Abstract
Background:
Symptom management remains a challenging clinical aspect of MS.
Objective:
To design a performance improvement continuing medical education (PI CME) activity for better clinical management of multiple sclerosis (MS)-related depression, fatigue, mobility impairment/falls, and spasticity.
Methods:
Ten volunteer MS centers participated in a three-stage PI CME model: A) baseline assessment; B) practice improvement CME intervention; C) reassessment. Expert faculty developed performance measures and activity intervention tools. Designated MS center champions reviewed patient charts and entered data into an online database. Stage C data were collected eight weeks after implementation of the intervention and compared with Stage A baseline data to measure change in performance.
Results:
Aggregate data from the 10 participating MS centers (405 patient charts) revealed performance improvements in the assessment of all four MS-related symptoms. Statistically significant improvements were found in the documented assessment of mobility impairment/falls (p=0.003) and spasticity (p<0.001). For documentation of care plans, statistically significant improvements were reported for fatigue (p=0.007) and mobility impairment/falls (p=0.040); non-significant changes were noted for depression and spasticity.
Conclusions:
Our PI CME interventions demonstrated performance improvement in the management of MS-related symptoms. This PI CME model (available at www.achlpicme.org/ms/toolkit) offers a new perspective on enhancing symptom management in patients with MS.
Introduction
The severity of disease and disability-related symptoms varies widely among individuals with multiple sclerosis (MS) and treatment plans are often highly personalized.1-4 In addition to non-pharmacological symptom management such as rehabilitation, several MS-related symptoms may also be improved with appropriate drug treatment. While many pharmacologic therapies used to alleviate symptoms of MS are approved to treat those individual symptoms, few agents for symptomatic relief have been studied specifically in persons with MS. 5 Many of these therapies come with their own associated sets of benefits and risks. Compounding this issue, symptoms of MS cross multiple disciplines of medicine and many clinicians do not have the experiential foundation or evidentiary awareness to assess and implement improvement strategies in many of these areas. Thus, management of MS-associated symptoms represents one of the most challenging clinical aspects of the disease.
Although the recognition and treatment of symptoms in patients with MS present special challenges, a systematic effort in care may result in positive outcomes. A performance improvement continuing medical education (PI CME) activity was designed to enhance the management of disease-related symptoms in persons with MS through the joint sponsorship of Purdue University College of Pharmacy, PeerView Institute for Medical Education (PVI), and The Academy for Continued Healthcare Learning (ACHL; an accredited medical education provider). The PI CME activity was funded by an educational grant provided by Acorda Therapeutics, Inc.
This article describes the ways PI CME allows MS clinicians to retrospectively assess their current clinical practice, select areas for improvement, implement interventions based on treatment guidelines and healthcare standards, and then re-evaluate their clinical practice.
PI CME design
The PI CME model used for this activity was based on the American Medical Association approved learning formats and other well-established models of performance and quality improvement from other healthcare domains6-8 and consisted in three stages: A) learning from baseline assessment; B) intervention and action plan implementation; and C) learning from reassessment (Figure 1). In this PI CME activity, aggregate data and performance were compiled for each participating MS center rather than individual clinicians. This iterative process is consistent with the Institute for Healthcare Improvement paradigm for rapid improvement of clinical practices and expert consensus recommendations to improve symptom management in persons with MS.9,10
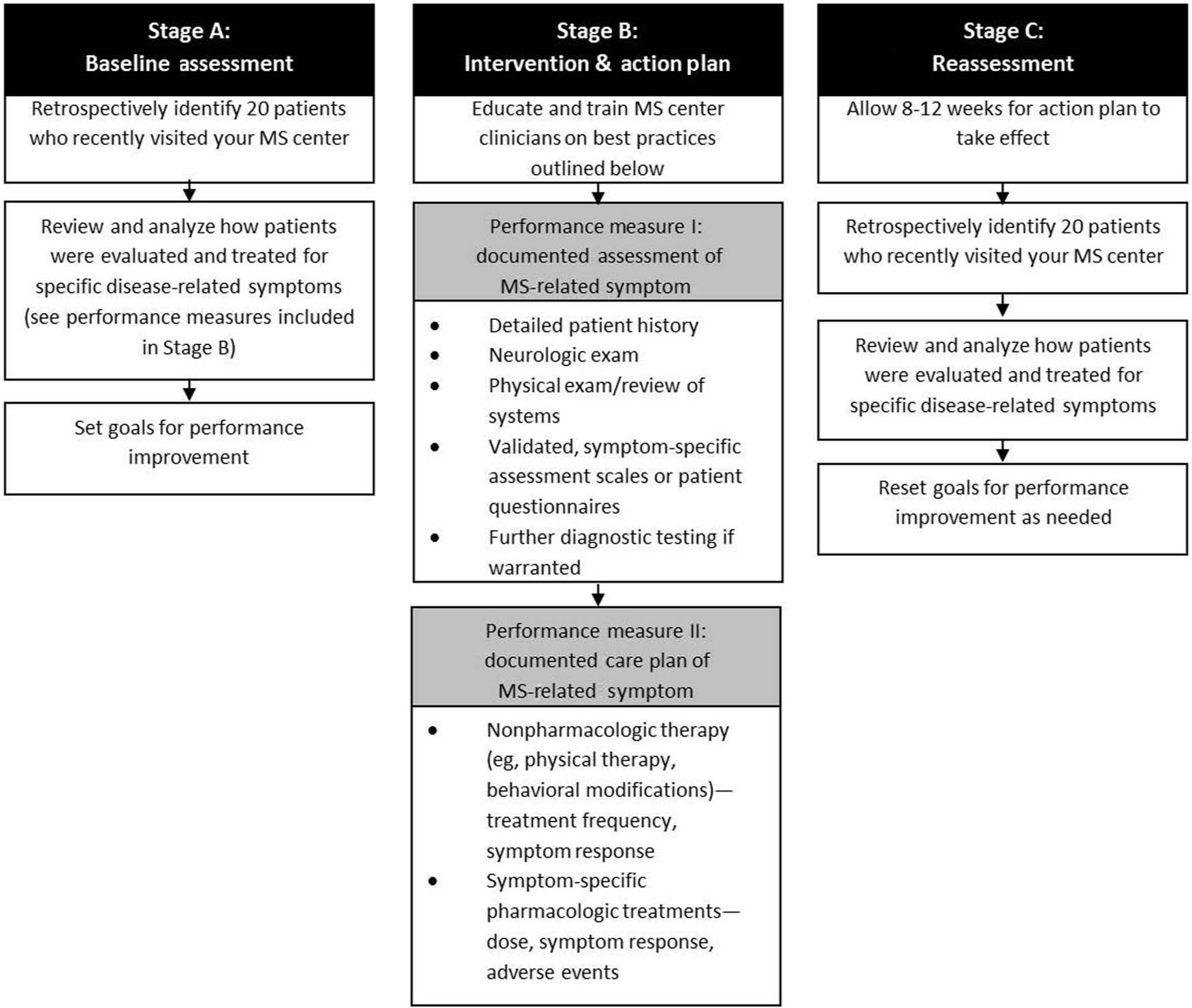
Algorithm for improved management of MS-related symptoms.
The planning phase consisted in inviting faculty regarded as experts in MS-related symptom management. An activity chair (AEM) was identified and led the efforts on recruiting additional faculty as well as developing the performance measures and interventions for the activity. Next, outreach was conducted to recruit volunteer MS centers with an interest in the PI CME activity. Upon confirmation, MS champions were identified for each facility who would be responsible for the overall management of participating clinicians at their respective MS center, reviewing charts and entering data (during baseline and reassessment Stages A and C), and coordination of a live in-service workshop (part of the Stage B intervention). Designated MS champions were then trained on the process including data entry into a web portal specifically designed for this activity.
The expert faculty also convened for several planning meetings at critical phases in the project: identification of performance measures; creation of the online data collection form; development of content for live in-service workshop meeting; and approval of toolkit components. The following symptoms were selected from the highly-rated MS care domains: 9 1) depression; 2) fatigue; 3) mobility impairments/falls; and 4) spasticity. Other common MS symptoms such as bladder dysfunction and cognitive dysfunction were considered but not selected because the multidisciplinary complexity of their assessment and management was considered to be beyond the scope of the activity. The two performance measures selected for the PI CME activity (percentage of patients with a “documented assessment” of the MS-related symptom and percentage of patients with a “documented care plan” for the MS-related symptom) were agreed upon by the faculty after scanning various quality measures and guidelines for symptom management in the MS setting.9,11 -15 “Documented” referred to written evidence indicating whether a procedure/discussion was indicated/conducted or not indicated/not conducted.
During Stages A and C, each MS champion was instructed to randomly select 20 patient charts and enter data into the web portal using a predefined data collection form. Because of a miscommunication with the first participating site, five additional charts were pulled and reviewed during Stage A; these are included in the analyses. Patient identifiers were automatically populated using serial numbers so that patient information could be blinded once entered into the online system. For Stage C, MS champions randomly selected 20 different patient charts for review eight weeks after implementation of the Stage B intervention(s) and action plans. The Stage C sample allowed measurement and recording of changes in performance compared with pre-intervention baseline measures via statistical analysis.
The Stage B intervention consisted in a live workshop, which was customized for each individual MS center. Prior to each visit, the faculty member would review a particular center’s Stage A data and assess any performance gaps in need of improvement; the didactic content and discussion of relevant cases and implementation challenges were then tailored to that specific MS center’s needs and goals. Throughout the activity, participating MS centers (champions and individual clinicians) were provided feedback to stimulate their participation in the PI CME activity as well as numerous tools and resources that could be modified and implemented in each facility’s processes of care. The toolkit components included clinical practice guidelines and best practices for symptom management in MS (developed by the Consortium of Multiple Sclerosis Centers (CMSC) and the National Multiple Sclerosis Society (NMSS)) as well as independent online CME activities.
Impact of PI CME on MS symptom management
Of the 10 MS facilities that participated in this PI CME activity, five were not-for-profit centers (located in California, Maine, Nebraska, and Ohio (n=2)) and five were private commercial centers (located in Florida, Georgia, Louisiana, Texas, and Utah). A total of 37 individuals participated in the activity: 22 physicians, five nurse practitioners, three physician assistants, two registered nurses, and five “other” (i.e. administrative) MS center employees.
The reported patient demographic data including sex, ethnicity, and age, mirroring those of reported MS epidemiological statistics. 16 Stage A and Stage C data revealed the majority of patients to be female (74.6%) and Caucasian (75.6%); the most common age group (31.1%) was between the ages of 41 and 50 years.
Stage A baseline assessment revealed the most prominent areas for improvement at each site. After assessing and reflecting upon their Stage A baseline data, participating MS center clinicians then entered Stage B, during which they were required to attend an in-service workshop led by external faculty and then develop and implement an action plan based on what they learned. As part of their action plan, participating MS centers could also select resources from a toolkit developed for this activity (available at www.achlpicme.org/ms/toolkit). Overall, the toolkit components that were reviewed most frequently by participating clinicians were the quality indicators for MS 9 (32.4%), followed by best practice assessment scales for MS-related depression and fatigue (29.7%), and a clinical practice algorithm for spasticity (29.7%). When reviewing the MS-related symptoms of interest for participants, depression was ranked the highest (86.5%), followed by fatigue (83.8%), spasticity (59.5%), and mobility impairment (40.5%).
Eight weeks after completion of Stage B, MS center champions were notified to again pull 20 random charts for Stage C data entry, which were then compared with baseline data (Figure 2).
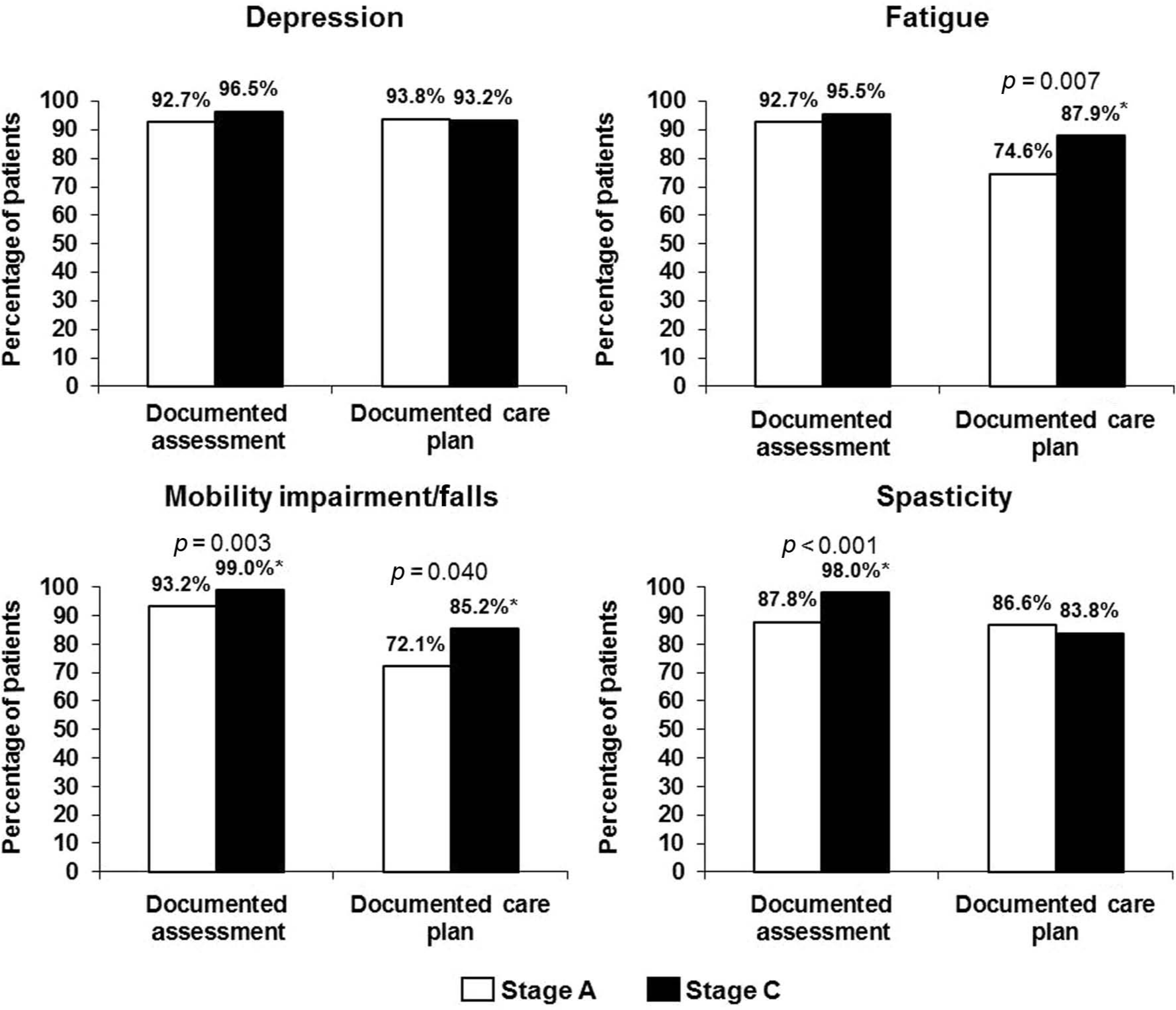
Aggregate performance change reported by 10 MS centers from baseline (Stage A) to eight weeks post intervention (Stage C) for prespecified MS-related symptoms reported by 10 MS centers: depression, fatigue, mobility impairment, and spasticity. White bars and black bars indicate Stage A and Stage C, respectively. *p=0.007 for change in fatigue documented care plan; p=0.003 for change in mobility assessment; p=0.040 for change in mobility documented care plan; p<0.001 for change in spasticity assessment.
The patient charts reviewed in Stage A also revealed the documentation of the following MS-related symptoms: 82 (40.0%) with depression; 132 (64.4%) with fatigue; 96 (46.8%) with mobility impairments/falls; and 89 (43.4%) with spasticity. Similar values were reported during the Stage C data collection: 74 (37.0%) with depression; 125 (62.5%) with fatigue; 82 (41.0%) with mobility impairments/falls; and 74 (37.0%) with spasticity. Statistical analysis showed no significant differences between Stage A and C sample populations for these specific MS-related symptoms.
When reviewing aggregate data from all 10 MS centers, improvement in performance was noted for the documented assessment of all the symptoms measured after the Stage B intervention; however, statistical significance was demonstrated only for mobility impairment/falls (5.8% performance improvement in documented symptom assessment; p=0.003) and for spasticity (10.2% performance improvement in documented symptom assessment; p<0.001). For the documented care plan performance measures, statistically significant improvements were reported for fatigue (13.3%; p=0.007) and mobility impairment/falls (13.1%; p=0.040) following intervention. In Stage C, the percentage of documented care plans for depression and spasticity slightly decreased compared with Stage A, although no statistical significance was shown.
Assessment scales for MS-related symptom(s) were reported for both baseline (Stage A) and post intervention (Stage C). These varied among the participating MS centers, with many using more than one scale for individual patients. Most symptoms were frequently assessed using a patient-detailed history for MS-related depression and fatigue and a neurological examination for MS-related mobility impairments and spasticity. However, a significant change (p<0.001) was reported for the use of the Timed 25-Foot Walk for mobility assessment: 24.9% during Stage A versus 45.5% during Stage C. A significant change was also noted in the use of a clinic questionnaire for spasticity assessment: 2.9% during Stage A versus 8.5% during Stage C (p<0.001).
The components of the care plans were also analyzed for MS-related symptoms that demonstrated a significant improvement in performance measures (i.e. fatigue and mobility impairment/falls). Documented care plans for patients with MS-related fatigue revealed an increased tendency in the use of behavioral modifications (e.g. pacing or relaxation; 54.3% during Stage A and 60.6% during Stage C) and a decreased tendency in the use of pharmacotherapy (76.6% during Stage A and 68.9% during Stage C). Documented care plans for MS-related mobility impairments showed decreased tendencies in both the use of physical therapy (75.8% during Stage A and 73.9% during Stage C) and of pharmacotherapy (35.5% during Stage A and 27.5% during Stage C). However, none of these changes were statistically meaningful.
In addition to the performance measures, information was also obtained from the reflections that the clinician participants completed after each stage of the PI CME activity. Some recurring challenges or barriers that were mentioned by the participants included time constraints and not having enough staff/personnel to address MS-related symptoms at every visit. When asked to summarize any practice, process, and/or outcomes changes that resulted from this PI CME activity, participating MS clinicians indicated that the education increased awareness of MS-related symptoms, allowed for comprehensive management of the disease, and improved patient care overall.
Conclusions
PI CME is an educational activity in which clinicians retrospectively assess their current clinical practice; choose areas for improvement; implement interventions based on treatment guidelines and healthcare standards; and then re-evaluate their clinical practice to assess the improvements made.6-8 Based on this rationale, the performance measures selected by the faculty for this PI CME activity addressed the topics of actively assessing and accurately diagnosing MS-related symptoms; selecting appropriate treatments; and monitoring the efficacy of non-pharmacologic and pharmacologic therapies. Our outcomes demonstrated that PI CME is a viable approach for quality improvement in MS as it resulted in significant increases in the frequency of assessment and care plan documentation for several important MS symptoms.
Based on our results, we developed an algorithm (Figure 1) for clinicians who wish to improve their performance in MS-related symptom management. In addition to the algorithm, a toolkit with resources from the PI CME interventions and action plans has been developed, which is available to the public at www.achlpicme.org/ms/toolkit.
Footnotes
Acknowledgements
The authors would like to thank the following MS centers for participating in the PI CME activity described in the paper: Augusta MS Clinic at Georgia Health Sciences (Augusta, GA); Louisiana State University MS Clinic (New Orleans, LA); MS Center of Maine Medical Partners Neurology (Scarborough, ME); MS Eye Care—University Eye Institute (Houston, TX); Mid Ohio MS Center at Neurological Associates, Inc. (Columbus, OH); Sutter Eastbay Medical Group MS Center (Berkeley, CA); University of Cincinnati Waddell Center for MS (Cincinnati, OH); University of Nebraska Medical Center (Omaha, NE); University of South Florida MS Clinic (Tampa, FL); and Western Neurological Associates MS Center (Salt Lake City, UT). We would also like to acknowledge Mark Van Buskirk (Data Reduction LLC; Chester, NJ) for the statistical analysis included in the manuscript. Finally, we would like to thank the accreditor Purdue University, and planning committee members Mindi Daiga, MBA and Lisa Keckich, MS (The Academy for Continued Healthcare Learning; Chicago, IL) and PeerView Institute for Medical Education for planning and managing the PI CME activity.
Conflict of interest statement
BAC has served as a consultant for Acorda, Astellas, Biogen Idec, EMD-Serono, Genzyme, Sanofi-Aventis, and Teva Neuroscience and has received research support though Northwestern University from Biogen Idec, Hoffman La Roche, and Novartis. SCK has received grant support from Bayer HealthCare Pharmaceuticals and consulting fees from work with Acorda Therapeutics, Bayer HealthCare Pharmaceuticals, Biogen Idec, EMD Serono, Genzyme, Novartis, Questcor, and Teva Neuroscience. CEM has received grants and research support from Bayer, Biogen Idec, EMD Serono, Novartis, Ono Pharmaceutical Group, and Teva; he has also served as a consultant for Acorda, Bayer, Biogen Idec, EMD Serono, Novartis, and Sanofi-Aventis. DHM has received research contracts and honoraria for speaking and consulting from Teva Neuroscience, Biogen Idec, and Novartis; research contracts and honoraria for speaking from Acorda Therapeutics and Genzyme; honoraria for speaking from EMD-Serono, Bayer, and Pfizer; and research contracts from MedImmune, Roche, ONO, Genentech, Sanofi-Aventis, Actelion, and Vaccinex. In the last year, AEM has received research support from Acorda, Biogen Idec, Genentech, Genzyme, Novartis, Osmotica, Questcor, Roche, and Sanofi-Aventis and has served as a consultant for Acorda, Biogen Idec, EMD Serono (Merck Serono), GlaxoSmithKline, Novartis, Nuron Biotech, Ono Pharmaceutical Group, Sanofi-Aventis, and Teva. HNT declares she was formerly employed by The Academy for Continued Healthcare Learning, which received unrestricted educational grants from Acorda Therapeutics.
Disclaimer
Full control of methodology, analysis, content, and manuscript creation resided with the faculty and authors in full compliance with ACCME® Accreditation Policies and the ACCME Standards for Commercial SupportSM.
Funding
This work was supported by Acorda Therapeutics, Inc. (unrestricted educational grant number: Acorda Grant ID number: 495937 CS2011CA38).
