Abstract
Background:
Ozanimod, an oral sphingosine 1-phosphate receptor 1 and 5 modulator, is approved in multiple countries for treatment of relapsing forms of MS.
Objective:
To characterize long-term safety and efficacy of ozanimod.
Methods:
Patients with relapsing MS who completed a phase 1‒3 ozanimod trial were eligible for an open-label extension study (DAYBREAK) of ozanimod 0.92 mg/d. DAYBREAK began 16 October 2015; cutoff for this interim analysis was 2 February 2021.
Results:
This analysis included 2494 participants with mean 46.8 (SD 11.9; range 0.033‒62.7) months of ozanimod exposure in DAYBREAK. During DAYBREAK, 2143 patients (85.9%) had treatment-emergent adverse events (TEAEs; similar in nature to those in the parent trials), 298 (11.9%) had a serious TEAE, and 75 (3.0%) discontinued treatment due to TEAEs. Serious infections (2.8%), herpes zoster infections (1.7%), confirmed macular edema cases (0.2%), and cardiac TEAEs (2.8%) were infrequent. Adjusted annualized relapse rate was 0.103 (95% confidence interval, 0.086‒0.123). Over 48 months, 71% of patients remained relapse free. Adjusted mean numbers of new/enlarging T2 lesions/scan and gadolinium-enhancing lesions were low and similar across parent trial treatment subgroups.
Conclusions:
This long-term extension of ozanimod trials confirmed a favorable safety/tolerability profile and sustained benefit on clinical and magnetic resonance imaging measures of disease activity.
Keywords
Introduction
Ozanimod, a sphingosine 1-phosphate (S1P) receptor 1 and 5 modulator, is approved in multiple countries for treatment of adults with either relapsing forms of multiple sclerosis (RMS) or moderately to severely active ulcerative colitis. Four clinical trials of ozanimod in RMS were completed, including a phase 1 study (unpublished), a phase 2 study with an extension period (RADIANCE),1,2 and two phase 3 trials (RADIANCE and SUNBEAM).3,4 In phase 3 trials, ozanimod 0.92 mg/d for up to 24 months significantly reduced clinical relapses and lesion counts on brain magnetic resonance imaging (MRI) and slowed brain volume loss relative to intramuscular interferon β-1a 30 μg/wk.3,4 Ozanimod was well tolerated, with fewer treatment-emergent adverse events (TEAEs) leading to discontinuation than interferon β-1a.3,4 Patients who completed any RMS trial were eligible to enroll in DAYBREAK, an open-label extension trial of ozanimod 0.92 mg/d (equivalent to ozanimod HCl 1 mg). This is an interim analysis of safety (primary objective) and efficacy (secondary objective) in DAYBREAK.
Methods
Parent trials
DAYBREAK participants originated from four “parent” ozanimod trials (Figure 1). The phase 1 study was a 12-week, open-label pharmacokinetic/pharmacodynamic study of ozanimod 0.92 or 0.46 mg/d. Phase 2 RADIANCE was a randomized, double-blind, 24-week study of ozanimod 0.92 or 0.46 mg/d versus placebo, 1 followed by a 24-month dose-blinded extension period in which patients initially assigned to ozanimod remained on their assigned dose while those in the placebo group were randomized to ozanimod 0.92 or 0.46 mg/d. 2 Two phase 3, randomized, double-blind, double-dummy trials compared oral ozanimod 0.92 or 0.46 mg/d with intramuscular interferon β-1a 30 µg/wk; phase 3 RADIANCE lasted 24 months, 3 whereas SUNBEAM continued until the last enrolled participant was treated for 12 months. 4 Parent trial participants were aged 18‒55 years with RMS, brain MRI lesions consistent with multiple sclerosis (MS), and an Expanded Disability Status Scale (EDSS) score of 0‒5.0 (phases 2 and 3) or 0‒6.0 (phase 1).1,3,4
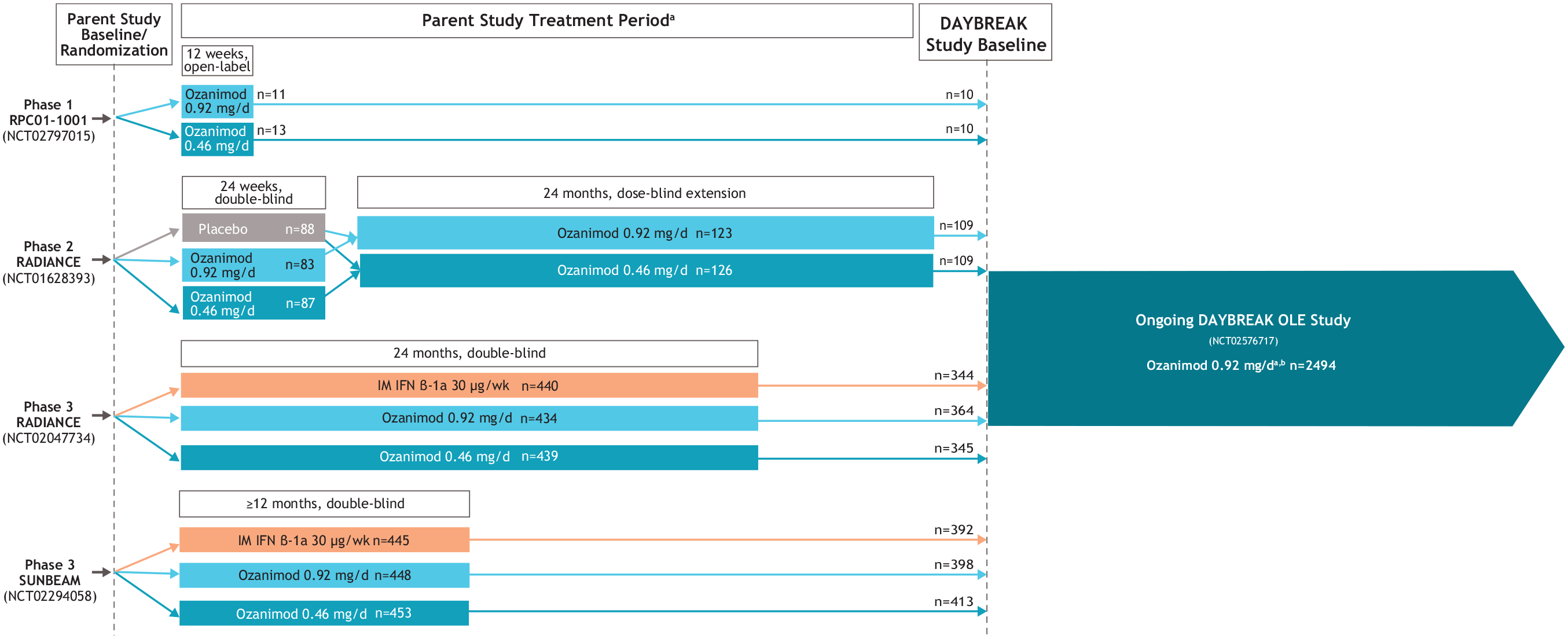
Parent trials feeding into the DAYBREAK extension.
DAYBREAK extension
Patients who completed a parent trial were eligible to enter the single-arm, open-label, phase 3 DAYBREAK trial (ClinicalTrials.gov ID: NCT02576717; EudraCT: 2015-002500-91). All DAYBREAK participants re-consented. DAYBREAK began 16 October 2015, and is being conducted in 25 countries in Europe and in North America, plus South Africa and New Zealand. Data cutoff for the primary safety and efficacy analyses was 2 February 2021.
Patients from the phase 3 trials underwent a 1-week dose escalation upon entry into DAYBREAK. Dose escalation was not performed for patients from the other trials unless there was a ⩾14-day gap in treatment.
Physical and skin examinations, electrocardiogram, pulmonary function tests (PFT), optical coherence tomography (OCT), and brain MRI were performed at baseline, every 12 months, and at termination. Laboratory testing was performed and vital signs were measured at baseline, every 3 months for 3 years and every 6 months thereafter, at termination, and during post-treatment follow-up. EDSS and neurologic examination were performed at baseline, every 3 months for 3 years and then every 6 months, upon suspected relapse, and at study termination. Unless otherwise specified, “baseline” refers to day 1 of DAYBREAK.
TEAEs, including those of special interest (infections, malignancies, macular edema, cardiac events, pulmonary and liver function test abnormalities), were monitored throughout the trial. Depression and suicide, lymphopenia, and post-treatment discontinuation relapse also are reported. Safety follow-up after discontinuation was expanded from 28 to 75 days in 2018 and to 90 days in 2019. External experts reviewed hepatic dysfunction cases. An independent Macular Edema Review Panel (MERP) reviewed TEAEs related to macular edema, cases of central foveal thickness greater than 20% of baseline, and OCT abnormalities.
COVID-19 cases were captured as TEAEs in the primary safety analysis using the 2 February 2021 data cutoff. In addition, we performed a separate post hoc analysis to better analyze any potential effects of ozanimod on COVID-19 outcomes. Confirmed and suspected COVID-19 cases occurring between 1 November 2019 and 10 May 2021 were identified based on adverse event preferred terms related to coronavirus infection or coronavirus test positive. Cases were assessed for seriousness, action taken with ozanimod, and infection outcome.
Efficacy outcomes included annualized relapse rate (ARR), time to first confirmed relapse, relapse-free rate, number of new/enlarging T2 lesions per scan relative to DAYBREAK baseline and number of gadolinium-enhancing (GdE) lesions on brain MRI, and time to onset of 3- and 6-month confirmed disability progression (CDP-3, CDP-6) (⩾1-point increase in EDSS from DAYBREAK baseline).
Statistical analyses
Patients who received ⩾1 dose of ozanimod in DAYBREAK composed the safety and intent-to-treat populations. Where provided, parent trial data are reported only for patients who entered DAYBREAK. Results are reported for the overall DAYBREAK population, and parent trial treatment assignment subgroups (Supplemental Figure S1). MRI lesion counts were analyzed in patients from the phase 3 parent trials. Unless otherwise specified, TEAEs and ARR are reported from DAYBREAK baseline through data cutoff; other efficacy outcomes and laboratory abnormalities are reported from DAYBREAK baseline through month 48 because data thereafter are limited to a small number of patients. Because malignancies are infrequent and have a variable latency period, all malignancies from date of first ozanimod exposure in any trial to DAYBREAK data cutoff are reported in patients exposed to either ozanimod dose; the subset of malignancies occurring in DAYBREAK are also reported.
TEAE incidence was summarized by Medical Dictionary for Regulatory Activities version 22.1 preferred term. Incidence rates (IRs) per 1000 person-years (PY) (100,000 PY for malignancies) were calculated to adjust for time on study.
ARR was calculated using a negative binomial regression model adjusted for region (Eastern Europe versus rest of world), age at parent trial baseline, and parent trial baseline number of GdE lesions. The natural log transformation of time on treatment was used as an offset term to adjust for patients having different exposure times. Time to first confirmed relapse and relapse-free rates were analyzed using Kaplan–Meier analysis, with censoring of patients for whom follow-up ended before a relapse occurred.
The adjusted mean (95% confidence interval (CI)) number of new/enlarging T2 brain MRI lesions per scan relative to baseline (parent baseline for parent trials, DAYBREAK baseline for DAYBREAK) was derived based on a negative binomial regression model, and adjusted for study, region, age at baseline, and baseline number of GdE lesions, with the log of the number of postbaseline scans as an offset term. The adjusted mean (95% CI) number of GdE lesions was derived using a negative binomial regression model, adjusted for study, region, age at baseline, and baseline number of GdE lesions. Time to onset of CDP-3 and CDP-6 was analyzed using Kaplan–Meier analysis.
Results
Disposition and demographics
Of 2639 eligible patients, 2494 (94.5%) enrolled and were treated in DAYBREAK; 2055 (82.4%) were continuing at data cutoff (Figure 2). Mean duration of ozanimod exposure in DAYBREAK was 46.8 months (standard deviation (SD) 11.9; range 0.033‒62.7; 9725.6 PY). Mean ozanimod exposure during parent trials plus DAYBREAK was 60.7 months (SD 16.5; 12,617.1 PY); maximum was 98.8 months.
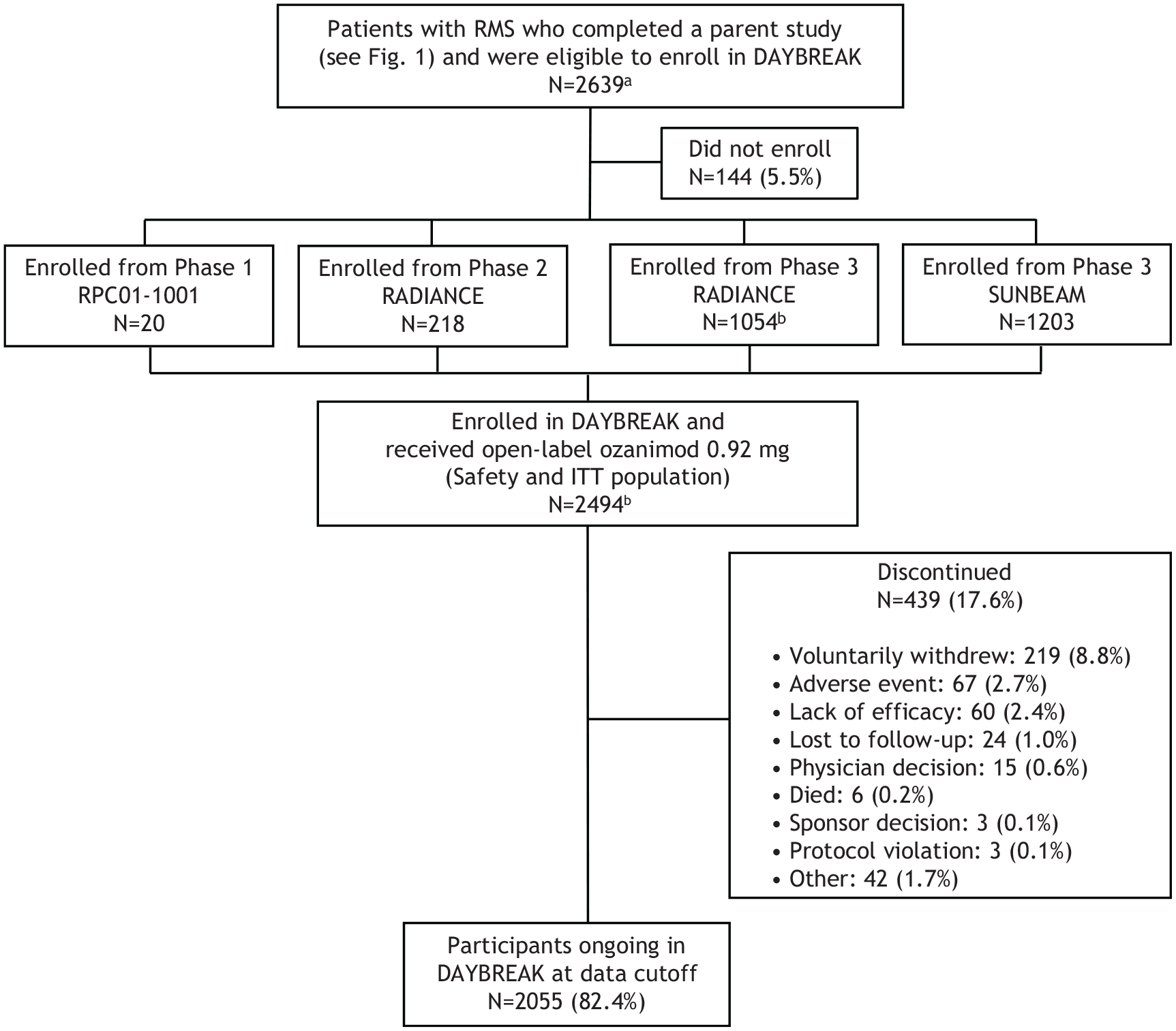
Flow diagram for DAYBREAK extension trial.
Baseline demographics and disease characteristics were generally consistent across parent trial treatment subgroups (Supplemental Table S1). Baseline characteristics of participants in DAYBREAK and those of participants in the phase 2 and phase 3 parent trials (who compose over 99% of the DAYBREAK population) are similar, with the exception that the DAYBREAK population is about 2 years older on average, as expected given the duration of the parent trials (Supplemental Table S2). In addition, mean number of GdE lesions was lower and mean number of T2 lesions was higher at DAYBREAK baseline than at parent trial baseline, which are expected findings given the treatment history and nature of the lesions.
Safety
During DAYBREAK, 85.9% of patients experienced ⩾1 TEAE. Most were mild or moderate; rates of severe TEAEs, serious TEAEs, and TEAEs leading to permanent treatment discontinuation were 7.6%, 11.9%, and 3.0%, respectively, and similar across parent trial treatment subgroups (Table 1). The most common TEAEs were nasopharyngitis, headache, and upper respiratory tract infection (Table 1). Prior to the 2 February 2021 data cutoff, three patients died from malignancies, two from accidents, and one each from community-acquired pneumonia, cerebral hemorrhage probably due to rupture of a preexisting occult aneurysm, sudden death due to unknown cause, and pulmonary embolism following leg fracture surgery. There were three deaths associated with COVID-19 (Supplemental Figure S2).
Overall safety during DAYBREAK by parent trial treatment subgroup and overall DAYBREAK population.
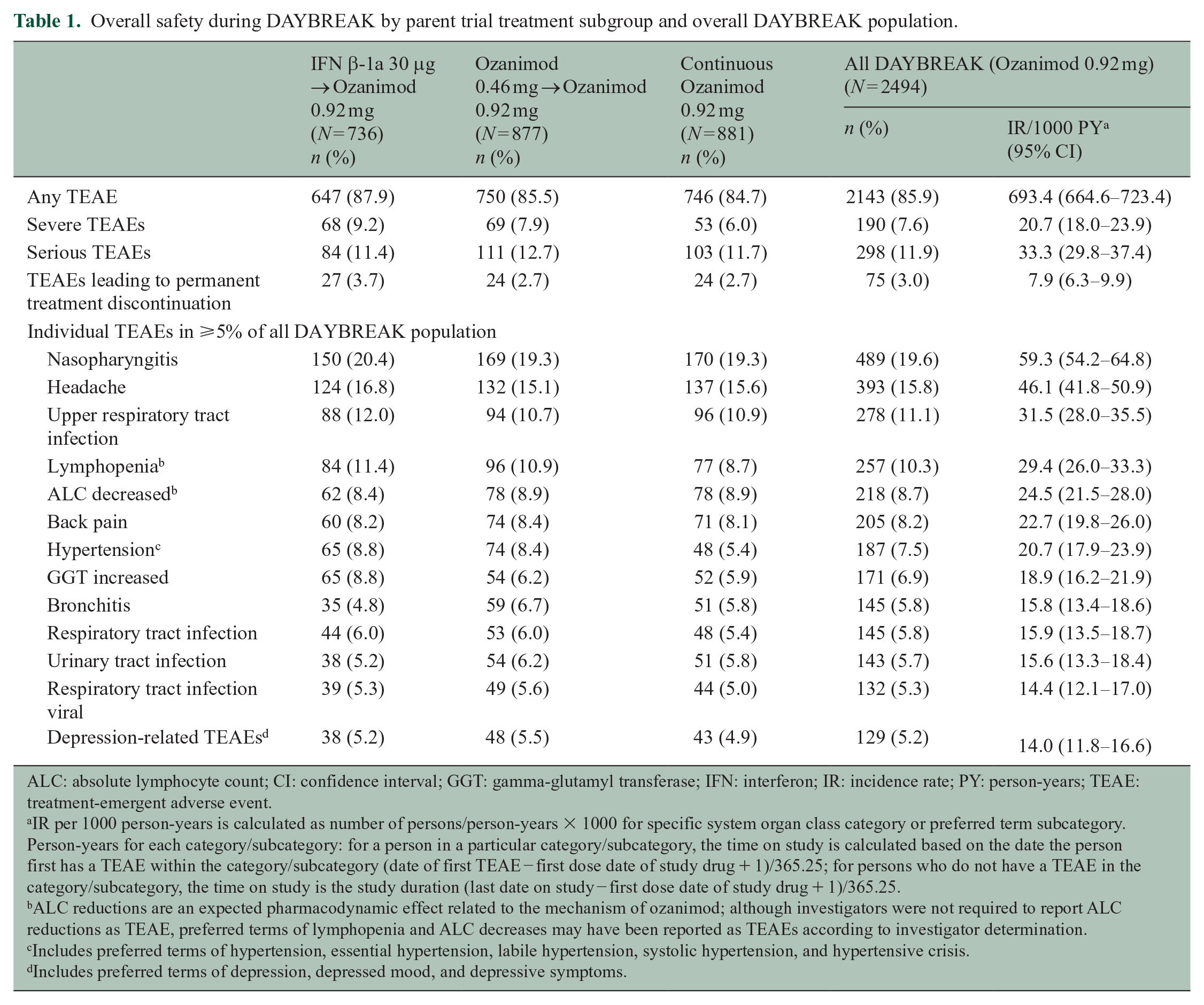
ALC: absolute lymphocyte count; CI: confidence interval; GGT: gamma-glutamyl transferase; IFN: interferon; IR: incidence rate; PY: person-years; TEAE: treatment-emergent adverse event.
IR per 1000 person-years is calculated as number of persons/person-years × 1000 for specific system organ class category or preferred term subcategory. Person-years for each category/subcategory: for a person in a particular category/subcategory, the time on study is calculated based on the date the person first has a TEAE within the category/subcategory (date of first TEAE − first dose date of study drug + 1)/365.25; for persons who do not have a TEAE in the category/subcategory, the time on study is the study duration (last date on study − first dose date of study drug + 1)/365.25.
ALC reductions are an expected pharmacodynamic effect related to the mechanism of ozanimod; although investigators were not required to report ALC reductions as TEAE, preferred terms of lymphopenia and ALC decreases may have been reported as TEAEs according to investigator determination.
Includes preferred terms of hypertension, essential hypertension, labile hypertension, systolic hypertension, and hypertensive crisis.
Includes preferred terms of depression, depressed mood, and depressive symptoms.
Infections―most commonly affecting the respiratory or urinary tract―occurred in 56.7% of DAYBREAK participants. The incidence of serious infection was 2.8% (Table 2). Opportunistic infections occurred in 5.6%; the most common were oral herpes (2.0%; IR 5.4/1000 PY) and herpes zoster (1.7%; IR 4.6/1000 PY) (Table 2). There were no serious opportunistic infections before data cutoff; however, a case of progressive multifocal leukoencephalopathy (PML) was reported in March 2021 in a 46-year-old woman who had received ozanimod 0.92 mg for about 4 years. 6 She discontinued ozanimod and had a nonfatal outcome with neurologic sequelae. Details regarding this case are provided in Supplemental Materials.
Serious and opportunistic infections during DAYBREAK by parent trial treatment subgroup and overall DAYBREAK population.
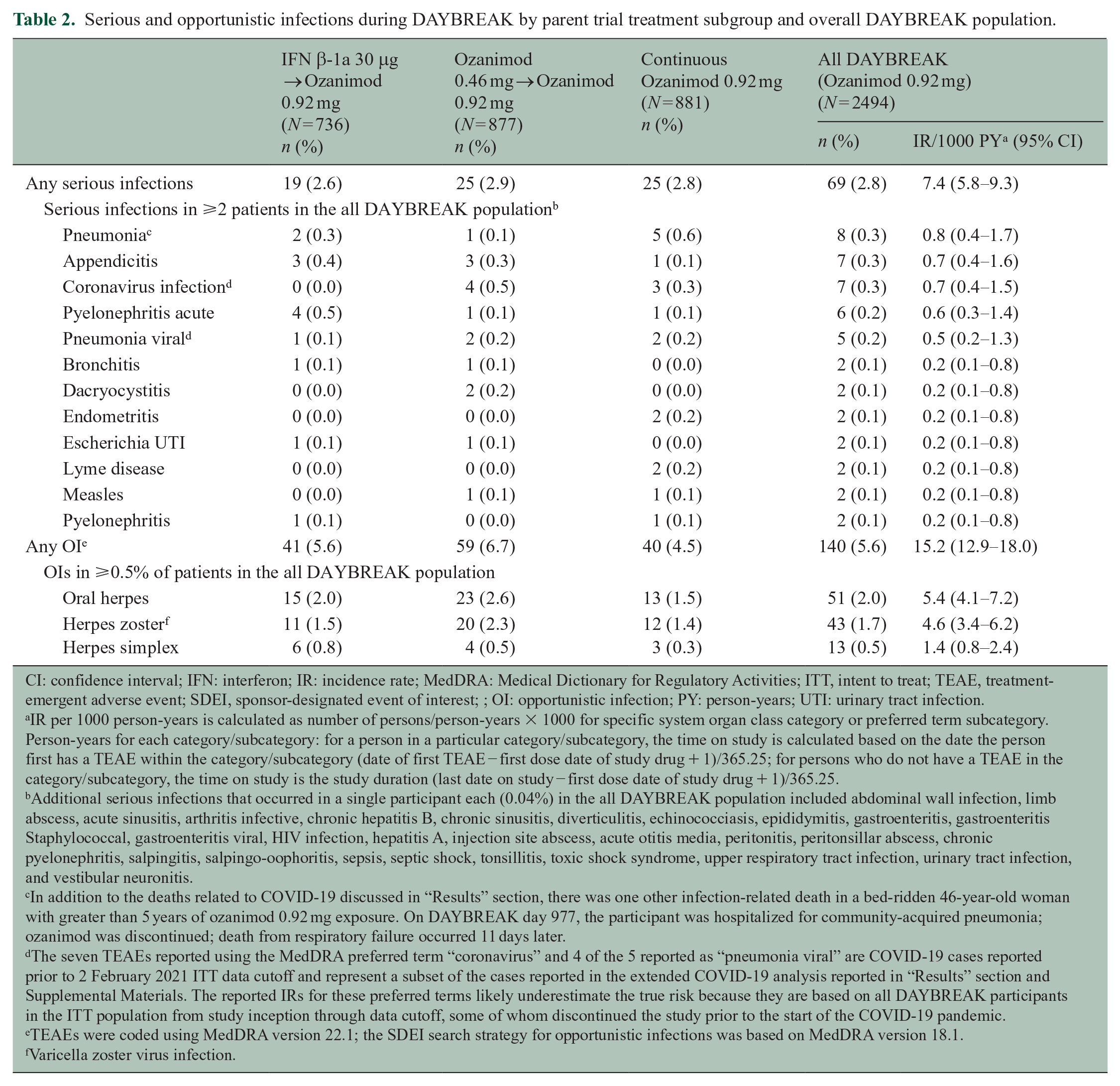
CI: confidence interval; IFN: interferon; IR: incidence rate; MedDRA: Medical Dictionary for Regulatory Activities; ITT, intent to treat; TEAE, treatment-emergent adverse event; SDEI, sponsor-designated event of interest; ; OI: opportunistic infection; PY: person-years; UTI: urinary tract infection.
IR per 1000 person-years is calculated as number of persons/person-years × 1000 for specific system organ class category or preferred term subcategory. Person-years for each category/subcategory: for a person in a particular category/subcategory, the time on study is calculated based on the date the person first has a TEAE within the category/subcategory (date of first TEAE − first dose date of study drug + 1)/365.25; for persons who do not have a TEAE in the category/subcategory, the time on study is the study duration (last date on study − first dose date of study drug + 1)/365.25.
Additional serious infections that occurred in a single participant each (0.04%) in the all DAYBREAK population included abdominal wall infection, limb abscess, acute sinusitis, arthritis infective, chronic hepatitis B, chronic sinusitis, diverticulitis, echinococciasis, epididymitis, gastroenteritis, gastroenteritis Staphylococcal, gastroenteritis viral, HIV infection, hepatitis A, injection site abscess, acute otitis media, peritonitis, peritonsillar abscess, chronic pyelonephritis, salpingitis, salpingo-oophoritis, sepsis, septic shock, tonsillitis, toxic shock syndrome, upper respiratory tract infection, urinary tract infection, and vestibular neuronitis.
In addition to the deaths related to COVID-19 discussed in “Results” section, there was one other infection-related death in a bed-ridden 46-year-old woman with greater than 5 years of ozanimod 0.92 mg exposure. On DAYBREAK day 977, the participant was hospitalized for community-acquired pneumonia; ozanimod was discontinued; death from respiratory failure occurred 11 days later.
The seven TEAEs reported using the MedDRA preferred term “coronavirus” and 4 of the 5 reported as “pneumonia viral” are COVID-19 cases reported prior to 2 February 2021 ITT data cutoff and represent a subset of the cases reported in the extended COVID-19 analysis reported in “Results” section and Supplemental Materials. The reported IRs for these preferred terms likely underestimate the true risk because they are based on all DAYBREAK participants in the ITT population from study inception through data cutoff, some of whom discontinued the study prior to the start of the COVID-19 pandemic.
TEAEs were coded using MedDRA version 22.1; the SDEI search strategy for opportunistic infections was based on MedDRA version 18.1.
Varicella zoster virus infection.
Between 1 November 2019 and 10 May 2021, 190 (8.7%) of 2181 DAYBREAK participants experienced confirmed (n = 160; 7.3%) or suspected (n = 30; 1.4%) COVID-19 while receiving ozanimod. Their demographic characteristics are shown in Supplemental Table S3. Most cases (n = 176/190 (92.6%)) were nonserious, and most patients (179/190 (94.2%)) recovered without sequelae while either continuing or temporarily interrupting ozanimod (Supplemental Figure S2). One patient died from pulmonary embolism associated with COVID-19; one died from suspected COVID-19 pneumonia; and one died from a lung abscess following COVID-19 (Supplemental Figure S2).
During the parent trials and DAYBREAK, 38 (1.4%) of 2787 patients exposed to either ozanimod dose–developed malignancies (Table 3). The most frequent was basal cell carcinoma (n = 11 (0.4%)). Nine (0.5%) of 1868 women developed breast cancer. Three malignancy-related deaths (from metastatic pancreatic carcinoma, disseminated cancer with unknown primary, and glioblastoma) occurred in DAYBREAK; there were no malignancy-related deaths in the parent trials. The subset of malignancies that occurred during DAYBREAK is also shown in Table 3.
Treatment-emergent malignancies during the parent trials or DAYBREAK (all patients who were exposed to ozanimod 0.46 or 0.92 mg) and treatment-emergent malignancies occurring in DAYBREAK only, by preferred term, in patients with RMS.
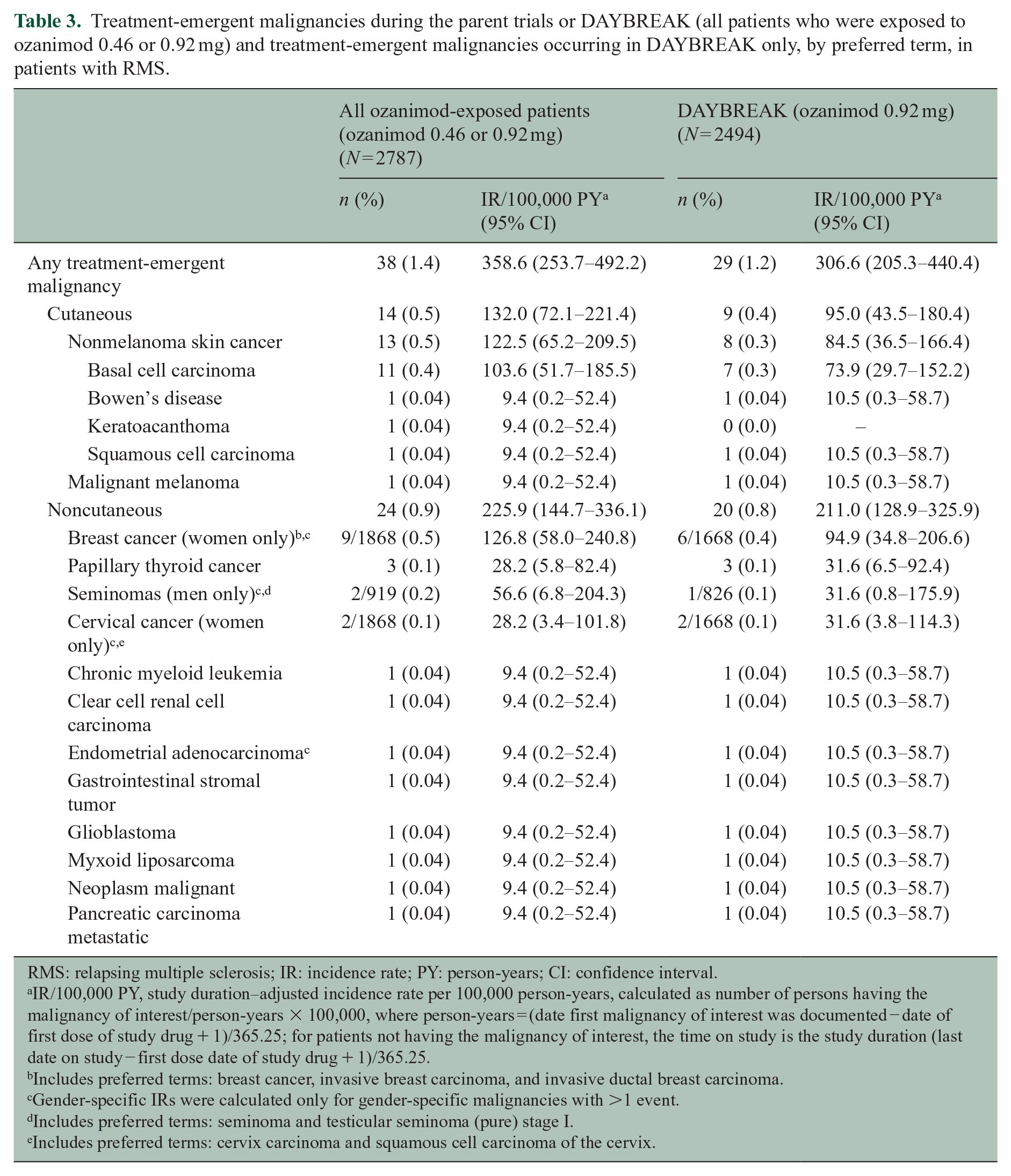
RMS: relapsing multiple sclerosis; IR: incidence rate; PY: person-years; CI: confidence interval.
IR/100,000 PY, study duration‒adjusted incidence rate per 100,000 person-years, calculated as number of persons having the malignancy of interest/person-years × 100,000, where person-years = (date first malignancy of interest was documented − date of first dose of study drug + 1)/365.25; for patients not having the malignancy of interest, the time on study is the study duration (last date on study − first dose date of study drug + 1)/365.25.
Includes preferred terms: breast cancer, invasive breast carcinoma, and invasive ductal breast carcinoma.
Gender-specific IRs were calculated only for gender-specific malignancies with >1 event.
Includes preferred terms: seminoma and testicular seminoma (pure) stage I.
Includes preferred terms: cervix carcinoma and squamous cell carcinoma of the cervix.
There were nine (0.4%) reports of macular edema during DAYBREAK―five in patients who switched from interferon β-1a to ozanimod. Based on review of TEAEs and OCTs, the MERP confirmed four macular edema cases (0.2%; IR 0.4/1000 PY (95% CI 0.2‒1.1)) during DAYBREAK. Three confirmed cases occurred within a year of switching from interferon β-1a to ozanimod in patients with preexisting risk factors or potentially predisposing conditions, as previously reported. 5 The fourth occurred in a patient who received ozanimod 0.92 mg in phase 3 RADIANCE and DAYBREAK for approximately 6 years total with no apparent risk factors for macular edema. Three cases resolved after ozanimod discontinuation; one was ongoing at data cutoff.
Cardiac TEAEs occurred in 69 (2.8%) patients (Supplemental Table S4), seven of whom had serious events (three myocardial infarctions (IR 0.3/1000 PY, 95% CI 0.1‒1.0), and one each of myocardial ischemia, unstable angina, bradycardia, and coronary artery stenosis). Six of seven patients who developed bradycardia had switched from interferon β-1a or ozanimod 0.46 mg to ozanimod 0.92 mg in DAYBREAK. Vascular TEAEs occurred in 236 (9.5%) participants (Supplemental Table S4). Hypertension was the most common (n = 187 (7.5%)) and included two serious cases of hypertension (0.08%) and four (0.2%) nonserious cases reported as “hypertensive crisis” (without sequelae). During DAYBREAK, 66/2490 (2.7%) patients had a recorded systolic blood pressure greater than 180 mm Hg or an increase of greater than 40 mm Hg from DAYBREAK baseline. From DAYBREAK baseline to month 48, mean (SD) change in systolic and diastolic blood pressure was 5.1 (11.8) and 1.7 (9.8) mm Hg, respectively, in the overall population.
Forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) were less than 80% of baseline at any postbaseline visit in 9.2% and 7.6% of patients, respectively. PFT abnormalities did not increase over time, and there were no differences by parent trial treatment subgroup. One patient each discontinued treatment due to pulmonary sarcoidosis, dyspnea, decreased FEV1, and decreased FVC.
Seventy-nine (3.2%) and 34 (1.4%) patients had a maximal alanine aminotransferase (ALT) or aspartate aminotransferase, respectively, ⩾3 times the upper limit of normal (ULN); 70 (2.8%) had bilirubin > 2 × ULN (Table 4). Eighteen patients (0.7% of the overall population) had ALT ⩾ 3 × ULN on consecutive postbaseline assessments (Table 4), suggesting that most elevations were transient. First occurrence of ALT elevations > 1 × ULN while patients were enrolled in DAYBREAK primarily occurred during year 1 (Figure 3). Hepatobiliary TEAEs were observed in 85 (3.4%) patients (Supplemental Table S5). There was one serious hepatic TEAE (chronic hepatitis). One patient discontinued ozanimod due to acute hepatitis (nonviral, nonautoimmune) and nine (0.4%) due to liver enzyme elevations. No cases of severe drug-induced liver injury occurred.
Hepatic laboratory abnormalities during DAYBREAK by parent trial treatment subgroup and overall DAYBREAK population.
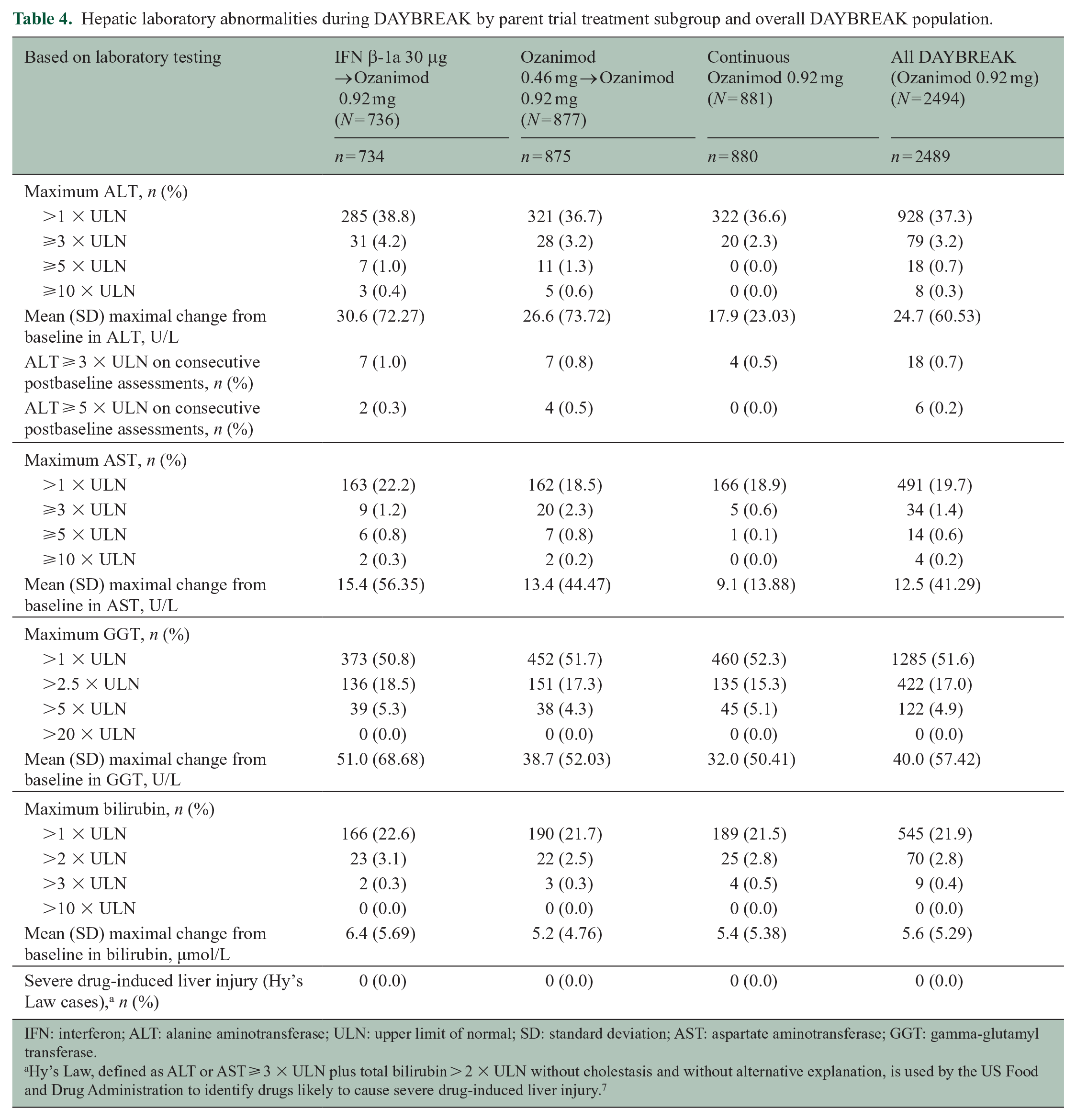
IFN: interferon; ALT: alanine aminotransferase; ULN: upper limit of normal; SD: standard deviation; AST: aspartate aminotransferase; GGT: gamma-glutamyl transferase.
Hy’s Law, defined as ALT or AST ⩾ 3 × ULN plus total bilirubin > 2 × ULN without cholestasis and without alternative explanation, is used by the US Food and Drug Administration to identify drugs likely to cause severe drug-induced liver injury. 7
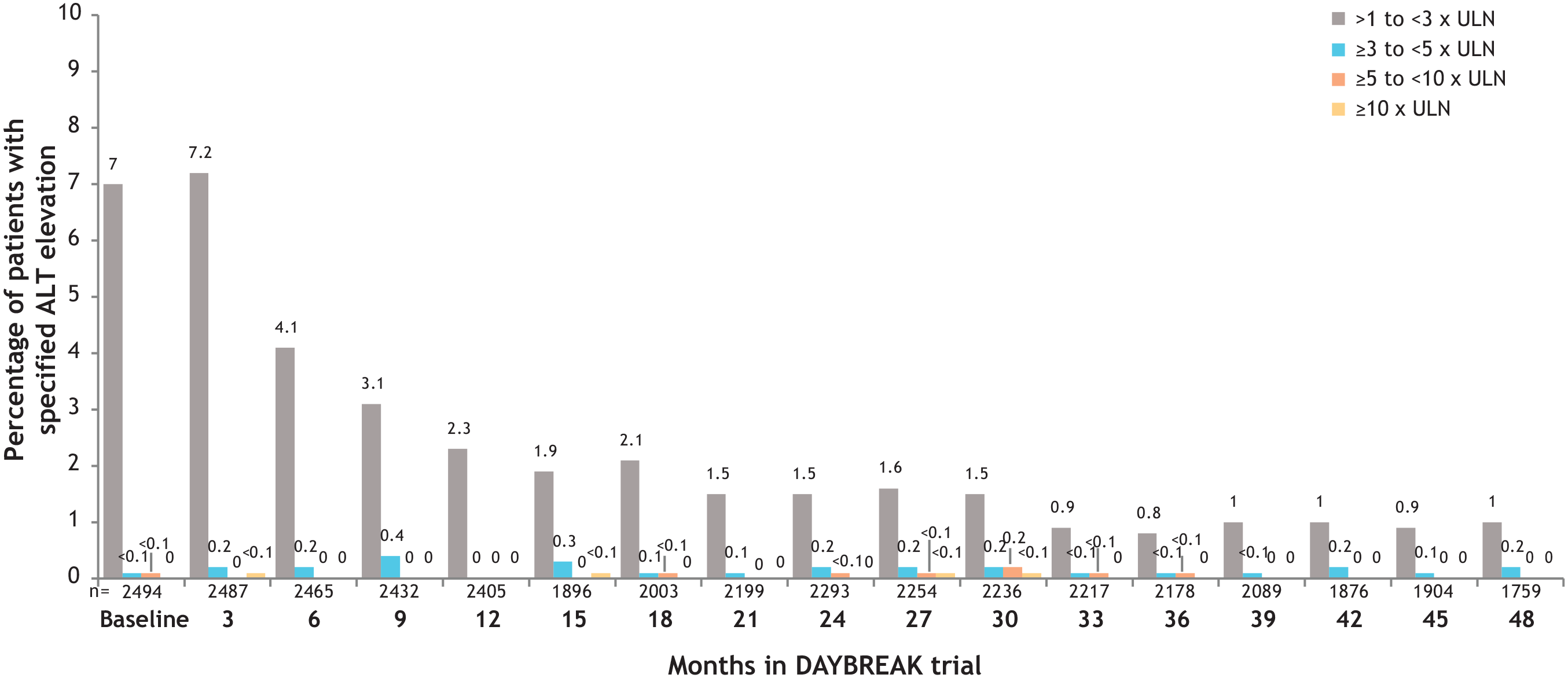
First occurrence of ALT elevations during DAYBREAK as a percentage of patients (ITT population).a
One hundred twenty-nine patients (5.2%) had a depression-related TEAE (Table 1). Five had suicidal ideation (IR 0.5/1000 PY (95% CI 0.2‒1.3)). Two attempted suicide (IR 0.2/1000 PY (95% CI 0.1‒0.8)).
Patients who received ozanimod 0.92 mg during the parent trials exhibited stable absolute lymphocyte counts (ALCs) (mean of 0.7‒0.8×109/L at each time point) throughout 48 months of DAYBREAK (Figure 4). After patients switched from interferon β-1a, mean ALC showed an expected decline over the first 3 months of ozanimod in DAYBREAK, to 43.9% of baseline; this reduction was largely sustained through month 48 (Figure 4). During DAYBREAK, ALC was less than 0.2 × 109/L in 244/2488 (9.8%) patients (Table 5). Persistent reductions were uncommon: 187 of the patients with ALC < 0.2×109/L had a repeat ALC assessment within 30 days, of whom 30 (16.0%, or 1.2% of the overall safety population (30/2488)) had a consecutive ALC < 0.2×109/L on retest, suggesting most instances were transient. Seven (0.3%) patients permanently discontinued treatment due to TEAEs reported as lymphopenia (n = 3) or lymphocyte count decreased (n = 4).
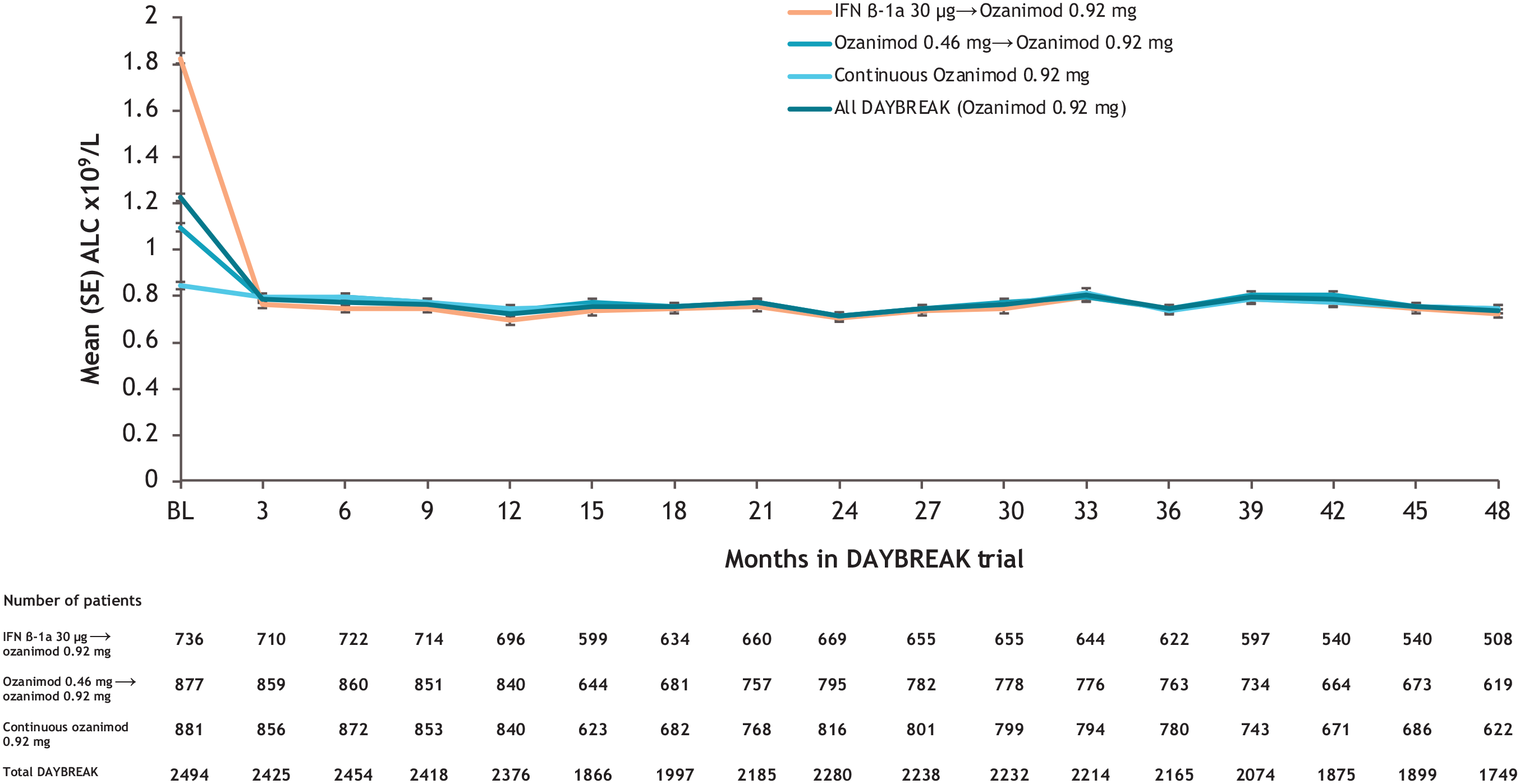
Mean ALC values during DAYBREAK in the overall population and by parent trial treatment subgroup.
Absolute lymphocyte count based on minimal value during DAYBREAK by parent trial treatment subgroup and overall DAYBREAK population.
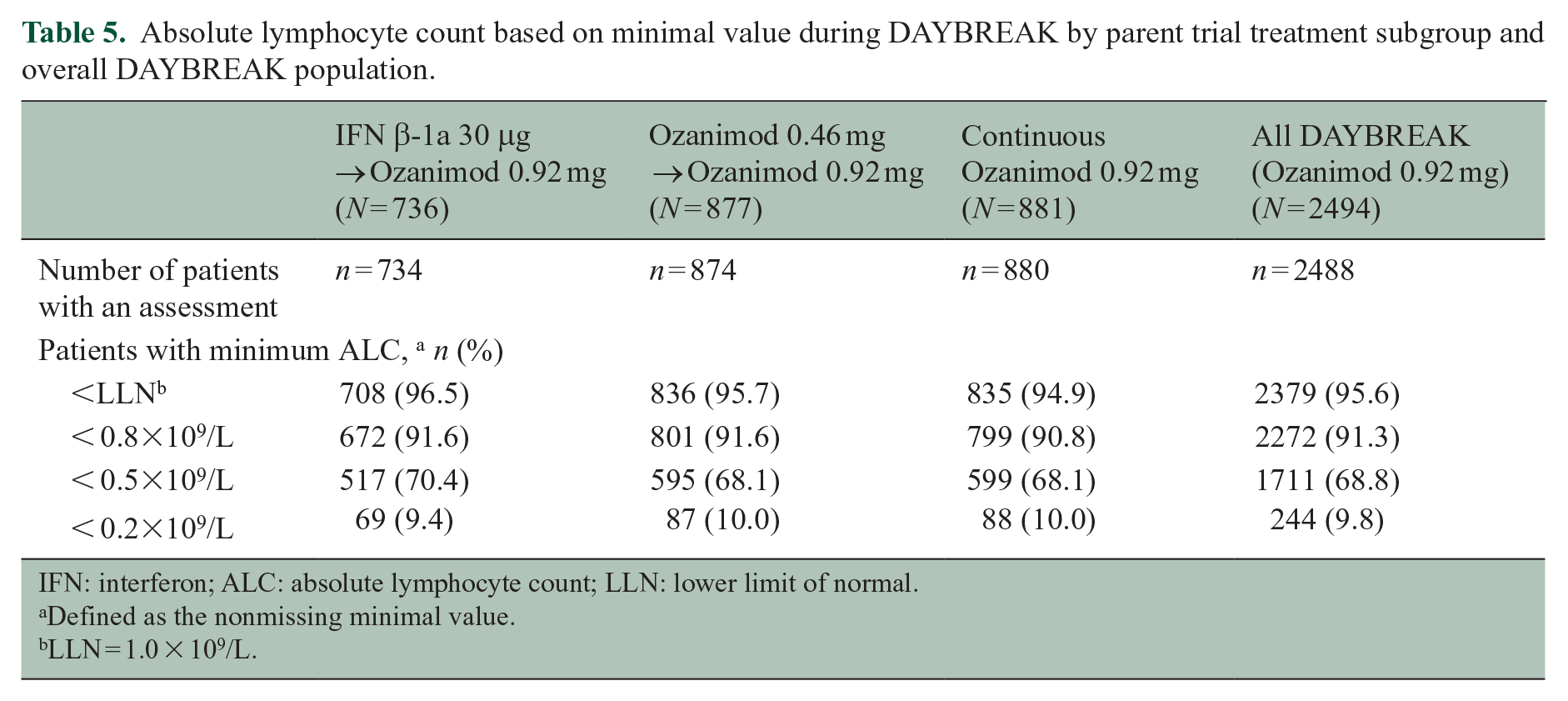
IFN: interferon; ALC: absolute lymphocyte count; LLN: lower limit of normal.
Defined as the nonmissing minimal value.
LLN = 1.0 × 109/L.
Of 439 (17.6%) patients who discontinued ozanimod in DAYBREAK for any reason, 10 (2.3%) subsequently experienced confirmed relapses, which all occurred >1 month (34‒141 days) after ozanimod discontinuation in the absence of any disease-modifying therapy (DMT). These relapses were associated with EDSS increases of 0.0‒2.5 points compared with the previous visit. Seven patients, including the patient with the 2.5-point EDSS increase, had complete recovery and three had partial recovery. Investigators did not consider any of the relapses to be severe, and no relapses resulted in a persistent severe increase in disability.
Efficacy
The adjusted ARR during DAYBREAK among all participants was 0.103 (95% CI 0.086‒0.123). ARR was similar across parent trial treatment subgroups (Figure 5(a)). Those who switched from interferon β-1a to ozanimod had the greatest reduction in ARR after entering DAYBREAK. During the first 48 months of DAYBREAK, 71% of patients remained relapse free (Figure 5(b)). Among those who continuously received ozanimod 0.92 mg in a parent trial and DAYBREAK, 64% remained relapse free throughout 60 cumulative months of treatment (the last assessment for which >50% had assessments; n = 493).
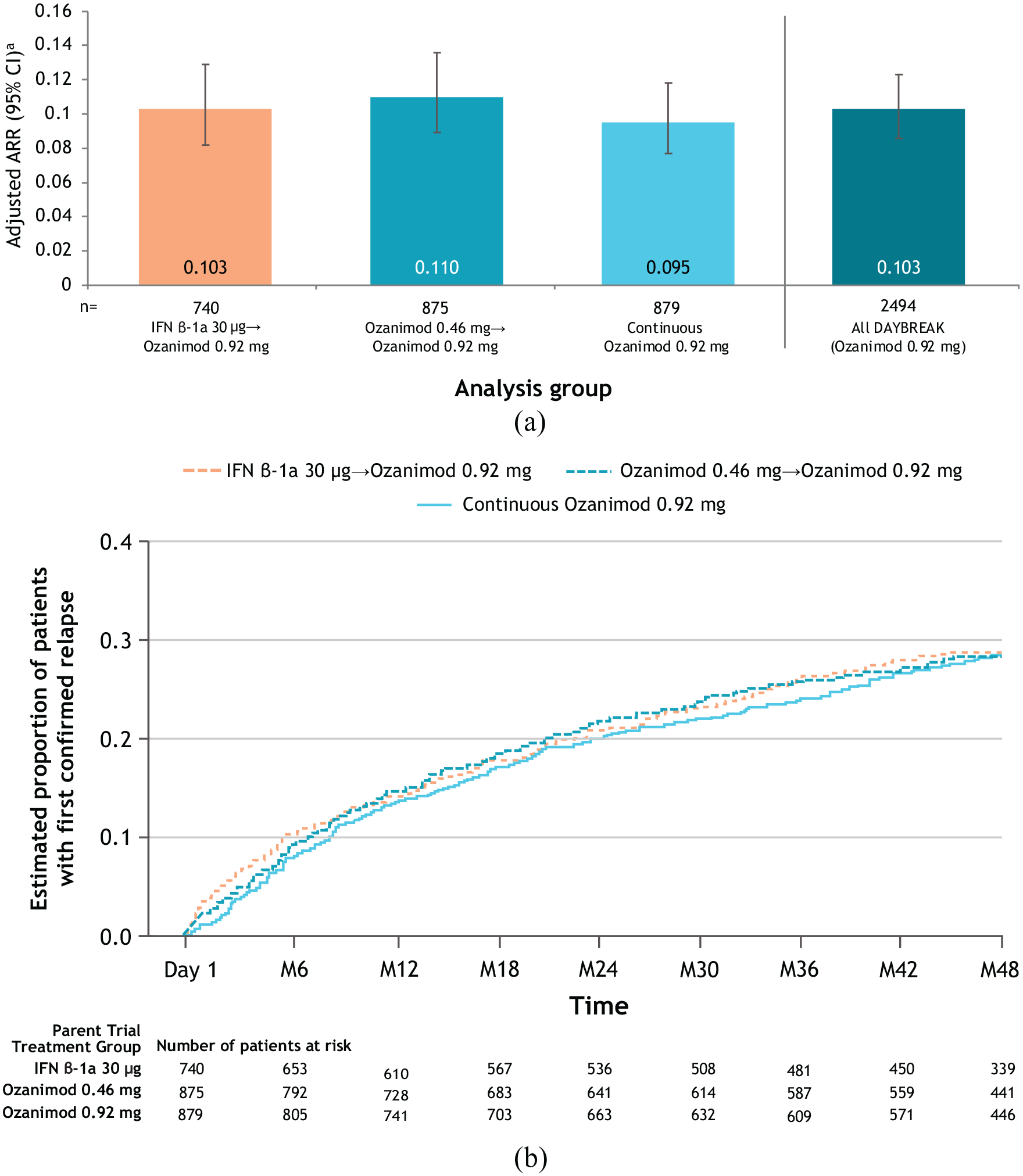
(a) Annualized relapse rate in DAYBREAK, ITT population. (b) Kaplan–Meier curve for time to first confirmed relapse during DAYBREAK, ITT population.
Adjusted mean number of new/enlarging T2 lesions per scan (Figure 6(a)) and GdE lesions (Figure 6(b)) at month 48 of DAYBREAK—when all patients had received ⩾4 years of ozanimod 0.92 mg—was low and similar across parent trial treatment subgroups. Patients initially treated with interferon β-1a experienced a decline in lesion counts upon switching to ozanimod in DAYBREAK. Among parent trial treatment subgroups, 35.0% of the interferon β-1a group and 37.7% of both the ozanimod 0.46 and 0.92 mg groups had no new/enlarging T2 lesions from DAYBREAK baseline through month 48; 88.9%, 88.5%, and 87.0%, respectively, had no GdE lesions on the month 48 scan (Supplemental Table S6).
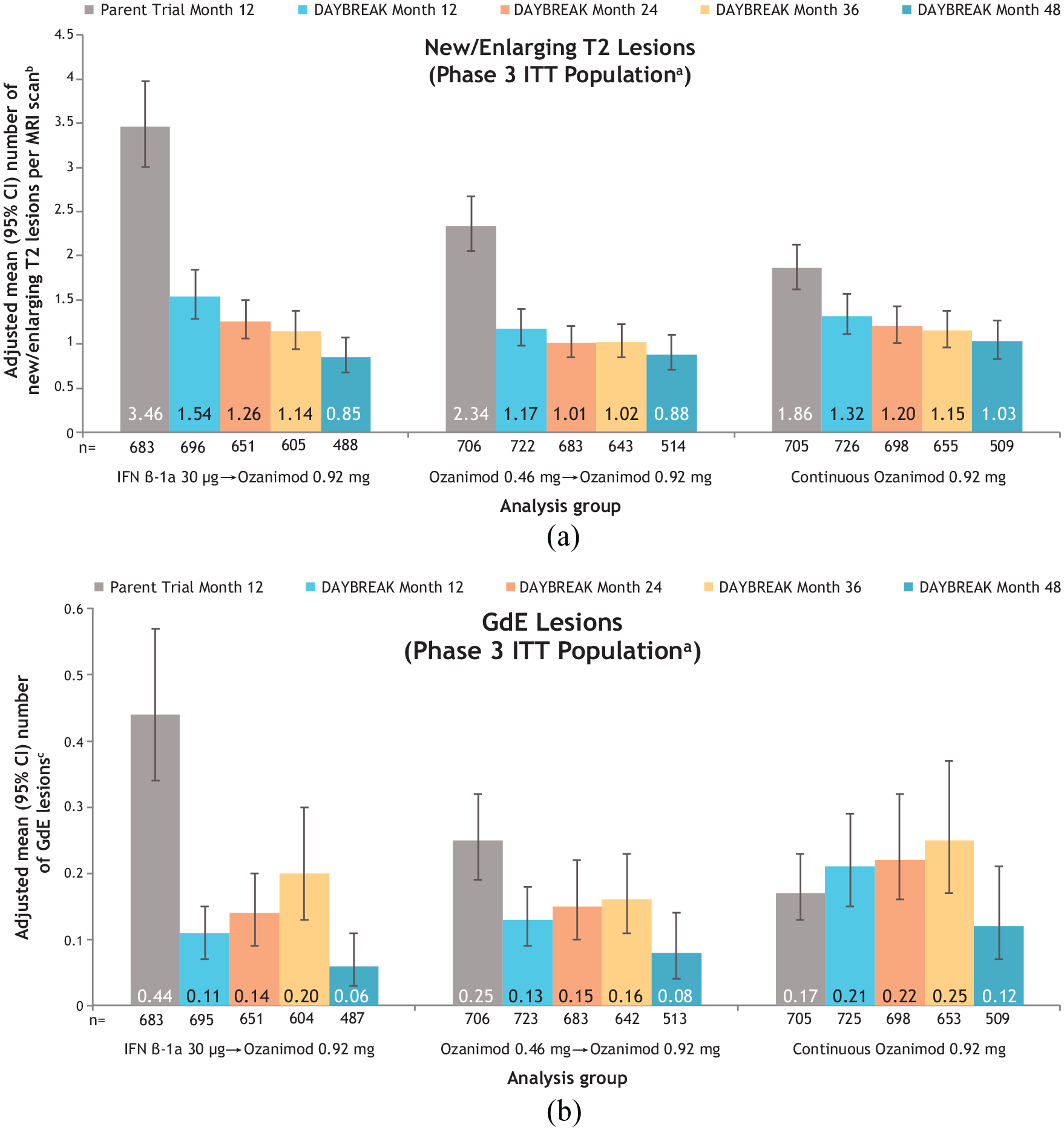
(a) Number of new/enlarging T2 lesions per scan, and (b) number of GdE lesions at month 12 of the active-controlled phase 3 parent trials and months 12, 24, 36, and 48 of the DAYBREAK extension by phase 3 parent trial treatment subgroup.
During DAYBREAK, 346/2494 (13.9%) patients had CDP-3 and 285/2494 (11.4%) had CDP-6 by data cutoff; rates were similar across parent trial treatment subgroups. Kaplan–Meier analysis of time to CDP-3 is shown in Figure 7.
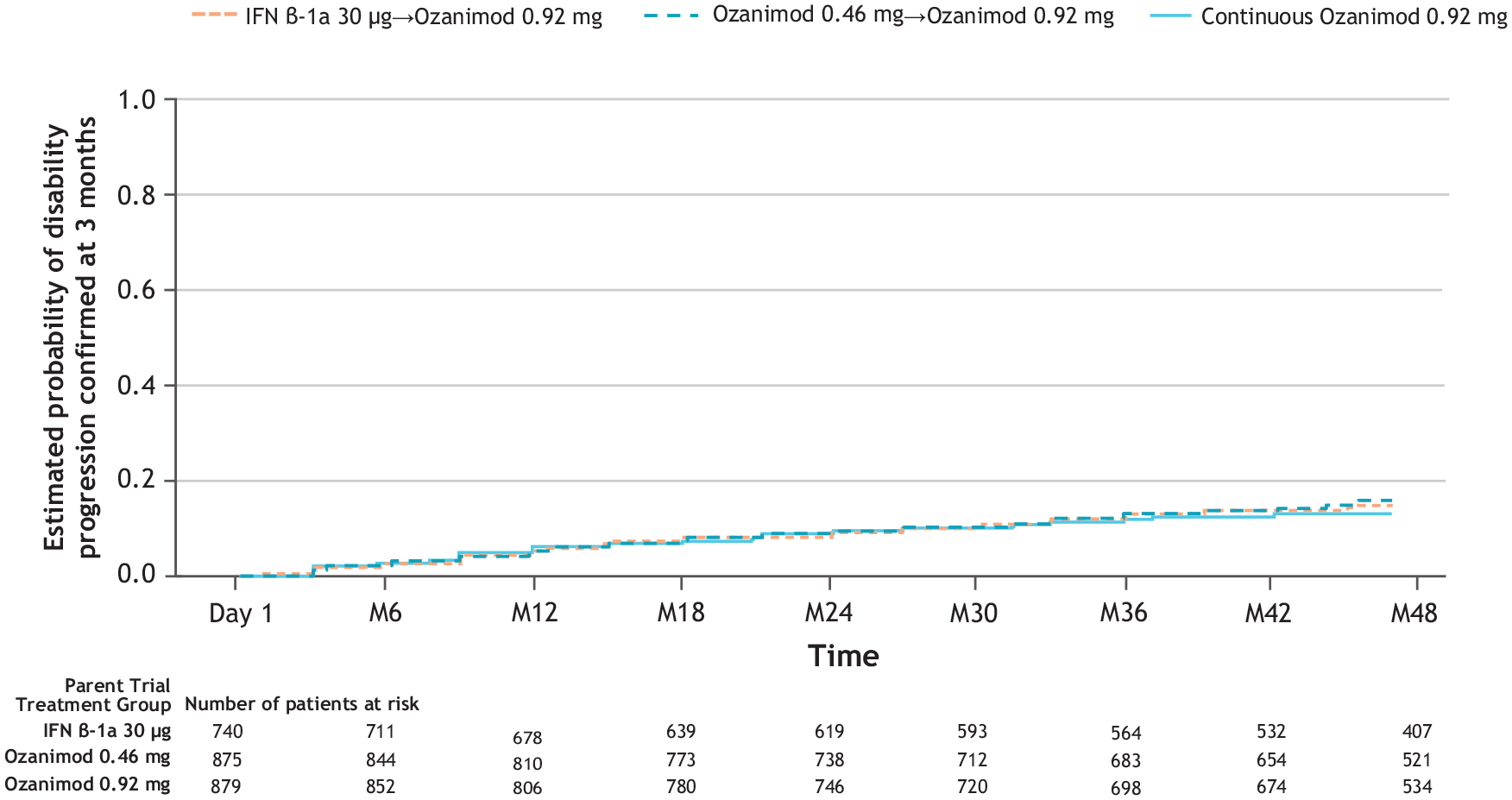
Kaplan–Meier analysis of time to onset of 3-month confirmed disability progression during DAYBREAKa (ITT population).
Discussion and conclusions
This interim analysis of the DAYBREAK extension study characterizes safety and efficacy of ozanimod for up to 5 years in DAYBREAK and up to 8 continuous years of exposure in patients with RMS, a longer duration than previously reported in randomized, controlled trials of ozanimod. The overall safety and tolerability profile of ozanimod was consistent with the phase 3 trials.3–5 MS clinical and radiologic disease activity remained low in patients who received ozanimod 0.92 mg continuously since the parent trials. Patients who switched to ozanimod 0.92 mg from either interferon β-1a or ozanimod 0.46 mg experienced reductions in ARR and brain MRI lesion counts.
Some DMTs with immunomodulatory effects, including S1P modulators, are associated with an increased incidence of herpes infection and reactivation.8–10 Herpes infections were the most common opportunistic infection; none were serious or disseminated. The IRs for oral herpes (5.4/1000 PY) and herpes zoster (4.6/1000 PY) in DAYBREAK were similar to values reported in an analysis of the overall ozanimod MS clinical development program that used an earlier DAYBREAK data cutoff (oral herpes: 5.7/1000 PY; herpes zoster: 5.3/1000 PY), suggesting that risk of these infections does not increase with longer ozanimod use. 5 Serious herpetic infections have occurred in patients taking other S1P receptor modulators.11,12 The IRs for oral herpes and herpes zoster were lower in DAYBREAK than previously reported with fingolimod (oral herpes: 10.3/1000 PY; varicella zoster virus: 7‒11/1000 PY).13,14
Among 2494 DAYBREAK participants, one patient developed PML shortly after data cutoff. 6 This is the first known case in an ozanimod-treated patient across 16,512 PY of exposure in the ozanimod development program, including MS and inflammatory bowel disease, and 6455 PY of postmarketing exposure to ozanimod, as of 19 November 2021. PML has been reported with MS DMTs, including S1P modulators.15–17
COVID-19 cases during DAYBREAK were largely nonserious and a vast majority of patients recovered from COVID-19 without sequelae. The 18-month cumulative incidence of total and confirmed COVID-19 among DAYBREAK participants was 8.7% and 7.3%, respectively. The cumulative incidence of COVID-19 varies worldwide, 18 and data show that MS patients do not have a higher risk of infection with SARS-CoV-2.19,20 As of 9 May 2021, the cumulative incidence of COVID-19 in the general population was 0.05%‒10.0% in countries participating in DAYBREAK.18,21 Over 90% of DAYBREAK participants are from Eastern Europe; published COVID-19 incidence data among MS patients in Eastern Europe are sparse or lack robust sample size. COVID-19-related mortality in DAYBREAK (3/190 (1.6%)) was similar to case-fatality rates in the general populations of participating countries (0.7%‒4.4%) 18 and within range of recent studies of COVID-19 mortality among MS patients (0.3%‒4.2%).22–26 In a review of MS patients, COVID-19 mortality based on data through 18 December 2020 was 3.0% overall and 0.5%‒2.5% among DMT users. 27
The IR for malignancy with ozanimod during the parent trials plus DAYBREAK is 358.6/100,000 PY (95% CI 253.7‒492.2), and the IR in an earlier analysis of the same trials was 289.3/100,000 PY (95% CI 187.2‒427.1). The IR in the phase 3 trial population was 298.2/100,000 PY (95% CI 128.7‒587.6). 5 No particular pattern in malignancy type occurred. Malignancies are a potential concern with any immune-modulating therapy, including S1P modulators.28–30 A study of S1P modulators suggests there may be an association with an increase in risk of skin cancers, particularly basal cell carcinomas. 31 Similar to the general population, basal cell carcinomas were the most frequent cancer type reported in ozanimod clinical trials (IR 103.6/100,000 PY). The estimated incidence of basal cell carcinoma in the general population ranges from 25 to 321 per 100,000 PY in European and US populations.32–35 There were nine cases of breast cancer among ozanimod-treated women with RMS in the parent trials or DAYBREAK; the expected incidence of breast cancer was seven events over the treatment period, derived by applying the breast cancer IR (92.4/100,000 PY) in an age-matched female population from the Surveillance, Epidemiology, and End Results to the female RMS participants with exposure to ozanimod (7096 PY). Malignancies continue to be monitored in the ozanimod clinical development program.
Macular edema is a TEAE with S1P modulators.12,13,30 The incidence of macular edema in DAYBREAK was low. Most confirmed cases occurred in patients with predisposing conditions or risk factors within 1 year of ozanimod initiation. One case without risk factors occurred after 6 years of ozanimod exposure; delayed onset of ME is unusual but has been reported after 7 years of fingolimod use.36,37
Bradycardia was infrequent, occurring mostly in those who switched from interferon β-1a or ozanimod 0.46 mg to ozanimod 0.92 mg. Similarly, bradycardia is uncommon after the first year of fingolimod use. 13 The IR for myocardial infarction in DAYBREAK (0.3/1000 PY (95% CI 0.1‒1.0)) was similar to that reported in the LONGTERMS study of fingolimod (IR 0.2/1000 PY). 13 Cardiovascular abnormalities associated with S1P modulators12,13,30 are thought to be mediated by S1P receptors expressed by cardiomyocytes. 38
PFT abnormalities, observed in a minority of patients, were rarely clinically relevant, as discontinuation for dyspnea or PFT abnormalities was uncommon. Dose-dependent reductions from baseline in FEV1 have occurred with other S1P modulators.39–41
The proportion of patients with hepatic laboratory abnormalities during DAYBREAK was similar to that in SUNBEAM 4 and RADIANCE. 3 These abnormalities were generally not of clinical consequence. Liver enzyme elevations are a known effect of S1P modulators, and cases of acute liver failure requiring transplantation have been reported in patients treated with fingolimod.40–42
ALC reduction to below normal (based on laboratory values), which occurred in 95.6% of patients, is an expected pharmacodynamic effect of ozanimod, and therefore was not required to be reported as a TEAE. For those who continued ozanimod 0.92 mg from parent trial into DAYBREAK, mean ALC remained stable and consistent with ALC levels at that dose in SUNBEAM 4 and RADIANCE. 3 In a prior pooled safety analysis of DAYBREAK and the parent trials, 182 patients had a minimal postbaseline ALC < 0.2 × 109/L, but ALC was less than 0.2 × 109/L around the time of onset of only 1/44 serious infections and 1/113 nonserious opportunistic infections; thus, no relationship was found between ALC < 0.2 × 109/L and serious or opportunistic infections. 5
Patients discontinuing any MS DMT are vulnerable to posttreatment relapse. Incidence of relapse after ozanimod discontinuation was low in DAYBREAK, and none of the relapses resulted in persistent, severe disability. Cases of severe disease recurrence have been reported after discontinuation of other S1P modulators.43,44
Limitations typical of open-label extension studies include lack of a control group or blinding; therefore, comparisons to blinded study data may be limited. Patients with less favorable outcomes may have dropped out of the parent trials, creating potential selection bias, but the number of subjects who did not enter the OLE was low (5.5% of eligible patients) and only 17.6% of enrolled patients discontinued from DAYBREAK.
Data from this ongoing, long-term observational study of patients with RMS treated with ozanimod 0.92 mg for up to 5 years are consistent with the established safety profile and demonstrate sustained control of disease activity and disability progression with continued ozanimod use. Adverse events, including COVID-19, were generally manageable, infrequently leading to permanent treatment discontinuation or adverse outcomes.
Supplemental Material
sj-docx-1-msj-10.1177_13524585221102584 – Supplemental material for Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial
Supplemental material, sj-docx-1-msj-10.1177_13524585221102584 for Long-term safety and efficacy of ozanimod in relapsing multiple sclerosis: Up to 5 years of follow-up in the DAYBREAK open-label extension trial by Bruce AC Cree, Krzysztof W Selmaj, Lawrence Steinman, Giancarlo Comi, Amit Bar-Or, Douglas L Arnold, Hans-Peter Hartung, Xavier Montalbán, Eva K Havrdová, James K Sheffield, Neil Minton, Chun-Yen Cheng, Diego Silva, Ludwig Kappos and Jeffrey A Cohen in Multiple Sclerosis Journal
Footnotes
Acknowledgements
Support for third-party writing assistance for this manuscript was provided by Lauren A Cerruto and Jessica D Herr, PharmD, of Peloton Advantage, LLC, an OPEN Health company, and funded by Bristol Myers Squibb. The authors thank Sonia Ariana Afsari, MD, and Jenna Hoogerheyde for reviewing the manuscript and also thank the members of the Data Monitoring Committee, the Macular Edema Review Panel, the expert hepatic safety advisors, the patients, their families, and trial investigators for their contributions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.A.C.C. reports personal compensation for consulting for Alexion, Atara, Autobahn, Avotres, Biogen, EMD Serono, Gossamer Bio, Horizon, Neuron23, Novartis, Sanofi, TG Therapeutics, and Therini, and received research support from Genentech. K.W.S. reports consulting for Biogen, Celgene, Genzyme, Merck, Novartis, Ono Pharma, Roche, Synthon, and Teva. L.S. reports consulting for AbbVie, Atreca, Celgene, Novartis, Teva, Tolerion, and EMD Serono, and research support from Atara, Biogen, and Celgene. G.C. reports compensation for consulting and/or speaking activities from Almirall, Biogen, Celgene, EXCEMED, Forward Pharma, Genzyme, Merck, Novartis, Roche, Sanofi, and Teva. A.B.-O. participated as a speaker in meetings sponsored by and received consulting fees and/or grant support from Atara Biotherapeutics, Biogen, BMS-Celgene, EMD Serono, Sanofi-Genzyme, Novartis, and Roche-Genentech. D.L.A. reports personal fees for consulting and/or grants from Albert Charitable Trust, Biogen, Novartis, Celgene, F. Hoffmann-La Roche, Frequency Therapeutics, MedDay, Merck Serono, Novartis, Population Council, and Sanofi-Aventis; grants from Biogen, Immunotec, and Novartis; and an equity interest in NeuroRx Research. H.-P.H. reports personal fees for consulting, serving on steering committees, and speaking from Bayer Healthcare, Biogen, Celgene, GeNeuro, Genzyme, Merck, MedImmune, Novartis, Octapharma, Roche, Sanofi, and Teva. X.M. has received speaking honoraria and travel expenses for participation in scientific meetings, has been a steering committee member of clinical trials or participated in advisory boards of clinical trials in the past 3 years with Actelion, Alexion, Bayer, Biogen, Bristol Myers Squibb/Celgene, EMD Serono, EXCEMED, Genzyme, Hoffmann-La Roche, Immunic, Janssen Pharmaceuticals, MedDay, Merck, Mylan, MSIF, Nervgen, NMSS, Novartis, Roche, Sanofi-Genzyme, Teva Pharmaceuticals, and TG Therapeutics. E.K.H. reports personal compensation for consulting and speaking for Actelion, Biogen, Celgene, Merck, Novartis, Roche, Sanofi, and Teva, and is supported by Czech Ministry of Education, project PROGRES Q27/LF1. J.K.S., N.M. C.-Y.C and D.S. are employees and shareholders of Bristol Myers Squibb. L.K. has received institutional research support: steering committee, advisory board, consultancy fees: Actelion, Bayer HealthCare, Biogen, Bristol Myers Squibb, Genzyme, Janssen, Japan Tobacco, Merck, Novartis, Roche, Sanofi, Santhera, Shionogi, and TG therapeutics; speaker fees: Bayer HealthCare, Biogen, Merck, Novartis, Roche, and Sanofi; support of educational activities: Allergan, Bayer HealthCare, Biogen, CSL Behring, Desitin, Genzyme, Merck, Novartis, Roche, Pfizer, Sanofi, Shire, and Teva; license fees for Neurostatus products; and grants: Bayer HealthCare, Biogen, European Union, Innosuisse, Merck, Novartis, Roche, Swiss MS Society, and Swiss National Research Foundation. J.A.C. reports personal compensation for consulting for Adamas, Atara, Bristol Myers Squibb, Convelo, MedDay, and Mylan; and serving as an Editor of Multiple Sclerosis Journal.
Some of the data in this manuscript were previously presented at the 37th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS); 13–15 October 2021; The Digital Experience.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ozanimod RMS trials were supported by Celgene International II. The sponsor was involved in data analysis and interpretation, and manuscript preparation, review, and approval. All authors vouch for data accuracy, reviewed all drafts, and approved the final manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
