Abstract
Background:
Siponimod-related lymphopenia in real-world clinical practice has implications for dose adjustment and infection risk.
Objective:
To characterise siponimod-related lymphopenia in people with secondary progressive multiple sclerosis (pwSPMS).
Methods:
This is a retrospective cohort of 188 pwSPMS. The development of grade 4 lymphopenia was interrogated with Kaplan–Meier survival analysis and binary logistic regression.
Results:
Lymphopenia develops soon after commencing siponimod. In total, 15 (8.5%) of 176 experienced grade 4 lymphopenia at 1 month after initiation. There were no clinically significant associations between patient characteristics and development of grade 4 lymphopenia.
Conclusion:
Grade 4 lymphopenia can occur soon after siponimod initiation and cannot be predicted.
Introduction
Siponimod is the only oral disease-modifying therapy approved for active secondary progressive multiple sclerosis (SPMS). 1 The approval for siponimod followed the EXPAND double-blind, randomised, phase 3 trial, 2 which demonstrated a reduction in confirmed disability progression.
Siponimod is a selective sphingosine 1-phosphate (S1P1,5) receptor modulator, reducing lymphocyte egress from secondary lymphoid organs. 3 A relationship has been observed between infections and siponimod-related lymphopenia (specifically lymphocyte count <0.4 × 109/L). 4 The development of grade 4 lymphopenia has particular importance in SPMS clinics: if grade 4 lymphopenia develops and is confirmed on a repeat blood test, it is advised to reduce the dose from 2 to 1 mg, or to interrupt therapy if the person is already receiving 1 mg. 1
It is unknown which people with secondary progressive multiple sclerosis (pwSPMS) are more likely develop grade 4 lymphopenia. Potential risk factors include age (immunosenescence 5 ), sex, 6 recent administration of a lymphopenia-associated disease-modifying therapy (L-DMT) 7 and baseline lymphocyte count. 8 Siponimod dose was also considered: the maintenance dose of 2 mg is reduced to 1 mg for CYP2C9*1/*3 or *2/*3 genotypes, 9 although this halving of dose does not equally adjust for the increased exposure with these genotypes.
This is a retrospective cohort analysis of pwSPMS receiving siponimod at three tertiary centres. The aims of this study are threefold. First, we aimed to characterise the degree of lymphopenia experienced by pwSPMS receiving siponimod. Second, we set out to investigate when grade 4 lymphopenia occurred. Finally, we explored whether the development of grade 4 lymphopenia within 6 months of initiating siponimod is associated with any patient characteristics, specifically age, sex, baseline lymphocyte count, dosage or L-DMT.
Methods
Participants
This multicentre retrospective clinical audit of pwSPMS receiving siponimod was undertaken by three UK tertiary centres: Nottingham University Hospitals NHS Trust (NUH), Sheffield Teaching Hospitals NHS Foundation Trust (STH), and University College London Hospitals NHS Foundation Trust (UCL). NUH was the audit sponsor and received anonymised data from the other centres. Full details are found in Supplemental Material 1.
Statistical analysis
Statistical tests included Kaplan–Meier survival analysis and binary logistic regression (BLR). A directed acyclic graph (DAG) was designed to inform adjustments for BLR. Full details can be seen in Supplemental Material 1.
Results
A total of 188 pwSPMS receiving siponimod were included in the analysis. Demographic and clinical characteristics are reported in Supplemental Material 2.
The proportion of pwSPMS experiencing each grade of lymphopenia at baseline and 1, 3, 6, 9 and 12 months after initiating siponimod is presented in Table 1 and plotted in Figure 1(a). Grade 4 lymphopenia was noted in 8.5% (15/176) at 1 month after starting siponimod and 5.9% (7/118) at 6 months.
Lymphopenia grading at baseline prior to initiation of siponimod and at follow-up after commencing siponimod.
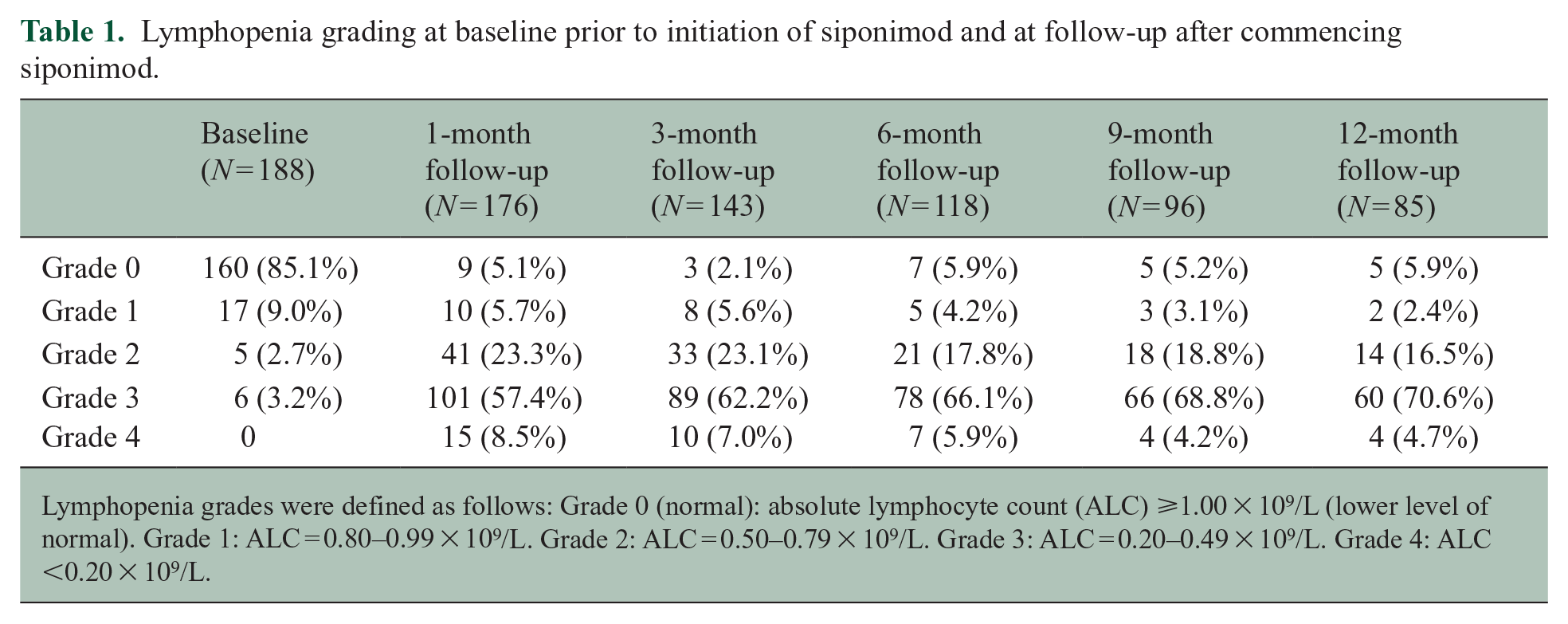
Lymphopenia grades were defined as follows: Grade 0 (normal): absolute lymphocyte count (ALC) ⩾1.00 × 109/L (lower level of normal). Grade 1: ALC = 0.80–0.99 × 109/L. Grade 2: ALC = 0.50–0.79 × 109/L. Grade 3: ALC = 0.20–0.49 × 109/L. Grade 4: ALC <0.20 × 109/L.
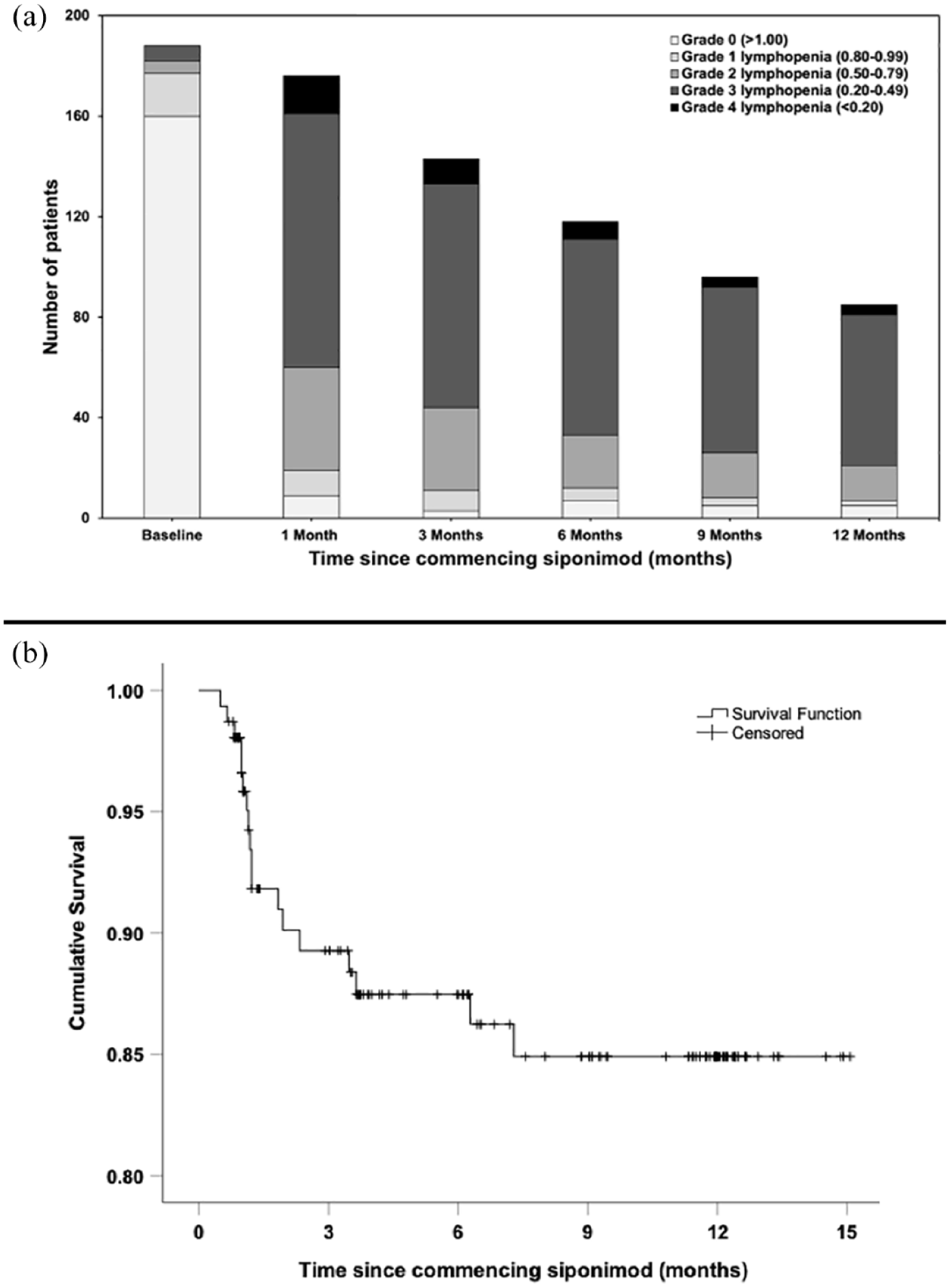
(a) Lymphopenia grading prior to initiation of siponimod and at follow-up after commencing siponimod (188 patients). (b) Kaplan–Meier analysis for the development of grade 4 lymphopenia with siponimod (155 patients with 18 events). Patients were excluded after initial event of grade 4 lymphopenia, with no re-counting.
The Kaplan–Meier curve in Figure 1(b) depicts the timing of onset for grade 4 lymphopenia in the study population. In total, 155 pwSPMS from NUH and STH were included in this analysis and 18 pwSPMS experienced grade 4 lymphopenia. Events tended to occur early, at mean 64 days after commencing siponimod. First grade 4 lymphopenia events did, however, occur as late as 7.3 months after commencing siponimod.
The occurrence of grade 4 lymphopenia over the first 6 months from initiating siponimod was investigated by BLR. In total, 125 pwSPMS from all sites were included in this analysis. There were no clinically significant associations identified for baseline lymphocyte count (odds ratio (OR) = 0.35; 95% confidence interval (CI) = (0.14–0.85)), sex (female vs male, OR = 0.87; 95% CI = (0.32–2.33)), age (OR = 0.99; 95% CI = (0.94–1.04)), initial maintenance dose of siponimod (2 mg vs 1 mg, OR = 2.39; 95% CI = (0.29–19.68)) or prior use of L-DMTs (OR = 0.98; 95% CI = (0.20–4.89)) by the time of, and including, the 6-month follow-up after commencing siponimod (Supplemental Material 3).
Discussion
This retrospective cohort analysis characterises the development of grade 4 lymphopenia with siponimod longitudinally across 12 months of data collection in real-world clinical practice (Table 1 and Figure 1(a)).
The timing for onset of grade 4 lymphopenia was investigated by Kaplan–Meier survival analysis. This demonstrated that in most pwSPMS, severe lymphopenia occurred soon after initiating siponimod (Figure 1(b)).
BLR analysis investigated whether baseline lymphocyte count, age, sex, dosage or L-DMT was associated with the development of grade 4 lymphopenia over the first 6 months of treatment. This model did not identify any clinically significant associations.
The lack of association with L-DMT is notable. Almost all L-DMT pwSPMS were previously receiving fingolimod (13/15), another S1P modulator. Substituting medications within the same drug class would not be expected to have a clinically significant impact on lymphopenia. Lymphocyte counts often return to normal between 1 and 2 weeks of discontinuing fingolimod, 10 and our analysis was of L-DMTs over the 6 months prior to initiating siponimod. The lack of association between initial maintenance dosage of siponimod (2 mg vs 1 mg) and the development of grade 4 lymphopenia may be accounted for by their differing cohorts: those receiving the lower dose of siponimod are genetically predisposed to its accumulation. In the general population, there is an overall reduction in lymphocyte count with age, 11 which occurs in the context of immunosenescence.5,12 It is reassuring, therefore, that age was not associated with grade 4 lymphopenia within 6 months of commencing siponimod. The lack of a clinically significant association for baseline lymphocyte count will likely surprise clinicians and may provide confidence in prescribing for pwSPMS with lower baseline counts. The lack of association for sex is similarly encouraging.
Strengths of our study include that it encompasses data from three tertiary multiple sclerosis (MS) centres, has a large cohort (188 pwSPMS), and characterises lymphopenia through regression analysis and Kaplan–Meier analysis. Our study may have been improved through investigating for associations between lymphopenia and other patient characteristics such as ethnicity or Expanded Disability Status Scale (EDSS). A limitation is that our study does not report infections: primary care data would be needed for accurate calculation of infection rates, and their interpretation would require comparison with a cohort not receiving siponimod. Infections are reported in the EXPAND randomised controlled trial 2 and in long-term safety monitoring. 4
This retrospective cohort study demonstrates that lymphopenia develops quickly after starting siponimod, occurs to a significant extent, and is present throughout 12 months of data collection. It is important for MS clinicians to be aware that the development of grade 4 lymphopenia within 6 months of commencing siponimod lacks clinically significant associations with baseline lymphocyte count, age, dosage, sex or L-DMT: these factors should not unduly influence treatment decisions.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231225711 – Supplemental material for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585231225711 for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis by Christopher GS Gilmartin, Natasha Hoyle, Afagh Garjani, Terri Dixon, Helena Jos, David Paling, Wallace Brownlee, Christopher Tench and Nikos Evangelou in Multiple Sclerosis Journal
Supplemental Material
sj-docx-2-msj-10.1177_13524585231225711 – Supplemental material for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis
Supplemental material, sj-docx-2-msj-10.1177_13524585231225711 for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis by Christopher GS Gilmartin, Natasha Hoyle, Afagh Garjani, Terri Dixon, Helena Jos, David Paling, Wallace Brownlee, Christopher Tench and Nikos Evangelou in Multiple Sclerosis Journal
Supplemental Material
sj-docx-3-msj-10.1177_13524585231225711 – Supplemental material for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis
Supplemental material, sj-docx-3-msj-10.1177_13524585231225711 for Real-world data on siponimod-related lymphopenia among people with secondary progressive multiple sclerosis by Christopher GS Gilmartin, Natasha Hoyle, Afagh Garjani, Terri Dixon, Helena Jos, David Paling, Wallace Brownlee, Christopher Tench and Nikos Evangelou in Multiple Sclerosis Journal
Footnotes
Data Sharing Statement
This is a clinical audit where NUH is the audit sponsor. Data have not been shared to an online repository.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: C.G.S.G.: nothing to disclose. N.H.: Honoraria for advisory boards and/or educational activities from Merck, Roche and UCB. A.G.: nothing to disclose. T.D.: nothing to disclose. H.J.: nothing to disclose. D.P.: He is a local principal investigator for commercial trials funded by Novartis and Janssen Pharmaceuticals, has received an investigator grant from Sanofi Genzyme and held roles on advisory boards/received consultancy and speakers’ fees from Biogen, Celgene, Janssen, MedDay, Merck, Novartis and Roche. W.B.: He has acted as a consultant and/or accepted speaker honoraria from Biogen, Celgene, Janssen, Merck, Novartis, Roche, Sanofi and Viatris. C.T.: nothing to disclose. N.E.: He has served as a member of advisory boards for Biogen, Merck, Novartis and Roche and has received grant income from the UK Multiple Sclerosis Society, Medical Research Council, Patient-Centered Outcomes Research Institute and National Institute for Health Research.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Statement and Patient Consent Information
This study is a clinical audit of routine care. Nottingham University Hospitals NHS Trust was the audit sponsor and received anonymised data from the other centres.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
