Abstract
The majority of patients with multiple sclerosis (MS) have symptoms of spasticity that increasingly impair function as the disease progresses. With appropriate treatment, however, quality of life can be improved. Oral antispasticity medications are useful in managing mild spasticity but are frequently ineffective in controlling moderate to severe spasticity, because patients often cannot tolerate the adverse effects of increasing doses. Intrathecal baclofen (ITB) therapy can be an effective alternative to oral medications in patients who have a suboptimal response to oral medications or who cannot tolerate dose escalation or multidrug oral regimens. ITB therapy may be underutilized in the MS population because clinicians (a) are more focused on disease-modifying therapies rather than symptom control, (b) underestimate the impact of spasticity on quality of life, and (c) have concerns about the cost and safety of ITB therapy. Delivery of ITB therapy requires expertly trained staff and proper facilities for pump management. This article summarizes the findings and recommendations of an expert panel on the use of ITB therapy in the MS population and the role of the physician and comprehensive care team in patient selection, screening, and management.
Keywords
Introduction
Spasticity is a frequent symptom of multiple sclerosis (MS) that increasingly impairs function and quality of life (QoL) as the disease progresses. 1 – 3 Spasticity typically is described as muscle stiffness, tightening, or spasm with associated involuntary jerking, pain, and weakness. It is a complex problem that can lead to significant disability and secondary complications such as decubitus ulcers and contractures. Therapeutic options include oral medications, but these have limited effectiveness and often cause side effects that limit dose titration. 2 Intrathecal baclofen (ITB) was approved by the US Food and Drug Administration (FDA) for treatment of severe spasticity in 1992. In carefully selected patients, ITB can reduce spasticity and improve patient function and comfort with fewer side effects than oral medications. 4,5
In 2008 and 2009 a group of clinicians with expertise in the care and management of patients with MS met to discuss the role of ITB as a treatment option for spasticity in MS. This expert panel included neurologists, physiatrists, a neurosurgeon, and a physical therapist from centers in the United States and Europe. Together the panelists have more than 100 years of clinical experience in the use of ITB for spasticity management in MS and have used ITB in the treatment of more than 1500 patients. This article reviews the literature on ITB therapy for spasticity in MS and provides the expert opinion of the panel regarding patient selection and management of ITB therapy to achieve optimal outcomes in MS patients.
Magnitude of the problem
In a recent survey of the North American Research Committee on Multiple Sclerosis (NARCOMS), 84% of 18,727 patients with MS reported at least some symptoms of spasticity, and 30% reported moderate to severe symptoms. 3 Only 38% were taking medications to control their spasticity symptoms, and one-third of respondents reported modifying or discontinuing daily activities because of spasticity. (In contrast, nearly 60% of the respondents were receiving disease-modifying therapy with immunologic drugs. 3 ) Inadequately controlled spasticity has a profound impact on QoL in patients with MS. Increasing severity of spasticity and related disability impacts patients’ mobility, sleep, sexual function, energy level, hygiene, employment, mood, and social function. 2,6 Spasticity also contributes to the development of decubitus ulcers in nonambulatory patients. 7,8 The severity of the consequences of inadequately treated spasticity underscores the urgency of closing the gap between treatment of underlying disease and treatment of spasticity in MS patients.
What is spasticity?
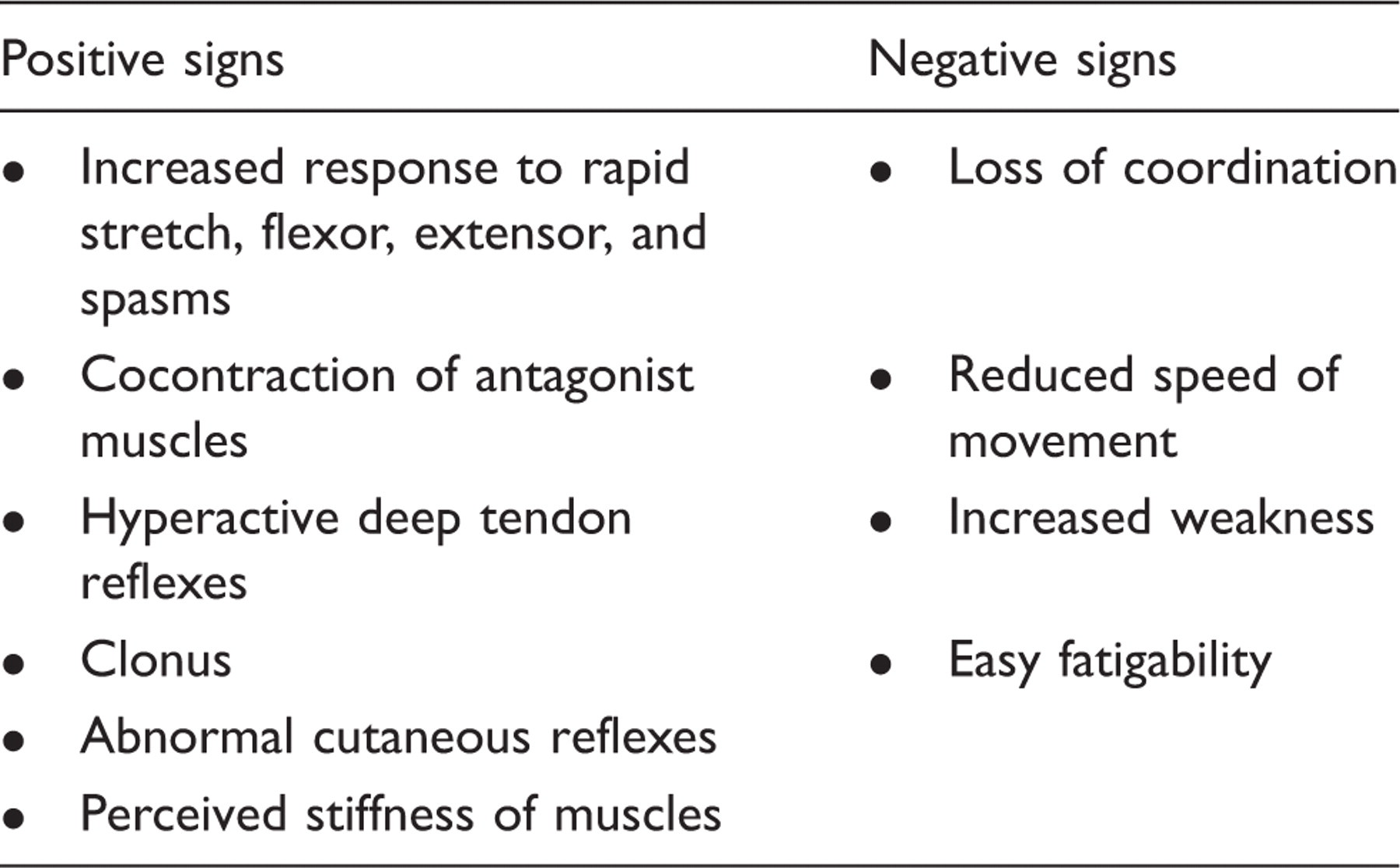
Positive and negative signs of spasticity 2
Modified Ashworth Scale 10
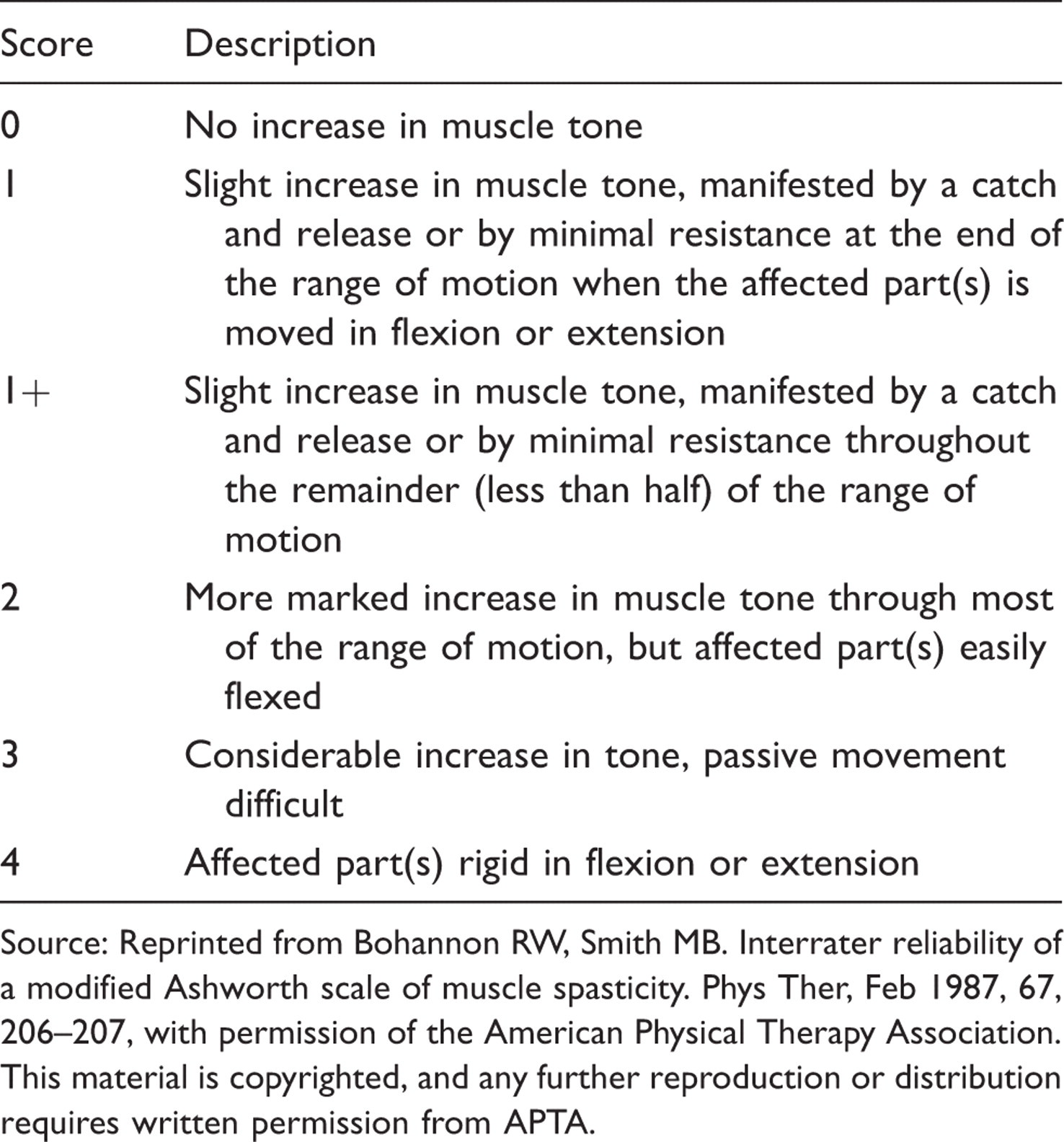
Source: Reprinted from Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther, Feb 1987, 67, 206–207, with permission of the American Physical Therapy Association. This material is copyrighted, and any further reproduction or distribution requires written permission from APTA.
Spasm Frequency Scale 7
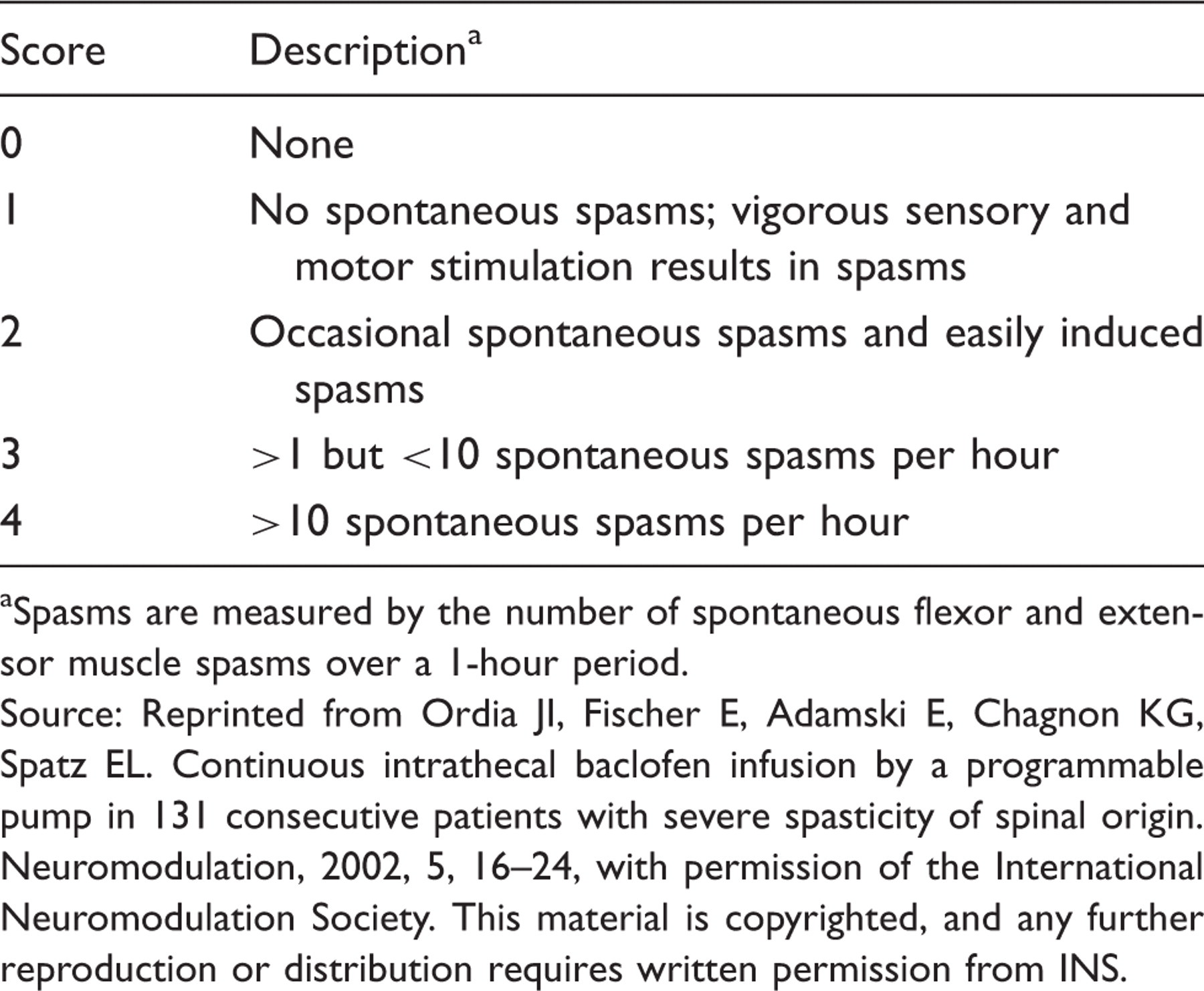
Spasms are measured by the number of spontaneous flexor and extensor muscle spasms over a 1-hour period.
Source: Reprinted from Ordia JI, Fischer E, Adamski E, Chagnon KG, Spatz EL. Continuous intrathecal baclofen infusion by a programmable pump in 131 consecutive patients with severe spasticity of spinal origin. Neuromodulation, 2002, 5, 16–24, with permission of the International Neuromodulation Society. This material is copyrighted, and any further reproduction or distribution requires written permission from INS.
In a more subjective sense, spasticity also includes patient reports of their experience of symptoms, such as pain, muscle cramping, and stiffness or tightness of the limbs. Data related to spasticity and its prevalence often is drawn from information collected directly from patients rather than from physicians or other clinical evaluators. When spasticity is reported in these subjective terms, symptoms become a surrogate indicator of the pathologic processes occurring in MS. 2
Management of spasticity
Lifestyle modifications (e.g., stretching, exercise, use of appropriate assistive devices) can and should be initiated early since they will be more difficult to implement when spasticity and overall disability are severe. Local treatments, particularly botulinum toxin injections, have been used to treat focal spasticity. However, this paper is primarily focused on more generalized spasticity.
Oral medications
Oral medications for spasticity include baclofen, tizanidine, dantrolene, and benzodiazepines. A systematic review of clinical trials involving these four medications revealed ‘limited evidence of [their] effectiveness’, although they were ‘approximately equally effective at reducing spasticity when assessed clinically’. 11 A Cochrane review of the same four medications did not yield enough definitive evidence of effectiveness for the authors to make any recommendations about prescribing them. 12 In its clinical practice guidelines for spasticity management in MS, the Consortium of Multiple Sclerosis Centers (CMSC) recommends initial therapy with oral baclofen or tizanidine but notes the need for careful dose titration to obtain the desired effect without unacceptable side effects. 13
Oral spasticity medications are associated with high rates of side effects. According to the FDA, drowsiness is reported by up to 63% of patients receiving oral baclofen, and dizziness occurs in 5% to 15%. 14 Similar side effects are reported for tizanidine and dantrolene, which also require monitoring of liver transaminase levels. 13 These and other side effects can interfere with the patient’s activities of daily life, causing the patient to stop taking oral spasticity medications or to scale back their use such that the spasticity remains poorly controlled. Consequently, a large population of patients with MS have suboptimally controlled spasticity and may benefit from alternative therapies.
Intrathecal baclofen
ITB therapy is administered through an implanted programmable pump with a reservoir and catheter that delivers baclofen directly into the thecal sac. 2,13 Baclofen reduces spasticity and spasms by binding to gamma-aminobutyric acid (GABAB) receptors and inhibiting the release of excitatory neurotransmitters, thereby inhibiting monosynaptic and polysynaptic spinal reflexes. 15 Baclofen is not taken up into the brain tissue GABA system, and very little reaches the fourth ventricle to have any effect on respiratory centers. 15 Instead, ITB has a ‘selective effect on spinal neurons, which control reflex excitation of motor neurons, sparing other sensory and motor input’. 15 The doses required for effective treatment of spasticity are much lower than the oral baclofen doses typically used, thereby reducing systemic side effects.
Barriers to use of ITB
In the NARCOMS survey, only 1% of patients receiving treatment for spasticity had an implanted pump for delivery of ITB, compared with the estimated 13% of all patients with MS who may be potential candidates for ITB. 3 Among a subgroup of patients in the NARCOMS registry, only 25% of users of oral spasticity medications reported that their physicians had discussed ITB therapy with them. 3 Thus, the principal barrier to the use of ITB therapy in MS patients appears to be the failure of physicians to present ITB as an effective and well-tolerated therapeutic option to many patients who would be suitable candidates.
Due to the increasing complexity of MS management, physicians sometimes focus on disease-modifying therapies without taking the additional step of providing adequate symptom control. Some may not recognize the impact spasticity has on QoL; others remain unaware of the potential benefits of ITB or are unsure how to select appropriate patients. In addition, fewer than half of all MS centers have comprehensive spasticity management programs. 9 Finally, misperceptions related to the safety and cost of ITB therapy may be barriers to its use.
Patient selection
Criteria for patient selection are described in the prescribing information for ITB and have been further delineated by the CMSC. 13,16 The goal is to identify patients suffering from the effects of spasticity despite standard therapies who are likely to experience improvement with a reduction in side effects as a result of ITB therapy. Careful assessment of disease and spasticity status at baseline is essential to monitor and optimize the level of muscle tone according to patients’ changing needs.
The CMSC spasticity treatment guidelines, which are consistent with patient selection criteria for many clinical trials, 7,8,17 – 19 recommend that patients with unresolved spasticity receive health promotion strategies such as range-of-motion exercise, stretching, and strengthening programs in combination with oral medications trialed sequentially. If oral medications are ineffective or poorly tolerated, patients should be considered for ITB therapy. 13 Some studies also specify that potential ITB candidates should have Ashworth/modified Ashworth scores ≥3 or spasm frequency scores >2. 8,19
Disability status in MS is most commonly assessed using the Expanded Disability Status Scale (EDSS), a 10-point global scale with half-point increments. 20 Low scores indicate no or minimal disability. Higher scores indicate increasing disability (10 = death due to MS). Above 4.0 (fully ambulatory without aid despite relatively severe disability), the EDSS rating is based mostly on mobility limitations. Patients with MS and spasticity can be categorized into three levels of mobility impairment: ambulatory (EDSS ≤ 5.5), ambulatory with assistance (EDSS 6.0 to 7.0), and nonambulatory (EDSS 7.0 to 9.5). The decision to offer ITB therapy to patients in these three groups will be based on very different goals. In their Cochrane Database review, Shakespeare et al. 12 note, ‘The immobile patient is treated for symptomatic relief (pain and spasms) and in order to make nursing care and seating easier, whereas the ambulant patient is treated with the additional aim of improving or preserving mobility.’ Patients who are ambulatory with assistance fall into a middle category. They may rely on extensor rigidity to remain upright and walk, requiring a weighted comparison of the benefit of reduced spasticity via ITB therapy with the possibility that reduced tone could impair ambulation.
Screening patients for their ability and willingness to return to clinic on a regular basis is an important part of determining their suitability as candidates for ITB therapy. Patients who do not return to clinic regularly for pump refills and adjustments are at greater risk for baclofen withdrawal and other complications of being unmonitored by the care team.
Screening trial and implantation
Before implantation of an ITB pump, a screening trial is recommended. The purpose of the trial is to determine whether ITB will be effective for the patient and to assess for any adverse reactions that would preclude its use. The trial, which can be done in an outpatient setting, typically involves intrathecal administration of a bolus test dose of baclofen by lumbar puncture, followed by observation of its effects. The FDA-approved test dose is 50 µg (25 µg for small pediatric patients), 16 but in clinical practice the dose may range from 25 to 100 µg. Cessation of oral spasticity medications is unnecessary during the screening trial, although patients may reduce or skip the morning dose on the day of the trial. 4,7,8,19
A decrease in one to two points in the modified Ashworth score and/or a decrease of at least two points in the spasm frequency score constitutes a positive response to the screening trial. 19,21 Because the test dose is not individualized or titrated, patients may experience a temporary decrease in function when their spasticity is suddenly removed. 2 Patients should be informed that the magnitude of response to the test dose does not reflect the level of change in tone they can expect once the pump is implanted and adjusted.
If the screening trial achieves spasticity or spasm reduction and appropriate goals are identified by the patient and treatment team, the patient may receive an implanted pump for delivery of baclofen by continuous intrathecal infusion. Implantation involves subcutaneous placement of a refillable pump. The pump contains a reservoir of baclofen, which runs through a catheter connected to the pump and into the subarachnoid space. The medication is delivered according to a programmable schedule, which should be adjusted incrementally over time to achieve the desired result. Pump refills depend on the daily dosage and concentration of baclofen used and on average are required every three to six months. 2,22
Treatment outcomes
A 2003 systematic review of treatments for spasticity in MS identified 15 studies of ITB in which at least 50% of participants had MS. 11 All participants had severe spasticity that was unresponsive to oral therapy. Ashworth scores were universally lowered by two to three points, and significant improvements were noted on other scales as well, most notably the Spasm Frequency Scale. Although there were few double-blind randomized controlled trials, the reviewers concluded that ‘there is evidence that intrathecal baclofen is effective in reducing spasticity in terms of both muscle tone and frequency of spasms’. 11
In a study of 131 patients with severe spasticity, including 63 patients with spinal or supraspinal MS, the mean Ashworth score dropped from a pre-implant score of 4.2 to 1.3 (p < 0.0005) after ITB therapy was implemented. 7 Reduced spasticity led to decreased pain, improvements in sleep, and increased physical activity. Eighteen ambulatory patients improved in their gait and balance.
In a more recent study of 30 patients with severe spasticity (22 with MS), clinical assessments conducted three days after pump implantation showed a drop in mean Ashworth score from 4.3 to 1.2 (p < 0.0005), reduced pain as measured by visual analogue scale (6.2 vs. 3.3, p < 0.005), and improved QoL (2 vs. 4 on a scale of 1 to 5, where 5 = best condition; p < 0.01). 8
The participants included in the 2003 systematic review by Beard et al. 11 were mostly nonambulatory. However, all 36 patients (27 with MS) studied by Sadiq and Wang 17 were ambulatory (with or without an assistive device) and had lower extremity muscle strength of at least 3 out of 5, with modified Ashworth scores of at least 3. All 36 patients retained ambulatory function as ITB was titrated for relief of spasticity, with improvement of an average of 1.5 points in Ashworth scores. Nine patients (25%) not only remained ambulatory but showed improvement in ambulatory function for two years or more of follow-up. 17 This improvement was attributed in part to an aggressive physical therapy program that accompanied the initiation of ITB, first for range of motion and safety postoperatively, then for optimal ambulatory function during the titration period. The authors concluded that ‘by physiologically decreasing the restriction of intractable spasticity, timely intervention with ITB therapy may result in maintenance of long-term ambulation’. 17
A 2004 retrospective chart review of ITB over a period of 10 years also described positive outcomes for ambulatory patients. 21 Among 50 subjects (19 with MS), 78% had achieved long-term goals such as elimination of the need for wheelchair, returning to work, or ambulating in the home without an assistive device. 21
Current data indicate that ITB therapy effectively and significantly reduces severe spasticity in ambulatory and nonambulatory patients with MS. Several factors contribute to successful use of ITB, including appropriate patient selection, education about realistic goals and expectations, and careful dose titration over time. Early involvement of rehabilitation therapists also helps to maximize patients’ clinical response to ITB therapy.
Safety and complication rate
The low systemic exposure to baclofen after intrathecal administration reduces the risk of side effects because plasma drug concentrations are reduced more than 100-fold. The 63% rate of drowsiness with oral baclofen is reduced to 21% in patients receiving ITB, and dizziness is reported in only 8%. The most prominent side effect is hypotonia, which occurs in 34% of patients. 16 This effect can be addressed in most cases by adjusting the rate of administration, assuming the hypotonia represents overshoot of the desired effect of alleviating spasticity. Other common side effects are headache and nausea/vomiting. 16
Device-related complications reported between August 2003 and October 2008 23

Most of the pump-related events (32 of 38) were reported with the discontinued SynchroMed EL pump. The SynchroMed II 20 mL and 40 mL are currently marketed (Medtronic, Inc, Minneapolis, MN, USA).
Although complications are rare when best practices are followed, clinicians should be prepared to recognize and treat them in a timely manner. Abrupt cessation of ITB can result in high fever, drowsiness and/or coma, return of spasticity, muscle rigidity, and in rare cases death. 16 Pump and catheter complications can result in baclofen overdose or underdose and may require surgery to correct. An acute massive overdose can cause coma; less severe overdoses can cause drowsiness, lightheadedness/dizziness, respiratory depression, seizures, rostral progression of hypotonia, and loss of consciousness. 16 Patients and caregivers should be educated to recognize potential symptoms and signs of complications and to contact their provider or seek urgent care appropriately. Providing patients and caregivers with emergency contact numbers is essential in case problems occur after hours or when patients are first treated.
Cost considerations
The 2003 systematic review 11 included five evaluations of the comprehensive cost of ITB therapy. These evaluations included mostly bed-bound patients with severe and disabling spasticity. The authors noted that despite the expense associated with initiation of ITB, its use ‘may be associated with significant savings in hospitalization costs’. 11 Cost savings occurred in several areas. First, pressure sores and ulcers were less common in patients receiving ITB, reducing costs in wound care and treatment, particularly in terms of hospitalization days and associated costs. Second, orthopedic surgery for muscle contractures and hip dislocations could be deferred when spasticity was more effectively managed. Third, the need for oral spasticity medications was reduced or eliminated in most patients receiving ITB. Fourth, improved spasticity control led to decreased costs for adaptive devices, orthotics, wheelchairs and other seating aids, and modifications of the home. Finally, the intensity of care required for patients with severe spasticity decreased in patients receiving ITB, thereby reducing caregiver and nursing home costs. 11 Similarly, a French study reported cost savings with ITB in patients severely disabled by spasticity, including those with MS. 24 Cost studies specific to ambulatory patients receiving ITB are not yet available.
Optimizing long-term therapy
ITB use extends beyond implementation of therapy to long-term management considerations. The expertise of a qualified surgeon is needed for pump implantation to ensure as low a complication rate as possible, as well as prompt follow-up for any problems that do arise. 25 A neurologist, physiatrist, or clinician trained in ITB must provide initial assessment of and counseling for potential candidates, ongoing monitoring of clinical status, incremental dose adjustments, and other needs. Nurses and nurse practitioners can provide long-term continuity of care in adjustment of pump settings and provide support for caregivers and patients when questions or concerns arise and when potential emergencies related to withdrawal or overdose occur. Rehabilitation therapists help to maximize the effect of ITB through training exercises, which improve functional status and ease of care. The availability of a well-trained, well-equipped, and responsive team of specialists will positively impact the effectiveness of ITB therapy as well as patient satisfaction with ITB for spasticity control. 18,21,22
Conclusions
Inadequately controlled spasticity has a profound impact on QoL in patients with MS. A broad range of patients with MS with spasticity at many disability levels can benefit from ITB therapy. Too often a gap in care is created when physicians utilize disease-modifying therapies but neglect symptom control. Oral medications do not fill this gap for all patients because they may be ineffective or have side effects that prevent further dose increases. Although ITB has been an FDA-approved therapy for severe spasticity since 1992, with wide availability and proven effectiveness, it remains underutilized.
ITB therapy has demonstrated benefit and should be evaluated as an option for patients with spasticity at all stages of MS who are intolerant of or unresponsive to oral therapies. Referral to a specialist with knowledge of options beyond oral medications should occur early in the disease process, preferably when the EDSS score approaches 4.0 rather than 6.5 and above, which is the more common clinical practice. Patient and caregiver education about options for spasticity management, including a clear understanding of appropriate treatment goals and realistic expectations, is a critical part of the clinician’s role in managing patients with MS.
More studies are needed to identify the functional gains, to address potential misconceptions, and to increase clinician awareness of what can be achieved in the MS population with ITB therapy. Additionally, good communication among members of the care team and patients and their caregivers is essential to achieve optimal therapeutic outcomes and satisfaction with ITB therapy.
Footnotes
Acknowledgments
Editorial assistance was provided by Colleen Gilbert, PharmD, and Julie A Linick, ELS, from CommGeniX, LLC, Tampa, FL, USA.
Funding
This work was supported by Medtronic, Inc., Minneapolis, MN, USA.
Conflict of interest statement
AE: no relationships to disclose. MG: Medtronic, Inc.: consultant. FB: Medtronic, Inc.: speakers’ bureau, consultant, clinical advisory board, and research support; Allergan: speakers’ bureau and clinical advisory board; IMPAX Laboratories: consultant. SEB: Medtronic, Inc.: speakers’ bureau; Teva Neuroscience, Inc.: speakers’ bureau; Acorda Therapeutics Inc.: speakers’ bureau and advisory board. SK: Allergan and Medtronic, Inc.: speakers’ bureau. RP: no relationships to disclose. SS: Medtronic, Inc.: advisory board. VLS: Bayer Healthcare and Medtronic, Inc.: advisory boards. AMT: Medtronic, Inc.: advisory board. CT: Biogen ldec and Teva Neuroscience: speakers’ bureau and consultant; Medtronic, Inc.: advisory board. MZ: Teva Neuroscience and Medtronic, Inc.: advisory board; Biogen Dompè: consultant. MH: Medtronic, Inc.: advisory board.
