Abstract
Postural imbalance, abnormal axial posture, and axial rigidity are the characteristic features of Parkinson’s disease (PD), and they are referred to as axial symptoms. The symptoms are difficult to manage since they are often resistant to both L-DOPA and deep brain stimulation. Hence, other treatments that can improve Parkinsonian axial symptoms without adverse effects are required. Vestibular dysfunction occurs in PD since neuropathological changes and reflex abnormalities are involved in the vestibular nucleus complex. Galvanic vestibular stimulation (GVS), which activates the vestibular system, is a noninvasive method. This review aimed to assess the clinical effect of GVS on axial symptoms in PD. To date, studies on the effects of GVS on postural instability, anterior bending posture, lateral bending posture, and trunk rigidity and akinesia in PD had yielded interesting data, and none of the patients presented with severe adverse events, and the others had mild reactions. GVS indicated a possible novel therapy. However, most included a small number of patients, and the sample sizes were not similar in some studies that included controls. In addition, there was only one randomized controlled clinical trial, and it did not perform an objective evaluation of axial symptoms. In this type of research, vestibular contributions to balance should be distinguished from others such as proprioceptive inputs or nonmotor symptoms of PD.
Keywords
Introduction
In addition to tremor, limb rigidity, and slowness of movement, postural imbalance, abnormal axial posture, and axial rigidity are the characteristic features of Parkinson’s disease (PD), and they are referred to as axial symptoms. Worsening of these symptoms is significantly associated with a risk for falling, and it can have a strong negative impact on quality of life. The symptoms are difficult to manage since they are often resistant to both l-3,4-dihydroxyphenylalanine (L-DOPA) and deep brain stimulation. 1 Dopaminergic treatments are sometimes correlated with balance problems, particularly when they cause dyskinesia. Further, the use of dopamine agonists can result in abnormal axial posture such as anterior bending posture, lateral trunk flexion, and Pisa syndrome. Deep brain stimulation can reduce the severity of axial symptoms. However, it is invasive. Hence, other treatments that can improve Parkinsonian axial symptoms without adverse effects are required.
Vestibular dysfunction occurs in PD. 2 Pathologically, Lewy bodies and neurites extend into the vestibular nucleus complex including the medial, superior, and lateral vestibular nucleus. 3 The reduction of non-phosphorylated neurofilament proteins, which could reflect degeneration that impairs motor function, was higher in the substantia nigra, and there was a 50% reduction in the lateral vestibular nucleus. 4 Patients with PD had impaired vestibular-evoked myogenic potentials, which were significantly correlated with contralateral rigidity, bradykinesia, and ipsilateral dyskinesia. 5 Moreover, peripheral ipsilateral vestibular paresis is associated with lateral trunk flexion. 6 These findings can provide substantial evidence regarding neuropathological changes and reflex abnormalities in the vestibular nucleus complex.
Galvanic vestibular stimulation (GVS), which activates the vestibular system, is a noninvasive method. Some studies have shown that GVS improves motor function in PD. 2 The exact mechanism underlying the effect of GVS on brain activity has not been elucidated. However, available human data support the notion that the basal ganglia receives vestibular input. 2 Positron emission tomography scan and functional magnetic resonance imaging studies have reported that the putamen and the caudate nuclei have higher activity after vestibular stimulation among healthy participants. 7 The vestibular nuclei directly project to the pedunculopontine nucleus (PPN) in monkey studies,8,9 and PPN deep brain stimulation can improve vestibular perceptual thresholds, in addition to postural control and balance. 10 Noisy GVS delivers currents with randomly varying amplitudes or direct currents to vestibular afferents. Subsequently, it influences resting state cortical electroencephalography activity or abnormal exaggerated beta synchronization throughout the basal ganglia–cortical network, which is a characteristic of PD.11-13 Theoretically, GVS may modulate such neuronal networks. In addition, noisy GVS increases gamma-aminobutyric acid levels in the substantia nigra pars reticulate, which is similar to that observed after L-DOPA treatment or subthalamic stimulation. 14 This result indicates that a neurochemical mechanism that can improve motor function might be involved in the inhibition of the nigra pars reticulate, which is overactive in PD.15,16
Axial symptoms in PD have been well known to include postural instability, bending posture, and trunk rigidity and akinesia, and each pathophysiological mechanism is different. GVS has some positive effects on the bending posture, and trunk rigidity and akinesia, but the effect on postural instability is minimal. This review aimed to assess the clinical effect of GVS on each axial symptom in PD.
Postural instability
Clinical studies on the effect of vestibular stimulation to Parkinsonian axial symptoms.
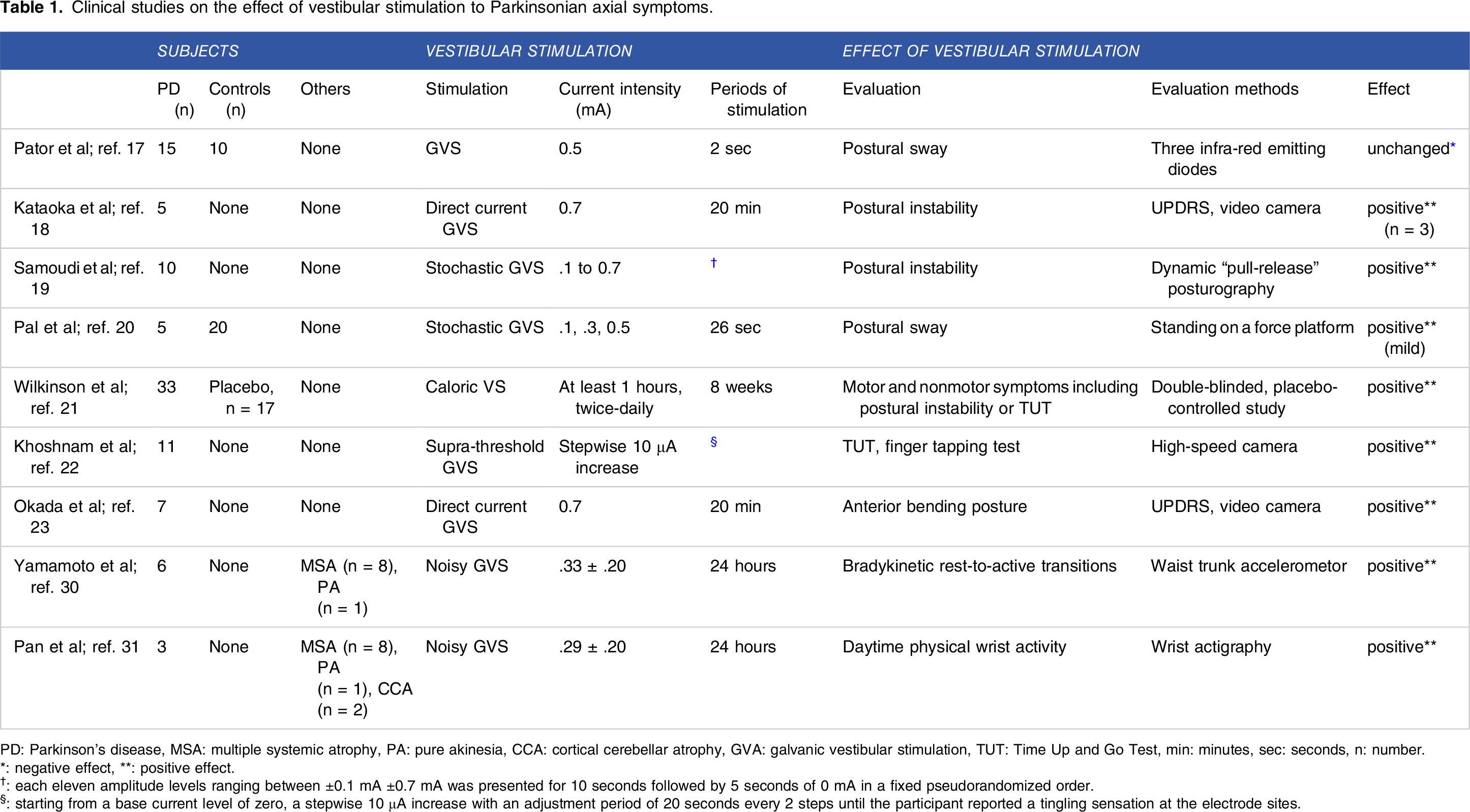
PD: Parkinson’s disease, MSA: multiple systemic atrophy, PA: pure akinesia, CCA: cortical cerebellar atrophy, GVA: galvanic vestibular stimulation, TUT: Time Up and Go Test, min: minutes, sec: seconds, n: number.
*: negative effect, **: positive effect.
†: each eleven amplitude levels ranging between ±0.1 mA ±0.7 mA was presented for 10 seconds followed by 5 seconds of 0 mA in a fixed pseudorandomized order.
§: starting from a base current level of zero, a stepwise 10 μA increase with an adjustment period of 20 seconds every 2 steps until the participant reported a tingling sensation at the electrode sites.
A randomized sham-controlled crossover study in 5 patients with PD showed that after the actual direct current GVS (.7 mA, duration: 20 min), the frequency of retropulsion decreased in 3 patients, independent of angle alternation in the anterior or lateral bending position, and the severity of postural instability did not change after the actual direct current GVS in 2 patients. 18 Postural instability was defined as a score of 3 for item 12 in the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part 3.
A double-blind placebo-controlled crossover pilot study using sham stimulation in ten patients with PD showed that stochastic GVS improved balance after a backward perturbation and shortened the postural response time. 19 Dynamic pull test posturography was performed. That is, a participant counterbalances 3% of his/her weight, which was suddenly released without prior notice. The overall dynamic posturography showed that the center of the pressure sway path reduced, and the maximum backward center of pressure sway path excursion decreased, particularly in the off-medication state. In addition, it has a small positive effect on motor symptoms. To evaluate the effects of stochastic GVS on postural sway in the medio-lateral and antero-posterior planes, Pal et al 20 assessed 5 patients with PD and 20 controls. 20 There was a minimal decrease in sway with low-intensity stimulation (.1–.5 mA, duration: 26 s), and the overall sway levels did not significantly differ between patients with PD and controls.
A double-blind, randomized, placebo treatmentcontrolled study using 8-week caloric vestibular stimulation in 33 participants with PD showed significant improvement in both nonmotor and motor functions based on the MDS-UPDRS. 21 Postural instability/gait difficulty according to the UPDRS can improve after stimulation. However, the result is not significant. The Timed Up and Go Test showed a decrease in time. Khoshnam et al have assessed 11 patients with PD to evaluate the effect of supra-threshold GVS using the Timed Up and Go Test. The current was applied with a stepwise increase of 10 μA increase during an adjustment period of 20 s every 2 steps. The result showed an improvement in the timing of steps and acceleration signal of the feet during the test. 22
Bending posture in PD
Anterior bending posture
A single-blind randomized sham-controlled crossover study of 7 patients with PD who had anterior bending posture showed that the anterior bending angles in the eye-open and eye-closed conditions significantly reduced immediately after the stimulation of direct current GVS (.7 mA, duration: 20 min)
23
(Figure 1). The mean degrees that can improve the anterior bending angle in the eye-open condition immediately after direct current GVS and sham stimulation were 5.4 and 4.4, respectively. The extent of improving the anterior bending angle between direct current GVS and sham stimulation was greater (8.2 vs 3.3) in the eye-closed than in the eye-open condition. There might be 2 mechanisms involved. First, direct current GVS activates the bilateral vestibulospinal tract, which may trigger the antigravity muscles. Second, it activates the temporoparietal junction including the vestibular cortex, and modified the body schema and/or vertical perception,24-26 which are integrated with visual, proprioceptive, and vestibular sensations and are important for postural control. PD duration and severity and postural deformities might not affect the anterior bending angles since there was no significant correlation between changes in the anterior bending angle and UPDRS motor score and duration of the disease and postural deformities. Further, direct current GVS might not affect gait disorders because there was no significant changes in the UPDRS gait subscore. Anterior bending angles significantly reduced after galvanic vestibular stimulation. Anterior bending angles were measured as the angle formed between the line jointing the C7 spinous process and the midpoint of the right and left posterior superior iliac spine.
Lateral bending posture
Peripheral ipsilateral vestibular paresis has been associated with lateral trunk deviation in patients with PD. 6 Subjective visual vertical, which is the ability of a person to perceive earth vertical with respect to gravity, is associated with lateral trunk deviation in patients with PD. 27 However, there is no study about the effect of GVS on lateral bending posture.
Posture overlap and other roles
Patients with PD generally exhibit both lateral and anterior trunk flexions, which are associated with Pisa syndrome.28,29 Regarding anterior trunk flexion, PD-specific axial rigidity or dystonia of the flexion muscles with erector spinal muscle weakness plays a role in the pathogenesis of Parkinsonian anterior trunk flexion. 29 Muscle diseases, such as focal myositis or secondary myopathies, which involve the axial muscles, are associated with not only neurodegenerative diseases but also injury caused by kyphotic postural changes. The age-dependent loss of tissue elasticity is involved. 29 Similarly, lateral axial rigidity or dystonia is primarily attributed to lateral trunk flexion, and secondary axial myopathy to stretching or muscle disuse, particularly in cases in which long-lasting trunk flexion could have caused lateral trunk flexion.6,27 These mechanisms can likely explain why the effect of GVS on anterior or lateral bending posture was short and mild.
Trunk rigidity and akinesia
Yamamoto et al 30 investigated the effects of 24-h noisy GVS at the current amplitude (.33 ± .20 mA) in 12 patients with PD and 7 with multiple systemic atrophy. 30 A portable ambulatory monitor comprising 2 shock sensors was used and attached around the waist, and it measured trunk acceleration in the vertical and horizontal axes. Noisy GVS increased the speed of bradykinesic rest-to-active transitions in patients with PD. Although trunk motor evaluation was not performed, the effect of 24-h noisy GVS was observed on a power-law temporal autocorrelation exponent of daytime wrist activity as the anti-akinesic effect. 31
Safety
Samoudi et al 19 reported that 4 of 10 patients presented with 6 adverse events, and 4 of which occurred during active stimulation of stochastic GVS (.1–.7 mA). Nausea and vomiting caused by levodopa were evident, and 1 patient reported mild headache and dizziness. 19 Wilkinson et al 21 studied 33 participants with PD using 8-week caloric vestibular stimulation. Results showed that the active stimulation group presented with 24 of 34 adverse events. The 3 adverse events were serious. However, these were not correlated with the device use. Four adverse events including ear discomfort, dizziness, motion sickness, and migraine might be associated with the device use. Nevertheless, none of the patients presented with severe adverse event, and the others had mild reactions.
Conclusion
To date, studies on the effects of GVS on axial symptoms in PD had yielded interesting data, and these findings indicated a possible novel therapy. However, the limitations of these studies must be considered. That is, most included a small number of patients, and the sample sizes were not similar in some studies that included controls. In addition, there was only one randomized controlled clinical trial, and it did not perform an objective evaluation of axial symptoms. In this type of research, vestibular contributions to balance should be distinguished from others such as proprioceptive inputs by measuring more broad vestibular functions including vestibular-evoked myogenic potentials or nonmotor symptoms of PD such as cognitive function decline and depression.
Footnotes
Authors Contribution
H Kataoka was responsible for the overall study design, and wrote the manuscript.
H Kataoka, T Kiriyama, Y Kita, J Nakamura, and Y Okada contributed to the acquisition of data.
H Kataoka and Y Okada contributed to analysis and interpretation of the data.
H Kataoka, K Shomoto, and K Sugie contributed to drafting and critical revision of part of the submitted materials.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
