Abstract
Direct oral anticoagulants (DOACs) are indicated by the European Medicines Agency and US Food and Drug Administration for stroke prevention in patients with nonvalvular atrial fibrillation (AF). The role of DOACs in patients with AF and concomitant valvular heart disease (VHD) is less clear. Recent subanalyses of randomized controlled trials and meta-analyses have evaluated the role of DOACs in patients with AF and VHD. Patients with native aortic valve disease, tricuspid valve disease, or mitral regurgitation will be the primary focus as these represent the majority of VHD represented in the DOAC AF trials. Limited data exist on the role in patients with rheumatic mitral stenosis, mechanical heart valves, and mitral valve repair. This review provides the current clinical and scientific data pertaining to the safety and efficacy of DOAC use in patients with VHD.
Introduction
Direct oral anticoagulants (DOACs)—apixaban, dabigatran, edoxaban, and rivaroxaban—are preferred alternatives to warfarin for the prevention and management of many thromboembolic diseases, including atrial fibrillation (AF). However, DOAC clinical trials typically excluded patients with certain types of valvular heart disease (VHD), and uncertainty remains as to whether the use of DOACs is safe or effective in this population. Valvular heart disease is a broad category of disorders characterized by damage to or a defect in one or more of the 4 heart valves. 1 Clinical trials of DOAC use in patients with AF included only a small proportion of patients with valve-related disorders (13%-26%), and yet as many as 60% of patients with AF have concomitant VHD. 2 Patients with mechanical heart valves were systematically excluded from DOAC clinical trials, and there have been very few clinical trials evaluating DOAC use in these specific populations.
It is hypothesized that thrombus formation in patients with nonvalvular AF may be different from that of patients with VHD. Patients with AF typically develop thrombi in the left atrial appendage. In patients with mechanical prosthetic heart valves, thrombi commonly form on the prosthesis or in the left atrium as a result of non-physiologic blood flow patterns due to prosthesis. 3 Patients with bioprosthetic heart valves are at a lower risk of thrombosis, but the risk is not zero. Risk of thrombosis in patients who receive a bioprosthetic valve increases in the setting of concurrent AF or mitral stenosis (MS). 4 Porcine valves confer a higher risk of thrombosis than pericardial valves. In both cases, thrombosis can be propagated by decreased cardiac output and valve deterioration. 5 While the exact mechanism is unclear, the presence of MS increases the risk of thrombosis as well, likely related to decreased blood flow through the left atrium. 6
This review aims to describe the subanalyses of major DOAC clinical trials that evaluated the safety and efficacy outcomes in enrolled patients with AF and VHD. This review also presents the data regarding the use of apixaban and dabigatran in animal models and human patients with artificial heart valves.
Direct Oral Anticoagulant Use in Patients With AF and VHD
Current European Medicines Agency (EMA) and US Food and Drug Administration (FDA) approval of apixaban, dabigatran, edoxaban, and rivaroxaban only includes the indication of patients with nonvalvular AF. However, all 4 anticoagulants have data to support their use in patients with certain types of valvular AF as well. Subanalyses of the DOAC trials for AF reviewed data on patients who had mitral regurgitation (MR), mild MS, aortic regurgitation (AR), aortic stenosis (AS), tricuspid regurgitation (TR), and previous valve surgery (Table 1).
Valvular heart disease subanalyses of direct oral anticoagulant atrial fibrillation trials.3,7–13
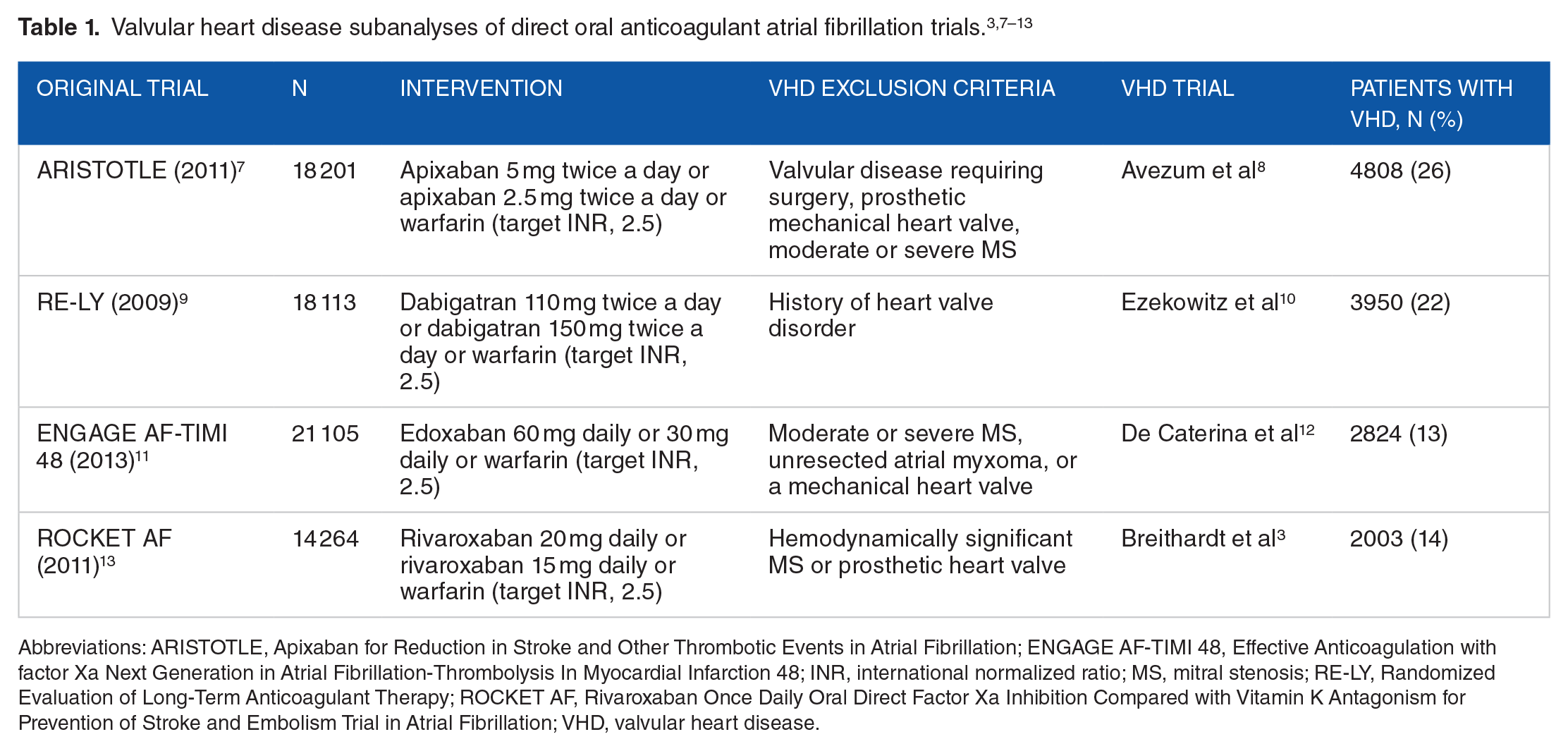
Abbreviations: ARISTOTLE, Apixaban for Reduction in Stroke and Other Thrombotic Events in Atrial Fibrillation; ENGAGE AF-TIMI 48, Effective Anticoagulation with factor Xa Next Generation in Atrial Fibrillation-Thrombolysis In Myocardial Infarction 48; INR, international normalized ratio; MS, mitral stenosis; RE-LY, Randomized Evaluation of Long-Term Anticoagulant Therapy; ROCKET AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; VHD, valvular heart disease.
The Apixaban for Reduction in Stroke and Other Thrombotic Events in Atrial Fibrillation (ARISTOTLE) trial enrolled 18 201 patients with AF and at least 1 risk factor for stroke to receive apixaban 5 mg orally twice a day and warfarin placebo or dose-adjusted warfarin (International Normalized Ratio [INR] target, 2.5) and apixaban placebo. Patients in the apixaban group had fewer strokes or systemic emboli, less bleeding, and improved mortality compared with those in the warfarin group.
7
The ARISTOTLE trial excluded patients with moderate or severe MS and mechanical heart valves, but enrolled a substantial number of patients with other types of VHD. Of the 18 201 enrolled patients, VHD status was available for 18 197, and 4808 (26.4%) of those patients had a history of moderate or severe VHD or previous valve surgery. The majority of patients with VHD had MR or TR. A secondary analysis of efficacy and safety outcomes in the subgroup of VHD patients found that those with VHD had higher rates of stroke or systemic embolism (3.2% vs 2.4%; hazard ratio [HR], 1.34; 95% confidence interval [CI], 1.10-1.62;
The Randomized Evaluation of Long-Term Anticoagulant Therapy (RE-LY) trial enrolled 18 113 patients with AF to receive dabigatran 150 mg orally twice a day (D150), dabigatran 110 mg orally twice a day (D110), or dose-adjusted warfarin (INR goal, 2.5). Patients in the D110 group experienced similar rates of stroke or systemic emboli as warfarin but with less bleeding, and patients in the D150 group experienced lower rates of stroke or systemic embolism than warfarin with similar rates of major bleeding.
9
The RE-LY trial excluded patients with significant MS, prosthetic heart valves, and VHD requiring an intervention, but enrolled a large number of patients with other types of VHD. A secondary analysis of the RE-LY trial found that 3950 patients enrolled (21.8%) had VHD, with MR being the most common. Rates of mortality were no different between patients with and without VHD, regardless of the treatment arm (propensity score–adjusted HR, 1.09; 95% CI, 0.96-1.23;
The Effective Anticoagulation with factor Xa Next Generation in Atrial Fibrillation-Thrombolysis In Myocardial Infarction 48 (ENGAGE AF-TIMI 48) trial enrolled 21 105 patients with moderate- to high-risk AF to receive high-dose edoxaban 60 mg orally once daily (E60), low-dose edoxaban 30 mg orally once daily (E30), or dose-adjusted warfarin (target INR, 2.5). Both the E60 and E30 regimens were found to be noninferior to warfarin with respect to the prevention of stroke or systemic embolism, with less risk of bleeding.
11
Patients were excluded from ENGAGE AF-TIMI 48 if they had moderate or severe MS or a mechanical heart valve. However, a limited number of patients with other types of VHD were included. Of the 21 105 patients enrolled, 21 046 had available VHD data. Of these, 2824 (13%) patients had a history of moderate or severe VHD, the most common being MR. Patients analyzed had moderate AR or MR, AS, or prior valve surgery (ie, bioprosthesis replacement, valve repair, valvuloplasty). Patients with VHD had increased rates of mortality (HR, 1.40; 95% CI, 1.26-1.56;
The Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) trial enrolled 14 264 patients with nonvalvular AF at moderate to high risk of stroke to receive rivaroxaban 20 mg orally once daily, rivaroxaban 15 mg orally once daily (in patients with a creatinine clearance [CrCl] of 30-49 mL/min), or dose-adjusted warfarin (target INR, 2.5). Rivaroxaban was found to be noninferior to warfarin with respect to prevention of stroke or systemic embolism with less risk of bleeding.
13
Patients with MS or artificial heart valves were excluded from the ROCKET AF trial, but a secondary analysis in 14 119 intention-to-treat patients was performed to assess the efficacy and safety outcomes in 2003 patients (14%) with significant valve disease defined as AS, MR, or AR. Stroke or systemic embolism occurred twice as often in the AS group (4.21) than either the MR or AR (2.01) or no VHD groups (2.09 events per 100 patient-years;
Between 13% and 26% of patients enrolled in the 4 landmark trials that supported the approval of each DOAC for cardioembolic stroke prevention in patients with nonvalvular AF had evidence of VHD. The most common form of VHD in these patients was MR. In the subpopulations of patients with VHD and AF, DOAC safety and efficacy were consistent with what was seen in the larger trial cohort. Data from 2 meta-analyses confirmed that pooling the results from the subanalyses of the 4 major trials described above demonstrated that DOAC use reduced the risk of stroke or systemic embolism to a similar degree in patients with AF and VHD compared with those with AF without VHD.15,16 Across the 4 trials, patients with AF and VHD were more likely to be female; have permanent or persistent AF; have a history of heart failure, myocardial infarction, or coronary artery disease (CAD); and previously used warfarin compared with patients with AF and no VHD. Patients with VHD were at higher risk of bleeding than those without VHD (HR, 1.24; 95% CI, 1.14-1.34;
Direct Oral Anticoagulant Use in Mechanical and/or Bioprosthetic Valve Replacement
Phase 3 clinical trials of apixaban, dabigatran, edoxaban, and rivaroxaban for AF excluded patients with mechanical heart valves. However, both apixaban and dabigatran have been studied in preclinical swine models in which individual swine were implanted with a bileaflet mechanical aortic valve. Swine are commonly used to model human thromboembolic disorders due to similarities to human hematologic and coagulation properties.
17
In the dabigatran preclinical study, 30 implanted swine were randomized to no anticoagulation (control), enoxaparin 2 mg/kg subcutaneous injection twice a day, or dabigatran 20 mg/kg orally twice a day. Postmortem valve thrombus measurements at day 30 demonstrated a significant reduction in thrombus size in the drug groups (enoxaparin 121 ± 128 mg and dabigatran 19 ± 31 mg) versus control (638 ± 895 mg). Thrombus size in the dabigatran group was significantly smaller than that of the enoxaparin group (
Following the preclinical study of dabigatran in swine with implanted mechanical heart valves, dabigatran was studied in human patients with mechanical heart valves. The Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients after Heart Valve Replacement (RE-ALIGN) study randomized 252 patients with a mechanical bileaflet mitral or aortic valve to receive dabigatran or warfarin in a 2:1 ratio. Based on pharmacokinetic data, patients with a CrCl <70 mL/min received dabigatran 150 mg orally twice a day, those with a CrCl of 70 to 109 mL/min received dabigatran 220 mg orally twice a day, and those with a CrCl >110 mL/min received dabigatran 300 mg orally twice a day. These doses were used to achieve a dabigatran trough level of >50 ng/mL. Patients randomized to warfarin had a target INR of 2 to 3 if they had a mechanical aortic valve with no additional risk factors and a target INR of 2.5 to 3.5 if they had a mechanical aortic valve with additional risk factors (eg, previous thromboembolism, MS) or a mechanical mitral valve. The study was stopped early due to an interim safety analysis that determined there was excess risk and no benefit for the patients in the dabigatran group. Stroke occurred in 9 dabigatran-treated patients (5%) compared with no strokes in the warfarin group. Bleeding occurred in 45 dabigatran-treated patients (27%) compared with 10 patients (12%) in the warfarin group (HR, 2.45; 95% CI, 1.23-4.86;
Despite the unfavorable outcome in the RE-ALIGN study, another group of investigators evaluated dabigatran use in human patients with bioprosthetic heart valves. The Dabigatran Versus Warfarin After Bioprosthesis Valve Replacement for the Management of Atrial Fibrillation Postoperatively (DAWA) study aimed to evaluate the efficacy and safety of dabigatran in patients with bioprosthetic mitral and/or aortic valve replacement and AF. Patients were randomized in a 1:1 ratio to receive dabigatran 110 mg orally twice a day or dose-adjusted warfarin (INR goal, 2.5) with a primary end point being the presence of new intracardiac thrombus at 90 days. The study was stopped early due to low enrollment. At 90 days, 1 patient (8.3%) in the warfarin group developed an intracardiac thrombus, 1 patient (8.3%) in the warfarin group developed an ischemic stroke, and 1 patient (6.7%) in the dabigatran group experienced a reversible ischemic neurological deficit. Bleeding occurred in 2 patients (16.7%) in the warfarin group and 1 patient (6.7%) in the dabigatran group. The DAWA study investigators concluded that this small pilot study is hypothesis generating for future studies to compare the use of dabigatran versus warfarin in a larger patient population with bioprosthetic heart valves. 21
A retrospective analysis of patients prescribed a DOAC for AF after bioprosthetic valve implantation yielded similar results to the DAWA study. Seventy-three patients at a single institution who were prescribed dabigatran (44; 60.3%), rivaroxaban (25; 34.2%), or apixaban (4; 5.5%) for AF after bioprosthetic valve replacement experienced no significant thromboembolic events. Patients were started on DOAC therapy for AF a mean of 990 ± 1029.1 days after valve replacement and had been receiving DOAC therapy for a mean of 511.8 ± 400.8 days at the time of the retrospective analysis. A majority of patients (72.6%) were concomitantly taking aspirin. One patient (1.4%) experienced a transient ischemic attack and there were 6 (8.2%) minor and 5 (6.9%) major bleeding events. Based on the data collected from this small retrospective cohort, study investigators concluded that DOACs are likely safe and effective for AF in patients with bioprosthetic valves. 22
Both ARISTOTLE and ENGAGE AF-TIMI 48 performed subanalyses of efficacy and safety in patients with bioprosthetic heart valves treated with apixaban and edoxaban, respectively. A subanalysis of 82 patients with bioprosthetic valves enrolled in ARISTOTLE (apixaban, n = 41 and warfarin, n = 41).
23
In this small cohort of patients, there were 2 strokes in the apixaban group and none in the warfarin group (
A pre-planned subanalysis of patients enrolled in ENGAGE AF-TIMI 48 who had experienced a bioprosthetic heart valve replacement >30 days prior to study randomization (n = 191; 0.9% of the total trial population) demonstrated that this population experienced similar rates of stroke or systemic embolism whether they received high- or low-dose edoxaban or warfarin (E60 vs warfarin: HR, 0.37; 95% CI, 0.10-1.42;
A case series of 27 patients with AF and a biological prosthesis, repaired mitral valve, or tubular aortic graft who were treated with rivaroxaban (mean dose, 16 ± 2 mg) found that no recurrent thrombotic events occurred in these patients. Minor bleeding (nose bleeding, n = 3 and hematuria, n = 2) resulted in a rivaroxaban dose reduction from 20 to 15 mg once daily. These patients were prescribed rivaroxaban instead of warfarin due to warfarin-related adverse effects, labile INR results, or cognitive impairment and received rivaroxaban for a mean of 15 months. The authors concluded that this case series is hypothesis generating for future studies of DOAC use in this patient population. 25
Transcatheter aortic valve replacement (TAVR) or transcatheter aortic valve implantation (TAVI) is becoming an increasingly prevalent procedure for patients with AS, and anticoagulation with DOACs in patients undergoing these procedures is of interest. One such study is the Anti-Thrombotic Strategy After Trans-Aortic Valve Implantation for Aortic Stenosis (ATLANTIS). The objective of this study is to demonstrate superiority of apixaban versus standard of care in patients who have undergone a successful TAVI procedure. 26 The Global Study Comparing a rivAroxaban-based Antithrombotic Strategy to an antiplatelet Strategy After Transcatheter aortic valve rEplacement to Optimize Clinical Outcomes (GALILEO) will assess whether a rivaroxaban-based regimen is superior in reducing death or first antithrombotic events compared with an antiplatelet-based regimen in patients who have undergone a successful TAVR procedure. 27 The results of these trials may influence anticoagulation in patients after TAVI or TAVR.
The limited data in patients with artificial heart valves make it difficult to draw uniform conclusions as to whether DOAC use is safe or effective in this population. Currently available data have demonstrated that DOAC use (specifically dabigatran) in patients with mechanical heart valves is neither safe nor effective. However, prospective and retrospective data analyses of DOAC use in patients with bioprosthetic valves are promising. In both cases—patients with mechanical or bioprosthetic valves—more data are needed to justify whether the use of DOACs for thromboembolic prevention is safe and effective.
Implications for Practice
Patients with AF and concurrent VHD have been underrepresented in clinical trials evaluating the safety and efficacy of DOAC therapy. Despite the lack of studies specifically evaluating DOACs in patients with VHD, there are subanalysis level data available for the major DOAC AF trials as discussed above. In addition, the increasing use of DOACs for anticoagulation therapy secondary to patient-specific factors (eg., ability to adhere to lab monitoring and/or dietary restrictions) has resulted in a need to further evaluate the role of DOACs in the VHD population with the currently available literature.
This review has highlighted the data across the major DOAC AF trials, which support the role of DOACs as safe and effective options in patients with AR, AS, MR, mild MS, and TR. Use of DOACs is likely safe and effective in patients with bioprosthetic valve replacements as well. Both the US and European guidelines recommend warfarin as the anticoagulant of choice in patients with mechanical heart valves or severe MS.4,28
With ARISTOTLE, ENGAGE AF-TIMI 48, RE-LY, and ROCKET AF trials demonstrating the DOACs had at least noninferior reductions in stroke and systemic embolism compared with warfarin, these trials are a driver for the expansion of DOAC use in the VHD population. The American College of Cardiology (ACC) and the American Heart Association (AHA) further recognized the expanded available literature in the VHD population with their 2017 guideline update. These updated guidelines reinforce the recommendation to anticoagulate patients with AF and a CHA2DS2-VASc score of 2 or greater with native aortic valve disease, tricuspid valve disease, or MR. They went on to add a new recommendation that “It is reasonable to use a DOAC as an alternative to a vitamin K antagonist (VKA) in patients with AF and native aortic valve disease, tricuspid valve disease, or MR and a CHA2DS2-VASc score of 2 or greater.” 4 The authors of the guidelines rated this recommendation as class IIa, with level of evidence C-LD indicating moderate benefit for risk with limited data consisting of subanalyses of randomized trials and meta-analyses of the major DOAC trials in patients with AF. With the recent release of these 2017 guideline recommendations, it has yet to be determined how the expanded recommendations will affect formulary decisions that will affect how and when patients with VHD will be able to get prescription coverage for DOACs. There will be a need to further clarify documentation of VHD status in patients with AF to optimize patients’ access to available indicated anticoagulation therapies (eg, DOACs).
This review has reinforced that the medical literature does not currently support a role for DOACs in patients with rheumatic MS and mechanical valve replacement based on limited data available evaluating the safety and efficacy in this patient population. The biggest driver for the preference of VKA use in these populations is secondary to the exclusion of these forms of VHD from the clinical trials evaluating DOAC safety and efficacy in patients with AF. The ACC/AHA 2017 updated guidelines reinforce this concept by continuing to recommend anticoagulation with VKAs and provide no mention of DOACs in their recommendations for these forms of VHD. Studies to date show promising data for DOAC use in patients with bioprosthetic valve replacements. Future studies are warranted to evaluate the safety and efficacy of DOAC use across all forms of VHD, including both patients with mechanical and bioprosthetic heart valves.
Conclusions
Apixaban, dabigatran, edoxaban, and rivaroxaban are indicated by the EMA and FDA for patients with nonvalvular AF defined as AF in the absence of rheumatic MS, a mechanical or bioprosthetic heart valve, or mitral valve repair. Evaluation of DOAC clinical trial data has demonstrated a role for DOACs as alternative anticoagulation options to VKAs in patients with AF and native aortic valve disease, tricuspid valve disease, or MR and a CHA2DS2-VASc score of 2 or greater. Recent VHD guidelines have incorporated these expanded DOAC indications, and thus, increased DOAC use in patients with AF and VHD is anticipated. A key take-away in managing patients with VHD receiving an anticoagulant is the known increased risk of bleeding in patients with VHD versus those without VHD, irrespective of the choice of the anticoagulant therapy.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SLA and JCM conceived and designed the experiments, analyzed the data, wrote the first draft of the manuscript, contributed to the writing of the manuscript, agree with manuscript results and conclusions, jointly developed the structure and arguments for the paper, and made critical revisions and approved the final version. Both authors reviewed and approved the final manuscript.
