Abstract
Purpose:
The establishment of early diagnostic methods for Parkinson disease (PD) is one of the key features to clinically control the rate of PD progression. This study aimed to give a first step toward recognizing the efficacy of multiple postural indices of balance control in differentiating medicated PD patients from health participants.
Methods:
Nine individuals with PD (Hoehn and Yahr Stage up to 2), 9 staged 2.5 and up, and 9 healthy age-matched Controls performed bipedal stances for 120 seconds with eyes either open or closed on a stable force platform. All participants with PD were under anti-Parkinsonian medication. Non-parametric tests investigated the effects of PD and visual input on postural indices extracted from the center of pressure coordinates.
Results:
Independent of the stage of the disease, individuals with PD presented faster and shakier body sway compared with Controls. Advanced stages of PD also revealed increased body sway length and variability. In addition, medio-lateral postural instability was more pronounced in all stages of PD when visual inputs were not allowed.
Conclusion and Significance:
Body sway velocity, jerkiness, length, and its variability revealed to be potential markers for subclinical signs of adjustments in the neuromechanisms of balance control and postural instability even at early stages of disease and under anti-Parkinsonian medication. Results produced here will direct future studies aiming to investigate the efficacy of these same indices on recognizing subclinical development of PD as well as those individuals susceptible to faster rates of progression.
Introduction
The rate of progression of Parkinson disease (PD) varies in its early stages of development and currently, there are no specific clinical markers or signs able to differentiate which patients will progress faster or slower through the stages of PD.1,2 This variability becomes an important factor in clinical management of PD cases as it promotes delays in establishing a reliable diagnosis as well as the implementation of early interventions to those patients developing PD at a slower rate. 1 Therefore, a pressing need arises to improve our ability to recognize and quantify the early and potential subclinical signs of PD development. However, the development of such indicators is dependent on several steps yet to be taken, including the recognition of early signs that are (1) common to patients in early stages of PD progression and (2) also immune to the effects of common PD medication used in these stages. This immunity to the medication will allow us to recognize the behavior of these indices on a larger spectrum of PD progression while representing a closer reality to the clinical practice.
Advances in the field of quantitative posturography have allowed for the development of comtemporary analytical approaches that are more sensitive to subtle impairments of balance performance caused by insults to the central nervous system (CNS).3-5 These methods rely on the quantification of indices of postural control directly linked to a large network of neuromechanisms dedicated to integrate sensory inputs from different sources (visual, vestibular, cutaneous, and proprioceptive) into motor outputs intended to control a large number of postural muscles involved in maintaning upright posture.6,7 These techniques employ the use of mutiple postural indices extracted from the body’s center of pressure (COP) dynamics and have been used not only to measure human postural stability in health but also to document rates of disease progression and efficacy of interventional protocols in different diseases.3,8,9 Previous studies using posturography assessed the effects of PD to posture control by using correlated methods and produced interesting but contradictory results. Some studies reported that PD affects postural behavior by decreasing one’s limits of functional stability 10 and increasing the amplitudes of body sway.10-12 Others reported no significant effect of PD to postural behavior13,14 or increased sway area when individuals close their eyes. 14 However, they have not been able to establish the reliability of these measures in early stages of medicated patients. In addition, Panyakaew et al 14 found a moderate correlation between Hoehn and Yahr (H-Y) Stage and both mean lateral sway and body sway area, and a strong correlation between disease duration and lateral sway, sway area, and lateral sway velocity. Taken together, these observations do not allow for a clear interpretation of the PD effects on postural behavior.
The disparity of results found across these studies may likely be due to the variability in populations with PD, experimental protocols, and postural indices used. Another important aspect from previous studies is the lack of standardization regarding the use or not of medication intended to reduce PD symptomatology. A recent study reported that dopaminergic regulation has an important impact on coordination of finger force production. 15 There are weaker synergy indices during steady-state force production in addition to delayed and reduced anticipatory adjustments of synergies prior to quick force pulse initiation in patients off-drug as compared with on-drug condition. These drug effects were observed in both the symptomatic and asymptomatic hands of patients in both H-Y Stages I and II, whose symptoms were limited to 1 side of the body. Based on these observations, it is expected that postural symptoms may also be masked in non-instrumented clinical testing of medicated patients even though the risks of falls may still be present.
To aid the development of reliable tools intended to assess balance in early stages of PD in medicated individuals, this study investigated postural behavior of PD patients by using contemporary postural indices in spatial, temporal, frequency, and structural domains. The authors hypothesized that individuals with PD would show increased body sway trajectory, area, velocity, shakiness, and irregularity in time compared with healthy age-matched individuals. The authors expect this report to facilitate future studies aiming to investigate the potential use of postural indices extracted from body sway dynamics as subclinical markers of early PD diagnosis and PD progression.
Methods
Participants
Twenty-seven volunteers participated in this study. Three distinct experimental groups were formed: Parkinson group 1 (PD1) consisted of 9 individuals diagnosed with PD and without clinical symptoms of postural instability (H-Y Stage ⩽ 2). Parkinson group 2 (PD2) consisted of 9 individuals diagnosed with PD and H-Y Stage ⩾ 2.5. Control group consisted of 9 healthy age-matched individuals with no history of any sensory, musculoskeletal, neurological, or cardiopulmonary disorder. Prior to participation, all volunteers gave their informed consent based on the procedures approved by the local Institutional Review Board (WMU-IRB # 19-04-10) and conforming to the Declaration of Helsinki. Table 1 presents general information about the participants’ characteristics.
General information about participants’ characteristics.

Means and standard deviations (SD) are reported.
Abbreviations: H-Y Stage, Hoehn and Yahr Stage; F/M, female/male sex; PD, Parkinson disease; PD1, Parkinson group 1; PD2, Parkinson group 2.
Apparatus
A force platform (Biomec, EMG Systems do Brasil Inc., Brazil) was used to acquire ground reaction forces and moments necessary to compute the body’s COP coordinates in both anterior–posterior (COPap) and medial–lateral (COPml) directions. Recordings were sampled at a frequency of 100 Hz with a 12-bit resolution.
Postural control assessment
All participants performed 2 simple postural tasks: bipedal stance for 120 seconds with eyes either open or closed (Vision and No Vision conditions). For both tasks, participants were instructed to stand barefoot in the force platform with their arms crossed and their feet 10 cm apart and parallel. For the Vision condition, participants were asked to focus their vision on a physical static point placed at eye level and at approximately 2 m. Conversely, they were asked to close their eyes for the No Vision condition. The duration of the entire experimental session was approximately 10 minutes and none of the participants reported fatigue or dizziness. Figure 1 illustrates the posture adopted by the participants on the top of the force platform along with the COP displacement at the screen of the computer. Participants could not see the screen.

Representation of the postural task on the force platform and the body’s COP displacement. COP indicates center of pressure.
Data processing
Center of pressure was analyzed off-line by customized software (BalanceLab vs 2.0, Synergy Applied Medical & Research Inc., USA). Ground reaction forces and moments of force around the 3 axes recorded by the force platform were used to compute COPap and COPml. The COPap and COPml time series were down-sampled to 10 Hz and de-trended by their mean. Twenty-one postural indices of interest computed from the COP signals included the following: area contained 95% of the COP displacement (Area95), the total length of the COP trajectory (TotalSway), the amplitude of the COP displacement in each direction (Rangeap and Rangeml), the variability of the COP displacement in each direction computed by its root mean square (RMSap and RMSml), the total mean velocity of the COP displacement (TMV), the mean velocity in each direction (MVap and MVml), the total mean jerkiness of the COP displacement (TMJerk), the mean jerkiness in each direction (MJerkap and MJerkml), the mean frequency on the power spectrum density (PSD) in each direction (Fmeanap and Fmeanml), the frequency peak on the PSD in each direction (Fpeakap and Fpeakml), the frequency band of the stabilogram containing 80% of the PSD in each direction (F80COPap and F80COPml), the sample entropy of the COP trajectory in each direction (SEntap and SEntml), and the cross-sample entropy (CrossSEnt) between COPap and COPml.
Innovative indices, such as body sway jerkiness and sample entropy, have been successfully used to detect subtle changes in postural control.16,17 Body sway jerkiness is defined as the rate of change in COP acceleration. In the study, it was computed as the third derivative of the COP position with respect to time.
The entropy of the body sway was computed to quantify the unpredictability level of the COP fluctuation in time by analyzing the probability of a particular sequence of COP values to repeat itself in time. Body sway entropy estimates were obtained through estimations of the correlation, persistence, and regularity of the COP signal for each direction (SEntap and SEntml) in time. In general, the algorithm includes the scalar time series (COPap or COPml displacement in time), the number of input data points for entropy computations (N), the length of compared runs (m), and the effectively filter level defining the criterion of similarity (r). The entropy of a scalar time series calculates the probability that a sequence of N data points, having repeated itself within a tolerance r for a window length m, will also repeat itself for (m + 1) points, without allowing self-matches. For m = 1 and 2, values of r between 0.1 and 0.25 standard deviation of the time series data produce good statistical validity of entropy. 18 For this study, the variables r (the tolerance) and m (the matching length) for sample entropy calculation were set at 2 and 0.2, respectively.
Resulting sample entropy estimation was represented by a single, nonnegative real number indicating the level of irregularity of the time series along a certain period. Smaller sample entropy estimates indicate many repetitive patterns of COP fluctuation in time, whereas larger estimates indicate a more irregular, random, and unpredictable pattern. Cross-sample entropy was obtained by estimations of the degree of asynchrony between COPap and COPml signals in time. Higher CrossSEnt estimates indicate larger levels of asynchrony of the postural sway between the 2 directions; lower values, in contrast, indicate more co-dependence of the postural sway dynamics between the 2 directions. See Richman and Moorman 19 for more details regarding sample entropy and cross-sample entropy.
Statistical approach
Statistical tests were performed using the IBM SPSS statistics software (version 22). For each task, non-parametric Kruskal–Wallis H and Mann–Whitney U tests were used to investigate the effects of Disease (Control, PD1, and PD2) on the response variables. For each group of participants, the effects of Visual Input (Vision and No Vision) were also investigated. For all response variables, medians and quartiles across participants were reported. For all statistics, P values and 95% confidence interval (CI) of the difference were reported. The significant level was fixed at .05.
Results
Vision condition: bipedal stance with eyes open
Figure 2 shows, on the left, the body’s COP displacement of a representative participant of each group (Control, PD1, and PD2) performing bipedal stance with eyes open. The similar body sway area among these 3 participants and a denser path for participants with PD visually show an increased total length of the COP trajectory.
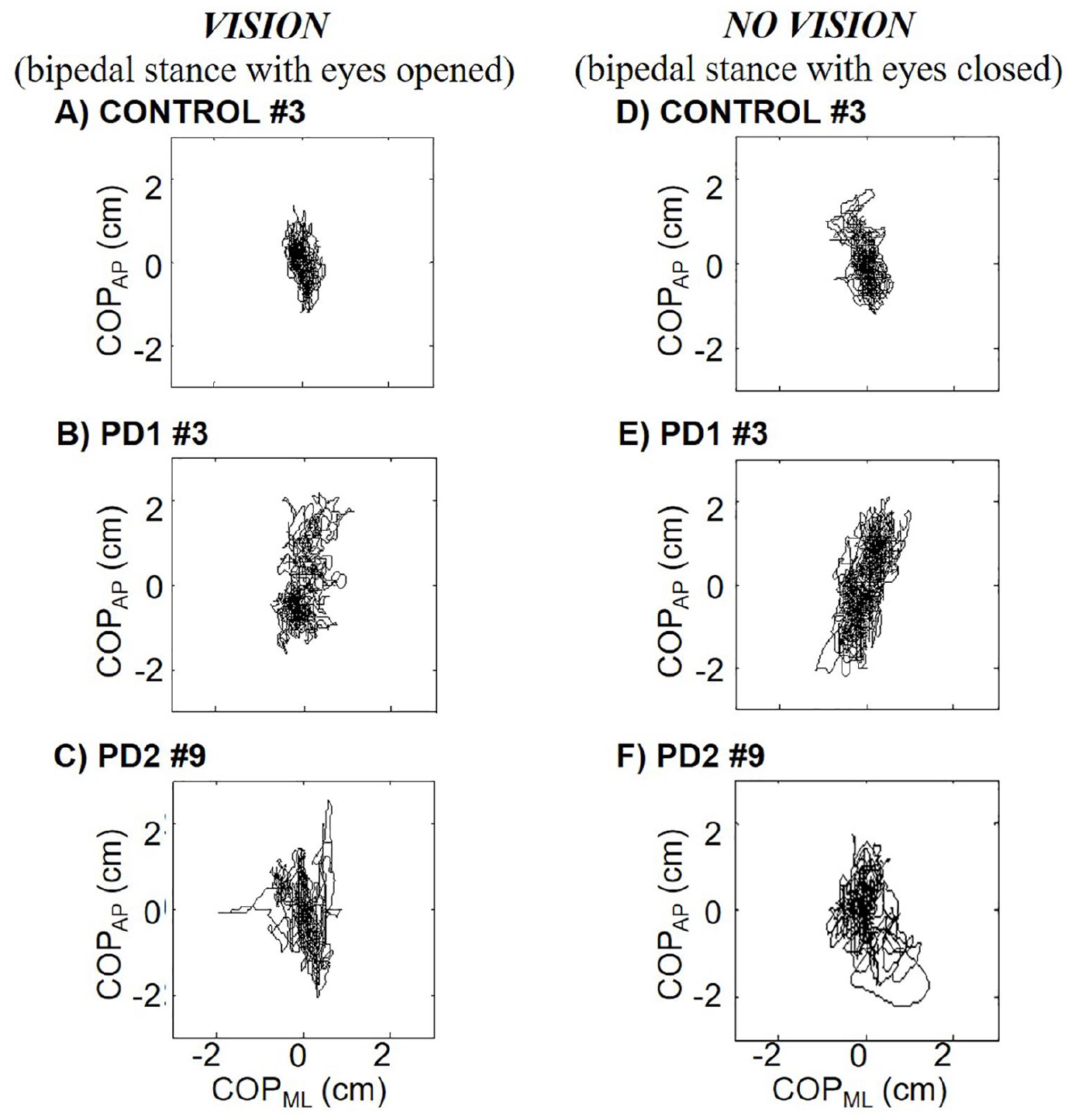
The COP displacement of a representative participant of each group (Control, PD1, and PD2) performing bipedal stance with eyes open (A, B, and C, respectively) and eyes closed (D, E, and F, respectively). COP indicates center of pressure; PD, Parkinson disease; PD1, Parkinson group 1; PD2, Parkinson group 2.
Static posturography revealed no significant effect of PD (Control, PD1, and PD2) for most of the postural indices when participants performed bipedal stance with eyes opened. However, PD1 participants presented significantly higher values for MVml, TMJerk, MJerkap, and MJerkml compared with Controls, P = .044, 95% CI of the difference, –0.05 to 0.01; P = .024, 95% CI of the difference, –55 to –14; P = .038, 95% CI of the difference, –46 to –10; and P = .031, 95% CI of the difference, –29 to –2, respectively, whereas PD2 participants presented significantly higher values for TotalSway, RMSap, MVml, TMJerk, MJerkap, and MJerkml compared with Controls, P = .047, 95% CI of the difference, –4499 to –574; P = .014, 95% CI of the difference, –0.39 to –0.07; P = .035, 95% CI of the difference, –0.41 to –0.01; P = .003, 95% CI of the difference, –455 to –50; P = .015, 95% CI of the difference, –368 to –48; and P = .003, 95% CI of the difference, –202 to –12, respectively. In addition, Mann–Whitney U tests revealed no significant difference in postural indices between PD1 and PD2 participants. Figure 3 shows box plots of COP area, total sway, velocity, and jerkiness of participants performing bipedal stance with eyes open. Table 2 shows all 21 postural indices across participants performing bipedal stance with eyes opened.

Box plot of the (A) COP area, (B) total COP sway, (C) mean COP velocity, and (D) mean COP jerkiness during bipedal stance with eyes open (Vision) and eyes closed (No Vision) conditions. COP indicates center of pressure; PD, Parkinson disease; PD1, Parkinson group 1; PD2, Parkinson group 2.
Median (first quartile, third quartile) values of postural indices across participants (Control, PD1, and PD2) while performing bipedal stance with eyes opened.
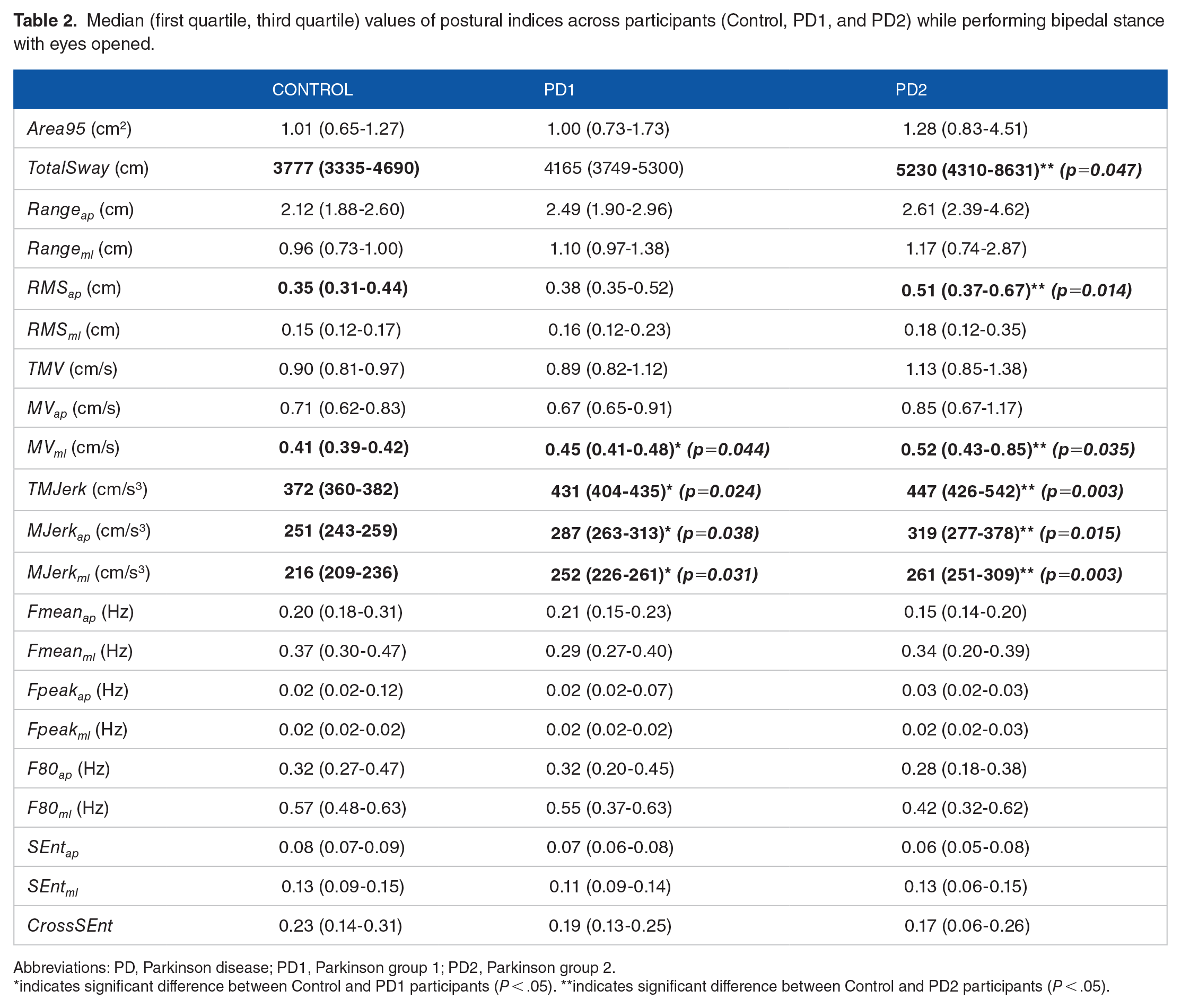
Abbreviations: PD, Parkinson disease; PD1, Parkinson group 1; PD2, Parkinson group 2.
indicates significant difference between Control and PD1 participants (P < .05). **indicates significant difference between Control and PD2 participants (P < .05).
No vision condition: bipedal stance with eyes closed
Figure 2 shows, on the right, the body’s COP displacement of a representative participant of each group (Control, PD1, and PD2) performing bipedal stance with eyes closed. When all participants performed bipedal stance with closed eyes, PD1 presented significantly higher MJerkml compared with Controls, P = .034, 95% CI of the difference, –48 to –8, whereas PD2 presented significantly higher RMSml, TMJerk, and MJerkml, and significantly lower Fmeanml and F80COPml compared with Control participants, P = .043, 95% CI of the difference, –0.38 to –0.00; P = .034, 95% CI of the difference, –820 to –15; P = .007, 95% CI of the difference, –339 to –15; P = .043, 95% CI of the difference, –0.18 to –0.19; and P = .016, 95% CI of the difference, –0.22 to –0.30, respectively. Table 3 shows all 21 postural indices recorded across participants performing bipedal stance with eyes closed. Figure 3 shows box plots of COP area, total sway, velocity, and jerkiness of participants performing bipedal stance with eyes closed.
Median (first quartile, third quartile) values of postural indices across participants (Control, PD1, and PD2) while performing bipedal stance with eyes closed.
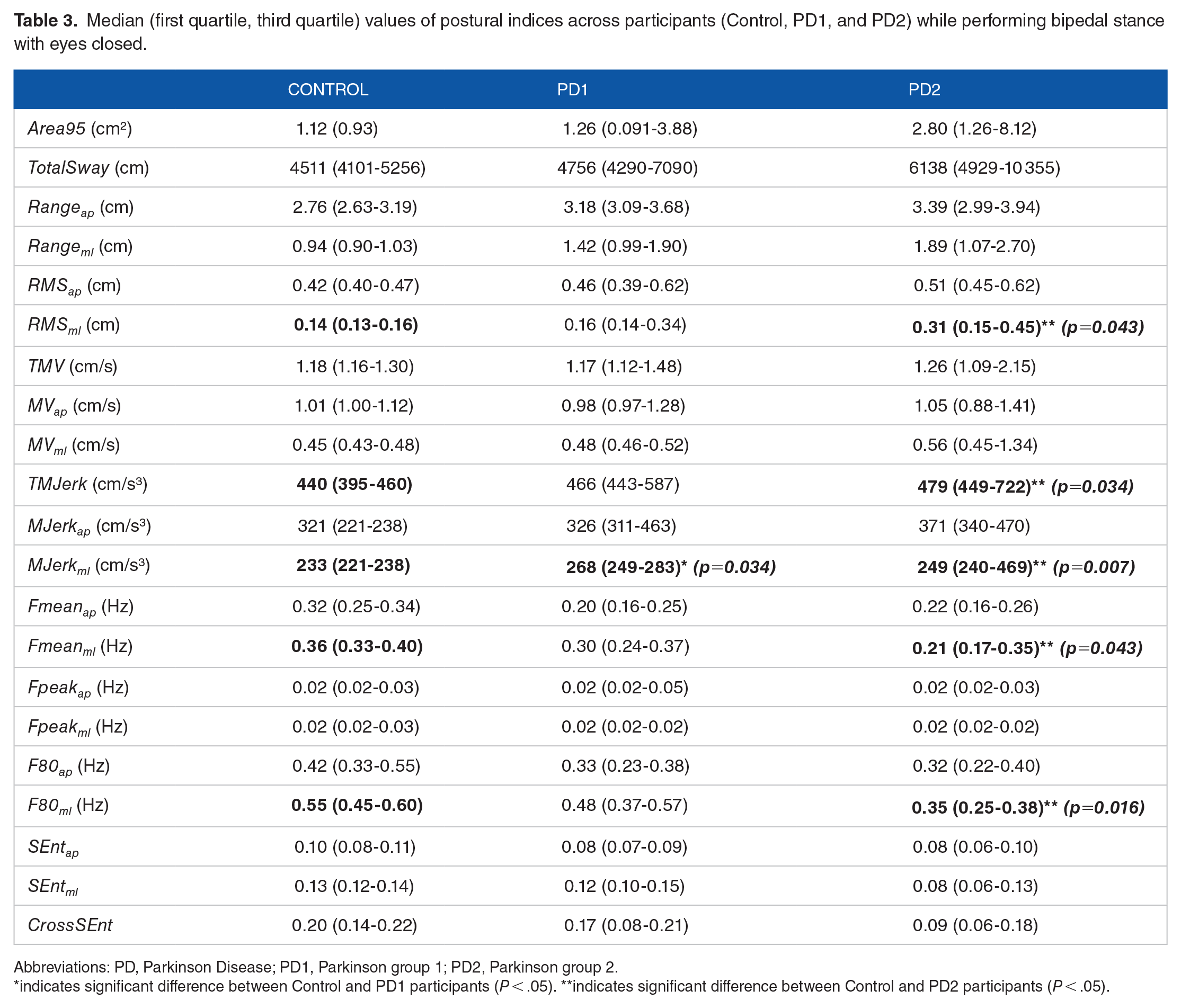
Abbreviations: PD, Parkinson Disease; PD1, Parkinson group 1; PD2, Parkinson group 2.
indicates significant difference between Control and PD1 participants (P < .05). **indicates significant difference between Control and PD2 participants (P < .05).
The effect of visual input on postural sway was also analyzed within each group of participants. When both Controls and PD1 closed their eyes, they increased their Rangeap, TMV, MVap, TMJerk, and MJerkap, P = .024, 95% CI of the difference, –0.87 to –0.63; P = .002, 95% CI of the difference, –0.49 to –0.29; P = .004, 95% CI of the difference, –0.49 to –0.29; P = .004, 95% CI of the difference, –110 to –46; and P < .001, 95% CI of the difference, –117 to –49, respectively, for Controls; P = .047, 95% CI of the difference, –1.44 to –0.36; P = .024, 95% CI of the difference, –0.35 to –0.19; P = .007, 95% CI of the difference, –0.35 to –0.18; P = .047, 95% CI of the difference, –136 to –42; and P = .047, 95% CI of the difference, –134 to –33, respectively, for PD1. No significant effect of visual input was observed for PD2.
Discussion
Motor impairments, such as tremor, slowness of movements, and increased muscle stiffness, often play an important role in the surfacing of abnormal postural behavior, postural instability, and its inherently higher risk of falling. The presence, timing, and intensity of these motor symptoms are also known to vary across individuals and change over time, which makes it difficult to assess postural impairments across this resulting spectrum of clinical scenarios. This present study was able to uncover features of postural behavior that seem to be independent of the severity of these symptoms. In general, neither group of individuals with PD (PD1 and PD2) showed significant differences regarding body sway area, frequency, sample entropy, or the irregularity level of body sway in time compared with healthy age-matched individuals. However, patients with PD using medication presented faster and shakier body sway when compared with Controls, independent of their H-Y Stage. In addition, the total length of body sway and the variability of the COPap displacement in individuals at advanced stages of PD (H-Y Stage above 2) were significantly higher compared with Controls. The present study found no significant differences for any postural index computed between early and late stages of PD. Finally, postural sway in PD on dopamine medications was laterally shakier compared with healthy age-matched individuals when visual input was withdrawn. In addition, healthy Controls and individuals at early stages of PD (PD1) swayed more, faster, and shakier when they stood still with eyes closed compared with eyes opened.
No significant differences regarding some postural indices in patients with PD and healthy Controls were also reported in previous studies.13,14 However, they are contradictory to a series of reports in which these indices seemed to be affected by PD.10-12 Despite expected changes in body sway indices as PD emerges and progresses, it seems that age-related changes in the sensory-motor system and anti-Parkinsonian medications might have counter-acted such effects of PD. A few studies have reported that the progressive and non-homogeneous degeneration of sensory, neural, and motor functions due to the process of aging by itself affects postural behavior by increasing body sway area17,20,21 and the unpredictability of COP displacement in time.17,22
In general, postural instability creates difficulties with transfers, gait, and activities of daily living. As a consequence, postural instability becomes the major cause of falls and fall-related injuries. Unfortunately, balance disorders in PD are clinically missed in the beginning of the disease. Falls at this moment seems to be associated to the age-related sensory-motor changes rather than to the disease. The present study was able to reveal non-age-related changes in the mechanisms of postural adjustments even at early stages of PD. The postural indices used in the study were able to detect subtle changes in body sway related with balance impairments that were not detected by regular clinical assessment. The body sway velocity and jerkiness were sensitive to PD, independent of its H-Y Stage and even under the effect of anti-Parkinsonian medication. The increased lateral sway velocity found for both PD1 and PD2 corroborates with reports of lateral postural instability in individuals with PD.14,16,23 Shakier body sway in PD detected by increased jerkiness of the COP displacement recorded by a force platform was also described in a previous report using an accelerometer positioned on the posterior trunk. 16 Considering that the process of aging alone increases both the velocity and jerkiness of postural sway,17,20,21 our findings suggest that the presence of PD will amplify these measures, therefore becoming clinically relevant to aid PD diagnosis. Most importantly, these indices were found to be altered even in those patients who did not report postural instability according to their H-Y level (PD1 group). Faster and shakier body sway reflects the effect of tremor, increased joint resistance (stiffness) and co-contraction of postural muscles, 24 impaired somatosensorial processing, 25 difficulty in scaling postural responses,26,27 increased voluntary postural corrections, 26 and impaired feedforward mechanisms controlling upright posture in individuals with PD, independent of its stage.
Regarding anti-Parkinsonian medications, Park et al 15 reported deterioration of synergistic patterns of finger coordination 12 hours after suspension of medication. This medication effect may also be present in the coordination of postural muscles. Therefore, we speculate that age- and medication-related effects on balance might have masked possible effects of PD on some postural indices. Recently, multi-muscle synergies stabilizing upright stance and anticipatory synergy adjustments to postural perturbations at early stages of PD and also in patients on dopamine-replacement drugs were reported.4,5 However, no effect of medication was observed on postural indices extracted from the COP displacement. Anticipatory postural adjustments were reported to be similar in individuals with PD in both “on” and “off” states of medication. 28 Patients in the “off” state improved their anticipatory postural adjustments with practice, while patients in the “on” state did not. Motor coordination of fingers are also affected by PD. Lower maximal finger forces, higher unintended force production, weaker multi-finger synergy, and delayed and reduced anticipatory synergy adjustments in early stages of PD have been reported, 29 in addition to improved multi-finger coordination during dopamine-replacement therapy. 15 It seems that anti-Parkinsonian drugs have different effects on posture control and fine multi-finger coordination. Taken all together, the present study strongly suggests the use of body sway velocity and jerkiness as sensitive markers to aid in early diagnosis of postural instability in individuals with PD.
The fact that the present study did not show significant differences on postural indices between the 2 Parkinson groups does not mean that postural sway does not change across all stages of the disease. The use of posturography to assess progress of PD was not the goal of the study. However, 2 groups of individuals with PD were formed to differentiate individuals with and without postural instability diagnosis according to their H-Y Stage. In general, studies have reported changes on postural control as the disease progresses such as increased postural sway in both static and dynamic stance conditions, 30 reduced postural control precision, 31 increased oscillations in both directions, 31 poorer balance performance, 32 slower movement velocity on the limits of stability test, 32 and changes in reactive postural control. 32 Taken together, it is suggested that total body sway and sway area, velocity, and jerkiness would be useful clinical markers to detect progress of postural instability in individuals with Parkinson diseases. Further studies including individuals with PD at different stages should be done to map progression of postural instability in this population.
Despite the effect of sensory conflict was not the focus of this study, a few findings regarding temporary withdrawal of visual input and postural control are worthy of discussion. The withdrawal of visual input leads to laterally shakier body sway in individuals with PD on dopamine medications compared with healthy age-matched individuals. A greater effect of PD on lateral postural sway indices when visual input is not allowed has also been reported in the literature.11,13,14,16,33 Panyakaew et al 14 compared postural sway of healthy individuals and individuals with early PD when standing with closed eyes. They reported increased mean medio-lateral sway, mean sway velocity, lateral sway velocity, and sway area. They also reported a moderate correlation between H-Y Stage and both mean medio-lateral sway and sway area, as well as a strong correlation between disease duration and mean medio-lateral sway, sway area, mean sway, and lateral sway velocity. In addition, measures of body sway at late stages of PD presented more variability and less frequency in the medio-lateral direction. A few studies suggested that lateral instability in PD might be related to the asymmetric aspect of the disease, 34 increased trunk and ankle stiffness reducing movements in the sagittal plane,33,35 and a possible greater decline in hip and trunk control compared with ankle control. 16 Therefore, the present study also suggests the use of static posturography with visual deprivation to assess postural instability. Moreover, the effect of visual manipulation affected postural sway amplitude, velocity, and jerkiness in both Controls and PD1 individuals. A few studies reported changes in postural sway indices under conditions of reduced or conflicting sensory information in both younger and older healthy adults.17,20,21,36 Despite reports of increased lateral body sway and sway area at early PD 37 and decreased lateral sway, 38 the present study showed no significant changes in postural indices when individuals at advanced stages of PD closed their eyes. The authors speculate a decrease in the reliance on visual input to control balance at later stages of PD. Previous studies of populations with PD have also reported the ability of appropriate postural responses to sensory conflict situations, such as visual input deprivation.10,39 Further studies focusing on the effect of temporary withdraw of visual input on postural control of individuals with PD would be of great importance to improve balance interventional tools for this population.
In general, findings from this study provided a step forward in the development of sensitive techniques and software to assess postural instability in PD by using posturography. Although postural instability is considered a clinical marker of PD at H-Y Stage 3, early detection of balance deficits is of extreme clinical importance. Many postural indices were not sensitive to the effect of PD on postural control, likely due to the combined effect of the variability of motor symptoms among patients, anti-Parkinsonian medication, and the natural process of aging. However, individuals with PD presented subclinical postural instability characterized by faster and shakier body sway even in the early stages of the disease. This finding indicates that balance control is affected even before clinical signs surface. In addition, lateral instability became more evident in late stages of the disease and when visual information was not allowed. A few postural indices revealed to be potential markers to aid in the diagnosis of PD even at early stages of the disease. First, the body sway velocity and jerkiness measured during simple standing tasks, such as bipedal stance with eyes either open or closed, revealed to be potential markers of postural instability in individuals with PD. Second, the total length of body sway and the variability of COP displacement in the anterior–posterior direction revealed to be objective measures of the progression of the disease. Third, the increased lateral sway in patients with PD compared with Controls when vision was occluded revealed to be another clinical biomarker of patients with PD regardless of severity. Therefore, postural markers used in this study are the great importance to improve early diagnosis of postural instability in PD, record progress of balance control, and assess fall risk. They should also be implemented in clinical trials of pharmacotherapy and balance training protocols specific to populations diagnosed with PD.
Footnotes
Acknowledgements
The authors would like to thank all volunteers for their motivated participation in this study.
Funding:
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Conception and design of the study (AMD, VSC, ATM, AD-d-S), data acquisition (VSC, ATM, ALSA, EdCS), data analysis and interpretation (AMD, VSC, ATM, AD-d-S), drafting the article (AMD), revising the article critically for important intellectual content (all authors), and final approval of the version to be submitted (all authors).
