Abstract
Traumatic brain injury (TBI) is a leading cause of pediatric morbidity and mortality. Recent studies suggest that children and adolescents have worse post-TBI outcomes and take longer to recover than adults. However, the pathophysiology and progression of TBI in the pediatric population are studied to a far lesser extent compared to the adult population. Common causes of TBI in children are falls, sports/recreation-related injuries, non-accidental trauma, and motor vehicle-related injuries. A fundamental understanding of TBI pathophysiology is crucial in preventing long-term brain injury sequelae. Animal models of TBI have played an essential role in addressing the knowledge gaps relating to pTBI pathophysiology. Moreover, a better understanding of clinical biomarkers is crucial to diagnose pTBI and accurately predict long-term outcomes. This review examines the current preclinical models of pTBI, the implications of pTBI on the brain’s vasculature, and clinical pTBI biomarkers. Finally, we conclude the review by speculating on the emerging role of the gut-brain axis in pTBI pathophysiology.
Keywords
Introduction
Traumatic brain injury (TBI) is a significant public health issue and a leading cause of mortality globally. The CDC 2017 surveillance report estimates that TBI-related hospitalization was 224,000 in the United States. 1 Moreover, TBI is a crucial contributor to prolonged disability and dependence. 2 Epidemiological studies and biomedical research have focused on understanding the pathophysiology of TBI in the military setting because military service members are at an increased risk of blast-related injuries from explosives or blunt force to the head during warfare. 3 An appreciation for TBI in the adult civilian population is becoming more prominent.4,5 However, TBI pathophysiology and progression in the pediatric civilian population is studied to a lesser extent than the adult population despite some studies showing that children have worse post-TBI outcomes and take longer to recover.6-8
Young children (0-4 years old) and adolescents (15-19 years old) are at an increased risk of developing TBI, predominantly resulting from falls or motor vehicle accidents.9,10 An epidemiological study showed that 28% of children who visited the emergency department were reported to have been struck by or against an object. 11 Likewise, the CDC revealed that over 812,200 children (age 17 or younger) were treated in the United States for a concussion or TBI in 2014. 11 Importantly, children that survive TBI-related events have an elevated risk of developing psychological, social, sensorimotor, and cognitive impairments in later childhood and into adulthood.12-17 When juxtaposed to an increased risk of death post-TBI, these findings highlight the significant economic and public health burden of pediatric traumatic brain injury (pTBI) on society. 10 Thus, it becomes paramount that adequate funding and research be directed towards understanding the pathophysiology of TBI in the pediatric population since no therapies currently exist that effectively mitigate the consequential effects of pTBI long-term. 8
Although TBI studies in adults - humans, pigs, and rodents have provided a functional understanding of TBI pathophysiology, several nuances exist and should be considered before extrapolating results from adult TBI (aTBI) to pTBI. For instance, many aspects of the central nervous system (CNS) in the pediatric population, e.g., as myelination and synapse formation, are in continual development, and brain injury in children could severely impact these brain maturation processes with lasting neurological consequences.18,19 In this review, we examine: 1) current preclinical models of TBI and their use in pTBI, 2) the implications of pTBI on the brain’s vasculature, and 3) clinical pTBI biomarkers. For simplicity, we will refer to the pediatric population in this review as the neonatal period to adolescence. We will conclude the review by briefly discussing an emerging role in pTBI pathogenesis’s gut-brain axis.
Research Models of TBI and Their Application in the Pediatric Population
The development of an experimental animal research model that recapitulates the mechanisms of TBI has been a persistent challenge for researchers.20,21 The multi-planar and heterogeneous physical forces involved, coupled with concussive, rotational, sheering, and ischemic intracerebral injuries that result, have proven challenging to recreate TBI in a rigorous and reproducible manner.22-24 For the past several decades, the modalities that have been developed for use in animal subjects generally focus on imparting a single component of these multifactorial injurious mechanisms and then studying the consequential brain injury pattern. Several such paradigms for studying TBI in animals have been described in the literature.22,25,26 These models have proven paramount in advancing our understanding of the complex pathophysiology that underlies TBI. However, studies on the applicability of these models to human head injuries, especially those involving the pediatric brain, are sparse.27-30
TBI models can be broadly grouped into 2 major classes: penetrating vs non-penetrating injury. Once differentiated into the above classes, the models have then been designed to simulate either focal or diffuse injury through specific modifications. The models vary in their ability to produce mild to severe injury. Some models are more amenable to adjusting severity gradients than others. Ease of implementation is also an important consideration when choosing the appropriate model due to the high throughput of animals often needed for TBI experiments. This section explores some of the more commonly used models in TBI research and discusses their use in pTBI research.
Closed Head Animal Models of TBI
“Closed head” animal models of TBI refer to the generation of brain injury by applying an impulse, i.e., force, through an intact skull with or without skin incision and outer table exposure. The major variations of this overarching category include
Non-Impact Models
In non-impact impulsive loading, angular momentum is imparted to the brain by inducing rapid head movement and slower deceleration without external loading. These models, therefore, recapitulate diffuse axonal or shear injury caused by the stretching and compression of anisotropic tissue.31,32 Interest in this specific modality has experienced renewed vigor, given that a vast component of modern human TBI is secondary to the primary impact from sporting collisions, motor vehicle accidents, and wartime blast injuries.
Non-impact blast impulse models have a conspicuous translation to the injuries experienced by soldiers in modern warfare. Replicating those conditions is usually accomplished by using a piston actuator to compress air or gas through an expansion chamber several feet in length instead of using a formal solid weight.33-35 Alternatively, some studies expose experimental animals to actual detonated explosives to replicate the desired wartime conditions with greater fidelity. 36 In models employing compressed air or gas via a shock tube, the imparted force can be titrated to achieve different severities of TBI, ranging from mild to fatal.33,35,37,38 To the best of our knowledge, there are no non-impact blast impulse models that have been validated in pediatric rodent animal models, and there is only one study that employed a non-impact model in young porcine subjects. The authors showed that 3- to 5-day old piglets receiving repetitive rapid axial rotation with a pneumatic actuator exhibited worsened composite cognitive function and increased mortality compared to single injury and sham piglets. 39 Given that 1 in 6 children live in a conflict zone frequently exposed to explosives, the absence of non-impact blast impulse models for pediatric populations is a critical barrier to progress pTBI research on a global scale.40,41
Impact Models
Impact-based TBI research models use a variable weight generally conducted through a tube propelled by gravity or a pneumatic or electromagnetic actuator to transmit a mechanical load directly onto the skull.42,43 The resultant injury (focal vs diffuse) is a function of both the mass of mechanical load used and applied to the skull, both of which can be easily modified. These models include the classic “weight-drop” model described by Marmarou et al., whereby a brass load is dropped through a two-meter Plexiglass tube and onto a murine skull. 42 Several closed head impact models recapitulate TBI in the pediatric animal subjects.27,28,44,45 The modifications to standard models for application to young subjects are minor, and mainly consist of altering the impulse amplitude or manner of fixation to the apparatus. The weight-drop impact model is less commonly used in pTBI research today than the controlled cortical impact model (discussed in the open head animal models of TBI section), which affords high precision and reproducibility. 8
Open Head Animal Models of TBI
Similar to closed head analogs, “open head” animal models of TBI, in which a craniectomy is performed so that impulses may be applied directly to the exposed dura, can be broadly divided into 2 schemas: fluid percussion and direct cortical impact. These models sacrifice the significant head movement of other models in exchange for in situ extradural impulses. The open head models can induce a focal cortical contusion of moderate to severe grade (direction cortical impact) or diffuse brain/axonal injury (fluid percussion).24,46
Fluid Percussion Models
The fluid percussion model has evolved since its inception by Denny-Brown and Russell in the 1940s. The original design relies on applying an impulse to the skull vertex alone after preparatory craniectomy. Historically, this model was developed in feline specimens, but researchers adopted the model for applications in other mammals.47,48 Some of the earliest adaptations to murine subjects were described in 1987 by McIntosh et al. and Dixon et al., who further modified the original design to allow lateral and midline testing.49,50 The Stalhammar apparatus was later developed, marking one of the seminal points in the model’s history. 51 In the Stalhammar method, a weighted pendulum is released from a known height to strike a piston attached to a fluid reservoir – often isotonic saline.51,52 The mechanical impulse is transmitted through the fluid to cause a localized deformation of the exposed brain.
Although these models have remained relatively unchanged since their inception, researchers have made refinements to allow for more precise digital manipulation of experimental parameters like pulse pressure, pulse velocity, and load duration.53-56 One crucial improvement was to perform the craniectomy and fluid percussion at sites other than the “standard” parasagittal location. In addition, the induction of apnea following percussion is a well-documented complication of this model and should be closely monitored to prevent mortality, particularly in pTBI models.52,54 Adaptions of the lateral and midline fluid percussion models to the pediatric rat have also been described.57,58 More recently, a study by Newell et al. was the first to demonstrate the feasibility of the lateral fluid percussion model in juvenile mice. 59 Fluid percussion models have also been utilized in newborn and young piglets to assess hemodynamic responses post-TBI.60-62
Cortical Impact Models
Controlled cortical impact (CCI) models share some of the general concepts of the weight drop, impact accelerator, and fluid percussion models. For this reason, they are commonly referred to as rigid percussion models. 25 In the most common paradigm for this model, a limited circular craniectomy is performed on an anesthetized subject where the head is securely affixed to the experimental apparatus by stereotactic pinning to prevent cranial motion. A direct extradural impulse is applied by a rigid piston that is driven electromechanically, pneumatically, or electromagnetically.63,64 Thus, the velocity and depth of the impulse can be scaled to affect both the severity of resultant brain deformation and the local contusion and axonal damage that follow. 63
While initially developed for studying aTBI, CCI is the most commonly used TBI model to study pTBI progression and pathogenesis. For pediatric applications, postnatal day (PND) 17-35 in rats and PND 21 in mice are frequently used in CCI studies. 8 There are, however, limitations to this model. The generated TBI is often too severe, confounding postoperative assessments and post-mortem analyses. To address this limitation, researchers have created closed head cortical impact models that allow for repetitive concussive impulses to the same subject.65,66 This refined model simulates a repetitive injury that mimics recurrent mild TBI; this novel feature will enable researchers to study the impact of mild, repetitive injuries that commonly occur in student-athletes.22,24
Models of Inflicted TBI
Shaken baby syndrome, also known as abusive head trauma or inflicted TBI, is often overlooked in the pTBI literature. As a result, very few models mimic the effects of shaken baby syndrome in babies less than one-year-old. Although models of shaken baby syndrome are utilized by a much smaller number of research groups, their studies can recapitulate the widespread cortical hemorrhage pattern suffered by human infant patients. At least 3 rodent models have been reported in the literature. Smith et al. were the first to describe such a model in 1998. 67 Their protocol involved exposing anesthetized PND 6 rat pups to one daily shaking episode for 3 consecutive days via a mechanical tabletop shaker set to 200 cycles per minute followed by euthanasia on PND 9. Bonnier and colleagues described another experimental model differing from the previous approach in that anesthetized PND 8 were exposed to shaking at 900 cycles per minute for 15 seconds on a tabletop shaker.68,69 Two decades later, Kawamata et al. described a refined apparatus and design consisting of lucent, fenestrated, and cushioned plastic tubes. PND 3 and PND7 mice were shaken by the same common tabletop shaker at 250 cycles per minute in single-minute bouts 5 times daily. The authors conclude that the model may help study the effects of cerebral microhemorrhages on behavioral outcomes in early development. 70
Research data on models of shaken baby syndrome in larger mammals is limited. Finnie and colleagues have published a few studies on their immature ovine model of inflicted TBI.8,71 This model involves manually grasping the axilla of anesthetized lamb subjects and vigorously shaking them in ten, 30-second bouts over a half hour. The development of a large animal model of pediatric inflicted TBI was galvanized by the hypothesis that the large volume gyrencephalic brains and relatively weak cervical muscles of ovine animals better replicated the forces and injuries experienced by human babies. Simulating abuse in anesthetized juvenile animals is controversial and presents a barrier to developing additional inflicted TBI models. 8 The reconciliation of these ethical concerns through the development of novel models should be a focus of future research.
Cerebrovascular Dysfunction Following TBI in Children
A major consequence following TBI is the damage to the brain’s vasculature. Cerebrovascular damage in animal and human TBI studies has been described in the context of hypoperfusion, hemorrhage, ischemia, edema, and blood flow abnormalities. 72 Cerebrovascular dysfunction is a hallmark finding in many pediatric conditions and often predicts cognitive outcomes. 73 For example, a systematic review by Bakker et al. found that decreased blood flow velocities in premature infants and children with sickle cell disease were associated with poor cognitive performance. 74 Likewise, Taylor et al. showed that vascular alterations in children increased the risk for cognitive impairment. 75 Moreover, recent studies suggesting the role of cerebrovascular dysfunction in neurodegenerative diseases further support the importance of vascular integrity in maintaining brain function and health. 76 This section briefly discusses the functional and structural vascular alterations evident following pTBI.
Cerebral Blood Flow Dysfunction Following pTBI
Transcranial doppler (TCD) ultrasound and magnetic resonance imaging arterial spin labeling (MRI ASL) has been used to measure cerebral blood flow (CBF) in children. TCD studies showed that newborns exhibit low (∼24 cm/s) cerebral blood flow velocity (CBFV); however, CBFV then rapidly rises (∼95 cm/s) and peaks at 6-9 years of age. 77 Beyond 10 years of age, CBFV declines and approximate adult values (∼50 cm/s).77-79 Furthermore, MRI ASL studies by Biagi et al. showed that CBF was highest in children 4-12 years of age and rapidly declined in adult subjects. 80 One explanation for the increased CBF values in children is the increased metabolic and energy demand needed by the developing brain. 73 Other vascular parameters such as cerebral vasoreactivity (CVR), which measures vascular responsiveness to vasodilation via changes in blood carbon dioxide (CO2), have also been shown to be elevated in children compared to adults.81,82 These findings make it apparent that cerebral hemodynamics in normal physiology differs significantly in early childhood compared to adults.
Cerebral autoregulation (CA) is crucial for maintaining a steady-state CBF under a precise range of cerebral perfusion pressure (CPP). 73 In normal physiology, Vavilala et al. showed that the cerebral autoregulatory index (i.e., how fast blood flow velocity returns to baseline after a transient decrease in mean arterial pressure) is lower in adolescents than in adults. 83 pTBI patients often reveal a significant reduction in CBF compared to aTBI patients. 73 It remains unclear whether the lower autoregulatory index in the pediatric population may be responsible for the more significant CBF alterations in pTBI compared to aTBI patients. Nonetheless, it is widely known that a drastic decrease in CBF puts the developing brain at risk for ischemia and neuronal death.81,84,85 Animal models of pTBI in piglets have revealed CA impairment couples a reduction in CBF and greater constrictions in pial vessels. Interestingly, CA impairment was more prominent in newborn TBI piglets (1-5 days old) compared to juvenile piglets (3-4 weeks old).60,86 The findings from the study mentioned above were corroborated by the results in another study demonstrating worsened and prolonged hypotension in PND 17 and PND 28 rats compared to adult rats following TBI. 87
Furthermore, human clinical studies in children have shown that CA impairment following TBI is a significant predictor of poor outcomes. More importantly, young age appears to be a risk factor for CA impairment.88-91 The mechanisms that lead to CA impairment and subsequent decrease in CBF are unclear; however, several pathways have been implicated. 73 For example, endothelin-1 (ET-1) has been shown to increase in pTBI animal models, and treatment with ET-1 antagonist, BQ-123, mitigates CA impairment by attenuating CBF decline and pial artery vasoconstriction.92,93 Similarly, a vasodilatory N-methyl-D-aspartate (NMDA) agonist (MK801) reduced pial vessel vasoconstriction and improved CBF following pTBI. 86 More recently, the c-Jun N-terminal kinase (JNK) intracellular signaling pathway has been implicated in pTBI-induced CA impairment. 73 These therapeutic targets that mitigate CA impairment are especially clinically relevant in the pediatric population since clinical studies have shown that CA impairment is present in about 17% of mild pTBI patients and 42% of moderate-severe pTBI patients.91,94
Blood-Brain Barrier Dysfunction Following pTBI
The brain’s vasculature’s primary structural unit is the blood-brain barrier (BBB). The BBB regulates a stringent transport of molecules and cells between the periphery and the brain parenchyma. This unique structure is composed of endothelial cells held together tightly by junctional proteins, astrocyte end-feet processes, surrounding pericytes, and a basal lamina. 95 Transporter and protein composition at the level of the BBB changes with brain maturity. For example, the immature BBB relies heavily on the inward transport of glucose and amino acids compared to the adult BBB. 96 Likewise, P-glycoprotein (P-gp) efflux transporter expression at the BBB has been shown to be increased at PND 7 compared to PND 28 in rats. 85 Moreover, Muramatsu et al. demonstrated that PND 7 rats had increased immunoglobulin G (IgG) compared to PND 21 rats following 24 h post-hypoxia-ischemia insult. Thus, an indication that the BBB is more responsive to hypoxia-ischemia insults in younger rats (PND 7) compared to older rats (PND 2). 97
Following TBI, the BBB becomes compromised. BBB perturbation post-TBI is evidenced by increased BBB permeability and loss of junctional proteins. 98 While most TBI patients tend to show acute BBB breakdown (days to weeks), some studies have shown that the breakdown of the BBB post-TBI may last for years.99-102 Models of pTBI, like aTBI, have revealed increased extravasation of dyes or IgG into the brain parenchyma. 103 Additionally, Badaut et al. showed increased BBB permeability to IgG coupled to the loss of junctional protein claudin 5 in PND 17 rats at day 3 post-pTBI. As measured via caveolin-1, cellular transcytosis was increased at days one and 7 following pTBI initiation in the same study. 104 Interestingly, claudin 5 levels have been shown to significantly increase at day 7 and up to 60 days post-pTBI compared to controls.103,105
Amyloid-beta accumulation is implicated in TBI pathogenesis and is thought to be mediated by BBB dysfunction at the transporter level. 106 Several studies have demonstrated that the P-gp efflux transporter is crucial for clearing amyloid-beta.107,108 Failure to remove amyloid-beta from the brain promotes neuroinflammation and neurodegeneration, which in turn impairs normal brain function. 109 Jullienne et al. and Pop et al. showed that P-gp transporter expression is decreased in PND 17 rats following pTBI. Consequently, the decrease in P-gp expression was coupled to increased brain amyloid-beta accumulation.105,110 Increased expression of perlecan and fibronectin perivascular matrix proteins post-pTBI is also thought to mediate the accumulation of brain amyloid-beta. 110
A complication of BBB damage seen earlier and more frequently in children than in adults is the accumulation of fluid in the brain (edema).111,112 Edema in pTBI patients is often associated with poorer outcomes and increased mortality114. In children, higher water content, a softer skull, and weaker cervical support are thought to be responsible for the increased risk of diffuse edema seen in children compared to adults. 113 Increased expression of water channel aquaporins (AQP4) present on the end-feet processes of astrocytes is thought to mediate the vasogenic edema formation seen post-pTBI. 73 This finding is supported by a study showing that inhibition of AQP4 via small-interfering RNA (siRNA) in PND 17 pTBI rats reduced edema and improved cognitive outcomes compared to controls. 104 Conversely, the increased expression of AQP4 in PND 17 rats is also thought to play a role in edema resolution; however, it appears that this benefit is present 3 days post-pTBI. 114
Breakdown of the BBB post-TBI is often associated with neuroinflammation, neuronal death, and long-term neurological deficits. Recent insights have generated new interest in studying the role of vascular integrity as a target for developing therapeutics that may be used to manage TBI acutely and long-term. For additional insights, readers are referred to this excellent review on vascular impairment post-pTBI. 73
Neuroimaging and Assessment of Clinical Biomarkers Following TBI in Children
Despite many proposed TBI therapies with encouraging early phase trials, none have made it through phase III clinical trials. 115 Discovering biomarkers is an important part of understanding the pathophysiology of any disease and identifying new ideas for therapies. While many biomarkers have been investigated and reviewed for the diagnosis, prognosis, and treatment of aTBI, and a few have focused on the pediatric population, no standard biomarkers have been widely adopted in the field.116-121 In general, TBI diagnosis is determined by the severity of primary cerebral lesions and secondary brain damage. Secondary brain damage can result from several biochemical and molecular mechanisms, including reactive oxygen species (ROS) production, lipid peroxidation, excessive glutamate release, and neuroinflammation. 122 The current standard for the assessment of TBI severity in both aTBI and pTBI is the Glasgow Coma Score (GCS), which classifies TBI as mild (13-15), moderate (9-12), or severe (≤8) 123 . Scoring is based on subsectional scales of the eye, verbal, and motor responses, with a score of 0 being no response (deep coma or death) and the highest score being “normal” ability to respond to the task, i.e., fully awake and alert). Iankova describes the clinical application of the GCS, including changes in the scale and the inconsistencies with scoring, as healthcare professionals are prone to subjective interpretation or discrepancies in the GCS assessment techniques. 124 Likewise, the Glasgow Outcome Scale (GOS) defines functional/neurological outcome and is typically scored as: 1 = death; 2 = persistent vegetative state; 3 = severe disability; 4 = moderate disability; and 5 = good recovery. 125 Although GOS is a global and nonspecific clinical score in infants and children with TBI, it is most widely used to assess late neurologic outcomes in this subset of patients.126-128 However, GCS and GOS are not specific to TBI, and these scales can be used to assess the severity of brain-related injuries, including stroke and Alzheimer’s Disease.129,130
Several reviews have described the use of brain imaging techniques following TBI in pediatric populations.131-135 Popular imaging techniques include computed tomography (CT) and MRI. TBI can result in physical changes of the brain structure, including axonal and white matter (WM) injury. The corpus callosum is a common area of injury in TBI, and its location permits the assessment of its structural integrity and function via imaging. Corpus callosum white matter integrity is measured using fractional anisotropy (FA). At the same time, the corpus callosum function can be assessed using interhemispheric transfer time (IHTT), which measures the time it takes for information to cross cerebral hemispheres. Dennis et al. have shown that callosal function is associated with disrupted white matter integrity in pTBI. 136 A recent study that examined post-TBI (mild TBI) cortical thickness, which varied by brain sub-region, compared to controls with orthopedic injury highlights the complexity of using neuroimaging techniques to predict TBI outcomes. 137 Brain imaging has been explored in the prediction of post-TBI behavioral deficits. 138 In addition, several metabolites detected during brain imaging have been examined for their applicability as biomarkers of TBI, including N-acetyl aspartate, creatine, choline, lactate, and myoinositol.139-142
Although CT scans are becoming increasingly common in head injury cases, physicians must consider the risk of unnecessary exposure to radiation, which is especially dangerous for children.143,144 Although MRI provides a better resolution of brain structure and function compared to CT, this technique has its own challenges in the pediatric population, including lack of proper child-sized equipment, increased movement-related artifacts, and the use of sedatives. 145 The goals of brain imaging are 2-fold and include detecting injuries that may require surgical or therapeutic intervention, as well as determining the prognosis of rehabilitative therapy or other long-term treatment plans. However, brain imaging techniques can be expensive, risky, and provide minimal information for targeting patient treatment or an assessment of patient prognosis. A biological fluid biomarker or panel of markers can provide a potentially faster, less expensive, and less stressful option to identify targeted therapeutic options for TBI patients. For the purposes of this review, we will briefly discuss some of these biomarkers that have been explored in TBI-defined pediatric clinical samples.
Potential brain-specific biomarkers in human pediatric Traumatic brain injury.
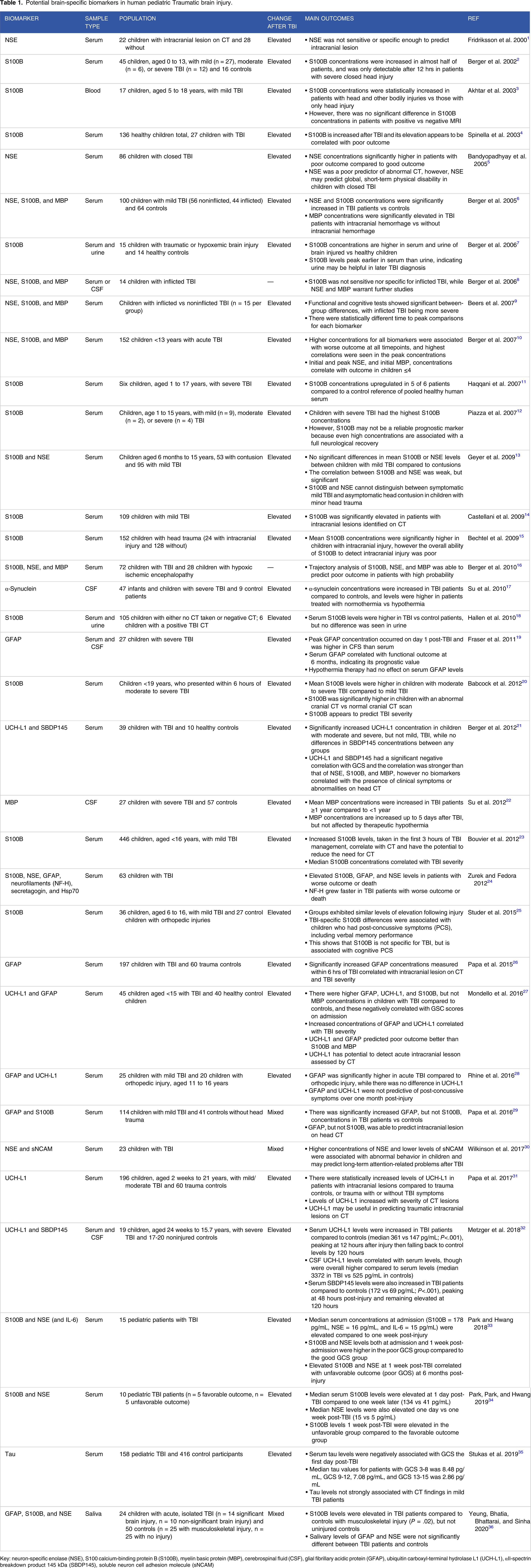
Key: neuron-specific enolase (NSE), S100 calcium-binding protein B (S100B), myelin basic protein (MBP), cerebrospinal fluid (CSF), glial fibrillary acidic protein (GFAP), ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), αII-spectrin breakdown product 145 kDa (SBDP145), soluble neuron cell adhesion molecule (sNCAM)
Nonspecific biomarkers potentially predictive of human pediatric traumatic brain injury.

Key: interleukin (IL), cerebrospinal fluid (CSF), vascular endothelial growth factor (VEGF), C-reactive protein (CRP), acute respiratory distress syndrome (ARDS), transforming growth factor-beta1 (TGF-1β), intracellular adhesion molecule (ICAM), nerve growth factor (NGF), doublecortin (DCX), brain-derived neurotrophic factor (BNDF), glial-derived neurotrophic factor (GDNF), neuron-specific enolase (NSE), gastrointestinal (GI), vascular cellular adhesion molecule (VCAM), tumor necrosis factor receptor 2 (TNFR2), matrix metalloproteinase-9 (MMP9), hepatocyte growth factor (HGF), high-mobility group box 1 (HMGB1), procalcitonin (PCT), triglycerides (TG), total cholesterol (TC), high-density-lipoproteins (HDL), low-density-lipoproteins (LDL), mitochondrial DNA (mtDNA), micro-ribonucleic acids (miRNAs), osteopontin (OPN), angiopoietin-2 (AP-2), endothelin-1 (ET-1), endocan-2 (EC-2), cardiac troponin (cTnI).
A Putative Role for the Gut-Brain Axis (GBA) in pTBI Pathophysiology
The gut microbiome plays an important role in pediatric health and disease, with strong evidence suggesting an influential part of the microbiome in maintaining brain function, homeostasis, and stress responses through the brain-gut axis. 151 This complex mutualistic ecosystem encompasses a diverse collection of microbial genomes that outnumber the cells in the human body. 152 Several studies have shown both intrinsic and extrinsic factors can shape the composition of the microbiome.153-155 For instance, children have a higher degree of interpersonal variation in their microbiome composition, especially within the first 3 years of life compared to adults. Additionally, the host’s geographical location also tends to have a significant effect on gut microbiome composition, with pronounced differences seen between individuals from different geographical and cultural backgrounds.153,155 Other factors that shape the gut microbiome include sex, genetics, lifestyle, and medications. 153 These factors should be considered when examining the bi-directional underpinnings of the gut-brain axis in maintaining overall health.
Disruption of the microbiome has been implicated in many neurodegenerative diseases, including Parkinson’s disease (PD), Alzheimer’s disease (AD), and multiple sclerosis (MS).156-160 Furthermore, recent studies have highlighted the role of the gut microbiome in maintaining healthy brain function. 161 For example, Braniste et al. showed that germ-free mice, beginning with intrauterine life, are prone to increased BBB permeability compared to pathogen-free mice with normal gut flora. However, the re-introduction of microbes to these germ-free mice decreased BBB permeability and improved cognitive outcomes. 162 Additionally, there is a growing body of preclinical and clinical evidence supporting microbiome dysbiosis in the pathogenesis and progression of aTBI.163-167 However, studies investigating the relationship between gut microbiome dysfunction and TBI progression in the pediatric population remain scarce.
The intestinal epithelium is a mechanical barrier that prevents commensal bacteria and pathogenic microbe translocation from the gut lumen into the bloodstream. However, following TBI, the permeability of the intestinal epithelium increases, providing a mechanism for bacterial translocation into systemic circulation.166,168-171 The translocated microbes are then propelled through blood vessels, ultimately gain access to other organs, and may usher in post-TBI complications such as sepsis, which in turn increases BBB permeability, promotes neuroinflammation, and worsens cognitive outcomes. 95 Furthermore, damage to the intestinal epithelium following TBI may lead to gastrointestinal ischemia, stress ulcers, and intestinal dysautonomia.165,172 Moreover, intestinal damage has been shown to correlate with the severity of brain injury and may be associated with TBI-related morbidity. 166 Furthermore, some studies have demonstrated in models of aTBI that specific bacterial species such as Lactobacillus acidophilus and Clostridium butyricum may be beneficial for post-TBI recovery.173,174
The pediatric microbiome is functionally and compositionally different from the adult microbiome 152 ; yet, to our knowledge, there are no preclinical studies that have examined the gut-brain axis in pTBI despite numerous aTBI studies demonstrating a promising role for gut dysbiosis in disease progression. Thus, preclinical and clinical studies that will examine microbiome dysbiosis in pTBI are urgently needed and could provide insights into the development of therapies that mitigate pTBI associated long-term neurological sequelae in children.175-236
Conclusion
Traumatic brain injury poses a significant economic and public health crisis. Despite the considerable advances in studying TBI in the adult population, fewer studies have examined the pathophysiology and progression of TBI in the pediatric population. Given the considerable differences in CNS development and cerebrovascular function between children and adults, as highlighted in this review, newer studies must investigate pTBI pathophysiology separate from aTBI. Furthermore, the diagnostic and prognostic value of neuroimaging and clinical biomarkers in pTBI needs to be explored. Ideally, optimal biomarkers should be easily accessible, minimally invasive, and rely on objective measures. Equally important is the emerging role of the microbiome in TBI pathophysiology. While some studies have examined the role of the microbiome in aTBI, to our knowledge, no studies have elucidated a role for the microbiome in pTBI. Taken together, this review addresses the current gaps in the pathophysiology of TBI in the pediatric population. Furthermore, we emphasize the importance of distinguishing between aTBI and pTBI in preclinical and clinical TBI research.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions:
DCN, CMB, and RQ contributed to manuscript design, compilation of final manuscript, and creation of figures and tables. DCN and ALB contributed to the section on clinical biomarkers. DCN, CHF, and BPL-W contributed to the section on pediatric TBI models. DCN contributed to the section on cerebrovascular dysfunction in pTBI. DCN, AA, and MAC contributed to the section on pTBI and microbiome. All authors reviewed the final documents and provided comments.
