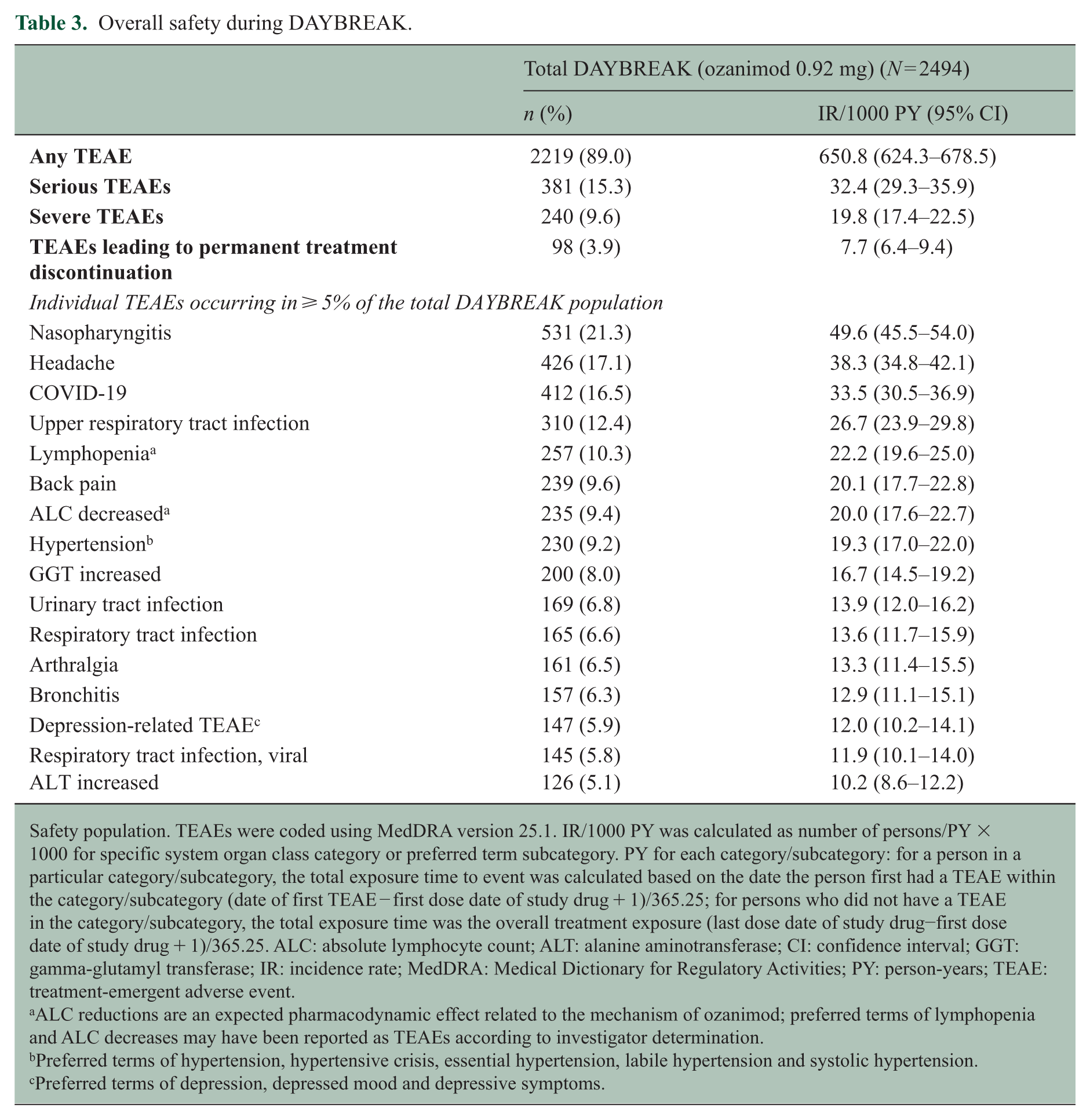
Safety population. TEAEs were coded using MedDRA version 25.1. IR/1000 PY was calculated as number of persons/PY × 1000 for specific system organ class category or preferred term subcategory. PY for each category/subcategory: for a person in a particular category/subcategory, the total exposure time to event was calculated based on the date the person first had a TEAE within the category/subcategory (date of first TEAE − first dose date of study drug + 1)/365.25; for persons who did not have a TEAE in the category/subcategory, the total exposure time was the overall treatment exposure (last dose date of study drug−first dose date of study drug + 1)/365.25. ALC: absolute lymphocyte count; ALT: alanine aminotransferase; CI: confidence interval; GGT: gamma-glutamyl transferase; IR: incidence rate; MedDRA: Medical Dictionary for Regulatory Activities; PY: person-years; TEAE: treatment-emergent adverse event.
ALC reductions are an expected pharmacodynamic effect related to the mechanism of ozanimod; preferred terms of lymphopenia and ALC decreases may have been reported as TEAEs according to investigator determination.
Preferred terms of hypertension, hypertensive crisis, essential hypertension, labile hypertension and systolic hypertension.
Preferred terms of depression, depressed mood and depressive symptoms.
Fifteen deaths occurred during the study or safety follow-up. Two participants died from malignancies (bladder cancer and disseminated cancer with unknown primary focus), two from accidents, two from pulmonary embolism and one each from abscess of the right lung, COVID-19, COVID-19 bilateral pneumonia, COVID-19 infection, COVID-19 pneumonia, heart failure, intracerebral haemorrhage, pneumonia and sudden death. Two additional deaths were reported after the required safety follow-up period due to malignancies (metastatic pancreatic carcinoma and glioblastoma).
Infections
Infections occurred in 1596 of 2494 (64.0%) participants, with an IR of 229.3 of 1000 PY (95% CI: 218.3‒240.8), the most common being nasopharyngitis (531 (21.3%)), COVID-19 (412 (16.5%)), and upper respiratory tract infection (310 (12.4%)). A total of 12 of 2494 (0.5%) participants permanently discontinued ozanimod due to infections, the most common reason being pneumonia (3/2494 (0.1%)). The percentages of participants with infections generally decreased over time (Supplemental Figure 2D).
Serious infections occurred in 106 of 2494 (4.3%) participants, with an IR of 8.5 of 1000 PY (95% CI: 7.0‒10.3), the most common being COVID-19 pneumonia (21/2494 (0.8%)), COVID-19 (19/2494 (0.8%)) and pneumonia (12/2494 (0.5%)) (Table 4). Opportunistic infections occurred in 159 of 2494 (6.4%) participants, with an IR of 13.1 of 1000 PY (95% CI: 11.2‒15.3), the most common being oral herpes (58 (2.3%)), with a 4.7 IR of 1000 PY (95% CI: 3.6–6.0) and herpes zoster (50 (2%)), with a 4.0 IR of 1000 PY (95% CI: 3.0–5.3) (Table 4). The percentages of participants with serious or opportunistic infections remained low and stable over time (Supplemental Figure 2EF). In addition, the percentages of participants with serious or opportunistic infections were similar across parent trial treatment groups (Supplemental Table 5). Three participants temporarily discontinued ozanimod due to herpes zoster; none of the oral herpes or herpes zoster cases were serious or resulted in permanent discontinuation from ozanimod. One serious opportunistic infection was reported in DAYBREAK: a confirmed case of progressive multifocal leukoencephalopathy (PML) that was previously reported in the interim analysis. 8
Serious and opportunistic infections during DAYBREAK.
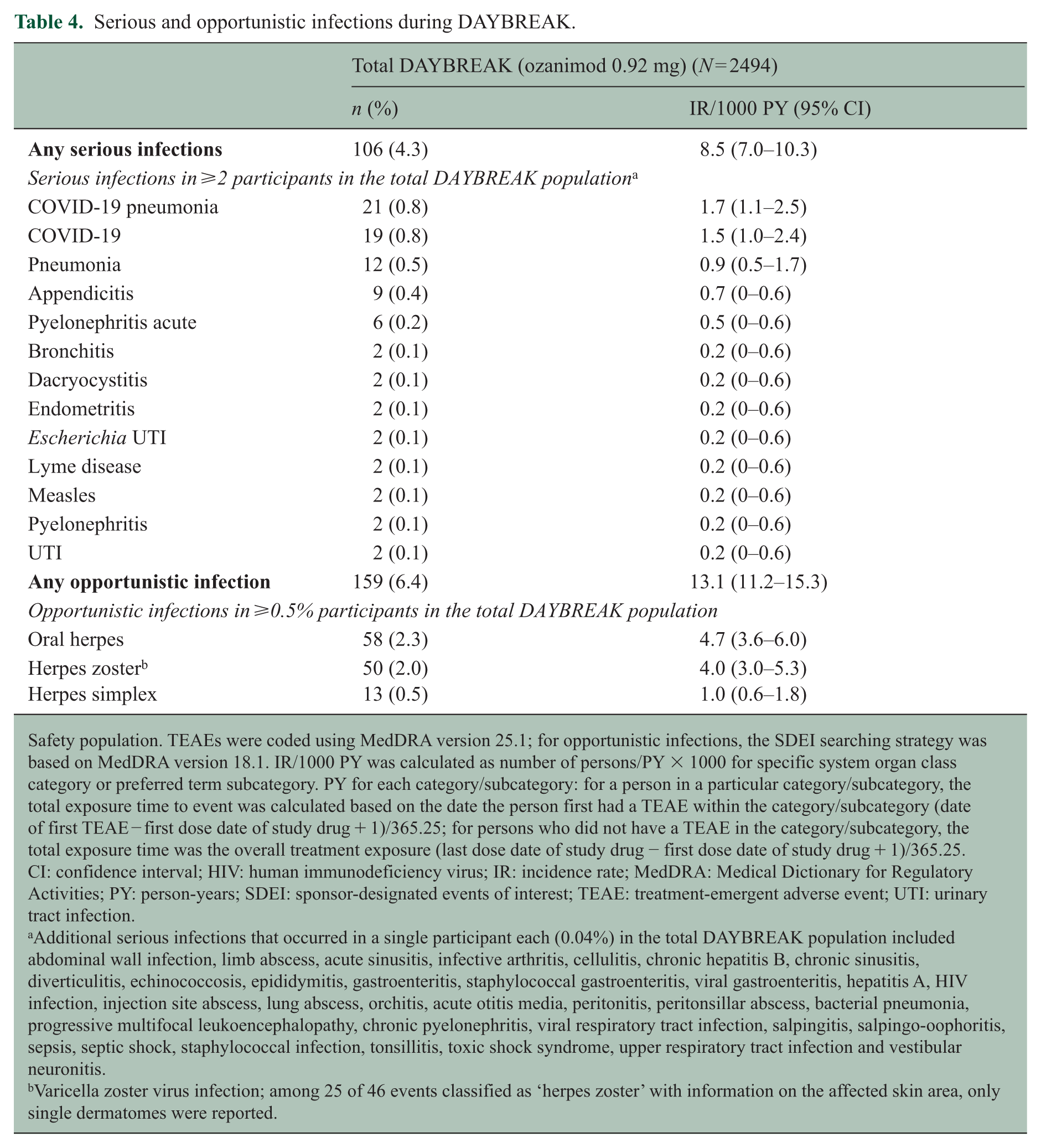
Safety population. TEAEs were coded using MedDRA version 25.1; for opportunistic infections, the SDEI searching strategy was based on MedDRA version 18.1. IR/1000 PY was calculated as number of persons/PY × 1000 for specific system organ class category or preferred term subcategory. PY for each category/subcategory: for a person in a particular category/subcategory, the total exposure time to event was calculated based on the date the person first had a TEAE within the category/subcategory (date of first TEAE − first dose date of study drug + 1)/365.25; for persons who did not have a TEAE in the category/subcategory, the total exposure time was the overall treatment exposure (last dose date of study drug − first dose date of study drug + 1)/365.25. CI: confidence interval; HIV: human immunodeficiency virus; IR: incidence rate; MedDRA: Medical Dictionary for Regulatory Activities; PY: person-years; SDEI: sponsor-designated events of interest; TEAE: treatment-emergent adverse event; UTI: urinary tract infection.
Additional serious infections that occurred in a single participant each (0.04%) in the total DAYBREAK population included abdominal wall infection, limb abscess, acute sinusitis, infective arthritis, cellulitis, chronic hepatitis B, chronic sinusitis, diverticulitis, echinococcosis, epididymitis, gastroenteritis, staphylococcal gastroenteritis, viral gastroenteritis, hepatitis A, HIV infection, injection site abscess, lung abscess, orchitis, acute otitis media, peritonitis, peritonsillar abscess, bacterial pneumonia, progressive multifocal leukoencephalopathy, chronic pyelonephritis, viral respiratory tract infection, salpingitis, salpingo-oophoritis, sepsis, septic shock, staphylococcal infection, tonsillitis, toxic shock syndrome, upper respiratory tract infection and vestibular neuronitis.
Varicella zoster virus infection; among 25 of 46 events classified as ‘herpes zoster’ with information on the affected skin area, only single dermatomes were reported.
A total of 402 of 2181 (18.4%) participants experienced confirmed (272 (12.5%)) or suspected (130 (6.0%)) COVID-19 while receiving ozanimod. Of the confirmed cases, most (250 of 272 (91.9%) were nonserious, and ozanimod was continued without interruption in 184 of 272 participants (67.6%); the majority (261/272 (95.6%)) recovered without sequelae (Supplemental Figure 3).
Malignancies
In the total DAYBREAK population, malignancies occurred in 41 of 2494 (1.6%) participants, with an IR of 324.8 of 100,000 PY (95% CI: 233.1–440.6), the most common being nonmelanoma skin cancer (13 (0.5%)) (Supplemental Table 6). Malignancies were reported in 50 of 2787 (1.8%) participants exposed to either ozanimod dose during the parent trials or DAYBREAK, with nonmelanoma skin cancer remaining the most common (18 (0.6%)). Overall, the percentages of participants in the total DAYBREAK population with cutaneous or noncutaneous malignancies were low (14/2494 (0.6%) and 27/2494 (1.1%), respectively). Malignant melanoma, seminomas, and adenocarcinoma (endometrial) were reported in 1 of 2494 (0.04%) participant each. Of the 1668 women in DAYBREAK, breast cancer was reported in six (0.4%) and cervical cancer in three (0.2%). The percentages of participants with malignancies remained low and stable over time (Supplemental Figure 2G).
Macular oedema
Macular oedema was reported in 10 of 2494 (0.4%) participants, with an IR of 0.8 of 1000 PY (95% CI: 0.4–1.5), of which, five cases with available optical coherence tomography data were confirmed as having macular oedema by a Macular Edema Review Panel. The percentages of participants with reported and confirmed macular oedema were similar across parent trial treatment groups (Supplemental Table 7). Of the 5 of 2494 (0.2%) participants with confirmed macular oedema (0.4 IR/1000 PY (95% CI: 0.2–0.9)), three permanently discontinued ozanimod (two cases were related to treatment and one was possibly related) and two remained on ozanimod (these cases were not deemed related to treatment). One of the participants with macular oedema who remained on ozanimod was diagnosed the day after their last ozanimod dose at the end of DAYBREAK, and even though they had stopped treatment, this case was not classified as discontinued due to timing. The other participant’s continuation was based on the trial investigator’s decision, considering the absence of symptoms, causality that was deemed unrelated and recovery with treatment while on ozanimod; however, the study protocol warranted discontinuation of ozanimod treatment in participants with confirmed macular oedema. Onset of macular oedema occurred after 15–2609 days of ozanimod exposure. At the end of DAYBREAK, 3 of 5 cases recovered/resolved; among the two participants that remained on ozanimod, one case was recovering/resolving and one case had not recovered/resolved by the end of the safety follow-up period. In 4 of 5 (80%) cases, confounding factors may have contributed to macular oedema onset (Supplemental Table 8).
Cardiac and vascular TEAEs
Cardiac TEAEs occurred in 103 of 2494 (4.1%) participants at any point in DAYBREAK, with an IR of 8.3 of 1000 PY (95% CI: 6.8–10.1), the most common being palpitations (14 (0.6%)), first-degree atrioventricular block (12 (0.5%)), bradycardia (10 (0.4%)), and extrasystoles (10 (0.4%)). The percentages of participants with cardiac TEAEs remained low and stable over time (Supplemental Figure 2H). In addition, the percentages of participants with cardiac TEAEs at any point in the trial were similar across parent trial treatment groups (Supplemental Table 9). A total of 2 of 2494 (0.1%) participants permanently discontinued ozanimod due to two events of QT prolongation and one event each of bradycardia and left bundle branch block. Serious cardiac disorders occurred in 11 of 2494 (0.4%) participants, including three events of myocardial infarction and one incidence each of Stokes–Adams syndrome, arrhythmia, bradycardia (separate from the discontinuation case), cardiac failure, coronary artery stenosis, myocardial ischemia, second-degree atrioventricular block, sinoatrial block and unstable angina.
During DAYBREAK, 89 of 2490 (3.6%) participants had systolic blood pressure >180 mm Hg or an increase of >40 mm Hg from DAYBREAK baseline. Mean systolic and diastolic blood pressure increased 5.7 and 2.2 mm Hg (n = 888), respectively, from baseline to DAYBREAK month 72.
Vascular TEAEs occurred in 289 of 2494 (11.6%) participants, with an IR of 24.6 of 1000 PY (95% CI: 22.0–27.6), the most common being hypertension (230 (9.2%). The percentages of participants with vascular TEAEs were low and decreased over time (Supplemental Figure 2I). In addition, the percentages of participants with vascular TEAEs were similar across parent trial treatment groups (Supplemental Table 9). Serious vascular disorders occurred in 10 of 2494 (0.4%) participants, including four events of varicose vein, three incidences of hypertension, and one incidence each of circulatory collapse, hypotension, hypovolemic shock and thrombophlebitis. A total of 4 of 2494 (0.2%) participants in DAYBREAK were reported to have hypertensive crisis (all moderately severe).
Pulmonary laboratory abnormalities and TEAEs
The percentages of participants with pulmonary TEAEs were low and stable over time (Supplemental Figure 2J). Overall median reductions of 170 and 140 mL were observed for forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) at DAYBREAK month 60, respectively. FEV1 and FVC < 80% of baseline at any postbaseline visit occurred in 345 of 2453 (14.1%) and 286 of 2453 (11.7%) participants, respectively. Dyspnoea was infrequent, occurring in 10 of 2494 (0.4%) participants. One participant each permanently discontinued ozanimod due to pulmonary sarcoidosis, dyspnoea and decreased FEV.
Hepatic laboratory abnormalities and TEAEs
The percentages of participants with hepatic TEAEs were low and generally decreased over time (Supplemental Figure 2K). Of the 2489 participants with a nonmissing postbaseline laboratory assessment, 91 (3.7%) and 42 (1.7%) had a maximal alanine aminotransferase (ALT) or aspartate aminotransferase (AST) ⩾ 3 × upper limit of normal (ULN), respectively (Table 5 and Supplemental Table 10), with similar rates across parent trial treatment groups (Supplemental Table 11). The mean (min, max) time to onset of ALT or AST increase ⩾ 3 × ULN was 27.3 (0.2, 72.6) and 30.2 (0.2, 68.9) months, respectively. Most elevations were transient, with only 22 of 2489 (0.9%) participants having consecutive ALT ⩾ 3 × ULN (Table 5). Initial occurrence of ALT elevations became more infrequent as DAYBREAK progressed (Figure 3). Hepatic enzyme elevation leading to permanent treatment discontinuation occurred in 10 of 2494 (0.4%) participants (IR: 0.8/1000 PY; 95% CI: 0.4–1.5); underlying events were ALT increase (7 (0.3%)), AST increase (5 (0.2%)), GGT increase (2 (0.1%)), and liver function test increase (1 (0.04%); not specified).
Hepatic laboratory abnormalities during DAYBREAK.
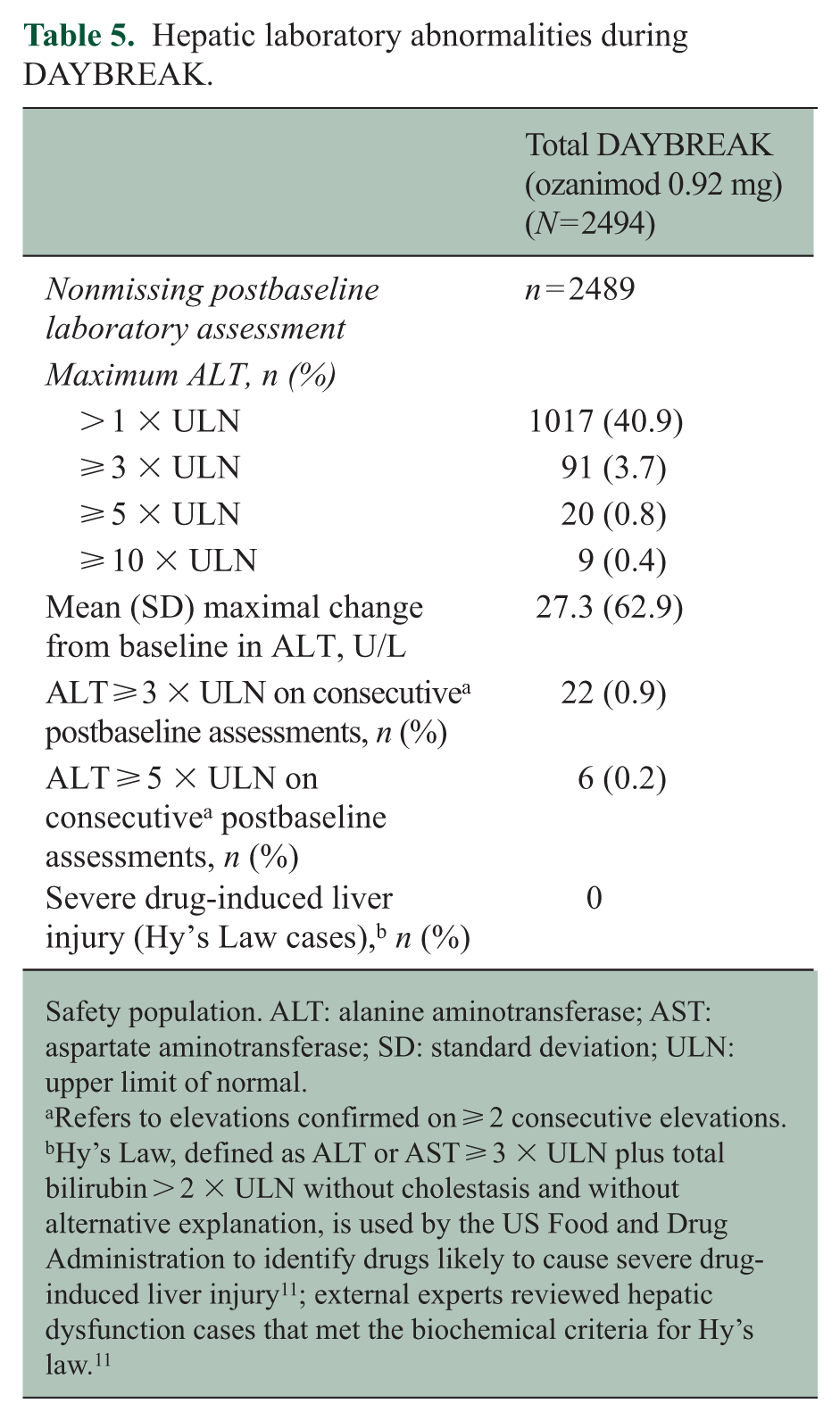
Safety population. ALT: alanine aminotransferase; AST: aspartate aminotransferase; SD: standard deviation; ULN: upper limit of normal.
Refers to elevations confirmed on ⩾ 2 consecutive elevations.
Hy’s Law, defined as ALT or AST ⩾ 3 × ULN plus total bilirubin > 2 × ULN without cholestasis and without alternative explanation, is used by the US Food and Drug Administration to identify drugs likely to cause severe drug-induced liver injury 11 ; external experts reviewed hepatic dysfunction cases that met the biochemical criteria for Hy’s law. 11
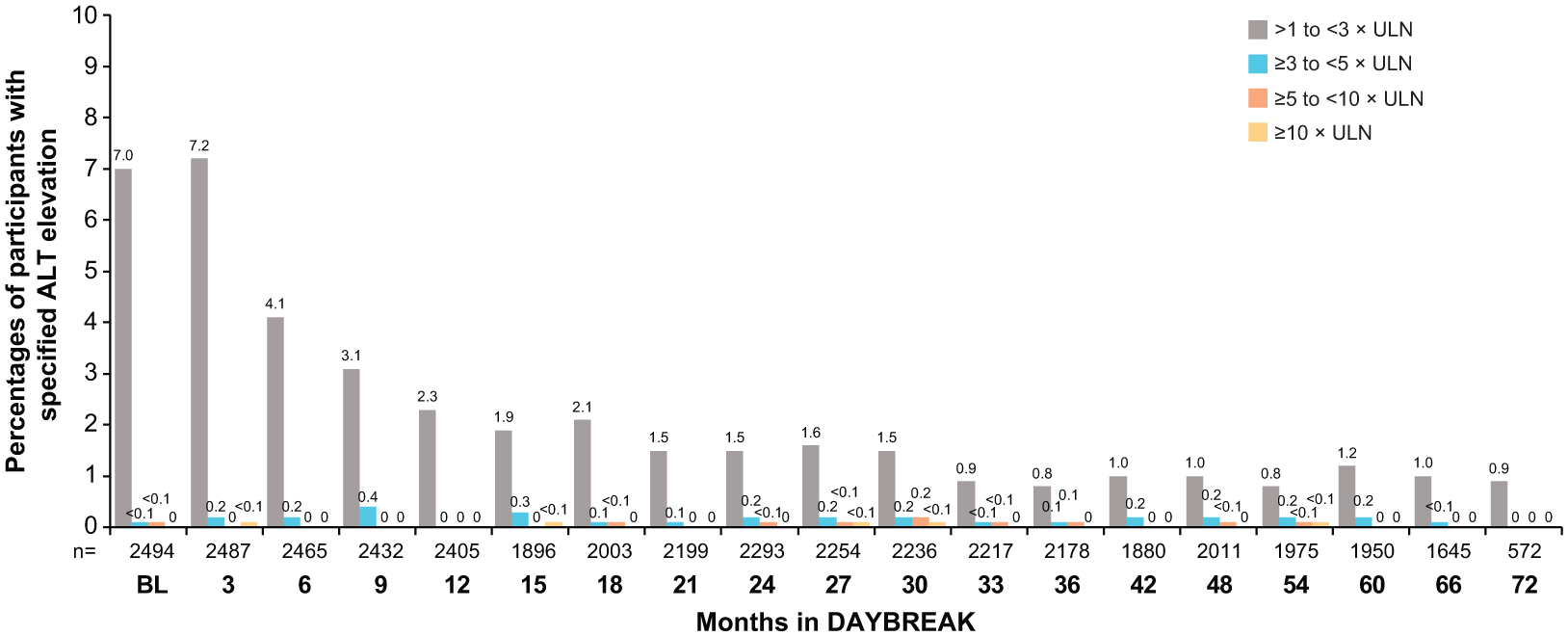
First occurrence of ALT elevations during DAYBREAK.
Hepatobiliary TEAEs were observed in 104 of 2494 (4.2%) participants, with an IR of 8.4 of 1000 PY (95% CI: 7.0–10.2), the most common being hyperbilirubinemia (27 (1.1%)), cholelithiasis (16 (0.6%)) and chronic cholecystitis (12 (0.5%) (Supplemental Table 12)). The percentages of participants with hepatobiliary TEAEs were low and stable over time (Supplemental Figure 2L). In addition, the percentages of participants with hepatobiliary TEAEs were similar across parent trial treatment groups (Supplemental Table 12). A total of 15 of 2494 (0.6%) events were serious. Two participants permanently discontinued ozanimod due to hepatobiliary TEAEs, one for acute hepatitis and the other for sphincter of Oddi dysfunction. Four participants had concurrent hepatocellular enzyme (⩾ 3 × ULN) and bilirubin elevations (⩾ 2 × ULN), but there were no cases of Hy’s law criteria-defined severe drug-induced liver injury per external hepatology expert review.
Absolute lymphocyte count
After an initial, expected decrease (particularly in participants receiving IFN β-1a in the parent trials), ALC remained relatively stable throughout DAYBREAK (Figure 4). Among the 2488 participants with ⩾1 ALC test in the DAYBREAK safety population, ALC was < 0.2 × 109/L in 369 of 2488 (14.8%) (Table 6), with similar rates across parent trial treatment groups (Supplemental Table 13). A total of 44 of 2488 (1.8%) participants had consecutive ALC < 0.2 × 109/L with repeated testing within 30 days. Four (0.2%) participants experienced TEAEs of decreased lymphocyte count that led to permanent treatment discontinuation.
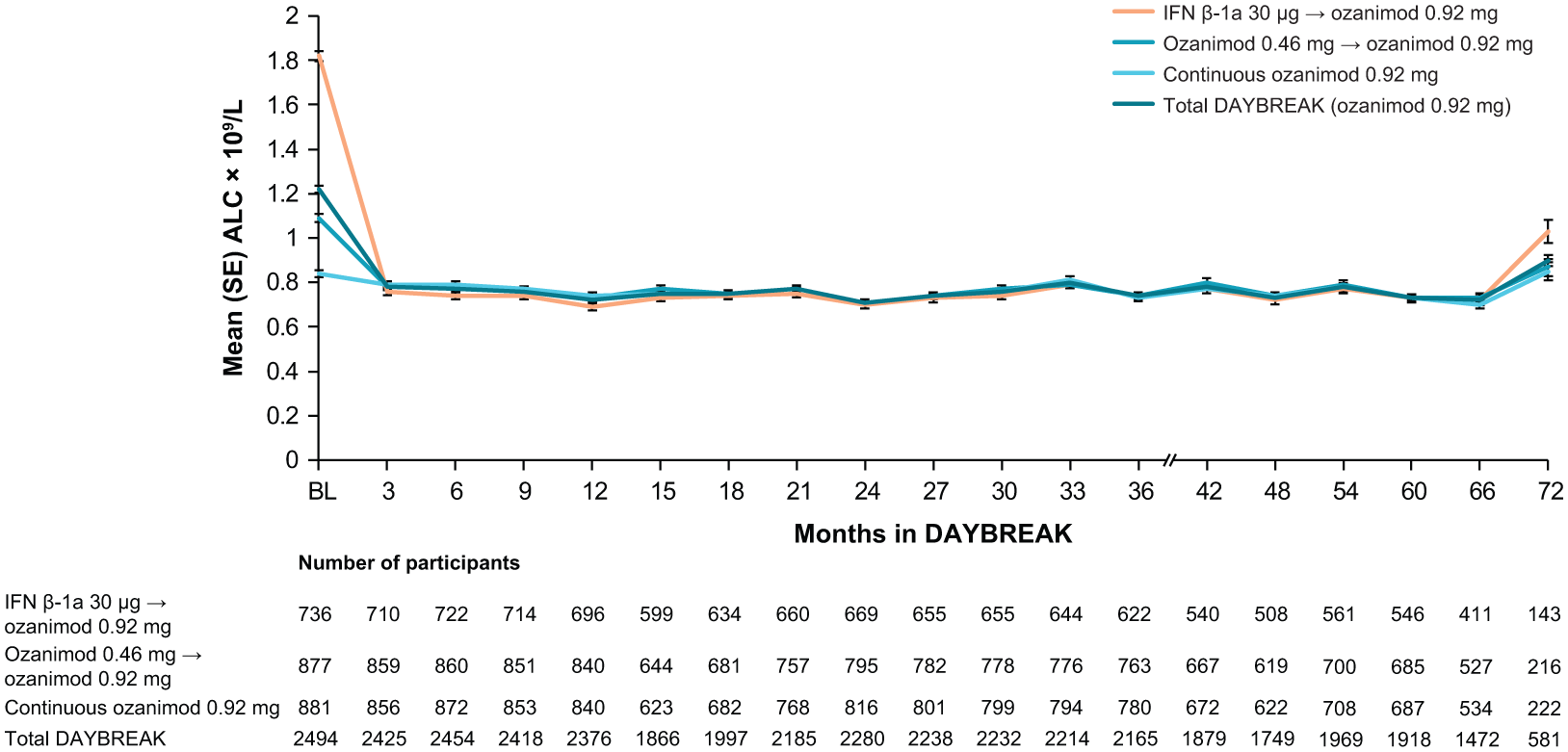
ALC over time during DAYBREAK by parent trial treatment group and in the total DAYBREAK population.
ALC minimum values during DAYBREAK.

Safety population. Per protocol, participants who had ALC < 0.2 × 109/L received a repeat test within approximately 7 days, and laboratory testing was repeated weekly until ALC was > 0.5 × 109/L. ALC: absolute lymphocyte count; LLN: lower limit of normal.
Defined as the nonmissing minimal value.
LLN = 1.0 × 109/L.
Efficacy
Relapses
Adjusted ARR (95% CI) in the total DAYBREAK population over the entire study was 0.098 (0.082–0.117), which was similar across parent trial treatment groups (IFN β-1a: 0.096 (0.077–0.120), ozanimod 0.46 mg: 0.109 (0.088–0.134), ozanimod 0.92 mg: 0.090 (0.073–0.111)). When observed by yearly interval, adjusted ARR initially decreased before remaining relatively stable after DAYBREAK month 24 (Figure 5a); this trend was similar across parent trial treatment groups (Supplemental Figure 4A). The majority of participants (1723 (69.1%) were relapse-free at the completion of DAYBREAK (Figure 5b), regardless of parent trial treatment group (Supplemental Figure 4B).
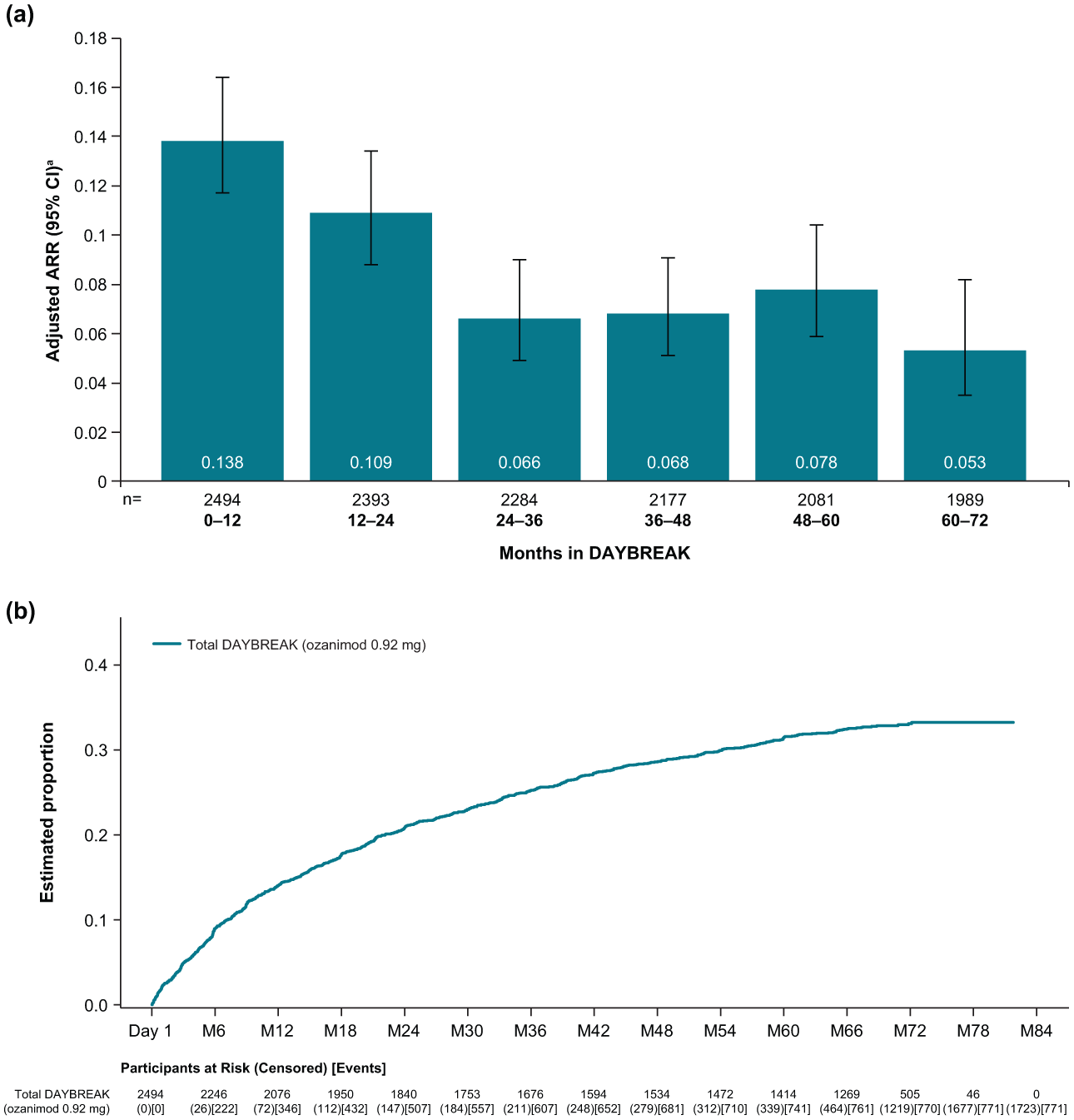
ARR by yearly interval (a) and Kaplan–Meier analysis (b) for the estimated proportion of participants with confirmed relapse during DAYBREAK
Disability progression
A total of 2066 of 2494 (82.8%) participants were free from 3-month CDP and 2115 of 2494 (84.8%) were free from 6-month CDP (Figure 6) at the discontinuation or completion of DAYBREAK; these rates were similar across parent trial treatment groups (Supplemental Figure 5).
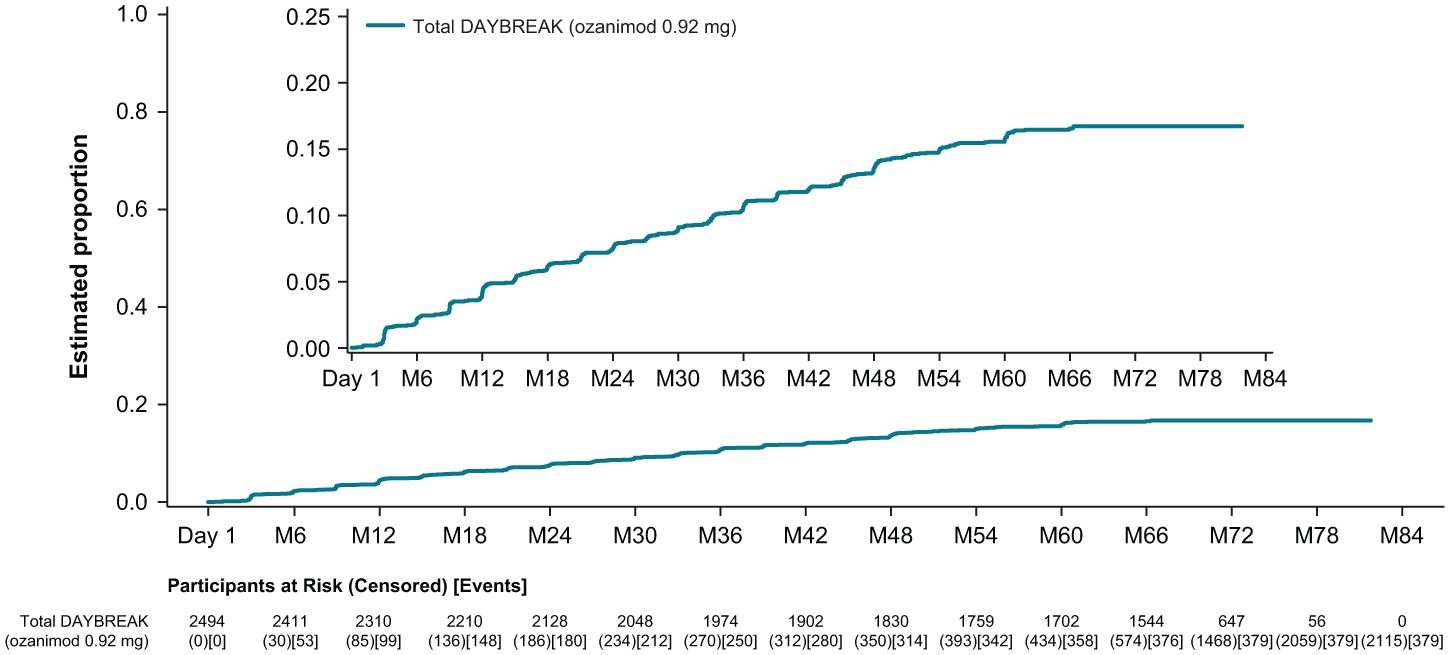
Kaplan–Meier analysis of time to onset of 6-month confirmed disability progression during DAYBREAK.
MRI lesions
Adjusted mean lesion counts were low and relatively stable throughout DAYBREAK (Figure 7 and Supplemental Figure 6). At DAYBREAK month 60, 599 of 1667 (35.9%) participants from the phase 3 ITT population were free of new/enlarging T2 lesions relative to DAYBREAK baseline and 1518 of 1666 (91.1%) were free of GdE lesions.
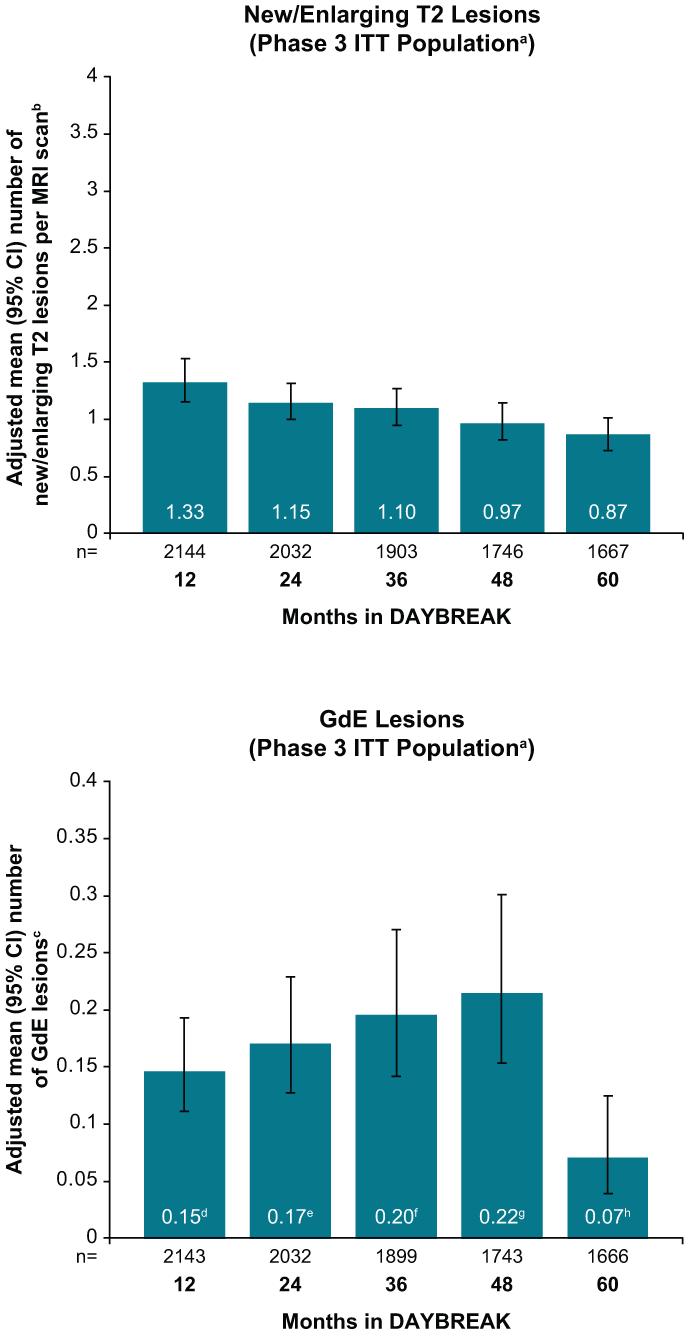
Number of new/enlarging T2 lesions per scan and GdE lesions during DAYBREAK.
Cognitive processing speed
Mean (SE) SDMT scores increased from 47.4 (0.7), 46.8 (0.7) and 48 (0.7) at SUNBEAM baseline to 48.5 (0.9), 47.5 (0.8) and 49.8 (0.8) at DAYBREAK month 72 in participants initially receiving IFN β-1a, ozanimod 0.46 mg or ozanimod 0.92 mg, respectfully, prior to switching to/continuing on ozanimod 0.92 mg in DAYBREAK (Figure 8a). The percentages of SUNBEAM participants with improved cognitive processing speed increased for 2‒3 years and were subsequently maintained with continuous ozanimod treatment in DAYBREAK (Figure 8b).
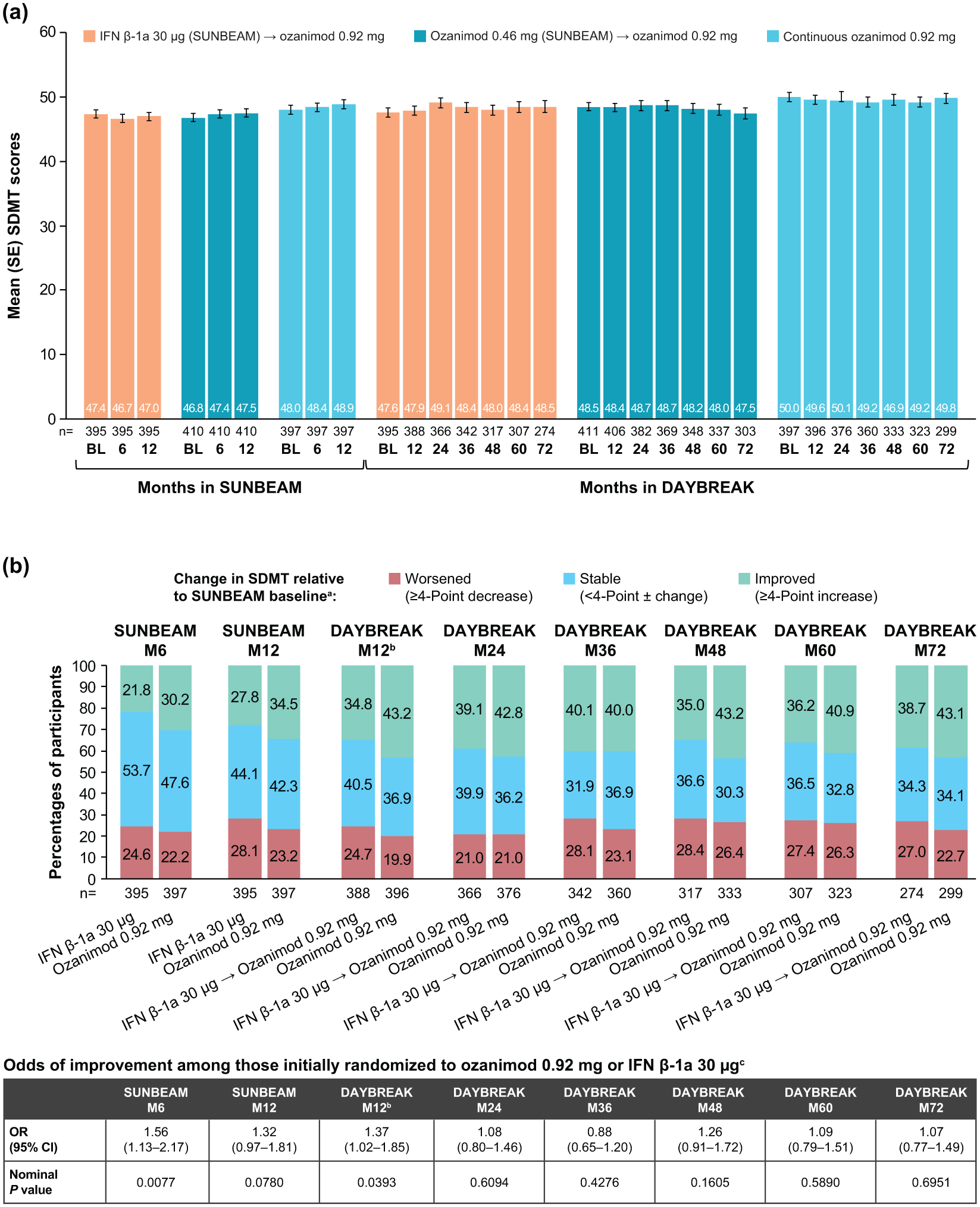
Mean (a) and change (b) in SDMT scores during SUNBEAM and DAYBREAK.
Discussion
The completed DAYBREAK OLE characterized the safety and efficacy of ozanimod 0.92 mg treatment in participants with RMS over a longer period (up to 6.8 years in DAYBREAK and up to 9.8 years of continuous exposure) than previously reported. Ozanimod was generally well tolerated, as most participants completed the study and the proportion experiencing TEAEs remained stable or decreased throughout the study period. Ozanimod treatment resulted in sustained efficacy with low ARR, most participants being free from 6-month CDP, and low numbers of new/enlarging T2 and GdE lesions. These long-term safety findings are consistent with phase 3 trials6,7 and the established safety profile of ozanimod.
When compared with the interim DAYBREAK analysis 8 or pooled safety results from the ozanimod clinical development programme, 12 exposure-adjusted IRs in TEAEs across all areas of concern were generally stable or lower, including declining rates of infections and relatively stable rates of serious infections and malignancies (including basal cell carcinoma and breast cancer). The age-standardized IRs of malignancies, including nonmelanoma skin cancer but excluding basal cell carcinoma, ranged from 220.9 to 293.8 of 100,000 PY in Eastern Europe as of 2020, 10 with malignancies, including basal cell carcinoma, occurring at an IR of 324.8 of 100,000 PY in DAYBREAK. The estimated age-standardized IRs of nonmelanoma skin cancers (including basal cell carcinoma) ranged from 22.1 to 795.7 of 100,000 PY in North American and European populations as of 2019, 13 with nonmelanoma skin cancers (including basal cell carcinoma) occurring at an IR of 102.9 of 100,000 PY in DAYBREAK. The IR of serious infections was relatively stable but numerically increased compared with the interim analysis, likely due to serious COVID-19–related AEs (8.5/1000 PY vs 7.4/1000 PY in the interim report). The incidence of opportunistic infections, particularly herpes infections, decreased over time, with numerical reductions in the final IRs (13.1/1000 PY vs 15.2/1000 PY in the interim report); there were no new serious opportunistic infections reported. One case of confirmed PML has been reported across 15,556.2 PY of exposure in DAYBREAK participants. Few participants had confirmed ALC < 0.2 × 109/L. There were no increased reports of pulmonary dysfunction TEAEs. Cardiac AEs at any point in DAYBREAK remained uncommon. The percentages of participants with systolic blood pressure > 180 mm Hg (or >40 mm Hg from DAYBREAK baseline) were low and stable over time (3.6% vs 2.7% in the interim report) and IRs of hypertension decreased (19.3/1000 PY vs 20.7/1000 PY in the interim report). Overall, these data suggest that the risk of AEs potentially associated with S1P receptor modulators does not increase with extended ozanimod use. The S1P receptor selectivity of ozanimod may thus contribute to a favourable risk/benefit profile. 3
Maintenance of RMS disease suppression was observed in participants previously treated with ozanimod, as demonstrated by sustained and further reductions in relapse rates, low rates of disability progression, and reductions in MRI lesion counts.
One limitation of this study was the lack of a control group and blinding, making cross-study comparisons challenging. Expedited site closures in Russia impacted the safety analyses, specifically follow-up after treatment discontinuation; however, overall data interpretations were not affected. Potential selection bias was possible because participants with less favourable outcomes may have discontinued the parent trials.
Conclusion
Long-term data from DAYBREAK are consistent with the established safety and tolerability profile of ozanimod and support sustained control of disease activity and disability progression over long-term treatment in participants with RMS.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251382796 – Supplemental material for Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Final analysis of the DAYBREAK open-label extension trial
Supplemental material, sj-docx-1-msj-10.1177_13524585251382796 for Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Final analysis of the DAYBREAK open-label extension trial by Krzysztof W Selmaj, Lawrence Steinman, Giancarlo Comi, Amit Bar-Or, Douglas L Arnold, Hans-Peter Hartung, Xavier Montalbán, Eva K Havrdová, James K Sheffield, Anthony Krakovich, Chun-Yen Cheng, Jon V Riolo, Chahin Pachai, Andrew Thorpe, Erik DeBoer, Ludwig Kappos, Jeffrey A Cohen and Bruce AC Cree in Multiple Sclerosis Journal
Footnotes
Acknowledgements
Writing, editorial and submission assistance was provided by Noud van Helmond, MD, PhD, Jessica Brunquell, PhD, and Cristina Silvestre of Peloton Advantage, LLC, an OPEN Health company, and was funded by Bristol Myers Squibb. Data analysis was provided by Zhaohui Liu, PhD and Tushar Nikumbh, PhD.
Author contributions
All authors vouch for data accuracy, reviewed all drafts and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article:
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Bristol Myers Squibb.
Ethical considerations
Consent to participate
All DAYBREAK participants or their legal representatives provided written consent before participation.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
