Abstract
Under normal physiological conditions, gut microbiota and host mutually coexist. They play key roles in maintaining intestinal barrier integrity, absorption, and metabolism, as well as promoting the development of the central nervous system (CNS) and emotional regulation. The dysregulation of gut microbiota homeostasis has attracted significant research interest, specifically in its impact on neurological and psychiatric disorders. Recent studies have highlighted the important role of the gut- brain axis in conditions including Alzheimer’s Disease (AD), Parkinson’s Disease (PD), and depression. This review aims to elucidate the regulatory mechanisms by which gut microbiota affect the progression of CNS disorders via the gut-brain axis. Additionally, we discuss the current research landscape, identify gaps, and propose future directions for microbial interventions against these diseases. Finally, we provide a theoretical reference for clinical treatment strategies and drug development for AD, PD, and depression.
Introduction
The impact of gut microbiota on Alzheimer’s Disease (AD), Parkinson’s Disease (PD), and depression.

Gut Microbiota and Neurodegenerative Diseases
The onset and progression of Alzheimer’s Disease (AD), Parkinson’s Disease (PD), and depression are closely associated with gastrointestinal microbial imbalance and significantly the “gut-brain axis.” Gut microbiota can disrupt disease development through the gut-brain axis mediated by the nervous system, immune system, and microbial metabolites (Figure 1). Mechanisms by which gut microbiota regulate neuropsychiatric disorders via the gut- brain axis.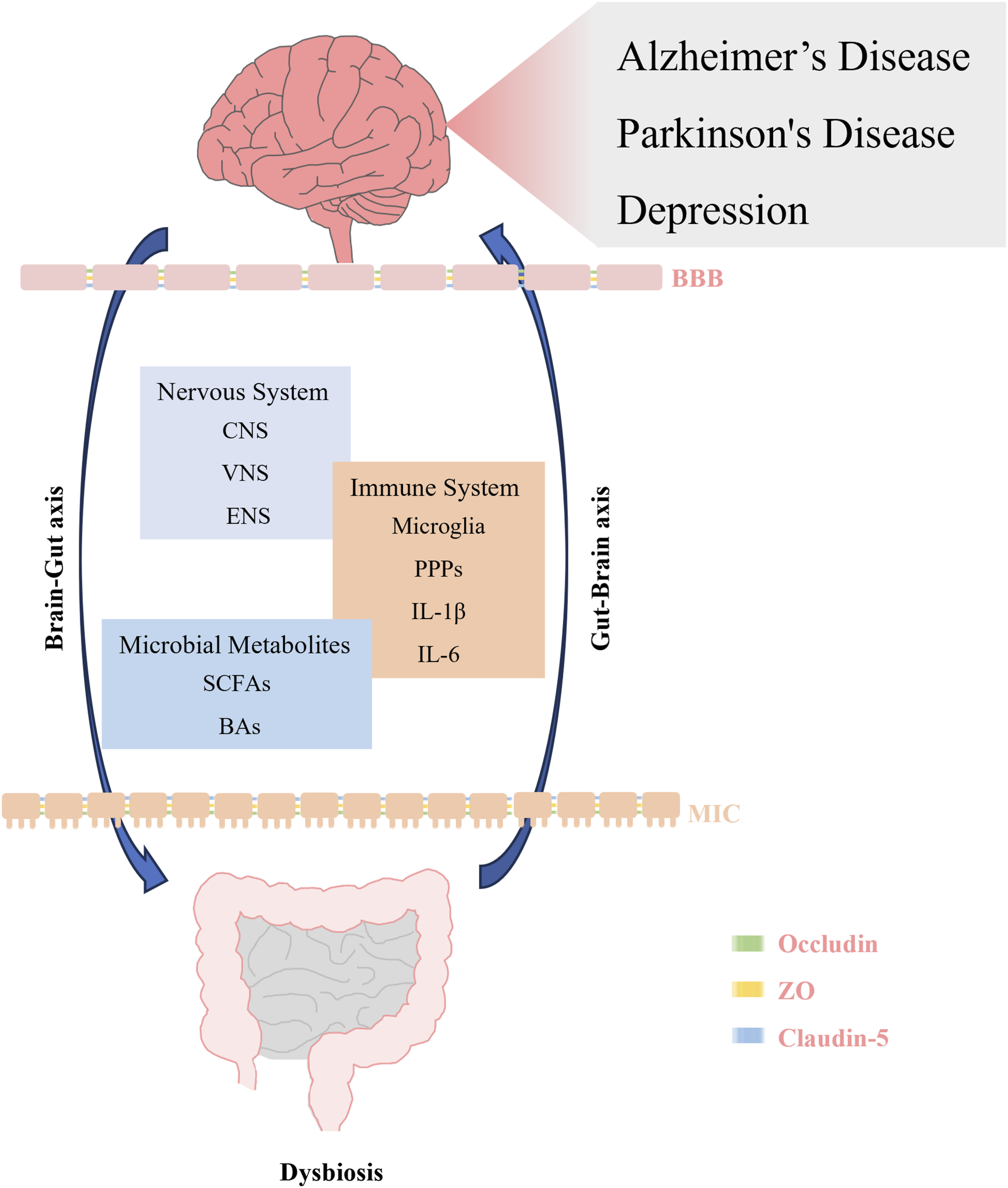
Alzheimer’s Disease
Alzheimer’s Disease (AD) is an age-related neurodegenerative condition of the central nervous system, characterized by irreversible impairments in learning and memory, progressive cognitive decline, as well as behavioral dysfunction. AD is pathologically marked by the presence of extracellular senile plaques formed by the deposition of β-amyloid (Aβ) protein and intracellular neurofibrillary tangles formed by hyperphosphorylated tau protein. 37 The Aβ42/Aβ40 ratio in cerebrospinal fluid is an indicator of amyloid load, inversely correlating with the amyloid deposition in the brain. P-tau is a marker of neurofibrillary tangles and positively correlates with the extent of tangles in the brain. The p-tau/Aβ42 ratio encompasses both aspects of AD pathology, such as neurofibrillary tangles and protein deposition. A higher ratio implies a more severe progression of AD. 38
Through 16S rRNA gene sequencing, Vogt et al unraveled that unlike healthy individual, AD patients exhibit a decreased abundance of Firmicutes and Actinobacteria, and an increased abundance of Bacteroidetes in their feces. Within Firmicutes, the genera SMB53 (family Clostridiaceae), Dialister, Clostridium, Turicibacter, and cc115 (family Erysipelotrichaceae) were less abundant, whereas the genera Blautia, Phascolarctobacterium, and Gemella were more abundant. The genera Bifidobacterium and Adlercreutzia were reduced among Actinobacteria. In Bacteroidetes, the genera Bacteroides and Alistipes displayed increased abundance. Within Proteobacteria, the genus Bilophila was more abundant in the feces of AD patients. 29 Intestinal microbiota induce the onset of diseases and predict disease severity. Vogt et al used Spearman’s rho statistical analysis to identify correlations between increased abundances of Bacteroides and Blautia genera and decreased abundances of SMB53 and Dialister genera in fecal samples from AD patients, and levels of cerebrospinal fluid (CSF) biomarkers (Aβ42/Aβ40, p-tau, and p-tau/Aβ42): bacterial abundances primarily negatively correlated with Aβ42/Aβ40 in CSF and positively correlated with p-tau and p-tau/Aβ42 in CSF. 29 Harach et al., in 2017 discovered that transgenic AD mice grown under germ-free conditions exhibited less Aβ in their brains compared to conventionally raised AD mice. 39 Through 16S rRNA gene sequencing, Liu et al observed that AD patients had reduced Firmicutes and increased Proteobacteria in their feces, unlike the control group. Interestingly, the abundance of Bacteroidetes significantly increased in the amnestic mild cognitive impairment (aMCI) group during the preclinical stage; however, there was no significant change in the AD group. 28 Unlike the aMCI group, the abundance of Bacteroidetes in the AD group significantly decreased, with similar trends in the changes of corresponding bacterial categories between the two groups. These results suggest that alterations in the gut microbiota of AD patients may result in pathophysiological changes in the brain and are associated with disease duration, particularly concerning Bacteroidetes bacteria. 28 Park et al discovered that AD patients treated with FMT significantly improved their cognitive function as assessed by MMSE, MoCA, and CDR scales. Moreover, AD scale scores further improved as well as the mood of patients upon continued FMT treatment after reinfection. 40
The aforementioned studies show that changes in the gut microbiota play a significant role in the onset and progression of AD. The inconsistencies in microbiota changes may be associated with the environment, region, and daily diet of the individuals. However, the underlying mechanisms remain to be explored.
Parkinson’s Disease (PD)
Parkinson’s Disease is a progressive, multisystem neurodegenerative condition caused by α-synuclein deposition in dopaminergic neurons of the substantia nigra in the brain. 41 This primarily involves Lewy bodies with α-synuclein and the loss of dopaminergic neurons in the substantia nigra, limiting voluntary movement. The major symptoms of Parkinson’s Disease include tremors, rigidity, and bradykinesia. 42 The pathological mechanisms involve neuroinflammation, 43 mitochondrial dysfunction, 44 and oxidative metabolism. 45 Subsequent investigations identified protein calcium homeostasis and endoplasmic reticulum stress as likely key mechanisms in the pathogenesis of PD. 46
Lin et al analyzed changes in relative abundance of microbiota through 16S rRNA gene sequencing. Consequently, unlike the normal population, the abundances of Tenericutes, Euryarchaeota, and Firmicutes at the phylum level were significantly reduced in PD patients. LEfSe analysis revealed that changes in microbiota abundance were linked to the duration of PD. In patients with PD for more than 5 years, Rikenellaceae and Deferribacteraceae were more abundant unlike those with a disease duration of less than 5 years. Conversely, the abundance of Phytobacterium significantly decreased. Early-onset PD patients (age of onset <50 years) had higher levels of Pasteurellaceae and Alcaligenaceae compared to late-onset PD patients (age of onset >50 years). At the genus level, early-onset PD patients had a significantly high abundance of Lactococcus, Facklamia, Clostridium, Sutterella, Faecalibacterium, Leptotrichia, and Haemophilus in their feces, whereas Comamonas and Anaerotruncus were more abundant in late-onset PD patients. 32 These results suggest that changes in gut microbiota abundance are associated with disease duration and represent a dynamic process. These changes are also attributed to individual lifestyle habits. 31
Through 16S rRNA gene sequencing technology, Zhao et al discovered that fecal microbiota transplantation (FMT) can improve the diversity and quantity of gut microbiota in a PD mouse model induced by oral rotenone (30 mg/kg) for 4 weeks.
Compared to the PD model group, the abundances of Akkermansia and Desulfovibrio significantly decreased, while Barnesiella, Butyricicoccus, and Helicobacter significantly increased after FMT administration. Furthermore, no significant differences were observed in microbiota abundance unlike the normal mouse group. 47
The aforementioned studies show that changes in gut microbiota are interconnected with the onset and progression of PD and these changes dynamically occur as the disease progresses.
Depression
Depression is a prevalent mental condition characterized by low mood, slow thinking, and reduced speech as well as movement. Its major pathological features involve neuroinflammation, 48 mitochondrial dysfunction,49,50 the HPA axis, 51 and more recently gut microbiota dysbiosis. 48
The microbial diversity and taxonomic composition of patients with mood disorders undergo significant changes. Chen et al utilized metaproteomics to analyze these changes and found that Firmicutes and Actinobacteria significantly increased in the feces of patients with depression compared to the control group, whereas Bacteroidetes and Proteobacteria also significantly increased. 34 Chung et al confirmed the aforementioned results by 16S rRNA gene sequencing of fecal samples from subjects. The relative abundance of Bifidobacterium, Blautia, and Clostridium XI significantly increased, whereas that of Prevotella and Sutterella significantly decreased. 36 According to a human case-control study by Cheung et al, the relative abundance of Anaerostipes, Blautia, Clostridium, Klebsiella, Lachnospiraceae incertae sedis, Parabacteroides, Parasutterella, Phascolarctobacterium, and Streptococcus were significantly increased in the feces of patients with depression compared to the healthy population. Conversely, Bifidobacterium, Dialister, Escherichia/Shigella, Faecalibacterium, and Ruminococcus were significantly decreased. 34 McGuinne et al reviewed 48 studies that used 16S rRNA gene sequencing and metagenomic methods to investigate changes in microbial abundance in the feces of patients with mental disorders (major depressive disorder, bipolar disorder, and schizophrenia). As a consequence, Bacteroides, Faecalibacterium, Prevotella, and Blautia exhibited differential changes in abundance among the cases. Most reports revealed lower relative abundance of Faecalibacterium, Coprococcus, Haemophilus, and Ruminococcus. Specifically, higher abundances of Alistipes and Parabacteroides and lower abundances of Prevotella were observed in the feces of patients with major depressive disorder. 19 Zhang et al used 16S rRNA sequencing to discover changes in gut microbiota in the feces of LPS-induced depression model mice compared to the control group. At the phylum level, Firmicutes were significantly increased, whereas Bacteroidetes and Proteobacteria were significantly decreased. At the genus level, Lactobacillus was significantly increased, whereas Bacteroides, Parabacteroides, Muribaculum, Butyricimonas, Erysipelatoclostridium, and Enterococcus were significantly decreased. 52
Cai et al observed that the fecal microbiota of the model mice had significant changes following fecal microbiota transplantation from normal mice into chronic unpredictable mild stress (CUMS)-induced depression model mice. We observed a significant increase in Firmicutes and a significant decrease in the relative abundance of Bacteroidetes and Proteobacteria. The mice displayed depression-like behaviors, induced inflammatory responses, and had significantly increased plasma levels of LPS and IL-6. Additionally, the expression of central neurotransmitters was significantly reduced, including 5-HT and GABA in the hippocampus. 53 Liu et al found that germ- free normal rats also had depression-like behaviors after receiving fecal microbiota from depressed patients, 54 suggesting that gut microbiota modulate depression development and can be a promising therapeutic measure. Nonetheless, the mechanisms by which gut microbiota exerts its antidepressant effects remain to be further explored.
Microbial Regulation Via the Gut-Brain Axis
Regulation Through the Nervous System
The gut microbiota and the brain can engage in bidirectional regulation via the gut- brain axis. This process involves the vagus nerve system (VNS), the enteric nervous system (ENS), and the CNS.1,55,56 The vagus nerve is a primary component of the parasympathetic nervous system, responsible for monitoring physiological homeostasis. It connects the emotional and cognitive brain functions with peripheral gut functions.
The abdominal vagus nerve afferents include mechanoreceptors, chemoreceptors, and tension receptors located in the mucosa of the stomach and proximal small intestine.
57
The afferent nerves do not pass via the intestinal epithelial cell layer but are distributed on the surface of the intestinal lumen without directly contacting the gut microbiota
58
(Figure 2). Signal transmission occurs through receptors on the intestinal epithelial cells and the secretions as well as metabolites of the gut microbiota, including NOD domain-like receptors (NLRs) and Toll-like receptors (TLRs).
59
Active components secreted by the microbiota include gamma-aminobutyric acid (GABA), dopamine, and acetylcholine,
60
and metabolites include short-chain fatty acids (SCFAs) and bile acids (BAs).
61
GABA is a major inhibitory neurotransmitter in the mammalian central nervous system, with neuroprotective properties. It regulates synaptic transmission, promotes neuronal development and relaxation, and prevents insomnia and depression by suppressing neuron degeneration.
13
The cell bodies of vagal sensory afferents are located in the nodose ganglion and send information to the nucleus tractus solitarii (NTS).
57
The NTS subsequently projects vagal sensory information to brain regions including the locus coeruleus, rostral ventrolateral medulla, amygdala, and thalamus in the central nervous system, hence influencing the expression and release of neurotrophic factors like brain-derived neurotrophic factor (BDNF). BDNF is involved in neuroplasticity, neuron survival, new synapse formation, dendritic branching, as well as regulation of excitatory and inhibitory neurotransmitter spectra, thereby affecting cognitive functions including learning and memory.62–64 Gut microbiota involvement in the regulation of the gut-brain axis through the nervous system.
Unlike the normal control group, O’Leary et al found that vagotomy significantly reduced BDNF mRNA expression in the dentate gyrus, CA1, and CA3 regions of the hippocampus, as well as cell proliferation in the dentate gyrus and the number of doublecortin-positive neurons. 65 Elsewhere, Shin et al. found that, compared to the chronic restraint stress-induced depression model group, vagus nerve stimulation significantly improved depression-like behaviors in model mice, reduced immobility time in the forced swim test (FST), increased climbing time, and significantly increased BDNF protein levels and 5-HT1B receptors in the hippocampus. 66 Unlike LPS (0.5 mg/kg, i.p.)-induced depression model mice, Zhang et al found that subdiaphragmatic vagotomy (SDV) significantly reduced immobility time in the FST, altered the relative abundance of gut microbiota, significantly decreased the relative abundance of Firmicutes, and significantly increased the relative abundances of Bacteroidetes and Actinobacteria. Moreover, SDV downregulated the expression of the synaptic protein PSD-95 and the AMPA receptor subunit GluR1 in the medial prefrontal cortex. Interestingly, following vagus nerve injury, a subsequent LPS injection failed to induce depression-like behaviors in normal mice. Additionally, it did not alter gut microbiota composition or affect synaptic protein expression. 52 Wu et al found that recombinant human atrial natriuretic peptide could relieve LPS-induced hippocampal inflammation and cognitive impairment via the gut-brain axis mediated by the subdiaphragmatic vagus nerve. 67 The bidirectional regulatory role of the vagus nerve between the microbiota, gut, and brain is a promising treatment approach for neuropsychiatric disorders.
Unlike the VNS, the ENS operates independently. The ENS primarily comprises two bundles of ganglia, i.e., one located beneath the intestinal epithelium (the submucosal plexus) and the other between the layers of the intestinal smooth muscle (the myenteric plexus) (Figure 2). 68 Interestingly, following vagus nerve injury, a subsequent LPS injection failed to induce depression-like behaviors in normal mice. Additionally, it did not alter gut microbiota composition or affect synaptic protein expression. 69 Sox2 is a transcription factor known as a marker of ENS progenitor cells and is majorly expressed by enteric glial cells in adult animals. HuC/D+/Sox2 + double labeling is used to examine enteric neurogenesis. 70 The ENS operates independently of direct regulation by the CNS but interacts with the vagus nerve system (VNS), along with the sympathetic and parasympathetic nervous systems, to coordinate gastrointestinal functions such as motility of the small and large intestines. This regulation alters gastrointestinal motility and fluid secretion, causing clinical phenomena such as dysphagia, gastroparesis, chronic intestinal pseudo-obstruction, constipation, and fecal incontinence. 69
Barberio uncovered that the overall prevalence of anxiety symptoms in patients with inflammatory bowel disease (IBD) stands at 32.1%, and the overall prevalence of depressive symptoms at 25.2%. Additionally, it was discovered that IBD patients often experience concurrent anxiety and depression symptoms. Unlike the general population, individuals with IBD are more likely to develop depression. 71
Zhao et al. discovered that rotenone (30 mg/kg)-induced Parkinson’s Disease (PD) model mice have symptoms of intestinal dysfunction, characterized by significantly shortened intestinal transit distance and colon length. 47 Vicentini et al administered a mixed antibiotic regimen (containing ampicillin 1 g/L, neomycin 1 g/L, vancomycin 0.5 g/L, and metronidazole 1 g/L) to normal mice for 4 weeks. As a consequence, they found an increase in the weight of the cecum and a significant reduction in the number of bacteria in the cecum despite no significant change in body weight in the model group mice. 72 Zhao et al developed an antibiotic-induced bacterial depletion mouse model (Abx) in 2021 to study the effects of microbial imbalance on the gut. 70 Compared to the control group, small intestinal motility was markedly reduced, and the integrity of the intestinal mucosal barrier was significantly compromised, as indicated by FSA leakage into the plasma. These results highlight that microbial imbalance impairs intestinal motility and enhances permeability. Following antibiotic treatment, there was a loss of HuC/D+ neurons and a reduction in Tuj1+ neuronal fiber density in the submucosal and myenteric plexuses of the ileum and colon. Additionally, several enteric glial cells in the ileal myenteric plexus were decreased, and cell bodies were reduced in size. This suggests that microbial dysbiosis increases enteric neuron loss, decreases enteric glial cells in the ileal myenteric plexus, and alters their function. 70 However, the mechanisms by which the gut microbiota influences the progression of neuropsychiatric disorders via the ENS pathway remain unstudied.
In summary, the gut microbiota can influence the onset and progression of neuropsychiatric disorders via the VNS. Disruption of the vagus nerve elicits changes in neurotransmitters and neurotrophic factors, reducing neuronal proliferation and maturation. It can also stimulate pro-inflammatory changes in microglia within brain regions, causing a release of pro-inflammatory cytokines and neuroinflammation. Conversely, vagus nerve stimulation can improve disease states, and the enteric nervous system also modulates gut function and alleviates gastrointestinal complications. However, the mechanisms underlying these effects remain unclear. Further investigations are essential to elucidate how the gastrointestinal system promotes and/or is altered by these disorders. This is geared towards developing more effective treatments for gastrointestinal comorbidities.
Immunoregulation
Gut microbiota can influence the metabolism and immune status of the host, thereby affecting neuronal pathways in the gut and CNS. 73 The gut microbiota interacts with the peripheral immune system through host factors to regulate local immunity within the gut. These host factors include bacterial cell wall components (peptidoglycans and LPS), microbial-derived metabolites (SCFAs, secondary bile acids, and amino acid metabolites), and other bioactive molecules (microbial-associated molecular patterns). Through the circulatory system, these interactions also influence microglia in the brain, thereby regulating the central nervous system. 74
Microglia are resident macrophages in the central nervous system with similar roles to peripheral macrophages. Microglia constantly monitor the brain for harmful signals, both internal (such as damaged DNA, RNA, or proteins) and external (like bacteria or viruses). These signals are primarily recognized by pattern recognition receptors (PRRs) located on the microglial cell surface. A series of receptor-ligand interactions causes the release of inflammatory mediators, causing neuroinflammatory responses. 75 PRRs include TLRs, NLRs, and TREMs, whereas inflammatory mediators include IL-1β, IL-6, TNF-α, COX2, NOS2, and ROS. 76 The release of inflammatory factors triggered by LPS can occur via various pathways, such as MyD88-dependent and MyD88-independent pathways. 76
Zhang et al discovered that stimulation with 50 μg/mL lipoteichoic acid (LTA) in primary mixed cultures (85 ± 5% neurons, 7 ± 3% astrocytes, and 5 ± 3% microglia) for 3 days significantly increased the extracellular inflammatory factors TNF-α, IL-1β, and NO, with excessive proliferation of microglia and approximately 40 ± 5% of neuron death due to apoptosis or necrosis. Selective microglia elimination using L-leucine methyl ester (50 mM) without reducing the number of neurons and astrocytes significantly improved neuronal death in LTA-stimulated primary mixed cultures. Delayed video recordings revealed that LTA stimulation improved the capacity of microglia to phagocytize neuronal cells. Phagocytosed neurons appeared normal without signs of nuclear condensation. This shows increased microglial phagocytic activity under acute inflammatory conditions causing phagocytosis of normal neurons and a reduction in neuronal numbers. 77 Neuronal death, a major pathological hallmark of neurodegenerative disorders, exacerbates the progression of neuropsychiatric disorders. 78
Braniste et al observed that unlike the normally raised adult mice (8-10 weeks old), adult germ-free mice (8-10 weeks old) displayed a significantly reduced expression of tight junction proteins occludin and claudin-5 in the frontal cortex, hypothalamus, and striatum tissues. Furthermore, germ-free mice exhibited a significant reduction in the number of intact tight junction structures within the striatum. Intravenous injection of monoclonal anti–N-methyl-D-aspartate receptor antibody (R4A, 250 μg) in germ-free adult mice revealed abnormal neuronal markers in the hypothalamic C1 region, with cytoplasmic condensation and cellular shrinkage, and a significant decrease in neuronal survival rate. This was accompanied by the penetration of Evans blue dye into the brain parenchyma. In contrast, comparable procedures in conventionally raised mice revealed no significant alterations in neuronal cells, with Evans blue dye remaining confined to blood vessels without penetrating the brain parenchyma. Subsequent transplantation of fecal microbiota from conventionally raised adult mice into germ-free adult mice significantly increased occludin and claudin-5 expression in the hypothalamus, striatum, and frontal cortex 14 days post- transplantation, unlike the control group. This transplantation restricted Evans blue dye penetration into the brain parenchyma, suggesting that intestinal microbiota depletion may be associated with increased blood-brain barrier permeability. 79
The mucosal immune system (MIS), comprising lymph nodes, the lamina propria, and epithelial cells, forms a protective barrier for intestinal integrity. 80 As an important component of the MIS barrier, intestinal epithelial cells extensively express classical pattern recognition receptors (PRRs), including NLRs and TLRs. These receptors mediate communication and signal transduction with microorganisms through the initial recognition of lipopolysaccharides (LPS), flagellin, bacterial peptidoglycan, and cell wall lipoproteins. 59 Upon disruption of the gut microbiota, toxic digestive metabolites, bacterial toxins, and small molecules breach the intestinal barrier and “leak” into the bloodstream. Once these substances cross the blood-brain barrier (BBB) into the central nervous system, they activate immune cells, including microglia, triggering an immune response within the brain. This activation induces neuroinflammation, exacerbating the development and progression of neuropsychiatric disorders. 81
Unlike the general population, Ohlsson et al found that patients with recent suicidal tendencies (rSA) and non-suicidal major depressive disorder (nsMDD) had significantly decreased levels of zonulin, a tight junction protein of intestinal epithelial cells, and significantly increased levels of intestinal fatty acid-binding protein (I-FABP), a marker of intestinal cell damage, in their plasma. The levels of IL-6 positively correlated with I-FABP and negatively correlated with zonulin. Additionally, I-FABP levels positively correlated with scores on the Montgomery-Asberg Depression Rating Scale (MADRS) and the Suicide Assessment Scale (SUAS). This indicates that depression is linked to inflammatory responses and pathological features of intestinal barrier damage, which correlate with disease progression. 82 Zhao Zhe et al discovered that, unlike the control group, rotenone-induced PD model mice exhibited dysbiosis of the fecal microbiota and compromised BBB as well as intestinal barriers. In these model animals, the levels of ZO-1, occludin, and claudin-5 in the substantia nigra (SN) and colon tissues were significantly reduced, causing leakage of inflammatory molecules and significantly increased serum levels of LPS, LBP, TNF-α, IL-1β, and IL-6. 47
Gut microbiota dysbiosis induces a release of inflammatory factors, which disrupt the MIS and the BBB. Bacterial metabolites, other active components, and inflammation-related factors enter the central nervous system, stimulating microglial cells and inducing neuroinflammatory responses, thereby exacerbating disease pathology. This shows that gut bacteria can disrupt the development and progression of neuropsychiatric disorders by regulating the immune system.
Gut-Microbiota-Derived Metabolites
Gut microbiota can regulate various metabolic reactions in the host and generate metabolites including short-chain fatty acids, bile acids, and choline which are vital for host health. Complex carbohydrates consumed daily (e.g., dietary fiber) can be digested by gut microbiota and fermented in the colon to generate neuroactive short-chain fatty acids (SCFAs), including butyrate, acetate, and propionate. 56
Liang et al administered Lycium barbarum polysaccharides (LBP, 200 mg/kg) to normal mice. Consequently, the total SCFA content in the feces and colon of the LBP group was significantly higher than that of the control group. Through 16S rRNA analysis, LBP significantly increased the relative abundance of Lactobacillaceae, with the abundance of Lactobacillus and Lactococcus increasing from 22.49% to 52.90%, whereas the relative abundance of Ruminococcus and Mucispirillum decreased. 83 Wang et al discovered that a composite polysaccharide (Lycium barbarum polysaccharide: Poria cocos polysaccharide: Lentinus edodes polysaccharide, 1:1:1) significantly increased the relative abundance of Bifidobacterium, Lactobacillus, Allobaculum, and Oligella in the feces of 21-day-old male SD rats, while significantly decreasing the abundance of Enterococcus; Heatmap and correlation network analysis revealed that changes in various microbial abundances were linked to metabolic changes. Fumarate in the urine, an intermediate of the tricarboxylic acid cycle positively correlated with Lactobacillaceae and Lactobacillus. Three SCFAs (acetate, butyrate, and propionate) in the feces positively correlated with Alcaligenaceae and Sutterella, whereas butyrate positively correlated with Lactobacillaceae and Lactobacillus. Glycolytic products (pyruvate and lactate) negatively correlated with Rikenellaceae, whereas pyruvate negatively correlated with Lactobacillaceae and Lactobacillus. Further, PICRUSt analysis confirmed that dietary intervention influenced the biological metabolic pathways in young rats, indicating that changes in microbial categories can influence the biological metabolic processes of the body, including energy metabolism and SCFA-related metabolism. 84
Short-Chain Fatty Acids (SCFAs)
SCFAs are major products of dietary fiber fermentation in the colon and are saturated fatty acids with one to six carbon atoms. Approximately 500-600 mmol of SCFAs are produced in the gut daily depending on the fiber content in the diet. 85 The major phyla in the gut-producing SCFAs are Firmicutes (Gram-positive), accounting for 60% of the total gut microbiota, and Bacteroidetes (Gram-negative), accounting for 20%. 86 Different phyla produce different SCFAs: Firmicutes (e.g., Clostridia, Enterococci, Lactococci, Staphylococci) mainly produce butyrate, whereas Bacteroidetes (e.g., Bacteroides, Prevotella) mainly produce acetate and propionate.87,88 Acetate, propionate, and butyrate are the most abundant SCFAs in the human body and the most prevalent anions in the colon. 89 A small portion of propionate and acetate are effectively metabolized in the liver to produce glucose as substrates for the tricarboxylic acid cycle, producing energy. A small fraction of SCFAs exists in the gut in non-ionized forms and directly crosses the intestinal epithelial barrier; however, most exist in ionized forms and are transported across the mucosa primarily through two receptor-mediated active transporters: monocarboxylate transporter 1 (MCT-1) and sodium-coupled monocarboxylate transporter 1 (SMCT-1). 86
Intracellular butyrate and propionate can suppress the activity of histone deacetylases (HDACs) and promote hyperacetylation of histones. Based on their location and function, HDACs have many subtypes and are classified into four classes (I, II, III, and IV). HDAC1 and HDAC2 are involved in neurodegenerative changes, whereas HDAC3-11 and class III HDACs have beneficial neuroprotective effects. HDACs can upregulate glutamate, tau phosphorylation, and glial fibrillary acidic protein, but downregulate BDNF, heat shock protein 70, and gelsolin, 90 hence affecting brain development and the progression of various neuropsychiatric disorders including depression, schizophrenia, and Alzheimer’s Disease. 91 SCFAs regulate immune and inflammatory responses by activating free fatty acid receptors type 2 (FFA2), FFA3 receptors, and G-protein coupled receptors (GPRs), or by suppressing the activity of HDACs, hence regulating the MAPK signaling pathway and NF-κB activation. 92
Besides their anti-inflammatory and HDAC-inhibiting effects, SCFAs protect the integrity of the blood-brain barrier (BBB). Mechanistically, the above protective effects of SCFAs on the BBB integrity are postulated to be mediated by G-protein coupled receptor signaling pathways, HDAC inhibition, and the acetylation of histones and non- histone proteins, as well as interactions between these signaling pathways and transcription factors NF-κB and Nrf2.93,94 Previous studies have shown that microbial deficiency can disrupt BBB integrity. Braniste et al demonstrated that administration of SCFA-producing bacteria, C. tyrobutyricum (mainly producing butyrate) or B. thetaiotaomicron (mainly producing propionate and butyrate), to germ-free adult mice caused marked inhibition of the Evans blue dye penetration into the brain parenchyma.
Oral administration of sodium butyrate (1 g/kg) to these mice induced similar effects, causing a significant upregulation of occludin protein expression in the prefrontal cortex and hippocampus. Furthermore, administering C. tyrobutyricum or sodium butyrate alone increased histone acetylation in the brain, but without significant changes. 79
Wenzel et al found that a high concentration (500 μM) of SCFA mixture (acetate, propionate, butyrate, valerate, and formate in a ratio of 16:8:8:1:1) reduced the levels of inflammatory cytokines IL-1β, MCP-1, and TNF-α released by LPS + INF-γ induced THP-1 monocytes in a pro-inflammatory state in vitro; a low concentration (25 μM) did not reduce cytokine levels, suggesting that the anti-inflammatory effects of SCFAs may be concentration-dependent. 95 Unlike the model group, Li et al found that pre- treatment with acetate (10 mM) for 16 hours significantly reduced the levels of extracellular pro-inflammatory cytokines upon the stimulation of human umbilical vein endothelial cells (HUVECs) with LPS (1 mg/mL) or TNF-α (1 ng/mL). Co-treatment with GPR41 antagonist SHB (5 mM) and/or GPR43 antagonist GLPG (0.1 mM) significantly reduced the inhibitory effect of acetate on LPS-induced IL-6 and IL-8. Pre-treatment with propionate (10 mM) and butyrate (5 mM) for 24 hours also significantly reduced IL-8 levels, increased the expression of vascular cell adhesion molecule-1 (VCAM-1), and the activity of HDAC. However, SHB or GLPG treatment did not improve the inhibitory effects of propionate and butyrate on IL-8, whereas HDAC inhibitor trichostatin A (TSA) significantly suppressed VCAM-1 expression and improved peripheral blood mononuclear cell (PBMC) adhesion to HUVECs. This shows that GPR41/43 activation mediates the effects of acetate on IL-6 and IL-8 production in an inflammatory state; butyrate and propionate influence IL-6 production by inhibiting HDAC activity, affecting IL-8 production, VCAM-1 expression, and PBMC adhesion to endothelial monolayers. 96
Liu et al found that SCFA-producing Clostridiaceae, Lachnospiraceae, and Ruminococcus in the Firmicutes phylum were significantly reduced in AD patients. In patients with amnestic mild cognitive impairment (aMCI), the relative abundance of Clostridiaceae and Lachnospiraceae was significantly reduced, whereas Ruminococcaceae did not show significant differences. The extent of microbial changes positively correlated with cognitive status parameters indicating AD, including MMSE and MoCA scores. 28 These studies suggest that the anti-inflammatory effects of SCFAs are concentration-dependent and may prevent endothelial inflammation by activating GPR41/43 and suppressing HDAC. Nevertheless, whether the ameliorative effects of SCFAs on psychiatric disorders are mediated by GPR or HDAC in neuroinflammation pathways remains to be further explored.
Bile Acids
Bile acids (BA) are amphipathic steroid molecules derived from cholesterol metabolism. They act as multifunctional signaling molecules within the body, regulating their synthesis and lipid, glucose, and energy metabolism via interactions with various receptors, including the Takeda G-protein coupled receptor 5 (TGR5), Farnesoid X Receptor (FXR), and Sphingosine 1-phosphate Receptor 2 (S1PR2).97,98 These BA receptors, including TGR5, 99 FXR, 100 and S1PR2 98 are not only present in liver and intestinal cells but are also in neurons, astrocytes, and microglia within the brain. This indicates that BAs can cross the blood-brain barrier (BBB) and bind to receptors on brain cells to convey signals.
The synthesis of BA occurs via two pathways and involves at least 17 enzymes. The classical (or neutral) pathway, which produces at least 75% of BAs, is initiated by the rate-limiting enzyme cholesterol 7α-hydroxylase (CYP7A1), primarily producing cholic acid (CA) and chenodeoxycholic acid (CDCA). The alternative (or acidic) pathway, initiated by sterol 27α-hydroxylase (CYP27A1) and catalyzed by oxysterol 7β-hydroxylase (CYP7B1), mainly produces CDCA.
101
Primary BAs undergo conjugation with glycine in humans or taurine in mice through the enzymatic actions of bile acid-CoA synthase (BACS) and bile acid-CoA:amino acid N-acyltransferase (BAAT). Once conjugated, these BAs are then released into the intestine postprandially. These primary conjugated BAs are converted to secondary BAs including deoxycholic acid (DCA), lithocholic acid (LCA), and ursodeoxycholic acid (UDCA) via 7α- dehydroxylation by intestinal bacteria.
102
Bacterial bile salt hydrolases (BSHs) deconjugate primary BAs in the intestine, forming free BAs which are then 7α- dehydroxylated to produce secondary BAs (DCA and LCA) (Figure 3). Gram-positive bacteria including Clostridium, Enterococcus, Bifidobacterium, and Lactobacillus secrete BSHs; Bacteroides is the only Gram-negative genus that secrete BSHs.
103
In the terminal ileum, most unconjugated BAs (including a small fraction of free and secondary BAs) are reabsorbed into enterocytes via the apical sodium-dependent bile acid transporter (ASBT) and returned to the liver via the portal circulation. The gut microbiota regulates the gut-brain axis through bile acids.
Genes encoding the cytochrome P450 enzymes essential for the alternative BAs synthesis pathway (CYP8B1, CYP27A1, and CYP7B1) are also expressed in the brain, suggesting that primary BAs can be locally synthesized within the brain.104,105 Moreover, BAs can be reabsorbed from peripheral circulation into the brain. Research has demonstrated that non-conjugated BAs, such as CA, CDCA, and DCA, can passively diffuse across the BBB due to their hydrophobic properties at low concentrations, allowing them to reach the brain. 106 Higashi et al detected the presence of CA, CDCA, and DCA in rat brain tissue using liquid chromatography/electrospray ionization-tandem mass spectrometry and chemical derivatization methods; their levels correlated with those in the serum, despite brain tissue levels being lower than serum levels. 107
Receptors for BAs are present on the BBB. Ursodeoxycholic acid (UDCA) and tauroursodeoxycholic acid (TUDCA) can physiologically cross the BBB; however, most BAs are restricted to the peripheral circulation. Because of their large structure and amphipathic nature, conjugated BAs may require transport proteins on the BBB, including organic anion-transporting polypeptides (OATPs), 108 ATP-binding cassette (ABC) transporters, bile salt export pump (BSEP), 109 and multidrug resistance- associated proteins (MRP1 and MRP4) 110 to enter the brain. However, no direct evidence shows that these conjugated BAs cross the BBB into the brain via BA transporters (Figure 3).
CDCA and TUDCA have been extensively studied in neuropsychiatric disorders and have demonstrated enhanced neuroprotective and cognitive properties. Ramalho et al found that 100 μM TUDCA can counteract Aβ-induced reductions in postsynaptic density protein-95 (PSD-95) in neurons and astrocytes, improve the frequency of spontaneous miniature excitatory postsynaptic currents, and increase dendritic spine density, which helps suppress neuronal death and reduce astrocyte reactivity as well as neurotoxicity. 111 Dionisio et al discovered that intraperitoneal injection of TUDCA (500 mg/kg) every 3 days for 3 months significantly reduced Aβ deposition in the prefrontal cortex and hypothalamus of 7-month-old APP/PS1 transgenic mice compared to the control group. TUDCA decreased levels of Aβ1-40 and Aβ1-42, as well as amyloid precursor protein (APP) and its β-secretase cleavage products sAPP-β and CTF-β. This suggests that TUDCA disrupts the amyloidogenic processing of APP and Aβ production. Additionally, TUDCA alleviated glial cell hyperplasia induced by amyloid deposition and had anti-inflammatory properties by reducing levels of pro- inflammatory cytokines IL-1β, IL-6, and TNF-α. 112 Rosa et al uncovered that TUDCA administration mitigated gait disturbance and tremors in MPTP-induced PD mice, improved motor dysfunction, and prevented the reduction of tyrosine hydroxylase- positive fiber density in the striatum and ATP levels in the midbrain. Furthermore, TUDCA increased the phosphorylation level of the energy sensor AMPK, indicating that it could prevent MPTP-induced dopaminergic neurodegeneration. 113 Furthermore, TUDCA modulated MPTP-induced nigrostriatal dopaminergic neurodegeneration. This neuroprotective effect may be mediated by the Akt signaling pathway, involving the phosphorylation of Bad and activation of NF-κB. 114
Microbial Interventions in Neurological Disorders
Therapeutic effects of FMT, antibiotics, and probiotics on neuropsychiatric disorders.
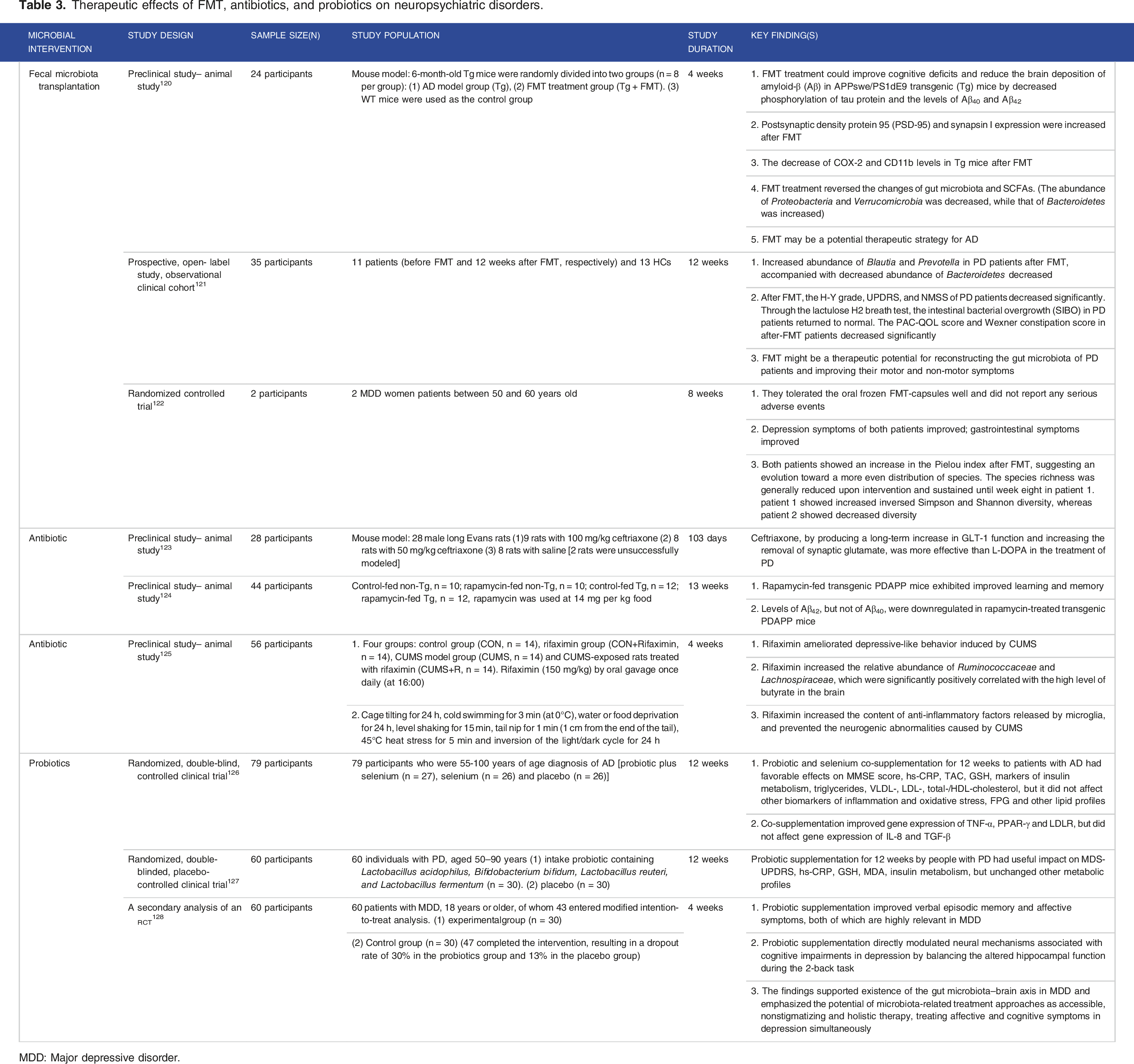
MDD: Major depressive disorder.
Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) involves transferring healthy microbiota into a diseased host to replace and proliferate, generating bioactive metabolites to treat diseases. 115 First used against Clostridium difficile infections, 116 the efficacy and persistence of transplanted microbiota in the host are influenced by factors such as the inflammatory state of the host and microbial composition.95,117 One key benefit of FMT is the absence of major side effects, even in high-risk patients. 118 FMT has recently demonstrated potential in treating Alzheimer’s Disease (AD), Parkinson’s Disease (PD), and depression. 119
Compared to untreated APP/PS1 transgenic AD model mice, mice administered with FMT extracted from wild-type mice showed improved cognitive function, reduced Aβ plaques, soluble Aβ40 and Aβ42, upregulated expression of proteins involved in the regulation of synaptic plasticity (e.g., PSD-95 and synaptophysin), and increased the composition of beneficial butyrate in the gut. 120 Several investigations have shown that FMT from elderly wild-type mice impaired spatial learning and memory in young wild- type mice, 129 whereas it improved age-related symptoms in elderly mice, 130 suggesting that the donor’s age significantly influences the effects of FMT. However, these findings need to be further investigated in human trials. In PD model mice, FMT derived from normal mice was found to enhance the balance in gut microbiota and PD symptoms via modulating the TLR4/MyD88/NF-κB pathway, thereby enhancing motor function. 47 Elsewhere, application of the FMT obtained from elderly to young animals induced hippocampal astrocyte proliferation, suppressed neurogenesis and activated neurons, accompanied with hippocampal-dependent memory dysfunction. Another study found that stimulating the vagal signaling in elderly animals abolished the negative impact on young animals’ hippocampus while improving elderly hippocampal function. 131 Other scholars have reported that administration of FMT from normal mice into AD model mice suppressed epithelial barrier loss, chronic gut, and systemic inflammation as well as other pathological features (e.g., reduced Aβ plaques and neurofibrillary tangles, reduced glial reactivity) and reversed abnormal gene expression in colonic macrophage activity and circulating inflammatory monocytes. 132
These findings from experiment on animal models and human studies highlight the therapeutic effects of FMT. A patients with PD demonstrated elevated levels of Akkermansia muciniphila and Prevotella following the application of FMT, leading to effective alleviation of motor and non-motor symptoms. 121 In two cases of women with depression, FMT intervention not only alleviated depression symptoms but also relieved constipation, but one patient experienced a relapse of constipation after four weeks, whereas the therapeutic effect was sustained for eight weeks in the other patient. 122 This phenomenon may be ascribed to individual differences among patients. Although the clinical sample size is small, these results suggest that FMT can treat neuropsychiatric disorders and further studies are needed to confirm this hypothesis.
Antibiotics
Antibiotics, as frequently used drugs to control infections, have recently been demonstrated to prevent the occurrence of neuropsychiatric disorders induced by microbial imbalance by conferring neuroprotection, anti-inflammation, anti-tau, anti- amyloid, and anti-apoptotic effects.133–135 Some of the widely studied anti-biotics are β- lactam and macrolide.
Mechanistically, the β-lactam antibiotics target glutamate transporter 1 (GLT-1) to prevent excitotoxicity of neurons. 136 Ceftriaxone (100 mg/kg) was reported to upregulate the expression level of GLT-1, thereby enhancing synaptic glutamate clearance, alleviating gait abnormalities in PD model animals, with the therapeutic effects lasting for 28 days post-treatment without causing significant toxicity. 123 On the other hand, ceftriaxone treatment prevented memory impairment, improved cognitive function, and increased neuronal density in AD model rat. 137 Rapamycin, a macrolide and mTOR inhibitor, decreased the formation of Aβ plaques and microglial activation, tau phosphorylation, but its Aβ-lowering effects were detected in the early stages of the disease.138,139 Further evidence from animal models has shown that rapamycin inhibits mTOR expression, reduces Aβ42 levels and prevents AD-like cognitive deficits. 124
Several other antibiotics were exerted therapeutic effects on neuropsychiatric disorders. Rifampicin inhibits the production of Aβ and inflammatory cytokines in AD animal models. 140 Rifaximin (150 mg/kg) treatment was found to alleviate CUMS-induced depressive behaviors, increasing Ruminococcaceae and Lachnospiraceae abundance in feces, accompanied with elevated levels of butyrate in the brain. It also mitigated microglial morphological changes, improved the secretion of anti- inflammatory cytokines, and prevented CUMS-induced neurogenic abnormalities. 125 Tetracyclines exert anti-amyloid, anti-inflammatory, antioxidant, and anti-apoptotic activities, making them ideal treatments for PD, but their safety needs to be confirmed.141,142
Antibiotics may potentially alter microbiota composition, suggesting that they are important drugs for improving the recovery of neuropsychiatric disorders induced by gut dysbiosis. However, they are also associated with systemic toxicity, may disrupt gut bacterial balance, 143 reduce microbial diversity, alter metabolic activity, and increase the risk of antibiotic resistance, triggering the side effects such as like antibiotic- associated diarrhea and recurrent Clostridium difficile infection. 144 This implies that antibiotics should be applied with caution.
Probiotics
Probiotics are live, non-pathogenic microorganisms with diverse benefits when administered in optimal quantities. 145 Common probiotics include Bifidobacterium, Lactobacillus, Bacillus, Streptococcus, and Enterococcus146,147 which are known to improve AD and PD pathology.127,148,149 Probiotics may improve the composition of gut microbiota, reduce pathogen colonization, enhance epithelial cell proliferation and differentiation, and strengthen the gut barrier, which modulate the development of immune responses. 150 The SCFAs produced by probiotics are known to enhance anti- inflammatory and neuroprotective effects, cross the BBB, modulate CNS immune cell activity, the production of inflammatory cytokines, influence the BBB integrity and neurogenesis, to improve brain health.146,151 Studies have demonstrated to stimulate neurotransmitter synthesis and release, altering BDNF levels, synaptic plasticity, and neuronal function. 152
Chronic application of Lactobacillus rhamnosus in adult male BALB/c mice with intact vagal nerves, resulted in a reduction of anxiety, depression, and stress responses by modulating central GABAAα2, highlighting the involvement of the vagal nerve in regulation of the gut-brain axis. 58 Bacillus can convert L-tyrosine to L-DOPA, a dopamine precursor, which then matures to dopamine. 153 Lactobacillus plantarum DP189 mitigates the MPTP-induced PD in mice by regulating oxidative stress, inflammation, and gut microbiota, thereby inhibiting α-synuclein aggregation. 154 Bifidobacterium breve CCFM1067 was reported to suppress dopamine neuron death and neurotransmitter loss in MPTP-induced PD mice, increase acetate and butyrate levels, which improved motor function. 155
Aβ plaques are produced through the cleavage of amyloid precursor protein (APP) β and γ-secretase. 156 The AppNL-G-F mice produce excessive levels of Aβ without overexpressing APP, which resulted in Aβ deposition at two months and memory impairment by six months, making them an ideal AD model. 157 Probiotic Bifidobacterium breve reduces Aβ production, mitigates microglial activation, decreases pro-inflammatory cytokine mRNA expression, inhibits neuroinflammation, and delays memory impairment which may prevent the occurrence of AppNL-G-F mice memory deficits by enhancing ADAM10 levels and downregulating hippocampal Aβ. 158 Prolonged probiotic use may have profound effects on normal brain activity and alleviate depressive behaviors in the model animals.159–162 Bifidobacterium breve CCFM1025 downregulates the pCREB-c-Fos pathway, upregulates BDNF expression, SCFAs, and 5-HTP levels, ameliorating gut dysbiosis as well as depressive and anxiety behaviors in chronic stress-induced depression mice. 163
Although probiotics hold potential in mitigating the progression of neuropsychiatric disorders, their safety profile warrants careful consideration. Potential risks include the induction of bacteremia, the horizontal transfer of antibiotic resistance genes, and the production of enzymes that may compromise the effectiveness of concurrent treatments.159,164 Being live microorganisms, they require proper storage conditions, 165 and factors such as purity, source, and contraindications for special populations need to be considered. 159 Although several clinical reports have demonstrated the clinical value of probiotics on neuropsychiatric disorders, their mechanisms need to be clarified, and the adverse reactions are not known, necessitating further safety validation.
Some traditional Chinese medicines have been reported to alter gut microbiota composition via targeting the immune system. Hypericum japonicum ethanol extract inhibited inflammation via the NF-κB and MAPK signaling pathways, regulating colonic bacterial abundance, and improving gut dysbiosis. 166
Conclusion
Preclinical studies have found that neuropsychiatric patients and those with AD, PD, and depression often present with gastrointestinal symptoms, 69 but the associated mechanisms are not well understood. Notably, the gut-brain axis’s has been implicated in the regulation of neuropsychiatric disorders, with the gut microbiota influencing the occurrence of AD, PD, and depression progression via targeting the nervous system (VNS and ENS), immune system (immune cells and gut mucosal barrier), and microbial metabolites (SCFAs and bile acids). 167 In future, researchers should investigate the exact mechanisms and signaling pathways activated by bacteria and their metabolites, as well as identify cells targeted by the bacteria, to better improve our understanding of the gut-CNS axis.
Given that gut microbiota can alter various phenotypes of AD, PD, and depression, several interventions such as FMT, antibiotics, and probiotics have been proposed to modulate microbial homeostasis and improve pathological conditions in patients or animal models. Although several clinical reports have uncovered the therapeutic value of FMT, they are limited by their small sample sizes. The outcomes of FMT requires optimal consideration to the health status of the donor and recipient. The occurrence antibiotic resistance is also a limitation of antibiotic use, suggesting that the use of probiotics need to consider the purity, source, and enzymatic effects on other treatments. Future research should aim to elucidate gut microbiota’s precise mechanisms and their safety in the treatment of neuropsychiatric disorders, to improve the therapeutic effects of gut microbiota.
Footnotes
Author Contributions
Yali Tang: Formal analysis; Methodology; Visualization; Writing - original draft; Writing - review & editing. Yizhu Zhang: Data curation; Software; Visualization; Writing - original draft. Chen Chen: Methodology; Visualization; Writing - original draft. Ying Cao: Data curation; Visualization; Writing - review & editing. Qiaona Wang: Conceptualization; Visualization; Writing - original draft; Writing - review & editing. Chuanfeng Tang: Conceptualization; Resources; Supervision; Writing - review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Research funding for Specially Appointed Professor at Nanjing University of Traditional Chinese Medicine (013038019032), and Taihe Hospital (2022JJXM073), (2023JJXM091).

