Abstract
Background:
Clinical practice, expert opinion, and evidence-based guidelines recommend daily stretching as first-line treatment for multiple sclerosis (MS) spasticity, but this has not been evaluated by fully powered clinical trials.
Objective:
To determine whether MS Spasticity: Take Control (STC), a guideline-based program of spasticity education and stretching exercises has different effects on the impact of spasticity than a control program of different spasticity education and range of motion (ROM) exercises.
Methods:
Ambulatory people with self-reported MS spasticity were randomly assigned to STC or ROM, delivered in same duration, facilitator-led, group classes, face-to-face (F2F) initially and later virtually, due to coronavirus disease 2019 (COVID-19). Multiple Sclerosis Spasticity Scale (MSSS) scores were compared between groups at 1 (primary outcome) and 6 months after interventions.
Results:
A total of 231 people enrolled. There was no significant difference in MSSS scores between STC and ROM at 1 month (mean difference = 0.28, 95% (confidence interval (CI)) = [−9.45 to 10.01], p = 0.955). There were significant group mean improvements in MSSS scores and most other outcomes at 1 and 6 months.
Conclusion:
Education with stretching exercises, the first-line recommended treatment for MS spasticity, and education with ROM exercises may both improve MS spasticity to a similar degree. This study debunks the belief that stretching is essential to managing MS spasticity.
Introduction
In total, 60% to 85% of people with multiple sclerosis (PwMS) experience spasticity with velocity-dependent involuntary resistance to stretch. Spasticity, a component of the upper motoneuron syndrome, is likely due to lesions in the descending motor pathways, including the reticulospinal paths, reducing inhibition of alpha and gamma motor neurons. Although spasticity is neural in origin, significant structural adaptations occur in the soft tissue, including the muscle cells, tendon, and extracellular matrix.1–4 Multiple sclerosis (MS) spasticity causes limitations in movement and pain which can result in problems with walking, working, daily activities, and other serious complications.1–3,5 These consequences and the interaction with other features of the upper motoneuron syndrome such as muscle weakness make the management of spasticity challenging. 4 First-line recommended treatments for MS spasticity are education, oral medications, and stretching.2,3,6 Stretching is thought to reduce spasticity by improving the viscoelastic properties of the muscle-tendon units and lengthening the muscles, thereby potentially normalizing muscle tone, reducing pain, and improving function. Stretching exercises are currently the most widely used physical intervention for spasticity management.4,7–9 Stretching takes time and effort, and its benefits are not proven.
In 1993, the top research priority of the spasticity panel of the multidisciplinary “What Do We Know in MS” conference was to answer the question, “Does a program of daily stretching result in decreased severity of spasticity?” 10 Ten years later, in 2003, based on expert opinion rather than evidence, the clinical practice guideline Spasticity Management in MS: Evidence-Based Management Strategies for Spasticity Treatment in MS (guideline) recommended stretching as foundational for managing MS spasticity.2,3 The need for high-quality research including randomized controlled trials (RCTs) with sensitive and valid outcomes was reiterated. There are now a number of valid outcome measures for MS spasticity, including the Multiple Sclerosis Spasticity Scale (MSSS)11,12 and the Numeric Rating Scale (NRS), 13 with the MSSS being more granular. Based on the guideline, our team developed MS Spasticity: Take Control (STC), a facilitated in-person group self-management program with spasticity education and instruction in standardized stretching exercises followed by daily home practice. STC demonstrated acceptance, feasibility, and potential for efficacy, as measured by mean changes in MSSS total and subscale scores, in a small (n = 38) randomized controlled pilot trial. 14
Here, we report the results of a full-scale RCT comparing STC to an active control for reducing the impact of spasticity, measured by the MSSS, in PwMS and self-reported spasticity. Due to the coronavirus disease 2019 (COVID-19) pandemic, we transitioned midway from face-to-face (F2F) to virtual study activities and also report these comparisons.
Materials and methods
This was a single-center, investigator-masked, parallel, two-arm, randomized, controlled trial with 1:1 allocation comparing STC to an active control, range of motion (ROM), for lower extremity spasticity in PwMS. Participants enrolled from September 2018 to May 2021. In 2020, F2F activities were interrupted for 6 months by the COVID-19 pandemic until resumed virtually. This study was approved by the joint Veterans Administration (VA) Portland Health Care System (VAPORHCS) and Oregon Health & Science University (OHSU) Institutional Review Board, registered with clinicaltrials.gov (#NCT03166930), and all participants signed and received a copy of the informed consent. Funding was provided by the VA Rehabilitation Research and Development Service of the VA Office of Research and Development.
Potential participants were identified from VAPORHCS and OHSU electronic medical records, support groups, advertisements, mailings, and email blasts, initially within 50 miles of Portland, Oregon, and then, when transitioned to virtual, throughout the United States. Inclusion criteria were physician confirmed diagnosis of MS of any subtype, ⩾18 years, able to participate in all aspects of the study including walking 25 feet independently with or without assistive devices, fluent in English, and having self-reported lower extremity spasticity using the North American Research Committee on Multiple Sclerosis (NARCOMS) definition: unusual tightening of muscles that feels like leg stiffness, jumping of the legs, repetitive bouncing of the foot, muscle cramping in the legs, or the legs going out tight and straight or drawing up. 1 Potential participants were excluded if they had spasticity from a cause other than MS. Men who were not also military veterans were excluded as required by the study sponsor.
Interventions
This study compared the active intervention, STC, with a control intervention, ROM. Participants were block randomized in groups of four separately for men and women in a 1:1 ratio using statistician generated randomization lists.
The active program, STC, was designed around an international evidence-based MS spasticity management guideline as an implementation and self-management program. STC’s education focuses on spasticity recognition and treatment and the exercises are stretching, defined as moving through active ROM and applying tension into passive ROM until a pull in the stretching muscle is felt but not to the point of feeling pain. Daily stretching is thought to prevent progressive complications from spasticity. Since evidence for this is lacking, our study was designed to provide high-quality evidence about the impact of stretching on MS spasticity. The program is standardized with two DVDs, a written participant manual with the information from the DVDs and photographs with written instructions for all the exercises, and a facilitator manual for running the two sessions. The first DVD is 28 minutes long and focuses on recognizing spasticity and triggers, impacts on quality of life, treatment, and encouragement to take control. The second DVD is 20 minutes long with models demonstrating stretching exercises for muscles of the trunk, hips, knees, and calves, with variations for ability and preference. In the first class, participants view and discuss the DVDs. In the second class, the facilitator demonstrates the stretches and participants try them and choose at least one for each body area for a daily 15- to 30-minute routine.
ROM was based on the National Multiple Sclerosis Society (NMSS) booklet Stretching for PwMS: An Illustrated Manual. 15 The NMSS has provided this or similar information for free to PwMS for more than 30 years. The education in the ROM control program focuses on exercise-related terminology and positioning for spasticity management, and most of the exercises involve moving through the readily available ROM without pain. Only one of the exercises, the heel cord stretch, was called a stretch. We made the information and the exercises in the booklet into the ROM program for this research study. We call this program ROM because, although the booklet’s title implies that it is about stretching, the exercises are mostly ROM exercises, with only one a stretching exercise. In the first session, participants discuss the information in the first third of the booklet: types of exercise, terminology, positions resulting from spasticity with suggestions for managing these positions, and precautions for the exercises. They then practice the sitting coordination exercises. In the second session, the facilitator demonstrates the remaining exercises and participants practice the exercises. Participants are instructed to do the exercises for 15–30 minutes per day, cycling through all the exercises over a few days if needed.
Education and exercise are both first-line recommendations for management of MS spasticity.2,3 To control for time and attention, both interventions in our study included MS spasticity education and daily exercises and were delivered in two 2-hour classes 1 week apart. Classes were led by trained facilitators who made reminder calls, confirmed attendance, and were retrained every 6 months and with transition to virtual classes. Class size was limited to 10 for F2F and to 6 for virtual. Exercise mats, straps, and class materials were provided before the first class. Participants were instructed to start daily home exercise the day after the second class, starting gradually if needed, and to record exercise practice in provided diaries. Participants were paid US$25 for the baseline visit and each of the classes and US$50 for each of the two outcome visits.
The aims of this study were to compare, in ambulatory adults with MS and self-reported spasticity, the effects of STC to those of ROM on the impact of spasticity, as measured by the MSSS, the severity of spasticity (NRS), functional walking tests, and other patient reported outcomes at 1 and 6 months after class completion. We also compared the effects of F2F and virtual delivery on all outcomes.
Measures
During the F2F phase, self-report questionnaires were completed in person or electronically using research electronic data capture (REDCap) survey 16 and walking tests were conducted by team members masked to allocation. During the virtual phase, questionnaires were completed via REDCap survey and walking tests not performed.
Medical history, demographics, disease course, disability level (patient determined disease step (PDDS)), 17 disease-modifying and spasticity medications, and other treatments used to manage spasticity were collected with questionnaires.
The primary outcome was the impact of spasticity after 1 month measured with the MSSS, an 88-item retrospective (past 2 weeks), self-report questionnaire answered on a 1 (not bothered at all) to 4 (extremely bothered) scale for each item, resulting in total scores ranging from 88 to 352. The MSSS total and its eight subscale scores are valid, reliable, and patient-focused measures of the impact of spasticity in PwMS.11,12 The MSSS takes about 10 minutes to complete.
Secondary outcomes were MSSS scores at 6 months, and, at 1 and 6 months, the NRS measuring the severity of spasticity, 13 the Multiple Sclerosis Walking Scale-12 (MSWS) measuring the impact of MS on walking, 18 the Modified Fatigue Impact Scale (MFIS) measuring the impact of fatigue, 19 the Pittsburgh Sleep Quality Index (PSQI) measuring sleep quality, 20 the Patient-Reported Outcomes Measurement Information System (PROMIS) Short Form 8a measuring emotional distress/depression, 21 the Multiple Sclerosis Impact Scale-29 (MSIS)measuring the impact of MS on day-to-day life, 22 the timed up and go (TUG) measuring timed walking and turning, 23 and the timed 25 foot walk (T25FW) measuring timed walking 24 taking 30–40 minutes to complete. Participants documented in daily diaries the number of minutes they spent each day doing the study program exercises. Diaries were provided in 6-week batches and returned when complete. If not received, participants were called and/or mailed reminders when the next batch was sent to them.
Statistical analyses
Power and sample size were derived from our pilot trial with 38 people of STC versus usual care (UC) with unguided use of the NMSS brochure in which the mean change in MSSS scores was −2% for UC and −14% for STC (standard deviation (SD) = 0.17 for both). We believed a three-fold improvement in change for STC versus UC (i.e. 12% decrease in the STC group vs 4% decrease in the UC group) would be clinically meaningful. We needed 100 participants/group (200 total) to achieve 80% power using a two-sided t-test at 5% significance, assuming the SD of the percentage change for all participants was 0.20. Allowing for 10% drop-out, our goal was to recruit 223 participants. The primary outcome, the MSSS total score 1 month following the interventions, was compared between groups using an independent t-test. Repeated measures analyses using linear mixed models assessed differences between interventions across time on all the secondary outcomes and provided supporting analyses for the primary outcome. To assess the impact of F2F versus virtual delivery, we used a mode of delivery by treatment interaction term in the repeated measures analyses. Descriptive statistics included frequencies and percentages for categorical data and sample size with an unstructured covariance matrix, means and SDs, and/or medians with minimum and maximum values for continuous data. Consistent with instrument guidelines, missing data were handled with mean substitution, except for the PSQI where the previous time point value was carried forward. We used a modified intent-to-treat (M-ITT) population of participants with at least 1 postbaseline measurement. Analyses were performed with Statistical Package for the Social Sciences (SPSS) v.27 using two-sided tests and α = 0.05 significance level.
All data are available from the authors.
Results
A total of 493 potential participants were approached and 232 consented. One did not complete the baseline assessment and was not randomized. Of the 231 enrolled and randomized, 86% were women, mean age was 54 (SD = 11.6) years, and median PDDS was 4 (range 0–6.0). There were no apparent differences in baseline characteristics between groups or when separated by the mode of delivery (Table 1). At baseline, 174 (75%) used MS disease-modifying therapies,147 (64%) used one or more prescription medications for spasticity, and 112 (48%) were stretching to manage spasticity (58 in STC and 54 in ROM).
Modified intent-to-treat baseline characteristics.
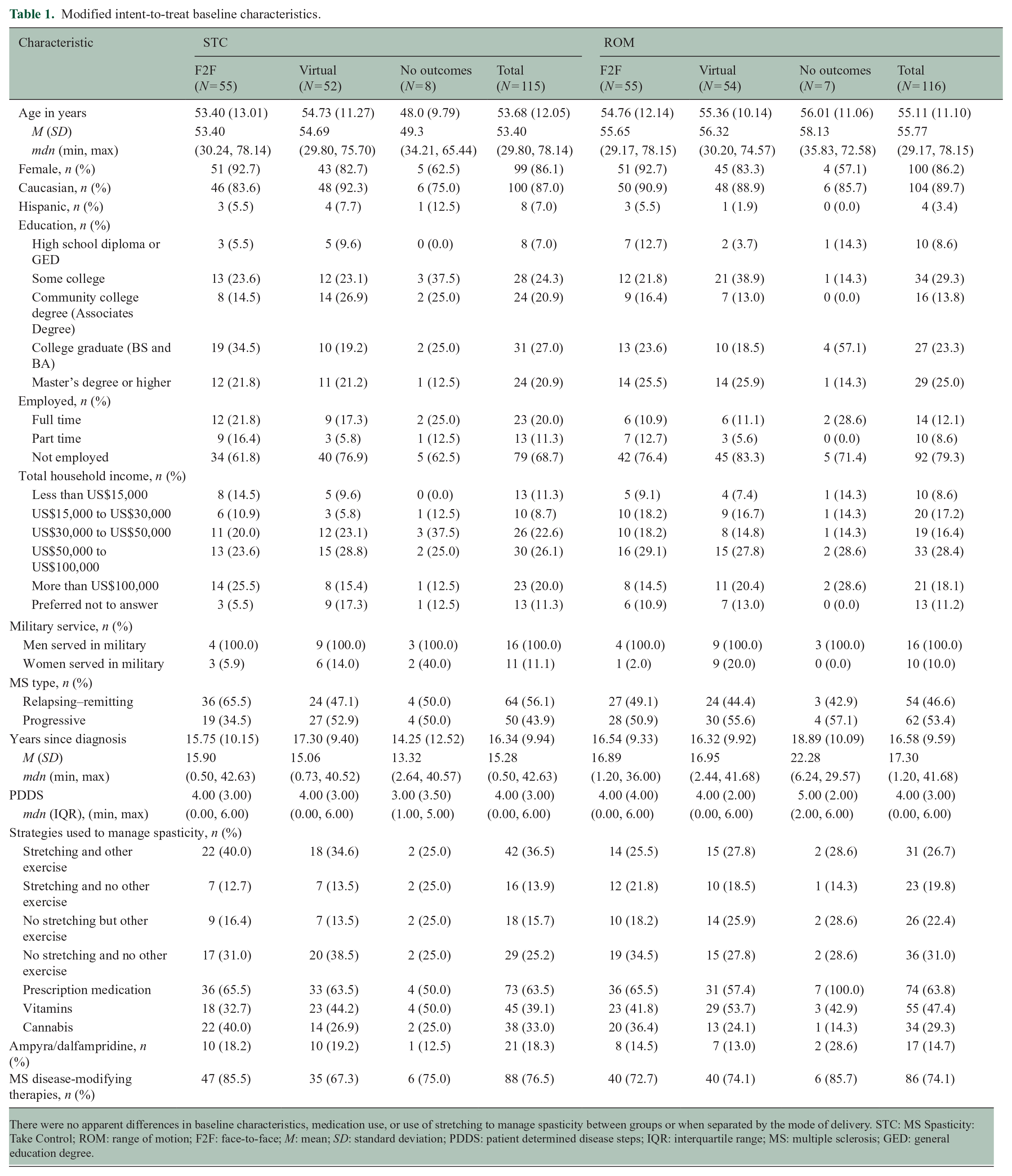
There were no apparent differences in baseline characteristics, medication use, or use of stretching to manage spasticity between groups or when separated by the mode of delivery. STC: MS Spasticity: Take Control; ROM: range of motion; F2F: face-to-face; M: mean; SD: standard deviation; PDDS: patient determined disease steps; IQR: interquartile range; MS: multiple sclerosis; GED: general education degree.
F2F class size ranged from 1–7 and virtual class size ranged from 1–6 (both median 4). A total of 214 (93%) of 231 people completed both classes. Class attendance, outcome completion (Figure 1), and exercise adherence were similar for both interventions and modes of delivery. Outcomes were completed by 210 (91%) at 1 month and 194 (84%) at 6 months. In the first month, the STC group performed the program exercises at least 15 minutes per day for a median of 16 days and the ROM group performed the program exercises at least 15 minutes per day for a median of 20 days; 6 in STC and 11 in ROM exercised at least 15 minutes every day; and 9 in STC and 4 in ROM never exercised at least 15 minutes. Over the entire 6 months, STC participants exercised for at least 15 minutes for a median of 55/182 days and the ROM participants exercised for at least 15 minutes for a median of 97/182 days.
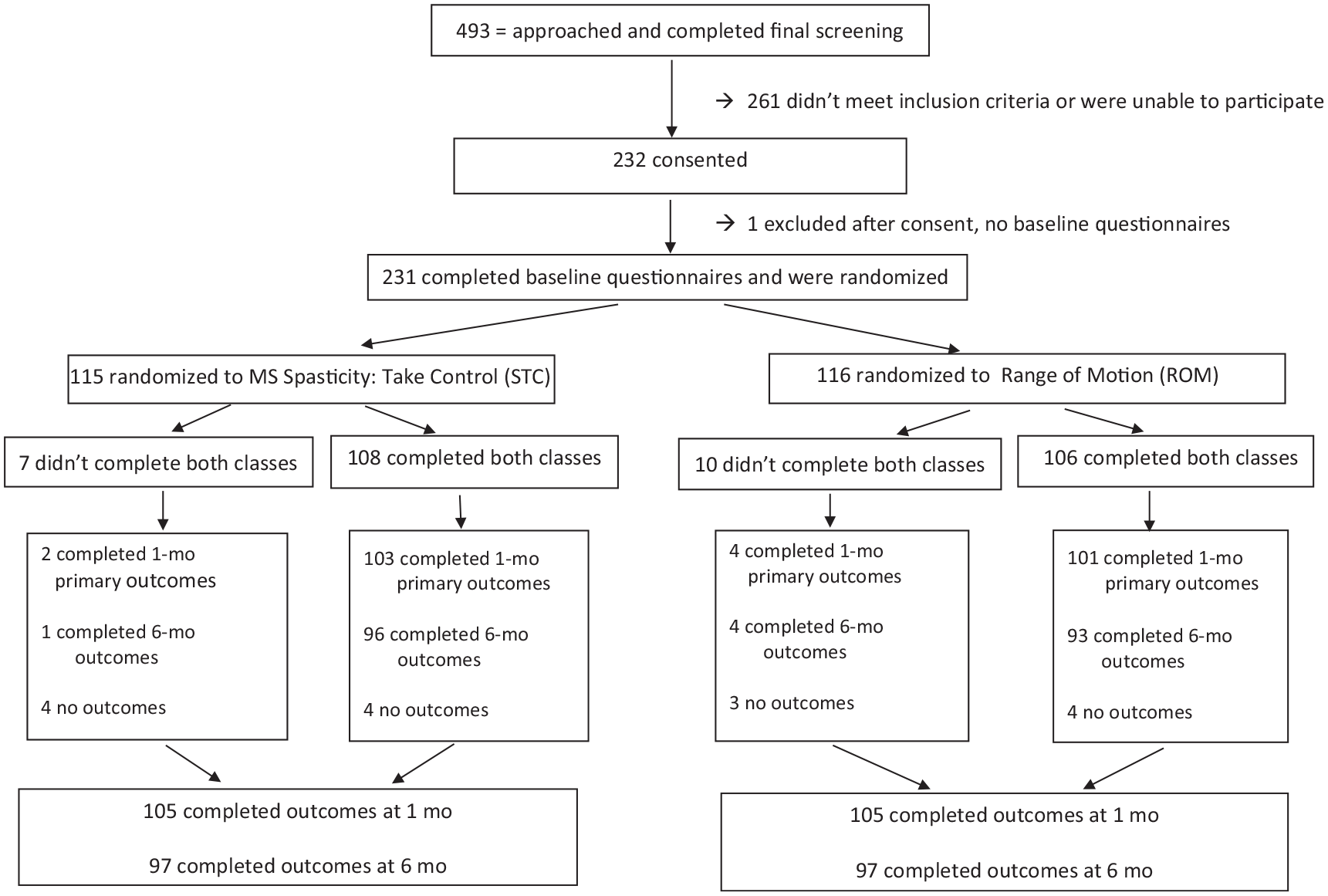
Consort diagram.
All statistical test assumptions were met, and results for intent-to-treat analyses were unchanged with per protocol analyses. There were no significant differences between the STC and ROM groups in MSSS total scores, MSSS subscale scores, or secondary outcomes at 1 month or 6 months (Tables 2 and 3). There were significant improvements in many outcomes over time (i.e. baseline to 1 month and baseline to 6 months) for the groups combined and individually (Table 4). The most consistent benefits were in the MSSS total scores, the MFIS total scores, and the MSIS psychological scores, which showed improvements from baseline to 1 month and to 6 months for the entire sample and for each group. In addition, there was significant improvement in MSSS total scores from 1 month to 6 months for the entire sample and for the ROM group alone but not for the STC group. The most consistent lack of benefit was for the TUG and T25FW which showed no statistically significant changes in means across any time interval, although these were only measured in the F2F cohort.
Modified intent-to-treat primary outcome measures.
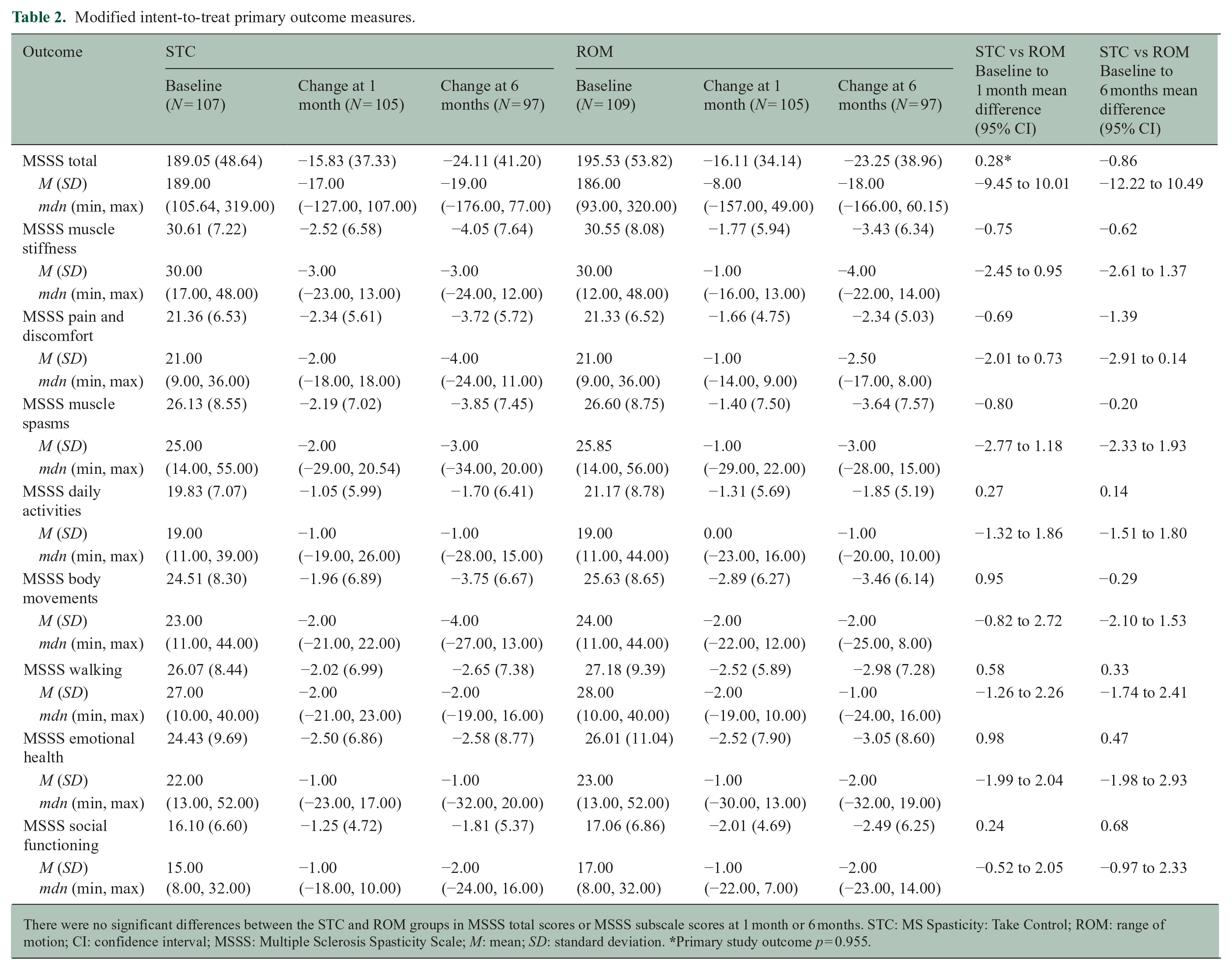
There were no significant differences between the STC and ROM groups in MSSS total scores or MSSS subscale scores at 1 month or 6 months. STC: MS Spasticity: Take Control; ROM: range of motion; CI: confidence interval; MSSS: Multiple Sclerosis Spasticity Scale; M: mean; SD: standard deviation.
Modified intent-to-treat secondary outcome measures.
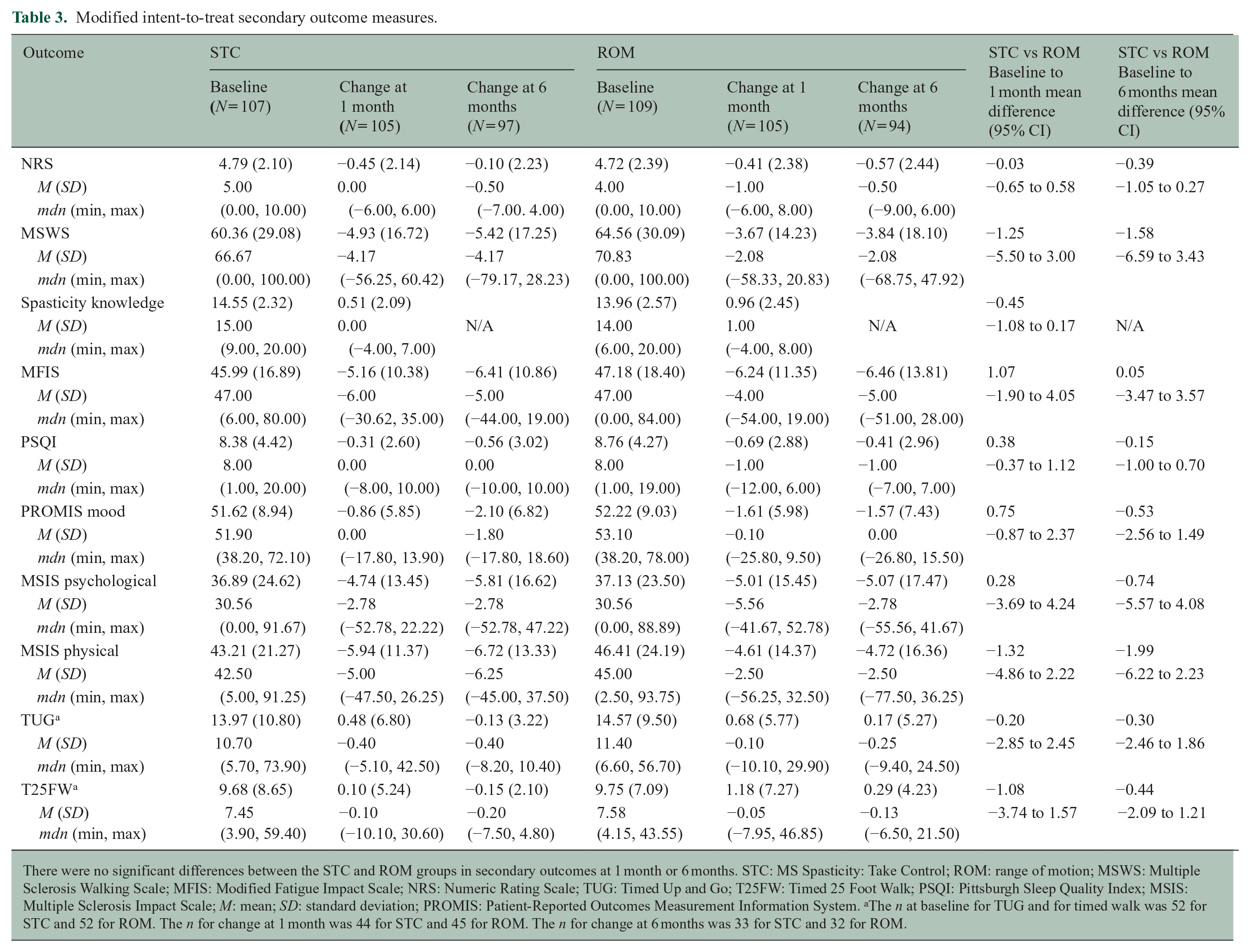
There were no significant differences between the STC and ROM groups in secondary outcomes at 1 month or 6 months. STC: MS Spasticity: Take Control; ROM: range of motion; MSWS: Multiple Sclerosis Walking Scale; MFIS: Modified Fatigue Impact Scale; NRS: Numeric Rating Scale; TUG: Timed Up and Go; T25FW: Timed 25 Foot Walk; PSQI: Pittsburgh Sleep Quality Index; MSIS: Multiple Sclerosis Impact Scale; M: mean; SD: standard deviation; PROMIS: Patient-Reported Outcomes Measurement Information System. aThe n at baseline for TUG and for timed walk was 52 for STC and 52 for ROM. The n for change at 1 month was 44 for STC and 45 for ROM. The n for change at 6 months was 33 for STC and 32 for ROM.
Different scores between STC and ROM over time with standard error and 95% confidence intervals.
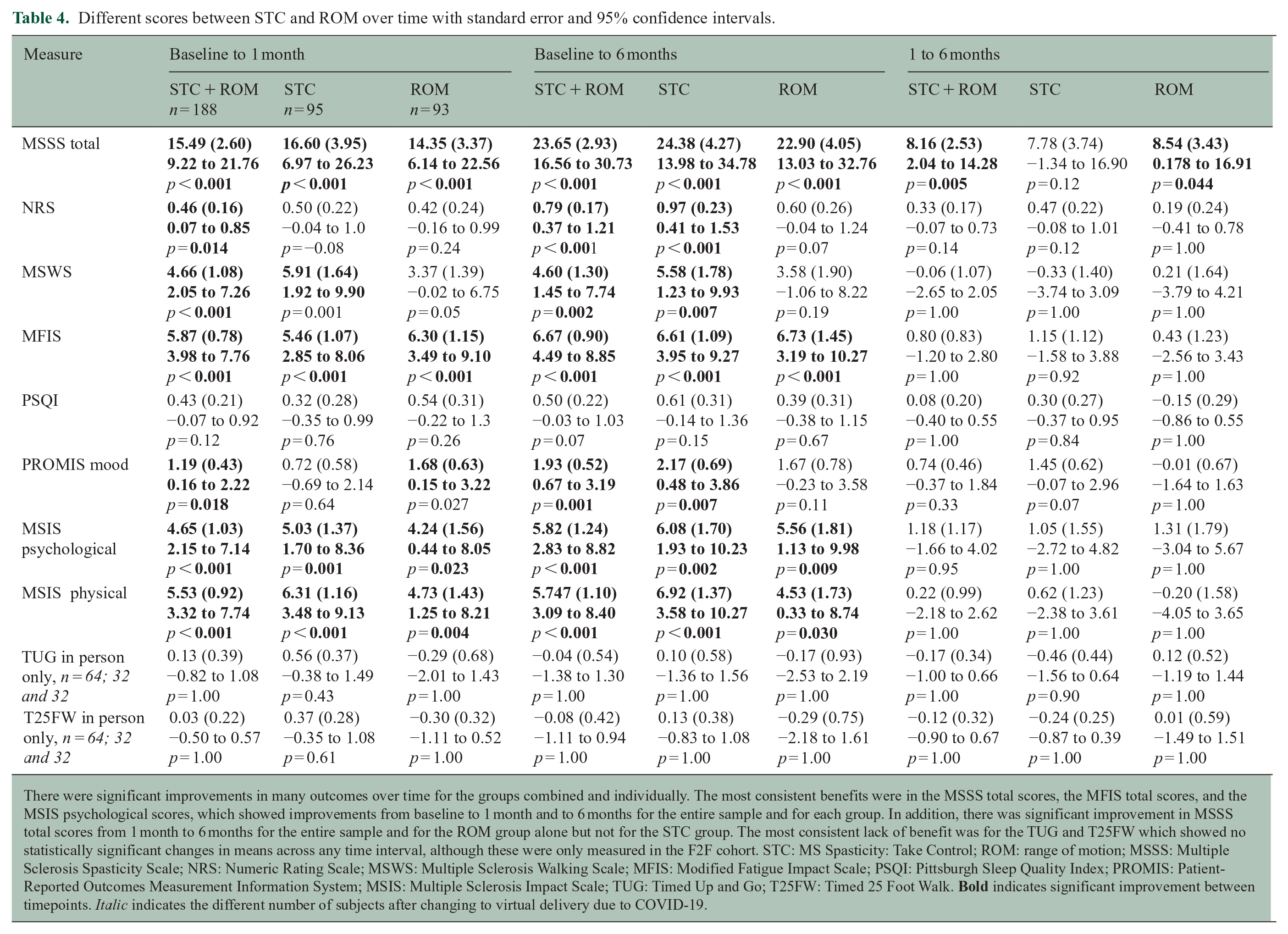
There were significant improvements in many outcomes over time for the groups combined and individually. The most consistent benefits were in the MSSS total scores, the MFIS total scores, and the MSIS psychological scores, which showed improvements from baseline to 1 month and to 6 months for the entire sample and for each group. In addition, there was significant improvement in MSSS total scores from 1 month to 6 months for the entire sample and for the ROM group alone but not for the STC group. The most consistent lack of benefit was for the TUG and T25FW which showed no statistically significant changes in means across any time interval, although these were only measured in the F2F cohort. STC: MS Spasticity: Take Control; ROM: range of motion; MSSS: Multiple Sclerosis Spasticity Scale; NRS: Numeric Rating Scale; MSWS: Multiple Sclerosis Walking Scale; MFIS: Modified Fatigue Impact Scale; PSQI: Pittsburgh Sleep Quality Index; PROMIS: Patient-Reported Outcomes Measurement Information System; MSIS: Multiple Sclerosis Impact Scale; TUG: Timed Up and Go; T25FW: Timed 25 Foot Walk.
There were no significant interactions between intervention group and mode of delivery. There were significant main effects of mode of delivery for some outcomes, but not in a consistent direction and effect sizes were small (Table 5).
Comparison of F2F and virtual differences in outcome measures.
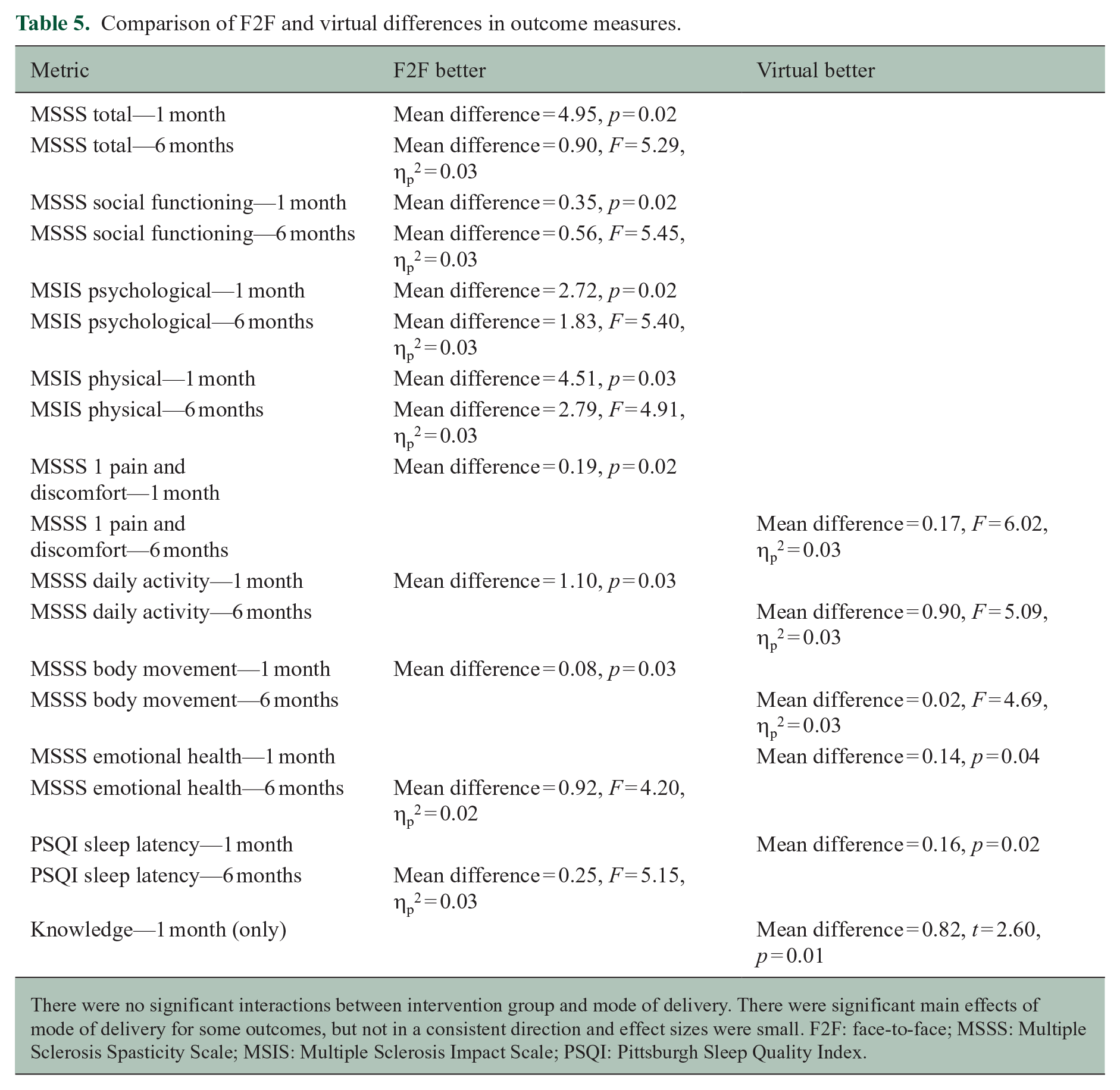
There were no significant interactions between intervention group and mode of delivery. There were significant main effects of mode of delivery for some outcomes, but not in a consistent direction and effect sizes were small. F2F: face-to-face; MSSS: Multiple Sclerosis Spasticity Scale; MSIS: Multiple Sclerosis Impact Scale; PSQI: Pittsburgh Sleep Quality Index.
Discussion
This fully powered study of education and exercise for management of MS spasticity found no statistically significant differences in the mean impact of spasticity, or other secondary outcomes, between an active stretching intervention and a control ROM exercise intervention. There were significant improvements in many outcomes with both interventions.
The lack of statistically significant differences in outcomes between groups in this fully powered RCT may be because the groups were too heterogeneous or because the interventions were too similar. Spasticity varies in severity, location, timing and triggers among individuals with MS. We included people with any self-reported spasticity in this trial. STC and ROM may have differential effects according to the characteristics of the person’s spasticity. In our pilot study, STC was associated with greater benefits than ROM, with STC delivered by a facilitator and ROM using a booklet without in-person facilitation. In the current trial to evaluate for specific effects of stretching, both STC and ROM were delivered by facilitators to control for time, attention, and daily exercise, and to maintain masking of study facilitators and participants. Our positive pilot results and negative results of this trial suggest attention and daily exercise may have greater impact on spasticity than the specific kind of exercise.
Our finding of significant improvements in most outcomes in both groups, individually and combined, with no difference between groups, supports either a nonspecific placebo effect or specific benefits from both interventions that involved education and daily exercise. Although nonspecific placebo effects are ubiquitous 25 and may be more pronounced for self-reported outcomes such as spasticity and pain,25,26 there are also reasons to wonder whether both stretching and ROM exercises help PwMS spasticity. Both involve actively moving involved muscles through available ROM. Whether the addition of tension to move into passive range involved in stretching is important is unknown. A number of studies suggest that movement through active range alone may reduce spasticity. For example, in a study in PwMS, 4 weeks of unloaded leg cycling was associated with significant improvements in spasticity. 27 In a survey of more than 1000 people with spinal cord injury (SCI), 48% reported stretching improved their spasticity and 45% reported exercise improved their spasticity. 9 A small clinical trial in people with SCI spasticity found passive movement through available range reduced spasticity as effectively as stretching. 28
Our results are similar to studies of other group single and multicomponent rehabilitation interventions for MS symptoms. The interventions demonstrate better outcomes when compared with waitlist, UC, or no control intervention, but demonstrate similar benefits to robust active control interventions.29–32 These findings are important. They demonstrate the acceptability, feasibility, and improvements over time with rehabilitation interventions. They also demonstrate the importance of selecting an appropriate control condition to answer a specific research question. For our study, the research question was, “Is an evidence-based program of stretching more effective than UC ROM exercise for reducing the impact of spasticity in PwMS?” Our study does not address the questions, “Are stretching or range of motion exercises effective for reducing spasticity?” or “Is stretching better than no exercise for reducing spasticity?”
Although we found no statistically significant differences in outcomes between our interventions, in post hoc analyses we found small but significant differences between F2F and virtual delivery for some outcomes. Although these results do not show one to be superior, they do suggest that clinicians and researchers should not assume them to be equivalent. Differences may be due to changes in participants due to recruitment, intervention delivery, or outcome measurement.
This study had a number of strengths. It was fully powered and fully enrolled with more than 200 ambulatory subjects with MS and self-reported spasticity, and 91% of them completed the primary outcome. All participants received an intervention with comparable time and attention and expectations of exercising for at least 15 minutes daily for the following 6 months. Information on exercise adherence with the number of minutes spent doing the exercises each day for the 6 months of follow-up and secondary outcomes related to activities and quality of life affected by spasticity were collected and analyzed.
The study also had limitations. We used self-reported spasticity, rather than clinician or instrument-based measures of spasticity, as the spasticity-related entry criterion and primary and secondary outcome. Self-reported spasticity measures have high precision and capture patients’ lived experiences of spasticity but do not provide information about potential mechanisms that could be derived from objective measures such as ROM or electromyography.33,34 As with any study, there may have been confounding from unmeasured factors (e.g. botulinum toxin injections, massage, and acupuncture) but these should be controlled by randomization. Generalizability of study results may be limited. Only people who could walk were included. The study was carried out in a region of the United States with low racial and ethnic diversity, used few facilitators, and was not powered to compare effects between facilitators. This study does not provide information on effects of either intervention beyond 6 months. Comparison between F2F and virtual delivery was not preplanned, although similar numbers of people participated by each mode of delivery.
Conclusion
This fully powered, double-blind, RCT found that a program of education and daily stretching exercises was not more effective than a program of education and daily ROM exercises for decreasing the impact of MS spasticity. This negative result debunks the long-held belief that stretching is essential to managing MS spasticity. Although there were improvements in the primary and secondary outcomes with both groups, there were no significant differences between groups in any outcomes at any time point. Given the tolerability and feasibility of the interventions, patients may continue stretching or ROM exercise but further research is needed to determine whether the improvements seen with both interventions in this study are specifically related to the interventions, differ by the characteristics of the individual’s spasticity, or reflect nonspecific placebo effects.
Footnotes
Acknowledgements
The authors would like to thank Lois Copperman, PhD, OTR, the Paralyzed Veterans of America, and Medtronic for assistance and support developing STC; our study coordinators and program facilitators ShienPei Silverman, Julia Norton, Nikita Sajeev, Sophie Samiee, Melissa Frederick, and Hannah Flegel, and all our patients and PwMS who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the Veterans Affairs Office of Research and Development to conduct this study.
