Abstract
Mammary Paget disease (MPD) is a rare form of breast cancer that accounts for just 1% to 4% of all cases and is often associated with underlying malignancies such as ductal carcinoma in situ and invasive ductal carcinoma. Its clinical presentation frequently mimics benign dermatologic conditions or malignant melanoma, leading to diagnostic confusion and significant treatment delays. This review explores the diagnostic challenges and patterns of misdiagnosis in MPD, as well as the consequences of delayed recognition. A comprehensive search of Embase and MEDLINE identified 29 studies reporting on 32 cases of MPD, all of which were initially misdiagnosed—most commonly as melanoma (44.4%) and atopic dermatitis (25.0%). The average diagnostic delay was 2.3 years. Most lesions were unilateral (93.8%) and involved the nipple-areolar complex (87.5%). Imaging modalities demonstrated limited sensitivity, reinforcing the importance of early biopsy for timely diagnosis. Surgical intervention was the predominant treatment approach, employed in 75% of cases, and no recurrences were reported during a mean follow-up of 1.3 years. These findings underscore the urgent need for heightened clinical suspicion, earlier tissue sampling, and the development of standardized diagnostic pathways to reduce misdiagnosis and improve outcomes in patients with MPD.
Introduction
Mammary Paget disease (MPD), also known as Paget disease of the nipple, is a rare form of breast cancer that accounts for ~1% to 4% of all breast carcinomas and predominantly affects postmenopausal women, with a median age of 57 years.1,2 MPD is strongly associated with underlying malignancies; more than 90% of MPD cases coexist with ductal carcinoma in situ (DCIS), invasive ductal carcinoma, or both. 3 MPD involves the invasion of malignant Paget cells into the epidermis of the nipple and areola, causing characteristic skin changes such as thickening, pigmentation, moistness, scaling, crusting, or ulceration, often with an irregular border. 4 These persistent and aggressive symptoms are frequently misinterpreted as eczema or rashes—contributing to diagnostic delays ranging from 1 month to 2 years. 5 An uncommon variant, pigmented MPD (PMPD), adds further diagnostic complexity as it clinically and histologically resembles malignant melanoma. Accurate diagnosis of MPD and PMPD often requires skin punch biopsies and immunohistochemical (IHC) panel analysis, though variability in staining patterns can complicate interpretation. 6 These challenges in recognition and timely diagnosis hinder effective management, adversely impacting treatment outcomes and prognosis. This systematic review aims to investigate diagnostic challenges, misdiagnosis patterns, and the implications of delayed recognition, addressing critical gaps to improve early detection and clinical outcomes for this rare yet significant disease.
Materials and Methods
Study Selection
A comprehensive search was performed in the Embase and MEDLINE databases using MeSH terms and free-text keywords related to “Paget disease” and “diagnostic error.” All English-language articles published up to November 2024 were included.
Inclusion Criteria
The inclusion criteria for the studies were (1) primary research articles, (2) studies that reported cases of MPD or PMPD initially misdiagnosed through trials of incorrect treatments (eg, topical medications) or diagnostic approaches (eg, biopsy, imaging, etc), and (3) cases where there was explicit mention of a time period during which an incorrect diagnosis was maintained before Paget’s disease was identified as the secondary (correct) diagnosis. Misdiagnosis was defined as the clinician’s initial conclusion being incorrect, such as a physical exam suggestive of melanoma with subsequent biopsy confirming MPD or PMPD. The exclusion criteria were (1) reviews or expert opinion articles, (2) studies that did not report specific details regarding misdiagnosis or the process of delayed diagnosis, (3) studies involving nonhuman subjects (cadaveric, laboratory, or animal models), and (4) cases where Paget’s disease was not confirmed as the correct diagnosis. Further details of the study selection process are presented in the Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flowchart (Supplementary Figure 1). The study protocol is registered in the PROSPERO database (CRD42025637270).
Data Extraction
Three independent reviewers assessed the retrieved abstracts and full-text articles. Relevant data was extracted by 3 reviewers and compiled into a Comma-Separated Values file. The extracted data included the author names, study design, publication date, patient demographics, symptoms and signs, lesion localization, initial health encounter, initial suspected diagnosis, lesion characteristics, initial treatment, correct diagnostic test, correct diagnosis, histology results, molecular markers, imaging, treatment, and follow-up. Any disagreements were resolved through consensus with the senior author. Of 744 studies screened, 150 full-text studies were assessed for eligibility. Following exclusion of studies due to wrong patient population, study design, outcomes, and setting, as well as inability to retrieve full-text and duplicated, a final dumber of 29 studies were included for extraction and retrospective analysis (Supplementary Figure 1).
Outcomes
Primary and Secondary Outcomes
The primary outcome of this study was identifying the accurate diagnosis of MPD following initial misdiagnosis. This included identifying the patterns and frequencies of misdiagnoses. The study also aimed to evaluate the clinical presentation of MPD, including common lesion characteristics and the extent of lesion involvement. The secondary outcomes focused on the implications of delayed diagnosis, including the emotional, financial, and clinical burden on patients, as well as the frequency and consequences of prolonged or inappropriate treatments. Additionally, the study assessed the impact of inadequate close monitoring during topical medication trials and the consequences of delayed or absent dermatologist referrals.
Results
A total of 32 cases of MPD were identified and analyzed. The cohort’s median age was 58.8 ± 15.2 years, comprising of 28 females (87.5%) and 4 males (12.5%). All 32 MPD cases were initially misdiagnosed as a mimicking condition. Among the 32 cases, there were a total of 36 misdiagnoses as some individuals had 2 or more initial misdiagnoses. The most frequently documented misdiagnoses included melanoma, which accounted for 44.4%, atopic dermatitis 25.0%, bacterial infection 8.3%, and fungal infection 5.6%. Other diagnoses include nevus, T-cell pseudolymphomatous infiltrate, enlarged areola, Darier disease, lichenoid keratosis, and one not specified, all accounting for 2.8% each, totaling 17% (Figure 1). The mean duration of misdiagnosis was 2.3 ± 2.7 years. The shortest recorded duration of misdiagnosis was 1 week, whereas the longest delay spanned 10 years before a correct diagnosis was made. Additionally, 21 were confirmed to have DCIS, while the remaining 11 comprised invasive carcinomas (n = 5) and cases with no underlying malignancy (n = 6). Lymph node involvement was reported in 5 cases. Of the 16 cases initially misdiagnosed as melanoma, all were ultimately diagnosed with PMPD due to the presentation of dark lesions (Figure 2).
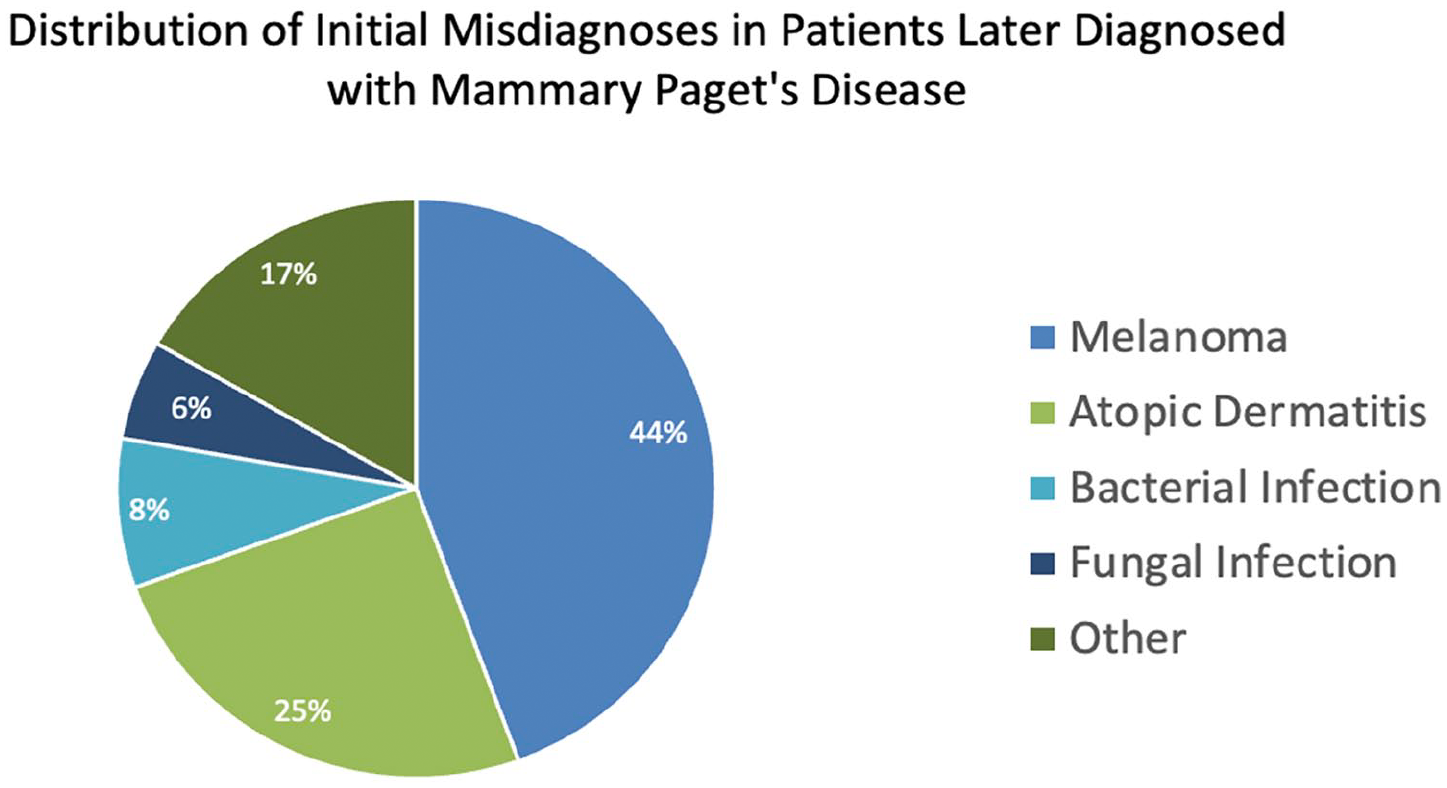
Distribution of initial misdiagnoses in patients later diagnosed with mammary Paget’s disease.
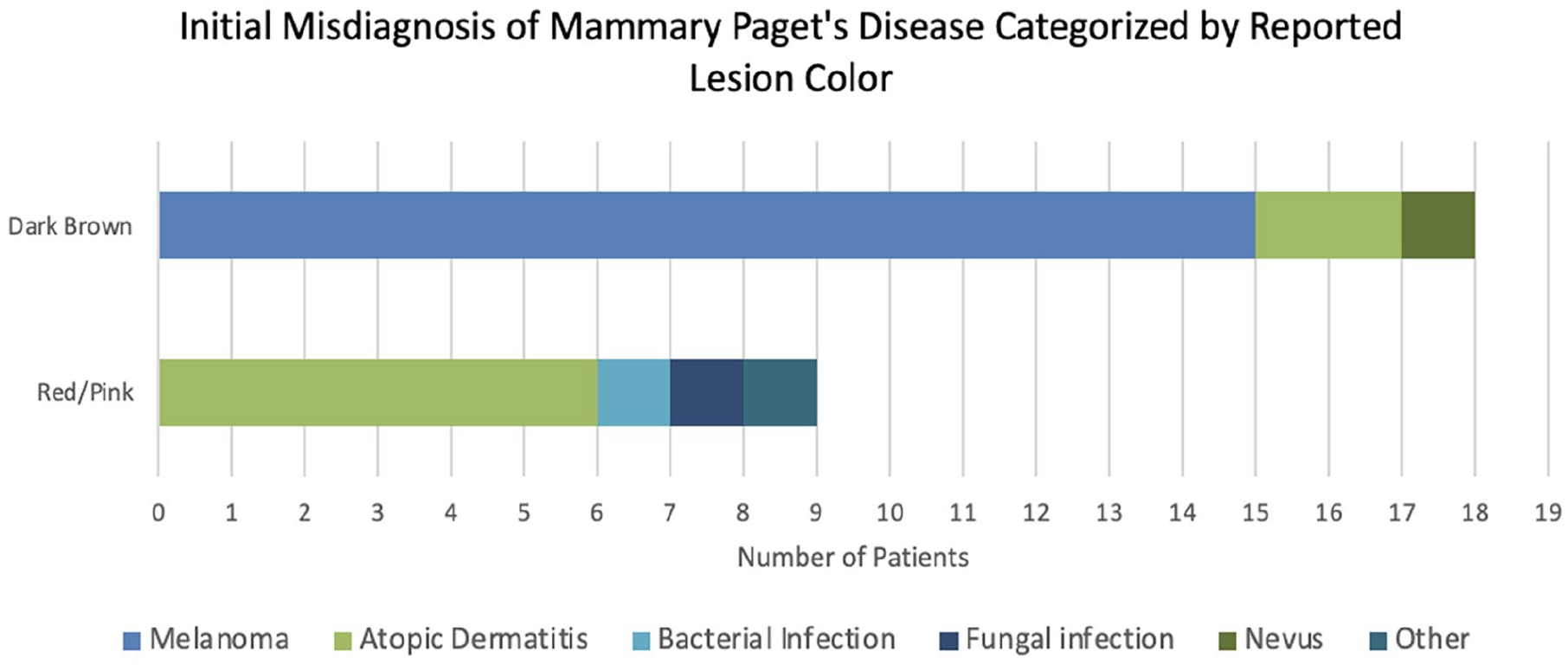
Initial misdiagnosis of mammary Paget’s disease categorized by reported lesion color.
Patients presented with a range of symptoms, with the most common being visible or palpable asymmetry of the nipple-areolar complex (53.1%), evolving lesions (43.75%), and erythema (40.6%). Other reported symptoms included plaque formation (34.4%), eczematous changes (25%), crusting (25%), ulceration (21.9%), pruritus (18.8%), discharge (15.6%), and bleeding (3.1%). Notably, no patients reported pain (Figure 3). Palpable masses were identified in 12.5% of cases, indicating that the majority of lesions did not present with a palpable component.
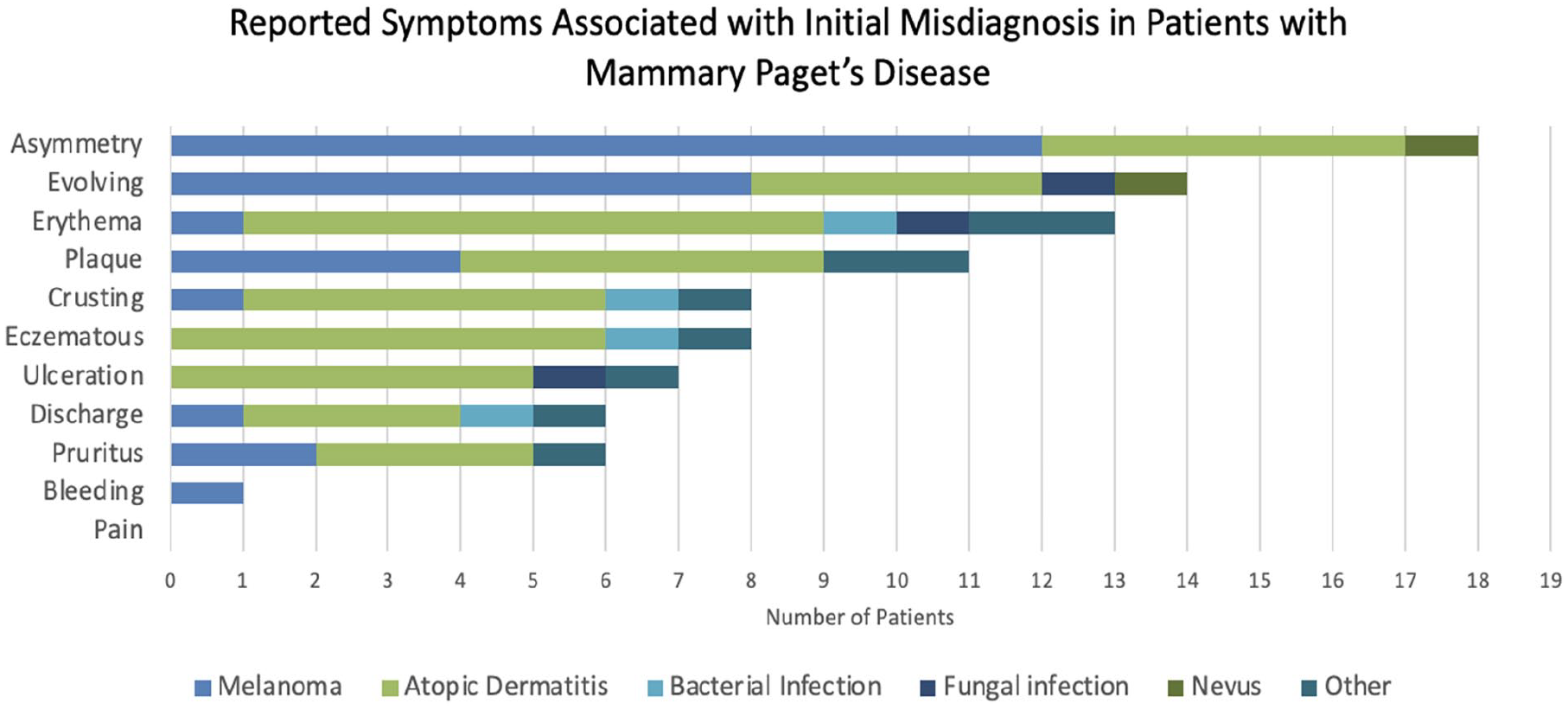
Reported symptoms associated with initial misdiagnosis in patients with mammary Paget’s disease.
Lesion localization predominantly involved the nipple-areolar complex, with nipple involvement observed in 87.5% of cases and areolar involvement in 53.1%. Unilateral presentation was the most common, seen in 93.8% of patients, while bilateral involvement was rare, documented in only 6.3% of cases. The bilateral cases were suspected to be atopic dermatitis, fungal infection, and bacterial infection, due to this unique presentation pattern. In a minority of cases (12.5%), lesions extended beyond the nipple-areolar complex, adding complexity to clinical evaluation.
Initial investigation strategies prior to a correct diagnosis of Paget disease varied widely, reflecting the challenges in clinical recognition and the reliance on limited diagnostic modalities. Biopsy was performed in 28.1% of patients and were completed only if melanoma was initially suspected. Imaging modalities were underutilized, with mammography performed in 21.9% of cases and ultrasonography in 12.5%. Magnetic resonance imaging (MRI) was rarely used (3.1%). The limited use of imaging and advanced diagnostic tools highlights the reliance on physical examination and biopsy for diagnosing Paget disease, underscoring the potential for missed or delayed diagnoses when more comprehensive testing is not employed.
Prediagnostic and postdiagnostic imaging included ultrasound, mammography, and MRI, with MRI showing the highest detection rate. Ultrasound was performed in 43.75% (14/32) of cases, with 50% (7/14) demonstrating pathology. Mammography was utilized in 53.13% (17/32) of cases, with 35.3% (6/17) showing pathology. MRI was conducted in 28.13% (9/32) of cases, with a notably high pathology detection rate of 88.9% (8/9).
Despite the widespread use of mammography and ultrasound, several cases demonstrated normal imaging findings, which further contributed to delays in diagnosis. These results suggest that while imaging is valuable in assessing underlying malignancy, it is not a substitute for biopsy in the diagnostic workup of MPD.
In addition to imaging, IHC analysis contributed to diagnostic confirmation and helped distinguish MPD from its clinical and histologic mimickers. GATA-binding protein 3 positivity on IHC was detected in 3/3 cases, supporting the mammary epithelial lineage of the disease. Melanocytic markers were assessed to differentiate MPD from malignant melanoma. HMB45 antibody was predominantly negative, with only 1/12 positive cases, and Melan-A protein was entirely negative (0/7 positive cases), helping to exclude melanoma in the differential diagnosis. S-100 protein was positive in 2/16 cases, with 14 negative results, demonstrating the potential for false-positive staining.
Additional markers, cytokeratin CAM 5.2 (4/4 positive) and carcinoembryonic antigen (7/9 positive), further supported the apocrine differentiation of PPD cells. The expression of epithelial membrane antigen (5/6 positive), cytokeratins AE1/AE3 (4/4 positive), and gross cystic disease fluid protein 15 (2/3 positive), contributed to the confirmation of the diagnosis. The predominantly negative findings for melanocytic markers, particularly HMB45 and Melan-A, emphasizes the accuracy of IHC in differentiating PPD from melanoma. These findings underscore the importance of human epidermal growth factor receptor 2 testing to guide targeted therapy and highlight the necessity of a comprehensive IHC panel in clinical practice (Supplementary Figure 2).
Treatment strategies for MPD and PMPD varied based on the presence of associated breast malignancies. Surgery was the most common treatment modality, performed in 75% (24/32) of cases. Of these, mastectomy was performed in 65.6% (21/32) of cases, while lumpectomy was conducted in 9.4% (3/32). Radiation therapy was administered in 21.9% (7/32) of cases. Chemotherapy was utilized in 18.8% (6/32) of cases. Systemic therapy and excision were both performed in 3.1% (1/32) of cases each. Only 1 case (3.1%) received no treatment. No cases reported the use of maintenance therapy.
The average follow-up period was 1.3 ± 1.3 years, with follow-up data available for 19 patients. Notably, an absence of recurrence was observed in all of these cases.
Discussion
This systematic review highlights the significant diagnostic challenges associated with MPD and PMPD. High rates of misdiagnosis and delayed detection highlight critical gaps in clinical recognition and effective management among primary care providers (PCPs).
The financial and personal toll of incorrect diagnoses is considerable. Many patients within our cohort underwent multiple failed treatment courses, including antibiotics, corticosteroids, and antifungal therapies, before a biopsy was performed. One patient in our review was initially prescribed antibiotics for a suspected bacterial infection, followed by corticosteroids and later, antifungal agents, leading to a prolonged delay of 2.5 years in accurate diagnosis before a biopsy confirmed MPD.
7
Such cases, in which MPD is misdiagnosed as benign inflammatory or infectious dermatoses, are particularly concerning due to the resulting treatment delays and avoidable healthcare costs. In contrast, misdiagnosis of PMPD as melanoma, while technically incorrect, often prompts biopsy at an earlier stage, enabling more timely diagnosis and management. Unnecessary treatments not only impose financial burdens on uninsured patients but also strain the healthcare system as a whole, leading to inefficiencies that compromise patient care quality without improving patient outcomes.
8
Initiatives, such as
Our study suggests that a critical aspect of improving diagnostic accuracy for breast lesions necessitates defined trial durations for topical therapies. If a lesion fails to resolve within the specified trial period, an immediate referral to dermatology for evaluation and biopsy is essential. Implementing clear clinical guidelines that define trial durations and escalation protocols for unresolved cases can prevent prolonged mismanagement. Furthermore, interdisciplinary collaboration among PCPs, dermatologists, and oncologists is crucial to ensure prompt referrals for appropriate diagnostic procedures. Lovén et al demonstrated that the integration of dermatologists into primary care practice, significantly reduced the time to confirmed diagnosis and treatment initiation for skin conditions (25 vs 49 days,
Imaging modalities, such as mammography, ultrasound, and MRI, are commonly employed to assess underlying malignancies in patients presenting with nipple-areolar abnormalities. 12 However, these investigations were often ineffective in detecting MPD among our cohort. Multiple patient cases exhibited normal imaging findings, with ultrasound detecting pathology in only 50% of cases in our cohort, which further contributed to delays in obtaining a definitive biopsy. MRI demonstrated the highest detection rate among the 3 modalities in our cohort, suggesting it may be underutilized and especially valuable when mammography and ultrasound are inconclusive despite persistent clinical concern. In cases where MPD was suspected but imaging findings are inconclusive, a stronger emphasis on clinical judgment, and biopsy should be advised to ensure accurate diagnosis. Emerging technologies, such as reflectance confocal microscopy (RCM), offer high-resolution, noninvasive imaging at a microscopic level, and have shown promise in the early detection of MPD.13 -15 Future research is needed to evaluate the efficacy of advanced imaging modalities, such as high-resolution dermoscopy or RCM, in facilitating earlier detection of MPD.
The high rates of misdiagnosis and diagnostic delays in MPD necessitate evaluation of whether these errors are attributable to individual provider oversight or systemic deficiencies in medical education. Broader institutional efforts, such as continuing medical education programs and public health initiatives, are warranted. In Canada, organizations like Public Health Ontario can play a pivotal role in developing and disseminating educational resources aimed at increasing provider awareness, leading to reduced MPD misdiagnosis rates. Integrating comprehensive information about MPD signs, symptoms, and investigation guidelines into medical curricula and professional development is essential to equip PCPs and dermatologists with the resources necessary for early recognition of the disease. Furthermore, implementing quality improvement initiatives that systematically track and audit diagnostic errors related to MPD can provide valuable insights into existing systemic gaps in care. Such data-driven approaches are crucial for informing targeted interventions and policy reforms designed to improve diagnostic accuracy and patient outcomes in MPD cases.
Although MPD is most commonly diagnosed in postmenopausal women, it is essential to consider male breast cancer in the differential diagnosis of nipple-areolar lesions.16,17 Male breast cancer remains a rare entity, accounting for 1% of all breast cancer cases globally. 18 Despite this, it necessitates vigilant monitoring, and clinicians should be cautious not to dismiss suspicious nipple lesions in male patients. In our cohort, male patients accounted for 12.5% (4/32) of all cases of MPD. As male breast cancer is often diagnosed at a more advanced stage due to lower clinical suspicion, increasing awareness of MPD in this population is crucial to ensuring timely diagnosis and intervention.19,20
Future research can also focus on developing adjunct diagnostic tools to facilitate early detection of MPD. Advances in dermoscopy, molecular diagnostics, and artificial intelligence-driven histopathologic assessment could offer promising avenues for improving diagnostic accuracy. Additionally, investigating the role of serum biomarkers and advanced imaging modalities may aid in differentiating MPD from benign dermatologic conditions. Integrating machine learning algorithms into pathology workflows could further enhance diagnostic standardization, offering more accurate interpretation of MPD histology, facilitating earlier, and more reliable detection.
This review has several limitations, primarily stemming from the heterogeneity in reporting across studies. In a substantial proportion of cases, the initial assessing healthcare provider was not specified, with reports often referring only to a general “clinic” or “initial visit.” This lack of detail limits our ability to determine whether the misdiagnosis originated with a PCP, dermatologist, or other specialist. Future studies should clearly document the clinical trajectory and provider type at each stage of assessment to better characterize diagnostic pathways and inform targeted interventions. Variability in case documentation, diagnostic criteria, and treatment approaches complicates direct comparisons and limits the generalizability of findings. Future studies should aim for standardized reporting of MPD cases to allow for more robust analyses of diagnostic trends and treatment outcomes. Additionally, studies with larger sample sizes and long-term follow-up data are needed to further evaluate the impact of early versus delayed diagnosis on patient survival and quality of life.
Conclusion
The findings of this review emphasize the critical need for improved early recognition of MPD, enhanced provider education, and systematic referral protocols. While MPD has a favorable prognosis when diagnosed early, delays in detection can lead to invasive disease and worse clinical outcomes. Addressing the gaps in primary care education, refining diagnostic protocols, and advancing research on novel diagnostic adjuncts are essential steps toward improving patient outcomes for this rare but significant breast malignancy.
Supplemental Material
sj-docx-1-cms-10.1177_12034754251364870 – Supplemental material for Dermatologic Mimickers of Paget’s Disease of the Breast: A Systematic Review
Supplemental material, sj-docx-1-cms-10.1177_12034754251364870 for Dermatologic Mimickers of Paget’s Disease of the Breast: A Systematic Review by Emily Volfson, Michal Moshkovich, Rebeca Yakubov, Jaycie Dalson and Carly Kirshen in Journal of Cutaneous Medicine and Surgery
Footnotes
Author Contributions
E.V. and M.M. contributed equally to this work and share first authorship. They conceptualized the review, developed the search strategy, conducted screening and data extraction, and drafted the initial manuscript. R.Y. contributed to study screening, data extraction, and critical revision of the manuscript. J.D. assisted with manuscript formatting and editing. C.K. supervised all aspects of the project, provided senior expertise in dermatology, resolved data extraction discrepancies, and critically revised the manuscript for important intellectual content. All authors reviewed and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
