Abstract
Background
Parkinson’s disease (PD) is a common degenerative disease caused by abnormal accumulation of α-synuclein. The glymphatic pathway is essential for removing macromolecular proteins including α-synuclein from the brain, which flows into deep cervical lymph nodes (DCLNs) through meningeal lymphatics. As a terminal station for the cerebral lymphatic system drainage, DCLNs can be easily assessed clinically.
Objectives
Although the drainage function of the cerebral lymphatic system is impaired in PD, the correlation between DCLNs and PD remains unknown.
Design
Single-center retrospective cross-sectional study.
Methods
The size of the DCLNs were measured using ultrasound. The Movement Disorder Society Sponsored Revision Unified Parkinson’s Disease Rating Scale and other scales were used to assess PD motor and non-motor symptoms.
Results
Compared with the healthy control (HC) and the atypical Parkinson’s disease (AP) groups, the size of the second and third DCLNs in the Parkinson’s disease (PD) group was significantly smaller (P < .05). The width diameter of the third DCLN (DCLN3(y)) was significantly smaller in the PD group than in the AP group (P = .014). DCLN3(y) combined with a variety of clinical features improved the sensitivity of AP identification (sensitivity = .813).
Conclusion
DCLNs were able to distinguish HC, PD and AP and were mainly located in Robbins ΙΙA level. PD and AP were associated with different factors that influenced the size of the DCLNs. DCLN3(y) plays an important role in differentiating PD from AP, which, combined with other clinical features, has the ability to distinguish PD from AP; in particular, the sensitivity of AP diagnosis was improved.
Plain Language Summary
Deep cervical lymph nodes, a potential marker for the diagnosis of PD and AP. Deep cervical lymph nodes (DCLNs) were able to distinguish HC, PD and AP and were mainly located in Robbins ΙΙA level. PD and AP were associated with different factors that influenced the size of the DCLNs. DCLN3(y) plays an important role in differentiating PD from AP, which, combined with other clinical features, has the ability to distinguish PD from AP; in particular, the sensitivity of AP diagnosis was improved. DCLN has potential diagnostic value in differentiating PD from AP.
Keywords
Introduction
Parkinson’s disease (PD) is a common neurodegenerative disease in middle-aged and older individuals. Pathological changes are characterized by a massive loss of dopaminergic neurons in the substantia Nigra. 1 Although the potential mechanisms associated with PD are still not fully understood, a large number of studies suggest that a-synuclein (a-syn) plays an important role in the occurrence and development of PD.2-4 A-syn is a small (140 amino acids long), naturally unfolded protein 5 that regulates the storage, transmission, 6 and release 7 of neurotransmitters. When pathological changes develop, a-syn monomers aggregate and form fibrillary intermediates, 8 leading to the generation of mature and insoluble fibrils in neurons. 9 Neuronal death can occur as a result of the abnormal aggregation and folding of a-syn. 10 Therefore, the promotion of the clearance of a-syn is a reliable approach for the prevention and treatment of PD.
Recent studies have confirmed that the lymphatic drainage system, which plays an important role in removing metabolic waste and macromolecules, is present in the brain. Metabolic waste in the brain is exchanged through the glymphatic system and discharged into the cervical lymph nodes via meningeal lymphatic drainage. Among them, deep cervical lymph nodes (DCLN) are the main and preferential injection sites of meningeal lymphatic vessels (MLV) 11 and are the terminal stations of perfusion of the cerebral lymphatic system.12,13 The brain metabolites collected at this site are more comprehensive than those in the cerebrospinal fluid (CSF). 14 Furthermore, the development of PD is closely related to the dysfunction of the brain lymphatic system.15-17 It is reported that after ligation of the deep cervical lymphatic vessels (DCLV), a-syn accumulates with reduced glymphatic system exchange which leads to glial cell activation and dopaminergic neuronal loss in the mouse brain. 18 The cerebral lymphatic drainage rate and perfusion of the DCLN were slower in patients with PD compared with controls. 19 In addition, lymphatic drainage in mice is affected by sleep 20 and vagal activity, 21 which are associated with PD. For example, sleep cycle disorders can cause the accumulation of macromolecular proteins, 22 and enhanced vagal activity can promote the exchange of cerebrospinal fluid and brain interstitial fluid. 21 Therefore, DCLN could be potentially linked to altered lymphatic drainage in PD.
Currently, invasive methods or complex image analysis techniques are mainly used to evaluate the brain lymphatic system metabolism in patients with PD, but this is difficult to apply in clinical practice. In comparison, caution should be exercised with regard to gadolinium-enhanced magnetic resonance imaging (MRI), as gadolinium deposition is observed in the capillary endothelium and neuropil of the intracranial area within several years of exposure. 23 Although there is no conclusive evidence for the biological toxicity of gadolinium deposition in the brain, free gadolinium is known to be neurotoxic and potentially mutagenic. 24 Therefore, many studies advocate alternative methods25,26 such as diffusion tensor image analysis along the perivascular space (DTI-ALPS) 27 and ultrafast magnetic resonance encephalography. 28 Owing to the complexity of data processing and the high requirements of medical equipment, these methods are difficult to apply widely.
This study evaluated the ability of ultrasound examination to distinguish PD-related diseases from healthy populations by measuring the size of the DCLNs, which are the main drainage areas for MLV. The main drainage areas and specific locations of the DCLNs were verified. Subsequently, the drainage characteristics of the DCLNs in the HC, PD, and AP groups were compared. Finally, DCLNs combined with clinical features could be used to distinguish between PD and AP.
Methods
Ethics statement
This cross-sectional study was performed at the Movement Disorder Clinic, Department of Neurology, The First Affiliated Hospital of Chongqing Medical University, from March 2022 to March 2023. This study was approved by the Ethics Committee of The First Affiliated Hospital of Chongqing Medical University (2022-56). This was a cross-sectional study, and patient information was anonymous. Informed consent was not required for this study.
Power calculation
The sample size was calculated prior to the start of the study. The mean of the average widthdiameter (y) of DCLNs in patients with PD, atypical Parkinson’s disease (AP), and healthy controls (HC) was expected to be 4.15 mm, 3.06 mm, and 3.74 mm, respectively, with a standard deviation of .96 (Power 1-β = .80, Test level α = .05). One-way analysis of variance (ANOVA) F tests was used to calculate the average y of DCLNs. The results indicated that a minimum sample size of 18 individuals was required per group.
Subject
The study included 121 subjects (105 men and 60 women): 64 patients with PD (29 men and 35 women), 24 patients with AP (15 men and 9 women), and 33 healthy controls (17 men and 16 women). The patients with PD met the Movement Disorder Society (MDS) diagnostic criteria for clinically established PD.
29
AP such as multiple system atrophy (MSA)
30
and progressive supranuclear palsy (PSP)
31
were included. Five patients had PSP (4 men and 1 woman), and 19 patients had MSA (11 men and 7 women). For each patient with AP, the diagnoses were based on current diagnostic criteria. None of the patients mentioned above had any other neurological or psychiatric disorders, or a history of head and neck inflammation or upper respiratory tract infections in the past month. There was no history of neck disease, neck surgery, or tumors among the patients. The detailed inclusion and exclusion criteria are shown in Figure 1. Subject screening flow chart. HC, healthy control; PD, Parkinson’s disease; AP, Atypical Parkinson’s disease; DCLNs, deep cervical lymph node.
Questionnaire assessment
Neurological examinations were performed by two experienced neurologists. The clinical characteristics of the enrolled patients, including demographics, severity of motor and non-motor symptoms, and DCLNs ultrasound results, were collected. The Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) and Hoehn and Yahr (H&Y) scores were used to measure the impairment levels of PD and AP. 29 The Rapid Eye Movement Sleep Behavior Disorder Screening Questionnaire (RBDSQ) 32 and Parkinson’s Disease Sleep Scale (PDSS) 33 were used to assess sleep. Autonomic function was evaluated using the Scale for Outcomes in Parkinson’s Disease for Autonomic symptoms (SCOPA-AUT). 34 Cognition was assessed by the Mini-mental state examination (MMSE). 35
Ultrasonography
Ultrasonography has proven to be a valuable tool for detecting lymph lymph nodes. Ultrasound examinations were performed using the ultrasound system (Aplio i800, Canon Inc, Japan). Patients were maintained in the supine position. Examinations were performed by an appointed ultrasound specialist with more than 5 years of experience and supervised by an appointed ultrasound specialist with more than 15 years of experience who was blinded to the clinical data of all patients. Deep cervical lymph nodes (DCLNs) are the main sites of MLV drainage in the central nervous system. The drained contents are mainly collected in the DCLNs, especially in the lymph nodes around the internal jugular vein36,37 (Robbins II A and III levels). The 2002 neck classification guidelines for nodal level delineation were used to define this level.
38
The Robbins II A level was surrounded by the skull base, inferior body of the hyoid bone, stylohyoid muscle, and spinal accessory nerve. Level III is surrounded by the following structures: the lower body of the hyoid bone, lower margin of the cricoid cartilage, sternohyoid muscle, sternocleidomastoid, and sensory branches of the cervical plexus. Therefore, we placed the probe close to the internal jugular vein and sequentially scanned and measured three lymph nodes in the Robbins II, A, and III level lymph nodes from the skull base (Figure 2). Schematic representation of the cervical lymph node division used in this paper. The blue box is the region of level Ⅱ and level Ⅲ of cervical lymph nodes.
All lymph nodes in the bilateral neck were scanned using a multifrequency linear transducer (i18LX5 probe (frequency 5-18 MHz, depth 4 cm; dynamic range 65Db). Because the shape of the nodes depends on the scan plane, scans were obtained with the transducer placed longitudinally and transversely until the plane showed the maximum cross-sectional area of the lymph node. On the longitudinal scan of the lymph nodes, the longitudinal diameter (x) and transverse diameter (z) were measured, whereas the width diameter (y) was measured on the transverse scan. DCLNs with the shortest longitudinal diameters (<2.5 mm) were not measured because smaller nodes are more prone to measurement errors.
39
All values of x, y and z were represented as millimeter (mm), and the lymph node volume (Figure 3) was calculated by the ellipsoid formula (volume = X·Y·Z·π/6). DCLNs measured on both sides of the neck were averaged and recorded sequentially. Schematic representation of DCLN measurements. (a). DCNL schematic representation of the meridians. x = Longitudinal diameter, y = Width diameter, z = Transverse diameter. (b-e). DCLN schematic representation of the longitudinal section under the ultrasound probe. Orange line represents longitudinal diameter. Green line represents transverse diameter. DCLN schematic representation of measurements in the Short-axis section ultrasound image. Blude line represents width diameter. (b-c) DCLNs in HC. d-e: DCLNs in PD.
Statistical analysis
Statistical analyses were performed using the IBM SPSS Statistics (ver. 26.0; IBM Corp., Armonk, NY, USA). Categorical variables are expressed as percentages and were statistically analyzed using the chi-square test. The Kolmogorov-Smirnov test was used to determine the normality of continuous variables. The Bonferroni method was used to compare statistical differences between two groups. Continuous variables were described by mean ± standard deviation (mean ± SD) and analyzed by one-way ANOVA. Other continuous variables are expressed as medians and interquartile ranges, and the Mann–Whitney U test was used for comparison. Spearman’s correlation analysis was used to examine the correlation between clinical data and DCLNs. The correlation coefficient, R, variance inflation factor, and tolerance were used to diagnose the collinearity of the independent variables included in the binary logistic regression. A P-value <.25 was taken into the ROC analysis. Due to the statistical difference in age between the PD and AP groups, accommodating covariates in the ROC analysis (AROC) were used in R 4.2.0. The AROC was used to measure the sensitivity and specificity of the DCLNs indices and clinical data. The Youden index was used to calculate the cut-off values. A P-value <.05 was taken to indicate statistical significance.
Results
Demographic characteristics and clinical features of HC, PD, and AP groups
Demographic characteristics and clinical features of HC, PD, and AP groups.

Abbreviations: HC, healthy control; PD, Parkinson’s disease; AP, Atypical Parkinson’s disease; HBP, Hypertension; DM, Type 2 Diabetes Mellitus; MMSE, Mini-mental State Examination; RBD-SQ, Rapid Eye Movement Sleep Disorders Screening Questionnaire; TS, Total score; GS, Gastrointestinal System; US, Urinary system; CS, Cardiovascular system; T, Thermoregulation; PDSS, Parkinson’s disease sleep scale; MDS-UPDRS, MDS-Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn-Yahr Bonferroni post-hoc tests were used to compare variables among the HC, PD, and AP groups. The HC group performed better than the PD and AP groups on the MMSE, RBD-SQ, SCOPA-AUT (TS), SCOPA-AUT (GS), and SCOPA-AUT (US). Compared with PD groups, patients with AP were younger and had higher SCOPA-AUT (GS), SCOPA-AUT (GS), SCOPA-AUT (T), and UPDRSΙΙ scores.
Analysis of volume and size of each DCLN in HC, PD, and AP groups
Comparison of the volume and size of each DCLN in HC, PD, and AP groups.

Note: Bonferroni post hoc tests were used to compare variables between the HC, PD, and AP groups. The volume (x, y, z) of DCLN2 and the volume (x, y, z, and x/z) of DCLN3 showed significant differences between the HC and PD groups. Y in DCLN3 could distinguish between PD and AP. HC, healthy control; PD, Parkinson’s disease; AP, Atypical Parkinson’s disease; DCLN, Deep cervical lymph node.
Comparison of the average volume and size of DCLN2 and DCLN3 in HC, PD, and AP groups.

Note: Bonferroni post hoc tests were used to compare variables among the three groups. The volume (x, y, and z) of the average DCLN size could be used to distinguish between HC and PD. X indicates a significant difference between the HC and AP groups. Y can distinguish between the PD and AP. HC, healthy control; PD, Parkinson’s disease; AP, Atypical Parkinson’s disease; DCLN, Deep cervical lymph node.
These results indicate that the size of the DCLNs differed among the HC, PD, and AP groups. Compared with the HC group, the number of DCLNs in the PD group decreased significantly. This change was also observed in DCLN2 and DCLN3 cells. DCLNs have a potential value in distinguishing HC from patients with PD. The y-value of DCLNs may play a role in the differentiation of PD and AP.
Correlation of DCLN3 width diameter with clinical features of PD and AP
To explore the correlation between the DCLN and clinical characteristics in the PD and AP groups, the size of DCLN3(y) was used as a variable, and Spearman’s correlation analysis was performed (Figure 4). Age is an important factor in cerebral lymphatic drainage.
13
In our study, the size of the DCLN3(y) in the PD and AP groups showed a negative trend with age (P < .05). Interestingly, SCOPA-AUT (CS) and SCOPA-AUT (TS) were positively correlated with the size of DCLN3(y), except in the PD group. These results showed a significant negative correlation between DCLN3(y) and age in patients with PD and AP. This finding is consistent with the characteristics of PD and AP as degenerative diseases. The reason for the significant difference in DCLN3(y) between the AP and PD groups may be the different correlations between clinical symptoms and DCLN3(y). The severity of GS and TS symptoms positively correlated with the size of the DCLN3(y). Correlation of DCLNs with clinical features in PD and AP groups. There was a significant negative correlation between DCLN volume and age in the PD and AP groups. The size of DCLN3(y) was negatively correlated with SCOPA-AUT (CS) and SCOPA-AUT (TS) in PD, but not in AP. Abbreviations: HC, Healthy control; PD, Parkinson’s disease; AP, Atypical Parkinson’s disease; UPDRS, Unified Parkinson’s Disease Rating Scale; TS, Total score; US, Urinary system; SCOPA-AUT, the Scale for Outcomes in Parkinson’s Disease for Autonomic symptoms; CS, cardiovascular system.
ROC curves for DCLNs and clinical features in the differentiation of PD and AP
Correlation analysis and collinearity diagnosis were used to screen the variables to explore whether DCLNs could differentiate PD from AP. Spearman’s correlation was used to analyze the correlation between the clinical characteristics of the PD and AP groups. Two variables with a correlation coefficient >.6 were determined to have a strong correlation. Variables with tolerance close to 0 and variables with a Variance Inflation Factor >10 were considered collinear (results not shown). DCLN 2 and 3 (y) and SCOPA-AUT (GS) were removed.
Independent factors analysis of PD and AP.

Note: Binary logistic regression was used to identify independent factors distinguishing PD from AP. To include more influencing factors, P < .15 was considered statistically significant. Thus, Disease duration, MDS-UPDRS ΙΙ, SCOPA-AUT (T) and DCLN3 (Y) were screened out. Abbreviations: OR, odds ratio; CI, confidence interval; CS: Cardiovascular system; T: Thermoregulation; MDS-UPDRS: MDS-Unified Parkinson’s Disease Rating Scale; DCLN3 (y), y-value in DCLN3.
ROC curves for DCLNs and clinical features in the differentiation of PD and AP.
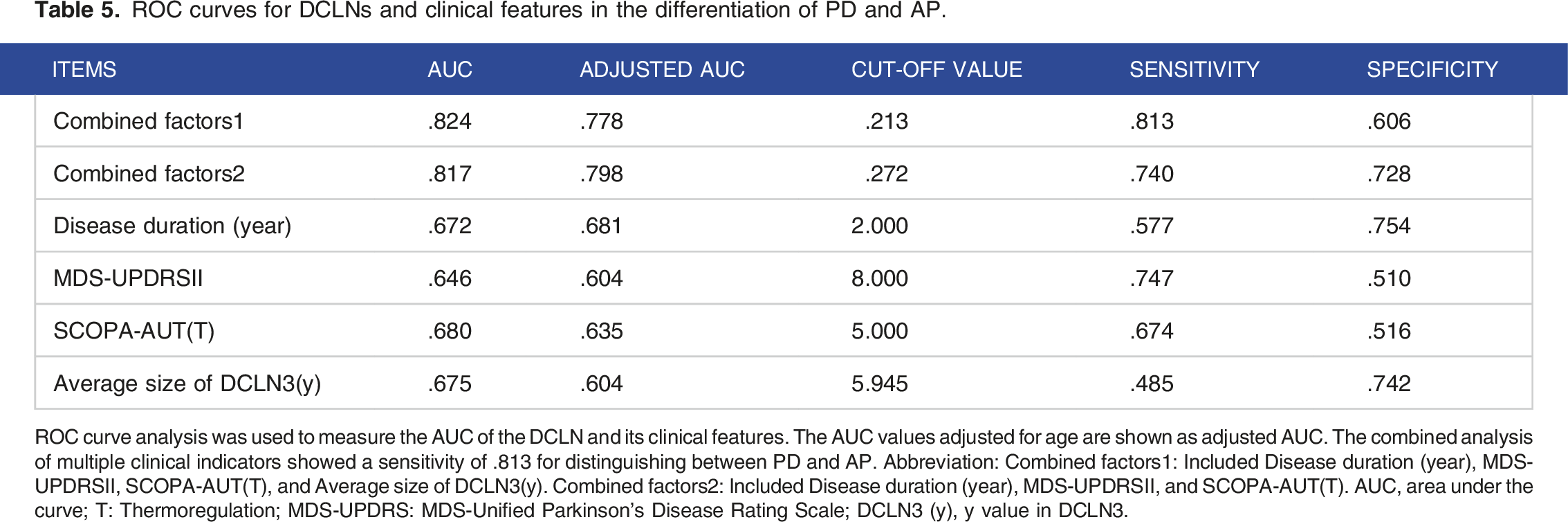
ROC curve analysis was used to measure the AUC of the DCLN and its clinical features. The AUC values adjusted for age are shown as adjusted AUC. The combined analysis of multiple clinical indicators showed a sensitivity of .813 for distinguishing between PD and AP. Abbreviation: Combined factors1: Included Disease duration (year), MDS-UPDRSΙΙ, SCOPA-AUT(T), and Average size of DCLN3(y). Combined factors2: Included Disease duration (year), MDS-UPDRSΙΙ, and SCOPA-AUT(T). AUC, area under the curve; T: Thermoregulation; MDS-UPDRS: MDS-Unified Parkinson’s Disease Rating Scale; DCLN3 (y), y value in DCLN3.

ROC curves of DCLN3(y) and clinical features to predict AP adjusted with age. ROC curves were used to measure the AUC of DCLN and laboratory markers to predict AP adjusted with age. (a): ROC curve was used to analyze the combined diagnostic efficacy of disease duration (years), MDS-UPDRSΙΙ, SCOPA-AUT(T) and DCLN3 average size (y). (b): ROC curve was used to analyze the combined diagnostic efficacy of disease duration (years), MDS-UPDRSΙΙ and SCOPA-AUT(T). (c): ROC curve for Disease duration. (d). ROC curve for UPDRSⅡscore. (e). ROC curve for SCOPA-AUT(T). f. ROC curve for DCLN3(y). The black lines are crude estimations of AUC. The red lines are AROC curves corrected age. After correction for age, the AUC of each index decreased. Abbreviations: ROC, receiver operating characteristic, AUC: Area under the curve; DCLN, deep cervical lymph nodes; AP, atypical Parkinson’s disease; MDS-UPDRSΙΙ, Movement Disorder Society Sponsored Revision Unified Parkinson’s Disease Rating Scale II; SCOPA-AUT, the Scale for Outcomes in Parkinson’s Disease for Autonomic symptoms; AROC, accommodating variables in the ROC analysis.
Discussion
This study aimed to explore potential connections between DCLNs and PD-related diseases. In this study, there was a significant difference in the DCLN between the HC and PD groups. The DCLN in the PD group were generally smaller than those in the HC group in terms of volume and each axis, and the difference was more significant for DCLN2 and DCLN3. Research on multiple sclerosis has shown that the size of DCLNs at Robbins IIA and III levels is more sensitive than other levels in reflecting brain lymphatic drainage. 40 The DCLNs are important terminal stations for the drainage of waste products from the brain lymphatic system. A study evaluating lymphatic system function using the DTI-ALPS method indicated that brain lymphatic drainage function in patients with PD was lower than that in HC subjects. 41 The above results confirm that the DCLN can distinguish between HC and PD. The change in DCLN size is positively correlated with cerebral lymphatic drainage function; that is, when the lymphatic drainage function is reduced, the DCLN is reduced. The location of the altered DCLN was consistent with that reported in previous studies. 40
Aging is an important factor that affects the drainage of the cerebral lymphatic system. Aging causes age-related morphological changes in the glymphatic system 42 and MLV, 43 resulting in a decrease in the flow rate of cerebrospinal fluid content to the lymphatic vessels surrounding the nodes, decreasing the drainage of macromolecules to the DCLNs. 44 Whether healthy or not, the older the age, the lower the cerebral lymphatic drainage function and the less macromolecular metabolic waste drained into the DCLNs. In the present study, the trend of DCLNs with age in each group of subjects was consistent with that reported previously. Furthermore, there was a significant negative correlation between DCLN size and age in the PD and AP groups, with the DCLN size changing most closely with age in the PD group. AQP4 is an important channel for the excretion of macromolecular proteins from the glymphatic system. The overexpression of a-syn in PD may lead to the dysfunction of the cerebral lymphatic system by reducing the expression of AQP4. 45 However, the overexpression and abnormal folding of a-syn may lead to the emergence of oxidative damage mechanisms, such as endoplasmic reticulum stress, and accelerate the aging of tissues via the a-syn clearance pathway. 46
In addition, PD duration was negatively correlated with DCLN3(y). This indicates that the DCLN3(y) diameter decreased as the disease progressed. PD is an irreversible disease, and this result is consistent with the characteristics of PD. Interestingly, the DCLN3(y) levels were higher in the AP group than in the PD and HC groups. Some researchers reached similar conclusions when analyzing meningeal lymphatic drainage using MRI. The cerebral lymphatic drainage rate and perfusion of the DCLN were slower in patients with PD compared with controls. 19 Impairment of meningeal lymphatic function in patients with PD is more obvious than in patients with AP, and the drainage efficiency of AP is higher than that of PD. 19 We believe that the above phenomena may be related to the pathophysiological differences between PD and AP. First, this result may be related to the differences in the composition and structure of the pathological proteins that need to be metabolized in the two diseases. In this study, MSA was predominant in the AP group, whereas PD and MSA are known as α-synucleinopathies. The characteristic inclusions of PD are Lewy bodies, which are spherical in shape and composed of filamentous α-syn, ubiquitin, and neurofilaments. The composition of MSA inclusions, called gliocytoplasmic inclusions (GCIs), is more complex than that of Lewy bodies. GCIs have a triangular or sickle-like morphology. 47 Studies have shown that inflammatory changes in AP are more significant than those in PD. The levels of neuroinflammation in PSP and MAS are much higher than in patients with PD. 45 Simultaneously, inflammation can accelerate the injury to the lymphatic vessel endothelium and increase endothelial permeability. It can also reduce lymphatic vessel activity through an nitric oxide (NO) dependent mechanism, resulting in the clearance of metabolites. 48 This leads to impaired excretion of pathological proteins and an inflammatory response in LN. We speculate that metabolic changes in the glymphatic system may occur before clinical symptoms and brain lymphatic metabolism may be the cause of the clinical symptoms of PD and AP. This may explain why there was no significant correlation between the clinical symptoms of PD, AP, and DCLN.
Some patients with AP may be diagnosed with ‘clinically probable PD’ in the early stages of the disease due to atypical symptoms. This may lead to difficulties in obtaining an accurate diagnosis and effective drug intervention during the early stages of the disease. The ROC curve was used to comprehensively evaluate diagnostic accuracy. Age was corrected for in this study because of the difference in age between the PD and AP groups, and the fact that the DCLN may be affected to some extent by age. ROC curve analysis showed that the combination of DCLN3(y), disease duration, MDS-UPDRS II score, and SCOPA-AUT (T) significantly improved the sensitivity of AP diagnosis. The sensitivity was better than that of the other methods. The pathophysiological changes in AP may be closely related to neuroinflammation. The size of DCLN3(y) in AP is significantly larger than that in PD, which may be related to the reduced removal of noxious molecules, increased inflammatory state, and deleterious neurotoxic stimuli. 49 In the correlation analysis, disease duration was significantly correlated with DCLN changes in patients with PD, and the size decreased with prolonged disease duration in patients with PD, possibly as a result of blockage of the brain lymphatic system by an increasing number of macromolecular proteins. However, the disease course of AP is shorter than that of PD and the disease progresses rapidly. Inflammation is a pathogenic mechanism underlying AP. A change in the DCLN size may occur earlier than that in the disease. In addition, the characteristic clinical manifestations that distinguish AP from PD are that patients with AP tend to have more psychiatric symptoms, such as increased MDS-UPDRSII scores, and more severe autonomic dysfunction, such as increased SCOPA-AUT(T) scores. Therefore, in commonly used clinical diagnostic means, the superposition of DCLN measurements can be considered to increase the pathophysiological diagnostic means to a certain extent, and the diagnosis of AP can be better achieved.
The present study had certain limitations. Although a minimum sample size was calculated, it was relatively small for this exploratory study. Follow-up studies with larger sample sizes at multiple centers should be conducted to further investigate the relationship between DCLNs, PD, and other related diseases. This study mainly involved patients with early- and mid-stage PD, and further studies are needed to investigate the characteristics of DCLNs in patients with late-stage PD. This was a cross-sectional study, and it was difficult to comprehensively understand the characteristics of DCLNs with disease progression.
Conclusion
In our study, the use of ultrasound to assess the size of DCLNs to indirectly understand the condition of cerebral lymphatic drainage in neurodegenerative diseases was an innovative approach that has not yet been reported. Similarly, the relationship between DCLNs and neurological diseases has been poorly studied. An important result of the present study is that we found the location of DCLNs that clinically distinguished healthy people from those with PD and AP. In addition, DCLNs can improve the sensitivity of AP discrimination.
Footnotes
Acknowledgements
We gratefully acknowledge Dr Zhen Zhang’s assistance in preparing this manuscript.
Author contributions
Zhaoying Dong: Conceptualization, Data curation, Investigation, Methodology, Writing - original draft, Writing - review & editing, Xinyi Du: Data curation, Formal analysis, Software, Writing - original draft, Ling Wang: Data curation, Investigation, Software, Writing - original draft, Xiaoya Zou: Investigation, Methodology, Writing - review & editing, Hongzhou Zuo: Data curation, Formal analysis, Software, Yong Yan: Conceptualization, Data curation, Visualization, Guojun Chen: Conceptualization, Project administration, Visualization, Writing - review & editing, Oumei Cheng: Project administration, Resources, Supervision, Yong Zhang: Data curation, Investigation, Methodology, Project administration, Writing - review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the National Natural Science Foundation of China (No. 81871002) and the Joint Project of the Chongqing Health Commission and Science and Technology Bureau (2020MSXM003;2022MSXM182).
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
