Abstract
Background
Appropriate treatment reduces the severity and duration of relapses in demyelinating diseases of Central Nervous System (CNS). If high-dose corticosteroids treatment fails, therapeutic plasma exchange (TPE) is considered as a rescue treatment.
Objectives
This study aimed to investigate early clinical response and complications of TPE and prognostic factors in CNS demyelinating relapses.
Design
This prospective observational study was designed in a tertiary center during one year.
Methods
All adult patients diagnosed corticosteroid-resistant Multiple Sclerosis (MS), NeuroMyelitis Optica Spectrum Disorder (NMOSD), idiotypic Transverse Myelitis or Clinical Isolated Syndrome relapses, were eligible. Clinical response is defined based on Expanded Disability Status Scale (EDSS) at discharge. Clinical and laboratory complications recorded.
Results
Seventy-two patients were analyzed which 58.3% patients were female. MS was diagnosed for 61.1% of cases. Thirty-five patients (48.6%) responded and the mean differences of EDSS significantly decreased 0.60 score (CI95%:0.44-.77). Electrolyte imbalances and thrombocytopenia occurred in 80.6% and 55.6% of cases respectively and 40.3% of patients had systemic reactions. However, 26.4% patients experienced moderate to severe complications. In patients with moderate to severe disability, responders were younger (MD: 8.42 years, CI95%: 1.67-15.17) and had lower EDSS score at admission (median:6, IQR: 5.5-6 against 7.5 IQR: 6.5-8). The risk of failure was higher in active progressive MS patients compared with RRMS patients (OR: 6.06, CI 95%:1.37-26.76). Patients with thrombocytopenia were hospitalized more than others (MD: 1.5 days, CI 95%: 0-3). Females were more prone to hypokalemia and systemic reactions (OR: 3.11, CI 95%:1.17-8.24 and OR: 6.67, CI 95%:2.14-20.81 respectively).
Conclusion
The most common indication of TPE was corticosteroid-resistant severe MS relapses. About half of the patients presented an early clinical response. Lower disability, younger age and RRMS diagnosis are prognostic factors of better response. One out of four patients experienced moderate to severe complications, mainly electrolyte imbalances and systemic reactions. Appropriate interventions against these complications should be considered during TPE, especially in females.
Introduction
The most widespread demyelinating diseases of the Central Nervous System (CNS) include Multiple Sclerosis (MS), Neuromyelitis Optica Spectrum Disorder (NMOSD) and Idiopathic Transverse Myelitis (iTM).1,2 These disorders are relapsing in nature. Relapse is a clinical situation wherein the neurological symptoms of the patient worsen for more than 24 hours without any fever or infection. Relapses have a major impact on the quality of life of sufferers. For instance, the rate of relapses in MS patients is estimated to be between .27-1.66 per year. While mild relapses can be observed, intervention is essential for severe ones. Although some cases of relapse can only be observed, appropriate treatment can reduce the severity of the relapse and its duration, thus improving the quality of life of the affected individuals. This makes the management of relapses an important intervention for people with CNS demyelinating diseases. 3
The primary treatment for relapses is high-dose intravenous corticosteroids. In cases of corticosteroid resistance (worsening or not improving of symptoms or improvement of symptoms without change in impaired function), 4 the second-line treatment is therapeutic plasma exchange (TPE) and immunoadsorption. TPE could remove auto-antibodies, immune complexes and cytokines from the blood. Previous studies has demonstrate the crucial role of IL-6, IL-10, IL-12, TNF-α and IFN-γ in MS relapses.5,6 Among the proposed options, TPE has a special place due to its availability and cost. Several studies have shown that MS and NMOSD patients can respond significantly to TPE, reducing their Expanded Disability Status Scale (EDSS) by .88-2.5 scores. Meanwhile, selecting the patients who can respond to this treatment is challenging.7–9
Identifying the predictive factors for TPE can help us select appropriate patients, prevent useless interventions and unnecessary complications. Despite the studies conducted to determine the factors that predict the response to TPE, it is still not possible to determine the effects of different factors and provide a model to predict the response to TPE.8,10 Therefore, in this study, we aimed first assess the early clinical response and complications of patients with CNS demyelinating disease who received TPE, then evaluate the relationship of different factors with the early clinical response.
Method and Materials
This article is reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Study Design and Setting
This prospective observational study was conducted from March 2021 to March 2022 in Kashani hospital, which is a referral center for neurological disease in central Iran.
Participants
All CNS demyelinating patients who underwent plasmapheresis and met the eligibility criteria studied. Inclusion criteria were age≥18 years, diagnosis of MS based on McDonald criteria 2017, NMOSD based on the 2015 International Panel of NMOSD Diagnosis, idiopathic transverse myelitis or Clinically Isolated Syndrome (CIS), the onset of new neurological symptoms associated with CNS damage lasting more than 24 hours and at least 30 days interval from the previous attack. Active progressive MS subtypes including primary progressive (PP) and secondary progressive (SP) were also included like relapsing-remitting (RR). 11 An experienced neurologist who examined the patients before and after the relapse, based on the severity of the relapse requiring intervention or failure of treatment with intravenous corticosteroid (.5-1 gr/day methylprednisolone for 3-5 days), made the patient a candidate for plasmapheresis and refer to this center. Patients who could not complete the TPE course in our center (due to complications or other reasons) were excluded for further analysis. Change of diagnosis to other causes was also one of the exclusion criteria.
In our plasmapheresis department, a neurologist evaluated the patients daily and recorded the symptoms, examinations and EDSS. Systemic infections and SARS-COV-19 were ruled out using lab data and physical examinations. The internal jugular or femoral vein was used for access based on the patients’ condition and the surgeon’s decision. The plasmapheresis machine was PCS®2 (model number: 06002) from Haemonetics® company, United States of America, which uses centrifuge system. Before each session the catheter and plasmapheresis machine were checked. Citrate was used for anticoagulation. During the sessions, an expert nurse evaluated patients’ signs and symptoms including hypotension, tachycardia, allergic reactions, chest pain and inability to continue the session. At the end of each session, 20% albumin vial and normal saline serum were used for replacement. Laboratory tests including Complete Blood Cell counts (CBC), Sodium (Na), Potassium (K), Blood Urea Nitrogen (BUN), creatinine, Partial Thromboplastin Time (PTT), Prothrombin Time (PT) and International Normalized Ratio (INR) were checked at baseline and daily and Calcium (Ca), Magnesium (Mg), Albumin and Transaminases were evaluated twice a week. To calculate individual plasma volume (PV), we used Hakim, 12 Kaplan 13 and ideal PV 14 formulas.
We recorded demographic data, information of the disease, treatment and complications. Demographic data included age, sex, weight and comorbidity of diabetes, hypertension, any cancer and any other autoimmune disease. Duration of the disease, disease-modifying drugs (DMDs), total number of relapses during the disease, number of relapses in the last year, duration of current symptom and total dose of corticosteroid received before TPE were also recorded. We gathered the total plasmapheresis output volume, number of plasmapheresis sessions and duration of hospitalization. Severity of complications is categorized as mild, moderate and severe. Complications do not required intervention, are classified as mild, those that required intervention or interruption of plasmapheresis as moderate and life threatening complications as severe. 15 We defined the response to TPE as decrease of ≥.5 score in patients whose EDSS at admission was ≥6, and decrease of ≥1 score in patients whose EDSS at admission was ≤5.5. 16
Statistics
The Kolmogorov-Smirnov test was used to assess the normal distribution of variables. Numerical variables which were normally distributed are presented as mean ± Standard Deviation (SD); those which were not normally distributed are shown as median (IQR: interquartile range). Categorical variables are reported as numbers (%). The unit of analysis was based on individual patients. We used the independent sample T-test, paired sample T-test, Mann-Whitney test and Chi-square test. Binary logistic regression was used to consider covariations. P-value <.05 is considered statistically significant. All data was analyzed using BMI SPSS version 22 software.
Results
Characteristics of patients with CNS demyelinating disease relapses who undergo therapeutic plasma exchange (TPE).

MS: Multiple sclerosis, NMOSD: Neuromyelitis optica spectrum disorder, iTM: Idiopathic transverse myelitis, CIS: Clinically isolated Syndrome, FS: Function system, EDSS: Expanded disability status scale. Data is presented as number (%), mean ± standard deviation or median (interquartile range).
Disease-modifying drugs (DMDs) treatment among patients with CNS demyelinating disease relapses who undergo therapeutic plasma exchange.

Data is presented as number (%).
Four (5.6%) patients had no complications and 19 (26.4%) patients experienced moderate to severe complications. One (1.4%) patient experienced severe hyponatremia and one (1.4%) patient had severe thrombocytopenia. Electrolyte imbalances occurred in 58 (80.6%) patients. The three most common complications were thrombocytopenia, hypokalemia and plasmapheresis systemic reactions (55.6%, 52.8% and 40.3% respectively) (Figure 1). Evaluating changes in laboratory findings before and after TPE course showed decrease in platelet count (mean difference: 81,000 number/millimeter3 CI95%: 63,000-99,000) and potassium and calcium concentrations (mean difference: .3 milliequivalent/litter CI95%: .18-.42 and mean difference: .5 milliequivalent/litter CI95%: .33-.7) (Table S3). Complications of therapeutic plasma exchange among patients with CNS demyelinating disease relapses (n = 72). Other complications consisted of hypernatremia (n = 7), hypomagnesemia (n = 3), hyperkalemia (n = 1), hypercalcemia (n = 1).
Demographic and clinical characteristics of patients with CNS demyelinating disease relapses based on their response to therapeutic plasma exchange (TPE).
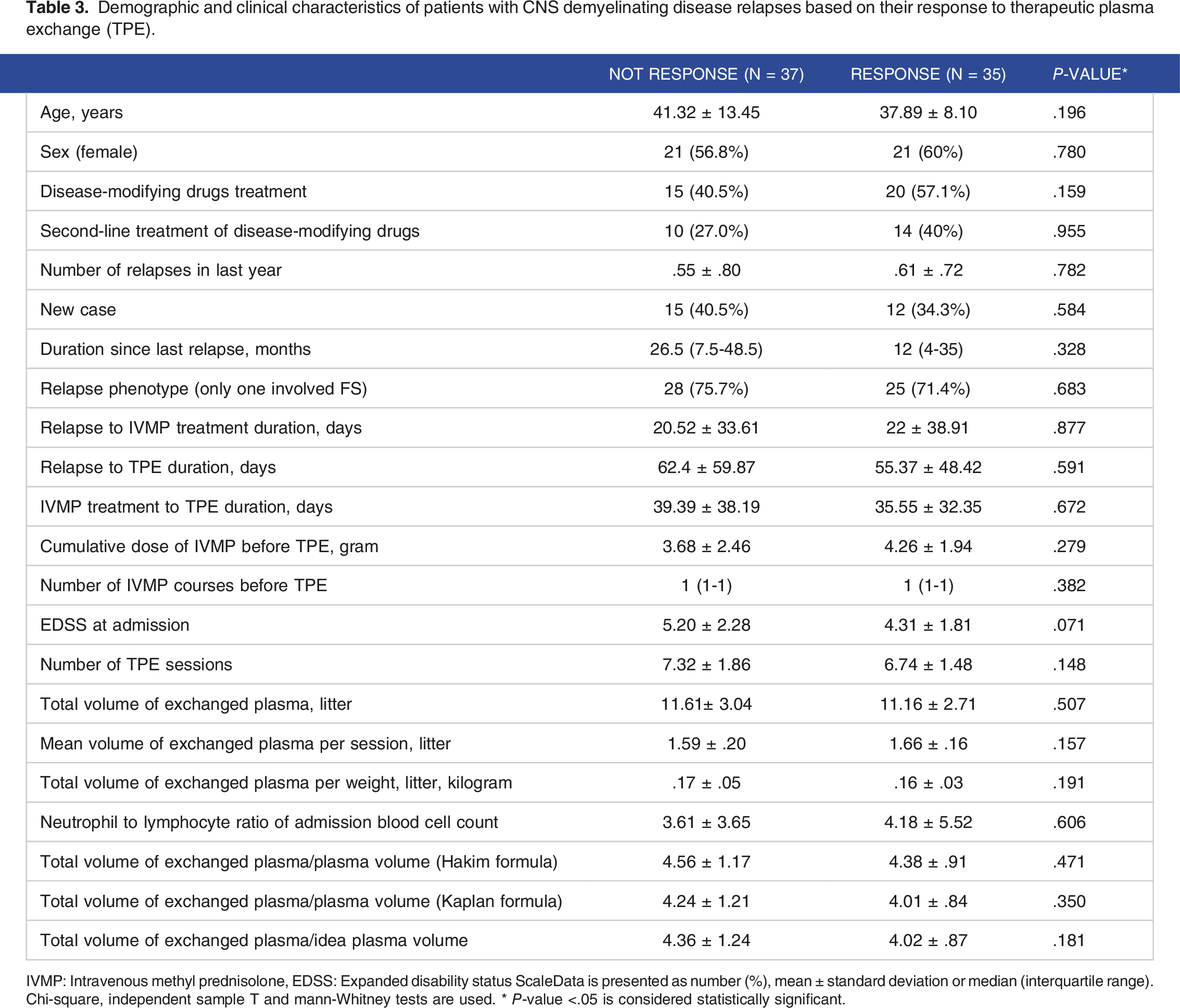
IVMP: Intravenous methyl prednisolone, EDSS: Expanded disability status ScaleData is presented as number (%), mean ± standard deviation or median (interquartile range). Chi-square, independent sample T and mann-Whitney tests are used. * P-value <.05 is considered statistically significant.
Demographic and clinical characteristics of CNS demyelinating disease patients with moderate to severe disability (EDSS at admission ≥4.5) based on their response to therapeutic plasma exchange (TPE) (n = 38).

EDSS: Expanded disability status scale, IVMP: Intravenous methyl PrednisoloneData is presented as number (%), mean ± standard deviation or median (interquartile range). Chi-square, independent sample T and mann-Whitney tests are used. * P-value <.05 is considered statistically significant.
To investigate the correlation of diagnosis with early clinical response, we grouped RRMS and CIS patients as one group and compared them with iTM patients. Patients with iTM were at higher risk of response failure compared with RRMS and CIS patients (OR: 2.5 with CI95%: .61-10.32). For subgroup analysis of MS subtypes, we categorized PPMS and SPMS as active progressive MS and compared them with RRMS based on the response. Results showed higher risk of failure in active progressive MS patients, with an OR: 6.06 (CI95%: 1.37-26.76).
We also analyzed the data to evaluate the relationship of each complication to other variables. Patients with thrombocytopenia were hospitalized for a longer period of time with a mean difference day of 1.5 (CI95%: 0-3). Hypokalemia and plasmapheresis systemic reactions were more frequent in females (OR: 3.11, CI95%: 1.17-8.24 and OR: 6.67, CI95%: 2.14-20.81 respectively). We found that patients with hypocalcemia had lesser weight (mean: 9.07, CI95%: 2.00-16.15), lesser total exchanged plasma (mean: 1.40, CI95%: .23-2.56) and lesser total exchanged plasma per session (mean: .13, CI95%: .04-.23).
Discussion
In this study, we assessed patients with CNS demyelinating disease receiving TPE in a tertiary hospital in Iran. Severe MS relapses with insufficient response to high doses of corticosteroids were the most common indication for TPE. We defined an early clinical response as a clinical improvement based on EDSS at the end of the TPE course. Finally, 48.6% of the patients were early clinical responders with a response rate ranging from approximately 35% to 55% depending on diagnosis. Totally, 68% of our patients were under treatment with second-line disease-modifying treatments (eg, fingolimod and rituximab), which showed that our study consisted of fewer patients with mild courses. This highlights that patients who require TPE to manage their relapses suffer from difficult-to-control relapses.
The response rate of patients with CNS demyelinating disease to TPE varies based on diagnosis, duration of follow-up and measurement of response. A review of 1191 patients with optic neuritis treated with TPE demonstrated that the response rate improved as follow-up duration increased. 17 This study also revealed that the response rate differs between various diagnosis; patients with CIS or MS, especially RR subtype, showed a better response to TPE. 17 Lipphardt et al. found that the response rate of patients with RRMS or CIS, active progressive MS and NMOSD patients was about 80%, 38% and 64% respectively in a 12-month follow-up. 18 Another study on NMOSD patients reported an early response rate of approximately 39%, which was slightly higher than our observations. 19 Ong et al. found that the response rate of NMOSD patients improved as they were followed up and reached 87% after 6 months based on their target neurological deficit. 20 Moreover, a systematic review in 2019 on MS reported that the response rate varies from 48% at discharge evaluation to 80% in 14-month follow-up. 8 Interestingly, Marrodan et al. claimed that there was no difference in EDSS score between RRMS patients with relapses who received TPE and those who observed after less than 6 months of follow-up. 21
In our patients, those with younger age and lower EDSS score at admission were prognostic factors for better response. Patients with moderate to severe disability profit better from TPE than patients with mild neurologic symptoms. Previous studies have also shown that younger age and female gender were favorable predicting factors in TPE response . 22 On the other hand, among patients with multiple sclerosis, a progressive phenotype was negatively associated with clinical response to TPE. Blechinger et. al. have previously observed that the pathology of demyelinating lesions, eg, active demyelination or complement deposition, may predict a better response to TPE. 23 Other predictive factors from previous studies were severity of relapse, duration of symptoms, presence of pathogenic auto-antibodies and specific phenotypes of MS can alter the response to TPE.10,16,22,24
Various clinical and demographic factors have been investigated for prognosis of outcome of TPE. Some studies have indicated that the delay in the start of TPE and old age can reduce the effectiveness of TPE.18,25–28 On the other hand, Bunganic et al. showed that in RRMS patients with aggressive relapses, there was no correlation between age and response. 29 However, there is no distinct definition regarding an appropriate time window or a favorable age for administering TPE. In line with our study, G´omez-Figueroa et al did not identify delayed time from relapse onset to TPE and age as predictive factors for response. 19 There are some reports that the severity of clinical symptoms can independently decrease the chance of response to TPE.10,28,30,31 However, patients who have well responded to TPE once, may be more likely to respond well to subsequent treatments. 22 Our results brought up lower severity of symptoms and RRMS or CIS diagnosis as prognostic factors of response. It seems that the interplay between different clinical and demographic factors alters the clinical outcome of TPE.
In our study, about one out of four patients experienced moderate to severe complications of TPE, which were mainly systemic reactions and electrolyte imbalances including hypokalemia, hypermagnesemia, hypocalcemia and hyponatremia. However, different studies have reported varying incidences of complications. A study in the Czech Republic reported that only 6% of RRMS patients who received TPE experienced clinical complications. 29 Another study evaluated both clinical and laboratory signs and symptoms and showed that 43% of patients were complicated. 21 Mavandadi et al. found that hypotension and skin rash, a plasmapheresis systemic reaction, were the most common clinical complications and hypokalemia was one of the most common laboratory complications, as our study confirmed. 32 Seyhanli et al. identified allergic reactions and hypotension as the two most common complications of TPE due to the treatment of different neurological diseases in a tertiary center in Turkey. 33 Anaphylactic reactions or mild toxic reactions to citrate, which were previously reported as self-limiting complications of TPE, did not occur in our study. 34 Previous studies have reported other serious complications, such as pneumothorax, thrombosis, and sepsis, in a percentage of patients. 35 These complications were not common in our patients. Considering the selected population and the specific neurological disorder, the occurrence and severity of TPE complications are variable in different studies. 34
Our findings also revealed that patients with thrombocytopenia were hospitalized for a longer period of time. In our setting we did not hospitalize the patients due to thrombocytopenia until the clinical indication of hospitalization meet, therefore it seems that patients who are hospitalized for a longer period of time due to any reason, are at higher risk of thrombocytopenia.
Our results showed that hypocalcemia and its symptoms were associated with lower total exchanged plasma volume and lower mean exchanged plasma for each plasmapheresis session. It seems that symptoms of hypocalcemia, especially in patients with lower body weights, were more likely to limit the time and consequently the volume of exchanged plasma during each session. Tingling or numbness are not rare during TPE and may be a consequence of hypocalcemia. 36 A clinical trial of 84 patients who underwent 581 plasmapheresis sessions has supported the use of calcium and potassium during plasmapheresis to reduce reactions and serve as the standard of care. 37
Female patients were more prone to develop hypokalemia and plasmapheresis-related systemic reactions. Similar to our findings, systemic reactions, presenting with dizziness, were reported in other studies and are commonly a symptom of low blood pressure or hypovolemia. 36
In this study we evaluate the early clinical response and complication of TPE in patients with CNS demyelinating disease. If it had been possible to follow up the patients for a longer period of time, we could have evaluated the long-term effects as well. Besides, the lack of information on patients prior to relapses prevented us from assessing the severity of relapses. For future investigations, we suggest long-term follow-up begins before relapse. On the other hand, CNS demyelinating disease consists of various disorders such as MS, NMOSD and iTM which our results revealed that they may influence how patients respond to TPE. However, we were not able to do more subgroup analysis based on diagnosis due to limited sample size.
Conclusion
In conclusion, we found that the most common indication for TPE in patients with CNS demyelinating disease is severe MS relapses with inadequate response to high corticosteroids doses. Considering early clinical response as a clinical improvement at the end of the TPE course, approximately half of the patients responded to TPE. However, about one out of four patients experienced moderate to severe complications, mostly electrolyte imbalances and systemic reactions. Although patients with iTM or active progressive MS have poor prognosis of response, but in general, less disability can predict better response. In addition, appropriate interventions should be implemented to prevent the most common complications, such as hypokalemia, plasmapheresis systemic reactions and hypocalcemia, especially in females.
Supplemental Material
Supplemental Material - Early clinical response and complications of therapeutic plasma exchange in central nervous system demyelinating diseases
Supplemental Material for Early clinical response and complications of therapeutic plasma exchange in central nervous system demyelinating diseases by Mehran Rashidi, Saba Naghavi, Neda Ramazani, Fereshteh Ashtari, Vahid Shaygannejad, Sayed Mohsen Hosseini and Iman Adibi in Journal of Central Nervous System Disease.
Footnotes
Acknowledgements
We want to appreciate Ms. Monireh Poostindooz, the nurse of plasmapheresis department of Kashani Hospital, for her assistance in sampling.
Author Contributions
Mehran Rashidi: data curation, formal analysis, investigation, methodology, project administration, validation, writing - original draft, writing - review and editing. Saba Naghavi: data curation, investigation, visualization, writing - review and editing. Neda Ramazani: investigation, methodology, writing - review and editing.Fereshteh Ashtari: conceptualization, methodology, writing - review and editing.Vahid Shaygannejad: conceptualization, methodology, writing - review and editing.Sayed Mohsen Hosseini: formal analysis, writing - review and editing.Iman Adibi: conceptualization, formal analysis, methodology, supervision, writing - review and editing.
Ethical Statement
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Isfahan University of Medical Sciences (grant number: 3401182).
Data Availability Statement
Some or all datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
