Abstract
Background:
Evidence suggests that efficacy and safety of disease-modifying treatments for multiple sclerosis may differ with age. We evaluate efficacy and safety of teriflunomide across age subgroups of patients from pooled clinical trials and real-world studies.
Methods:
Post hoc analyses of patients who received teriflunomide 14 mg in the pooled phase II and III TEMSO, TOWER, TENERE, and TOPIC core and extension studies (n = 1978), and the real-world Teri-PRO (n = 928) and TAURUS-MS I (n = 1126) studies were conducted. Data were stratified by age at study entry: ⩽25, >25 to ⩽35, >35 to ⩽45, and >45 years. In Teri-PRO and TAURUS-MS I, an additional group, >55 years, was assessed.
Results:
In the pooled core studies, teriflunomide reduced annualized relapse rate (ARR) versus placebo across all ages. Unadjusted ARRs remained low across age groups in pooled extensions (0.18-0.30), Teri-PRO (0.10-0.35), and TAURUS-MS I (0.14-0.35). Baseline Expanded Disability Status Scale scores were higher with age, but stable through core and extension studies (mean increases over 7 years: ⩽25 years, +0.59; >25 to ⩽35 years, +0.46; >35 to ⩽45 years, +0.35; >45 years, +0.81). Across age groups, adverse event (AE) incidences were 78.4% to 90.7% in pooled core and extension studies and Teri-PRO, and 29.2% to 37.7% in TAURUS-MS I; serious AE incidences were ⩽21.3% in all studies. In pooled phase III and Teri-PRO studies, lymphocyte count decreases over 1 year after initiating teriflunomide, and proportions of patients developing lymphopenia, were small across age groups.
Conclusions:
Teriflunomide efficacy was demonstrated regardless of age. Safety was generally consistent across age groups.
Introduction
Multiple sclerosis (MS) is the most common chronic neurologic condition in young adults worldwide, and is typically diagnosed between the ages of 20 and 40 years.1,2 Since MS usually extends over decades of a person’s life and has a minimal effect on life expectancy, its prevalence increases in older age groups, and disease activity may differ by age.3,4 It has been estimated that, in the United States, the prevalence of MS is highest in adults 55 to 64 years of age, underscoring the importance of understanding how age affects MS. 2 In addition, there is accumulating evidence that, with age, the efficacy of disease-modifying therapies (DMTs) may decrease and age-related adverse events (AEs) may emerge, altering the benefit-risk ratio of DMTs.5-7 Moreover, given that DMTs target the immune system, there is a potential for lymphopenia and increased infection risk, 8 which is an important clinical consideration in older people with MS (pwMS), who often present with various comorbidities.
Teriflunomide, a once-daily oral immunomodulator, is approved in more than 80 countries for the treatment of relapsing forms of MS (RMS), including clinically isolated syndrome (CIS), relapsing-remitting MS (RRMS), and in some jurisdictions, active secondary progressive MS.9,10 The efficacy and safety of teriflunomide 14 mg have been established in patients with RMS or RRMS in the phase II and phase III TEMSO, TOWER, and TENERE clinical trials, and in those patients with CIS in the TOPIC study.11-16 Long-term efficacy and safety of teriflunomide 14 mg was demonstrated in extension studies of the phase II and III trials through 9 years.11,17 In the real-world Teri-PRO and TAURUS-MS I studies, teriflunomide 14 mg—treated patients reported high levels of treatment satisfaction, with a stable safety and tolerability profile over 48 weeks and over 2 years, respectively.18,19
In this study, our objective was to evaluate whether age had an effect on the efficacy and safety of teriflunomide using pooled data from the phase II and III clinical trials and from 2 real-world studies of teriflunomide. In the safety analysis, we specifically evaluated the association of lymphocyte counts and risk of infection across age groups, as this is a safety concern of interest in aging pwMS.
Methods
Patients and study design
In this post hoc analysis, data were obtained from the pooled phase II and phase III TEMSO, TOWER, TENERE, and TOPIC clinical trials and their extension studies, the phase IV Teri-PRO study, and the TAURUS-MS I study. Patients who were treated with teriflunomide 14 mg were stratified by age at core study entry. Individuals who only started teriflunomide 14 mg during the extension studies were stratified by age at time of starting the extension study. Age stratification subgroups were as follows: ⩽25, >25 to ⩽35, >35 to ⩽45, and >45 years. Additional cohorts of patients aged >55 years were included from the Teri-PRO and TAURUS-MS I studies since these studies included a significant proportion of older patients (>55 years).
Complete study designs for the phase II and phase III TEMSO, TOWER, TENERE, and TOPIC studies have been published previously11-17 and are briefly summarized here.
The phase II study and phase III TEMSO and TOWER studies (for RMS patients) and the TOPIC study (for patients with CIS diagnosis; McDonald 2005 criteria 20 ) were double-blind, placebo-controlled studies in which patients were randomized 1:1:1 to receive placebo, teriflunomide 7 mg, or teriflunomide 14 mg. Age ranges for eligible patients were >18 to 65 years for the phase II core study and its extension, >18 to 55 years for TEMSO, TOWER, and TOPIC studies, and ⩾18 years for TENERE. Patients were treated for 36 weeks in the phase II trial, ⩾48 weeks in TOWER, and 108 weeks in TEMSO; a 108-week treatment duration was planned for TOPIC, but the study ended approximately 1.7 years earlier than planned due to the 2010 revisions of the MS diagnostic McDonald criteria.21,22 TENERE was a phase III, rater-blinded study in which patients with RMS were randomized 1:1:1 to receive teriflunomide 7 mg, teriflunomide 14 mg, or subcutaneous interferon beta-1a (IFN β-1a) 44 μg thrice weekly, and completed 48 weeks after the last patient was randomized. In the extension studies of the phase II and III trials, patients received teriflunomide 14 mg regardless of their original dose (TOWER, TENERE) or continued their original core study dose of teriflunomide (phase II, TEMSO, TOPIC). Patients who initially received placebo or IFN β-1a received teriflunomide 14 mg (TOWER, TENERE) or were reassigned 1:1 to teriflunomide 7 or 14 mg (phase II, TEMSO, TOPIC).
Details of the Teri-PRO study design have been published previously. 18 Briefly, this phase IV trial conducted in 14 countries (Austria, Belgium, Canada, Chile, Finland, France, Germany, Greece, Italy, Norway, Spain, Sweden, the United Kingdom, and the United States) was a prospective, single-arm, open-label study that assessed treatment satisfaction, disease outcomes, and safety in patients with RMS receiving teriflunomide per local prescribing guidelines. Patients received teriflunomide (7 or 14 mg once daily in the United States, teriflunomide 14 mg in the other countries) and were followed up for 48 weeks.
TAURUS-MS I was a noninterventional, prospective, longitudinal study in Germany. 19 Patients were eligible for enrollment if they were ⩾18 years old, had diagnoses of RRMS, were capable of completing questionnaires, and had no existing contraindications. To avoid selecting a nonrepresentative study population, no explicit exclusion criteria were specified. Documentation started approximately 4 weeks after treatment initiation with teriflunomide, and follow-up visits were scheduled after 3, 6, 12, 18, and 24 months.
Study endpoints
Although there were differences across the studies, the following common endpoints were assessed across all studies: annualized relapse rate (ARR), Expanded Disability Status Scale (EDSS) scores, and occurrence of AEs. Lymphocyte counts in the pooled core and extension studies and Teri-PRO studies were obtained using standard clinical laboratory assessments at baseline and Year 1 (Day 336-Day 378). Analyses could not be completed for lymphocyte counts in the TAURUS-MS I population.
Statistical analysis
All available data through the duration of each study were analyzed without imputation in patients who received teriflunomide 14 mg separately for each age group. In the TENERE core and extension studies, only patients who received teriflunomide 14 mg were analyzed, including patients switching from IFN β-1a 44 μg.
For the clinical trials, efficacy analyses (ARR and EDSS) were based on the combined intent to treat population, including all patients who were randomized and who received at least 1 dose of study drug from the phase II and phase III TEMSO, TOWER, and TENERE core and extension studies (maximum study durations: phase II extension, 12.8 years; TEMSO, 10.8 years; TOWER, 6.3 years; TENERE, 5.7 years). Data were included up to the last time point at which there were ⩾10 participants in each age category. Safety analyses were based on all patients grouped according to the dose of treatment they received, rather than the group to which they were randomized, and included data from TOPIC.
In the pooled core studies, adjusted ARRs were derived using a Poisson model with the total number of confirmed relapses onset between randomization date and last dose date as the response variable; poolcode (study), treatment, region, and EDSS strata at baseline as covariates; and log-transformed standardized study duration as an offset variable. Relapse rates were calculated for teriflunomide 14 mg versus placebo according to the age subgroups. In the pooled core and extension studies, Teri-PRO, and TAURUS-MS I, the unadjusted ARR and confidence intervals were calculated using the normal approximation for Poisson confidence interval; data in the core and extension studies were included up to the last time point at which there were ⩾10 patients in each group. EDSS scores were analyzed using year-wise descriptive statistics.
AEs were reported as incidences defined as the percentage of patients with at least 1 event. In the pooled core studies and Teri-PRO, mean lymphocyte levels were quantified using 1.0 cells/L × 109 cells/L as the lower limit of normal (LLN). Lymphopenia grades, categorized on a scale of 1 to 4 (Grade 1: 9 cells/L; Grade 2: <0.8 to ⩾0.5 × 109 cells/L; Grade 3: <0.5 to ⩾0.2 × 109 cells/L; Grade 4: <0.2 × 109 cells/L), were derived from 2 consecutive visits for the pooled phase III core studies (TEMSO, TOWER, TOPIC, and TENERE), and from the baseline and Year 1 visits for the Teri-PRO study.
Standard protocol approvals, registrations, and patient consents
All studies were conducted in accordance with the International Conference on Harmonisation Guidelines for Good Clinical Practice and the Declaration of Helsinki. All patients provided written informed consent before participating in the trials. All procedures were approved by local institutional ethics review boards of participating sites (names of the Institutional Review Boards and Independent Ethics Committees that approved the procedures at each participating site for all studies are listed in the Supplemental Appendix).
The phase II and III clinical trials and the real-world Teri-PRO study are registered with www.clinicaltrials.gov (ClinicalTrials.gov identifiers: phase II core: NCT01487096; phase II extension: NCT00228163; TEMSO core: NCT00134563; TEMSO extension: NCT00803049; TOWER: NCT00751881; TENERE: NCT00883337; TOPIC: NCT00622700; Teri-PRO: NCT01895335). The TAURUS-MS I study is registered in the Bundesinstitut für Arzneimittel und Medizinprodukte public database under number 2075.
Results
Patients
In the phase II, TEMSO, TOPIC, TOWER, and TENERE core and extension studies, data were pooled from 1978 patients treated with teriflunomide 14 mg: 209 (11%) aged ⩽25 years, 593 (30%) aged >25 to ⩽35 years, 719 (36%) aged >35 to ⩽45 years, and 457 (23%) aged >45 years. Mean ever exposed teriflunomide 14 mg treatment time during the pooled core and extension studies was 3.3 years (median 2.9 years; range 0.002-12.8 years).
At baseline, time since first symptom or MS diagnosis and mean EDSS score was higher with increasing age, whereas mean number of relapses in the 2 years preceding enrollment was lower with increasing age (Table 1). Baseline demographics and disease characteristics were similar by age group for patients on placebo and teriflunomide 14 mg (mean treatment time, 1.5 years) from the pooled core studies (Supplemental Table S1).
Baseline characteristics of patients treated with teriflunomide 14 mg in the pooled phase II, TEMSO, TOWER, TOPIC, and TENERE core and extension studies, Teri-PRO study, and TAURUS-MS I study by age group.

Abbreviations: DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; MS, multiple sclerosis; SD, standard deviation.
Values are mean (SD) unless indicated otherwise.
Patients initiated teriflunomide 14 mg at either core study baseline or extension baseline; bExcluding patients from the TOPIC study; cn = 146; dn = 485; en = 629; fn = 435; gn = 208; hExcluding patients in the phase II study and TOPIC study; in = 144; jn = 462; kn = 593; ln = 412; mn = 463; nn = 413; on = 279; pn = 285; qn = 211; rPer protocol set; sn = 39; tn = 163; un = 336; vn = 384; wn = 142; xn = 138; yn = 294; zn = 344; *n = 131; †n = 167; ‡n = 350; ≠n = 406; �n = 149.
In the Teri-PRO study, data were obtained from 928 patients treated with teriflunomide 14 mg: 23 (2%) aged ⩽25 years, 125 (13%) aged >25 to ⩽35 years, 280 (30%) aged >35 to ⩽45 years, 288 (31%) aged >45 to ⩽55 years, and 212 (23%) aged >55 years (percentages do not add up to 100% due to rounding). In TAURUS-MS I, data were obtained from 1126 patients treated with teriflunomide 14 mg: 43 (4%) aged ⩽25 years, 168 (15%) aged >25 to ⩽35 years, 357 (32%) aged >35 to ⩽45 years, 408 (36%) aged >45 to ⩽55 years, and 150 (13%) aged >55 years. As with the pooled core and extension studies, the duration since first symptom or MS diagnosis and the mean EDSS score at baseline was higher with increasing age in both the Teri-PRO and TAURUS-MS I studies (Table 1).
Clinical efficacy by age group
In the pooled phase II, TEMSO, TOWER, and TENERE core studies, teriflunomide 14 mg significantly reduced ARR compared with placebo across all age groups (relative risks of 0.445, 0.732, 0.641, and 0.642 in patients aged ⩽25 years, >25 to ⩽35 years, >35 to ⩽45 years, and >45 years, respectively; Figure 1A). Through the pooled core and extension studies, the unadjusted ARRs over 13 years were lower in patients aged >35 years (0.18-0.20) than in those aged ⩽35 years (0.27-0.30; Figure 1B). In the Teri-PRO study, the unadjusted ARRs were generally lower in the older age groups over 48 weeks (ARRs ranged from 0.35 in patients >25 to ⩽35 years old to 0.11 in patients >55 years old [Figure 1C]); however, the unadjusted ARR for the ⩽25 years age group was 0.10. In the TAURUS-MS I study, the unadjusted ARR was likewise lower over 2 years in older patients (ARRs were between 0.35 in patients >25 to ⩽35 years old to 0.14 in patients >55 years old, and was 0.29 in patients aged ⩽25 years, Figure 1D).
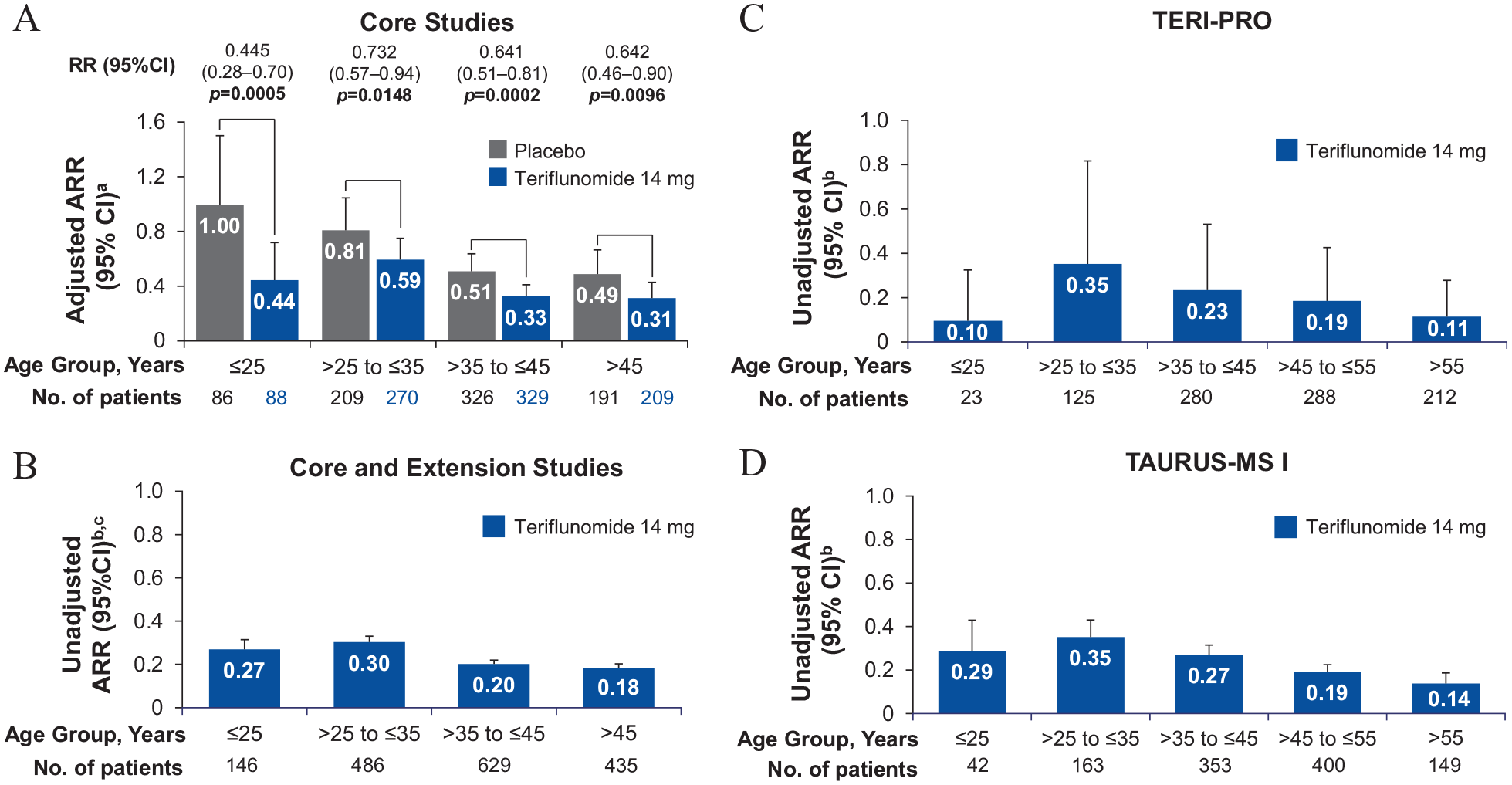
Relapse rates stratified by patient age at study entry: (A) ARR of placebo- and teriflunomide 14 mg-treated patients in the pooled phase II, TEMSO, TOWER, and TENERE core studies, and relative risk reductions with teriflunomide 14 mg versus placebo in all age groups, (B) unadjusted ARR in patients treated with teriflunomide 14 mg in the pooled phase II, TEMSO, TOWER, and TENERE core and extension studies through year 13, (C) unadjusted ARR in patients treated with teriflunomide 14 mg in the Teri-PRO study over 48 weeks, and (D) unadjusted ARR in patients treated with teriflunomide 14 mg in the TAURUS-MS I study over 2 years.
In the pooled phase II, TEMSO, TOWER, and TENERE core and extension studies, EDSS scores were stable over 7 years, with mean EDSS score changes observed at Year 7 versus baseline as follows: patients aged ⩽25 years: +0.59, aged >25 to ⩽35 years: +0.46, aged >35 to ⩽45 years: +0.35, and aged >45 years: +0.81 (Figure 2). Data from 8 to 13 years in the extensions were not included in the analyses due to low patient numbers (<10 participants in each age category). In the real-world Teri-PRO and TAURUS-MS I studies, mean EDSS scores were stable across all age groups, with mean score changes from baseline ranging from −0.18 to +0.13 at Week 48 and from −0.07 to +0.33 at Year 2, respectively (Table 2).

EDSS scores stratified by age in patients treated with teriflunomide 14 mg in the pooled phase II, TEMSO, TOWER, and TENERE core and extension studies over 7 years. Baseline for those receiving teriflunomide 14 mg in the core studies was core study baseline, while baseline for those receiving teriflunomide 14 mg after switching from placebo, teriflunomide 7 mg, or IFN β-1a in the core studies was extension study start. Due to low patient numbers (<10 participants in each age category), data from years 8 through 13 were not included.
EDSS scores stratified by age in patients treated with teriflunomide 14 mg in the Teri-PRO and TAURUS-MS I studies.

Abbreviations: CI, confidence interval; EDSS, Expanded Disability Status Scale; SD, standard deviation.
Values are mean (SD) unless indicated otherwise.
Safety by age group
In the pooled phase II, TEMSO, TOPIC, TOWER, and TENERE core and extension studies, the safety profile of teriflunomide over 13 years was generally similar across age groups. The frequencies of any AE were 87.0% to 90.7%, frequencies of any serious AEs were 14.2% to 21.3%, and frequencies of AEs leading to permanent treatment discontinuation were 12.0% to 15.0% (Table 3). In the core studies, frequencies of AEs were numerically equal or higher in teriflunomide 14 mg—treated patients versus placebo across all age groups; this trend was also observed for frequencies of serious AEs and AEs leading to permanent discontinuation across age groups, except in patients aged >45 years (Supplemental Table S2). In the core and extension studies, the most frequently reported AEs were nasopharyngitis, headache, increased alanine aminotransferase, hair thinning, and diarrhea, observed by ⩾10% of patients across all age groups (Supplemental Table S3). Of these AEs, hair thinning occurred more frequently in the >25 to ⩽35 years and >35 to ⩽45 years age groups than it did among patients aged ⩽25 and >45 years. Diarrhea, back pain, and hypertension were generally more common in the older age groups. Rates of malignancy were low in all age groups, with no malignancies reported in patients ⩽25 years old. Of the 10 deaths that occurred across age groups, 7 were deemed not related to treatment, while 3 (1 in the ⩽25 years old group and 2 in the >45 years old group) were deemed to be potentially related to treatment; however, medical evaluations have not established a formal causative role of teriflunomide as a risk factor explaining these fatal outcomes (Table 3).
AEs in patients treated with teriflunomide 14 mg in the pooled phase II, TEMSO, TOWER, TOPIC, and TENERE core and extension studies, Teri-PRO study, and TAURUS-MS I study by age group.
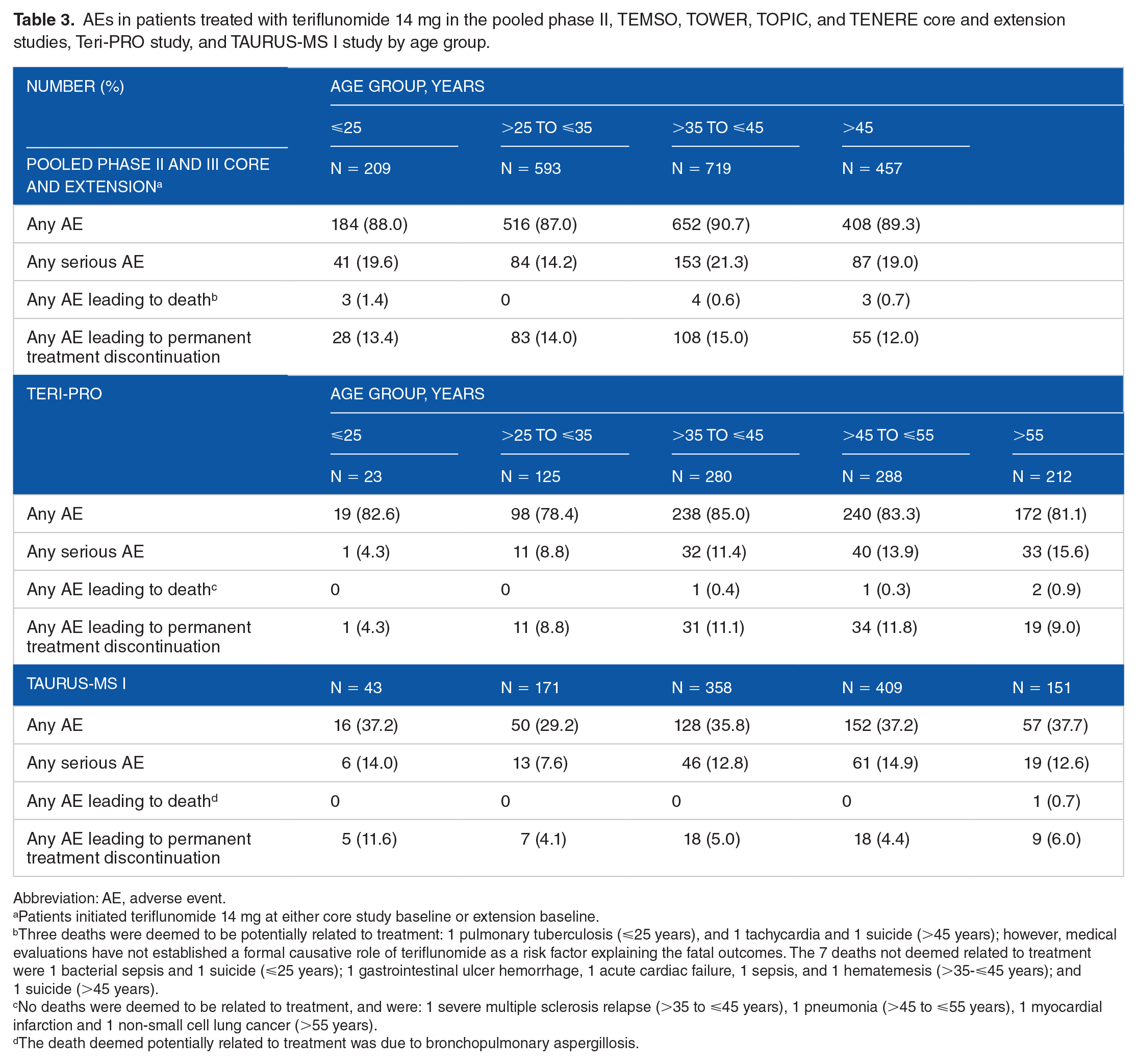
Abbreviation: AE, adverse event.
Patients initiated teriflunomide 14 mg at either core study baseline or extension baseline.
Three deaths were deemed to be potentially related to treatment: 1 pulmonary tuberculosis (⩽25 years), and 1 tachycardia and 1 suicide (>45 years); however, medical evaluations have not established a formal causative role of teriflunomide as a risk factor explaining the fatal outcomes. The 7 deaths not deemed related to treatment were 1 bacterial sepsis and 1 suicide (⩽25 years); 1 gastrointestinal ulcer hemorrhage, 1 acute cardiac failure, 1 sepsis, and 1 hematemesis (>35-⩽45 years); and 1 suicide (>45 years).
No deaths were deemed to be related to treatment, and were: 1 severe multiple sclerosis relapse (>35 to ⩽45 years), 1 pneumonia (>45 to ⩽55 years), 1 myocardial infarction and 1 non-small cell lung cancer (>55 years).
The death deemed potentially related to treatment was due to bronchopulmonary aspergillosis.
In the Teri-PRO study, frequencies of any AE were similar across age groups (78.4%-85.0%), while frequencies of serious AEs and AEs leading to death (none of which were deemed related to treatment) were higher with age (Table 3). Frequencies of AEs leading to permanent discontinuation in patient groups >25 years old were similar (8.8%-11.8%). The most commonly reported AE was hair thinning, which had lower incidence with increasing age (Supplemental Table S3). In patients ⩽25 years old, incidences of fatigue and headache were higher, and incidence of diarrhea was lower versus the other age groups. Patients >55 years old reported the lowest incidences of fatigue and headache. None of the 4 deaths reported in the study (all occurred in patients aged >35 years) were deemed related to teriflunomide treatment (Table 3).
In the TAURUS-MS I study, frequencies of any AE were similar across age groups (29.2%-37.7%), and were lower than those observed in the pooled studies and the Teri-PRO study (Table 3). Frequencies of serious AEs and AEs leading to permanent discontinuation were similar across age groups, although AEs leading to permanent discontinuation were approximately twice as high in the ⩽25 years old group than in the older age groups. One death was observed in a patient >55 years old and was deemed potentially related to treatment. Patients ⩽25 years old reported a higher incidence of MS relapse compared with patients in the other age groups (Supplemental Table S3).
As an AE of special interest, we investigated the association of lymphocyte counts and risk of infection across age groups. In the pooled TEMSO, TOWER, TOPIC, and TENERE core studies and the Teri-PRO study, mean baseline lymphocyte counts in patients who received teriflunomide 14 mg were 1.9 to 2.2 × 109 cells/L across age groups, and were lowered to 1.6 to 2.0 × 109 cells/L after 1 year of teriflunomide 14 mg treatment (Figure 3). All mean values pre- and posttreatment were above the LLN. The proportion of patients free from lymphopenia through Year 1 was similar across age groups in the pooled core studies (89.1%-96.3%; Supplemental Table S4). Most occurrences of lymphopenia were Grade 1 (2.9%-9.3% across age groups) or Grade 2 (0%-1.6% across age groups), with no cases at Grade 3 or 4. In the Teri-PRO study, the proportion of patients free from lymphopenia through Year 1 was likewise similar across age groups (92.6%-100%). There were 2 cases of lymphopenia that were Grade 3, and none that were Grade 4.

Mean lymphocyte counts stratified by age in patients treated with teriflunomide 14 mg, and measured at baseline and year 1: (A) in the pooled TEMSO, TOWER, TOPIC, and TENERE core studies and (B) in the Teri-PRO study.
In the pooled phase II, TEMSO, TOWER, TOPIC, and TENERE core studies, the incidence of infections was similar across age groups (39.5%-45.5%), and serious infections were less common in patients aged >35 years, versus those aged ⩽35 years (Table 4). Infections led to permanent treatment discontinuation in <1% of patients in each of the age groups ⩽45 years and none in those >45 years, and no deaths resulting from infections occurred in any age group. In the Teri-PRO study, infection incidences were lower in patients ⩽35 years old (17.4%-24.8%) versus patients >35 years old (27.8%-37.9%; Table 4). Incidences of serious infections were only observed in patients >35 years old and were similar across age groups >35 years. Infections led to death or permanent discontinuation in 0.3% and 1.7% of patients >45 to ⩽55 years old, respectively. In the TAURUS-MS I study, incidences of infection were generally lower with age (18.6% in patients ⩽25 years old, 13.1%-13.5% in patients >25 to ⩽55 years old, and 10.6% in patients >55 years old; Table 4). There was 1 patient >55 years old who had an opportunistic infection that led to death. 19
Infections in patients treated with teriflunomide 14 mg in the pooled phase II, TEMSO, TOWER, TOPIC, and TENERE core studies through 48 weeks, Teri-PRO study, and TAURUS-MS I study by age group.

Discussion
PwMS diagnosed at an older age generally have a poorer prognosis than their younger counterparts, and diminishing effectiveness of DMTs with age, which could be due to lower ARRs in patients with older age, may contribute to this observation.5,7,23,24 In this post hoc analysis of the pooled pivotal teriflunomide clinical trials and their extensions and real-world studies, efficacy of teriflunomide 14 mg on clinical outcomes was maintained regardless of age at study entry, with a safety profile that was generally consistent across age groups. In the pooled core studies, teriflunomide 14 mg showed greater efficacy on relapse rates compared with placebo over 2 years regardless of age at baseline, with a lower ARR observed in teriflunomide-treated patients >45 years old (0.31) than in younger patients (0.33-0.59). Through the extension studies, ARR was likewise lower in patients >45 years old (0.18) versus in patients ⩽45 years old (0.20-0.30). In the real-world studies, teriflunomide-treated patients ⩾55 years old also had lower ARR (Teri-PRO, 0.11; TAURUS-MS I, 0.14) compared with patients in younger age groups, in keeping with the natural history of MS. However, the observed stability of ARR in our study suggests that the effectiveness of teriflunomide persists even in older pwMS over extended treatment durations. Similarly, in the pooled core and extension studies conducted over 7 years, and in the real-world studies, there was relatively little disability progression in pwMS treated with teriflunomide over time irrespective of age (mean increase from baseline ⩽0.81 EDSS points) despite the age-related differences in baseline disability levels. Since disability progression is typically accelerated in older pwMS,25,26 the stability observed in those >45 years old in the pooled studies (n = 457; mean age = 50) and in those >55 years old in the real-world studies (n = 362, mean age = 61) is noteworthy, as few studies on MS treatments have specifically investigated a large number of older pwMS from these age groups.
The safety of teriflunomide treatment was generally consistent across age groups in both clinical trials and real-world studies. Incidence of AEs in the pooled core and extension studies and the real-world Teri-PRO study ranged from 78% to 91% and did not vary across age groups. Incidence of serious AEs and AEs that led to permanent discontinuation was likewise stable across age groups. PwMS in the real-world TAURUS-MS I study reported lower frequencies for AEs (29%-38%) and the safety outcomes did not vary across age groups. While it is unclear why the incidence of AEs was lower in TAURUS-MS, given that this was a study evaluating real-world effectiveness and safety, these results are reassuring.
Teriflunomide exerts immunomodulatory effects by selectively inhibiting mitochondrial dihydroorotate dehydrogenase, which results in inhibiting the proliferation of rapidly dividing cells such as lymphocytes.8,27,28 It is known that the immune system changes with age, and age-related immune deficits can include slowed cellular repopulation following teriflunomide-induced lymphopenia, and reduced numbers and diversity of newly generated T lymphocytes, which may limit responses to infections and malignancies in older adults.29-31 In the pooled core studies and the real-world Teri-PRO study, teriflunomide treatment over 1 year did not result in significant reductions in lymphocyte levels or in an increase in lymphopenia across age groups. None of the teriflunomide-treated patients >45 years old in the pooled phase II and III studies had Grade 3 or 4 lymphopenia, and only 2 patients in Teri-PRO had Grade 3 lymphopenia. During the same 1-year treatment duration, incidences of infections (39.5%-45.5%) and serious infections (1.0%-2.4%) in the pooled studies were similar across age groups, and no patients >45 years old had infections that led to death or permanent treatment discontinuation. In the Teri-PRO study, patients >55 years old had fewer infections (27.8%) than patients >35 to ⩽45 years old (37.9%) or those >45 to ⩽55 years old (33.3%), and there were no infections that led to death or permanent treatment discontinuation in this oldest age group. However, there was 1 patient in the 45-55 years age group in Teri-PRO who had a serious infection (pneumonia) leading to death. 18 In addition, in TAURUS-MS I, there was a single patient >55 years old who had a fatal opportunistic infection (bronchopulmonary aspergillosis), 19 illustrating that serious AEs can occur, but only rarely. Overall, the observed low incidences of serious infection and lymphopenia leading to treatment discontinuation in older pwMS suggest that teriflunomide is generally safe for use across age groups. This is an important consideration in the modern MS clinical practice at which DMTs are being utilized in an increasingly immunosenescent population. 32
This study has several limitations. First, it was conducted post hoc; thus, this analysis was not powered to compare across age groups for the treatment time, study duration, and total number of patients, and comparisons were descriptive. Another limitation of our study, in particular regarding the phase II and III extensions, is the potential of bias toward patients with favorable efficacy and tolerability outcomes, who are more likely to enroll in the extensions. Additionally, the pooled data from the phase II and III clinical trials only included 30 patients > 55 years (corresponding to 7% of the >45 years subgroup), making the oldest subgroup in this analysis (>45 years) not representative of the growing population of MS patients >55 years old.2,33,34 Moreover, analyses of lymphocyte counts in TAURUS-MS I patients could not be completed, excluding this population from our study of the association of lymphocyte counts and risk of infection across age groups. Finally, there were clearly observed baseline age-related differences in clinical characteristics, including higher EDSS scores and disease durations in older groups, and more inflammatory disease in younger groups. These factors need to be considered when interpreting our findings.
The importance of analyses such as ours that evaluate outcomes by age group is highlighted when considering recent real-world comparative studies that have found teriflunomide-treated patients to be older than those receiving dimethyl fumarate.35-37 These studies have supported the effectiveness and safety of both teriflunomide and dimethyl fumarate in MS patients in the real-world setting.35-37 These 2 oral DMTs also had similar discontinuation rates 38 and were associated with higher levels of persistence compared with injectable DMTs.39,40 With the inclusion of data from Teri-PRO and TAURUS-MS I, our study adds to this growing body of evidence for efficacy and safety of oral DMTs in real-world settings.
In conclusion, our findings suggest that the efficacy and safety of teriflunomide is similar across the age groups that were enrolled in the clinical trials reported. Furthermore, there was no increased incidence of infection and death in older patients. Collectively, these findings indicate teriflunomide treatment provides clinical benefit and an acceptable safety profile across a spectrum of ages in adults.
Supplemental Material
sj-pdf-1-cns-10.1177_11795735211028781 – Supplemental material for Efficacy and Safety of Teriflunomide in Multiple Sclerosis across Age Groups: Analysis from Pooled Pivotal and Real-world Studies
Supplemental material, sj-pdf-1-cns-10.1177_11795735211028781 for Efficacy and Safety of Teriflunomide in Multiple Sclerosis across Age Groups: Analysis from Pooled Pivotal and Real-world Studies by Jiwon Oh, Sandra Vukusic, Klaus Tiel-Wilck, Jihad Said Inshasi, David Rog, Darren P Baker, Yelena Pyatkevich, Elizabeth M Poole and Patrick Vermersch in Journal of Central Nervous System Disease
Footnotes
Acknowledgements
The authors and Sanofi thank the patients who participated in these studies, as well as the steering committees and the investigators in the phase II, phase III TEMSO, TOWER, TENERE, and TOPIC trials and their extension studies, and the Teri-PRO and TAURUS-MS I studies. Critical review of the manuscript was provided by Sarah Strattman, MS, of Sanofi. Editorial assistance was provided by George Gomez, PhD, and Panos Xenopoulos, PhD, of Elevate Scientific Solutions, and was funded by Sanofi.
Funding:
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The phase II, phase III TEMSO, TOWER, TENERE, and TOPIC trials and their extension studies, the Teri-PRO study, and the TAURUS-MS I study were funded by Sanofi. The sponsor was involved in the study design, collection, analysis, and interpretation of data, as well as verification of the data presented in the manuscript. The authors had unrestricted access to study data, were responsible for all content and editorial decisions, and received no honoraria related to the development of this publication.
Declaration of conflicting interests:
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JO received consulting or speaking fees from Alexion, Biogen Idec, Celgene, EMD Serono, Genzyme, Novartis, and Roche, and received research support from Biogen Idec, EMD Serono, and Roche. SV has received grants, personal fees, and nonfinancial support from Biogen, Celgene, Genzyme, MedDay, Merck Serono, Novartis, Roche, Sanofi, and Teva. KT-W received honoraria for lectures, studies, and consultancy from Almirall, Bayer, Biogen, Genzyme, Ipsen, Merck Serono, Merz Pharma, Novartis, Roche, Sanofi, and Teva. JSI reports nothing to disclose. DR received consulting fees from Bayer, Biogen, Celgene, MedDay, Merck Serono, Novartis, Roche, Sanofi, and Teva, and received research support from Actelion, Biogen, Genzyme, GW Pharma, Merck Serono, Mitsubishi, Novartis, Teva, and TG Therapeutics. DPB is an employee of Sanofi with ownership interest. YP and EMP were employees of Sanofi at the time the analysis was done. PV received consulting and/or speaking fees, and research support from Biogen, Celgene, Merck Serono, Novartis, Roche, Sanofi, and Teva.
Author Contributions
Author contributions for this study are as follows: substantial contributions to the conception or design of the work (DB, EMP, JSI, JO, PV, YP), acquisition of data for the work (DR, SV), statistical analysis (EMP), interpretation of data (all authors), and drafting the manuscript (all authors). All authors have read and given their approval for this version to be published, and agree to be accountable for all aspects of the work.
Previous Presentation of Data
Some of the data included in this manuscript were presented at the 71st Annual Meeting of the American Academy of Neurology (AAN), May 4–10, 2019; the Annual Meeting of the Consortium of Multiple Sclerosis Centers (CMSC), May 28–June 1, 2019; the 5th Congress of the European Academy of Neurology (EAN), 29 June–2 July, 2019; and the 6th Congress of the EAN, May 23–26, 2020 (Virtual Congress).
Availability of Data and Materials
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient-level data will be anonymized and study documents will be redacted to protect the privacy of the trial participants. Further details on Sanofi’s data-sharing criteria, eligible studies, and process for requesting access can be found at ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
