Abstract
Background:
Ischemic stroke or transient ischemic attack (TIA) occurs in 1.4% of patients with atrial fibrillation (AF) per year despite treatment with direct oral anticoagulants (DOACs). This group of patients is poorly studied, and possible causes for this are not fully understood.
Objectives:
The aim of the study was to analyze DOAC plasma levels in patients who had ischemic stroke or TIA despite treatment with DOAC.
Design:
Monocentric retrospective study.
Methods:
We selected consecutive DOAC-treated patients with acute ischemic stroke or TIA, admitted during a 2-year period, with measurement of DOAC levels ⩽24 h after hospital admission. Patients with and without low DOAC levels (<50 ng/mL) were compared.
Results:
We included 163 patients with median age of 80 years, 46.6% of female sex. AF was the most frequent indication for DOAC (83.4%). Low DOAC levels were found in 42.3% of patients. Patients with low DOAC levels more frequently were being treated with an inappropriate low DOAC dose (29.4% vs 16.0%, p = 0.004), more frequently had unknown last DOAC intake or intake >48 h before admission (72.5% vs 57.5%, p = 0.004), and had higher baseline NIHSS (8 vs 4, p = 0.001). Lower DOAC levels were associated with large or medium vessel occlusion (LVO/MeVO; odds ratio per 10 ng/mL-increase = 0.89, 95% confidence interval = 0.85–0.94, p < 0.001). Cardiac pathology as the only potentially causal mechanism was more frequent in low DOAC levels patients (76.8% vs 60.6%, p = 0.029). Thirty patients (18.4%) had ⩾2 potentially causal mechanisms, 18 (11.0%) had potentially causal mechanisms other than cardiac pathology, and 5 (3.1%) had no identifiable potentially causal mechanism.
Conclusion:
In DOAC-treated patients with ischemic stroke or TIA, low DOAC levels were found in 2/5 of patients. Lower DOAC levels are associated with increased stroke severity and the presence of LVO/MeVO. The profile of stroke etiology in DOAC-treated patients varies between groups with and without low DOAC levels.
Introduction
In the past 15 years, four non-vitamin K direct oral anticoagulants (DOAC) were approved for patients with non-valvular atrial fibrillation (AF). 1 Large prospective international registries show an increasing use of DOACs in patients with non-valvular AF, 2 and their use is expected to further increase due to expansion of the indications for their use and due to the future availability of generic drugs. 3 Despite the efficacy of DOACs in reducing the incidence of ischemic stroke, DOAC-treated patients can still suffer ischemic stroke, with a risk of 1.4% per year in randomized controlled trials. 1 There are several reasons for patients with non-valvular AF suffering a thromboembolic event despite prior DOAC use, including treatment failure, poor compliance, underdosage, concomitant drug–drug interactions, competing thromboembolic mechanisms, and pharmacogenetic factors. Understanding the causes of breakthrough strokes is clinically relevant, as evidence from multiple prospective cohort studies shows that ischemic stroke recurrence in patients with AF who are taking anticoagulants at the time of the first event is twice as high as in patients with AF who were not taking anticoagulants at the time of the first event. 4 There is currently a high degree of uncertainty concerning the best management of these patients. 5 A better understanding of the reasons for AF-related ischemic stroke occurrence despite anticoagulation can help to tailor individualized secondary stroke prevention strategies. The measurement of DOAC plasma levels in the hyperacute phase of ischemic stroke in patients taking anticoagulation may be useful to identify specific causes of breakthrough stroke, to support decisions regarding intravenous thrombolysis, 6 and to support recommendations regarding antithrombotic therapy. 7 It may also provide information regarding possible pathophysiological mechanisms of stroke, as it was shown that lower DOAC plasma levels in acute ischemic stroke patients taking DOACs are associated with more severe strokes and higher prevalence of large vessel occlusion (LVO). 8 The goals of our study were (1) to calculate the proportion of patients with ischemic stroke despite taking DOAC with low DOAC plasma levels; (2) to analyze the association of DOAC plasma levels with the clinical severity of stroke and presence of LVO or medium vessel occlusion (MeVO); (3) to describe the profile of ischemic stroke etiology despite anticoagulation with DOAC.
Methods
In this retrospective monocentric observational study based on a local stroke register of consecutive stroke patients, all patients admitted to the stroke unit and neurointensive care unit of the Department of Neurology of a university hospital during a period of 2 years (January 2022–December 2023) were screened to identify patients in whom DOAC plasma levels were measured within 24 h of hospital admission. Among these patients, only patients with a final diagnosis of acute ischemic stroke or transient ischemic attack (TIA) were included in the final study population. Individual patient records were reviewed to collect the following information: demographic data, vascular risk factors, comorbidities, CHA2DS2VASc, indication for anticoagulation with DOAC, specific DOAC used, reported DOAC dose used, last DOAC intake, hospital admission time, baseline National Institutes of Health Stroke Scale (NIHSS), presence of LVO or MeVO at admission, acute reperfusion treatments (intravenous thrombolysis, mechanical thrombectomy), DOAC plasma levels (ng/mL), time of availability of DOAC plasma level in the laboratory informatics system, glomerular filtration rate at admission (using the CKD-EPI 2009 formula, mL/min/1.73 m2), stroke or TIA etiology, functional status at discharge, in-hospital mortality. Inappropriate low DOAC dose was defined as a dose inferior to the one recommended by the manufacturer according to the clinical indication, age, weight, and renal function. The presence of vessel occlusion was collected from the neuroimaging reports from computed tomography angiography or magnetic resonance angiography performed in the emergency department in the hyperacute phase of ischemic stroke or TIA. LVO was defined as an occlusion of the cervical or intracranial internal carotid artery, of the M1 segment of the middle cerebral artery, and of the basilar artery. MeVO was defined as occlusion of A1 or A2 segments of the anterior cerebral artery, M2, M3, or M4 segments of the middle cerebral artery, of P1, P2, or P3 segments of the posterior cerebral artery, and of V4 segment of the vertebral artery (without concomitant LVO). The phenotypic characterization of patients with ischemic stroke and TIA was defined according to the ASCOD classification. 9 Functional status at hospital discharge was collected from patient records and classified according to the modified Rankin Scale (mRS). Favorable functional outcome was defined as mRS of 0–2.
Measurement of DOAC plasma levels
Laboratory measurements of dabigatran, apixaban, rivaroxaban, and edoxaban were conducted using the ACL TOP® coagulation analyzer (Instrumentation Laboratory, Bedford, MA, USA). Each DOAC was quantified using either chromogenic or clot-based assays specific to its inhibitory target. All reagents were stored and handled according to the manufacturer’s guidelines, and the analyzer was calibrated and maintained according to the laboratory’s standard operating procedures. Edoxaban, apixaban, and rivaroxaban (direct factor Xa inhibitors), were measured using chromogenic anti-factor Xa assays. Specific calibrators and controls were used to ensure measurement accuracy. Dabigatran (direct thrombin inhibitor) was measured using a clot-based assay with a modified thrombin time. The recorded clotting time was proportional to the dabigatran concentration in the sample, with calibration curves employed for quantification. Citrate plasma samples were collected and centrifuged at 2500g for 10 min to obtain platelet-poor plasma and analyzed immediately. Low DOAC plasma levels were defined as <50 ng/mL.10,11
Statistical analysis
We performed a descriptive analysis of the final study population. We calculated the frequency of breakthrough ischemic stroke or TIA, defined by the presence of cardiac pathology as the only potentially causal mechanism according to the ASCOD classification, absence of any additional pathology for which a causal link is uncertain according to the ASCOD classification, treatment with adequate DOAC dose, and presence of DOAC plasma levels >100 ng/mL. We grouped our final study population in the group of patients with and without low DOAC plasma levels (<50 ng/mL), and compared these two groups of patients using chi-square tests (for categorical variables) and Mann–Whitney U tests (for continuous variables). Because there is currently no consensus regarding range of DOAC plasma levels which represent a clinically significant anticoagulation or high risk of hemorrhage under specific clinical conditions, we performed a sensitivity analysis by defining low DOAC plasma levels as <100 ng/mL. 12 We performed univariable and multivariable logistic binary regression analyses using the presence of LVO/MeVO as the dependent variable, and we included age and baseline variables with p < 0.1 in the univariable analysis in the model. We performed a sensitivity analysis by excluding patients with unknown last DOAC intake from the univariable and multivariable logistic binary regression analyses. We analyzed the relationship between baseline NIHSS and DOAC plasma levels using Spearman correlation. We compared DOAC plasma levels in patients with cardiac pathology as the only potentially causal mechanism for stroke or TIA with DOAC plasma levels in patients with other etiologies using the Mann-Whitney U-test. No imputation of missing data was performed. Threshold for statistical significance was set at an alpha level of 0.05. All statistical analyses and visualizations were performed in SPSS® Statistics (version 28.0.1.0; IBM Corporation, Armonk, NY, USA). This report follows The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Results
During the study period, 2891 patients were admitted in the stroke unit or neurointensive care unit. A total of 252 patients received DOAC plasma level measurement within 24 h after admission. After exclusion of 33 patients with intracranial hemorrhages and 56 patients with other diagnoses, 163 patients were included in the final study population (ischemic stroke = 146 patients; TIA = 17 patients). Among the initial patient population, we found 50 patients who experienced acute ischemic stroke and TIA despite DOAC, who did not receive DOAC plasma level measurements within 24 h of admission. In comparison with the final study population of 163 patients, these 50 patients were of similar age (p = 0.403), had a similar distribution of sex (p = 0.433), and had similar frequency of acute ischemic stroke as the index event (p = 0.560). Median age of the final study population was 80 years (interquartile range (IQR) 72–84 years), and 76 patients were female (46.6%). The most frequently used DOAC was apixaban (n = 82, 50.3%), followed by edoxaban (n = 45, 27.6%), rivaroxaban (n = 35, 21.5%), and dabigatran (n = 1, 0.6%). The most frequent indication for treatment with DOAC was AF (n = 136, 83.4%), followed by pulmonary embolism (n = 6, 3.7%), deep vein thrombosis (n = 4, 2.5%), cryptogenic embolic stroke (n = 4, 2.5%), and other indications (n = 13, 8.0%). Inappropriate low DOAC dose was found in 35 patients (21.5%). Only 53 patients (32.5%) were known to have taken DOAC in the previous 24 h before admission, 6 patients (3.7%) reported last DOAC intake longer than 24 h before admission but no later than 48 h, and 104 patients (63.8%) had taken DOAC longer than 48 h before admission, or last intake was unknown. The median time between admission and availability of DOAC plasma levels was 50 min (IQR 35–90 min), and 90% of patients had available DOAC plasma level results within 176 min of hospital admission. Low DOAC plasma levels (<50 ng/mL) were found in 69 patients (42.3%). Acute arterial occlusions (LVO/MeVO) were detected in 64 patients (39.3%), 38 of whom had LVO (23.3%) and 26 had MeVO (16%). Breakthrough ischemic stroke or TIA (defined by the presence of cardiac pathology as the only potentially causal mechanism according to the ASCOD classification, absence of any additional pathology for which causal link is uncertain according to the ASCOD classification, treatment with adequate DOAC dose, and presence of DOAC plasma levels >100 ng/mL) was found in 25 patients (15.3%).
Comparison of groups with and without low DOAC plasma levels
The groups of patients with and without low DOAC plasma levels were compared (Table 1). Patients with low DOAC plasma levels more frequently were being treated with an inappropriate low dose of DOAC according to the recommendation of the manufacturer (29.4% vs 16.0%, p = 0.004), had higher admission NIHSS (median 8 vs 4, p = 0.001, after exclusion of patients with TIA), more frequently had LVO/MeVO (56.5% vs 26.6%, p < 0.001), and more frequently underwent mechanical thrombectomy (46.4% vs 17%, p < 0.001). Among patients who underwent intravenous thrombolysis, no patient received reversal of the anticoagulation before treatment with thrombolytic agent. DOAC plasma levels were significantly lower in patients with LVO/MeVO in comparison to patients without LVO/MeVO (median 41 ng/mL (IQR 4–82 ng/mL) vs 99 ng/mL (IQR 38–178 ng/mL), p < 0.001). Distribution of time since last DOAC intake was different between the groups with or without low DOAC plasma levels (p = 0.004), with patients with low DOAC plasma levels more frequently having the last DOAC intake longer than 48 h before admission or with unknown last intake. The results of the sensitivity analyses (with low DOAC plasma levels defined as <100 ng/mL) did not change significantly, with exception that TIA as index event was more frequent in the group of high DOAC plasma levels (21.7 vs 3.9%, p < 0.001), that inappropriate low DOAC dose was similar in both groups, and that favorable functional outcome at discharge was more frequent in the group of patients with high DOAC plasma levels (55.0% vs 37.9%, p = 0.034; Supplemental Table 1).
Baseline characteristics, therapy, and outcome of the study population according to the presence of low DOAC plasma levels (<50 ng/mL).

DOAC, direct oral anticoagulant; IQR, interquartile range; mRS, modified Rankin Scale; TIA, transient ischemic attack.
A detailed distribution of DOAC plasma levels in the groups of patients with TIA, ischemic stroke without LVO/MeVO, ischemic stroke with MeVO, and ischemic stroke with LVO is presented in Figure 1. We found a significant correlation between baseline NIHSS and DOAC plasma levels (rho −0.268, 95% confidence interval (CI) −0.398 to −0.127, p < 0.001), indicating higher NIHSS scores in patients with lower DOAC plasma levels (Figure 2).

DOAC plasma levels (ng/mL) at admission according to the final diagnosis.

Correlation between DOAC plasma levels and baseline NIHSS.
In the multivariable logistic regression analysis, lower DOAC plasma levels were independently associated with the presence of LVO/MeVO (odds ratio per 10 ng/mL increase = 0.89, 95% CI = 0.84–0.94, p < 0.001; Table 2). The exclusion of patients with unknown last DOAC intake did not change the results significantly (Supplemental Table 2).
Univariable and multivariable logistic regression model using the presence of large or medium vessel occlusion as the dependent variable.
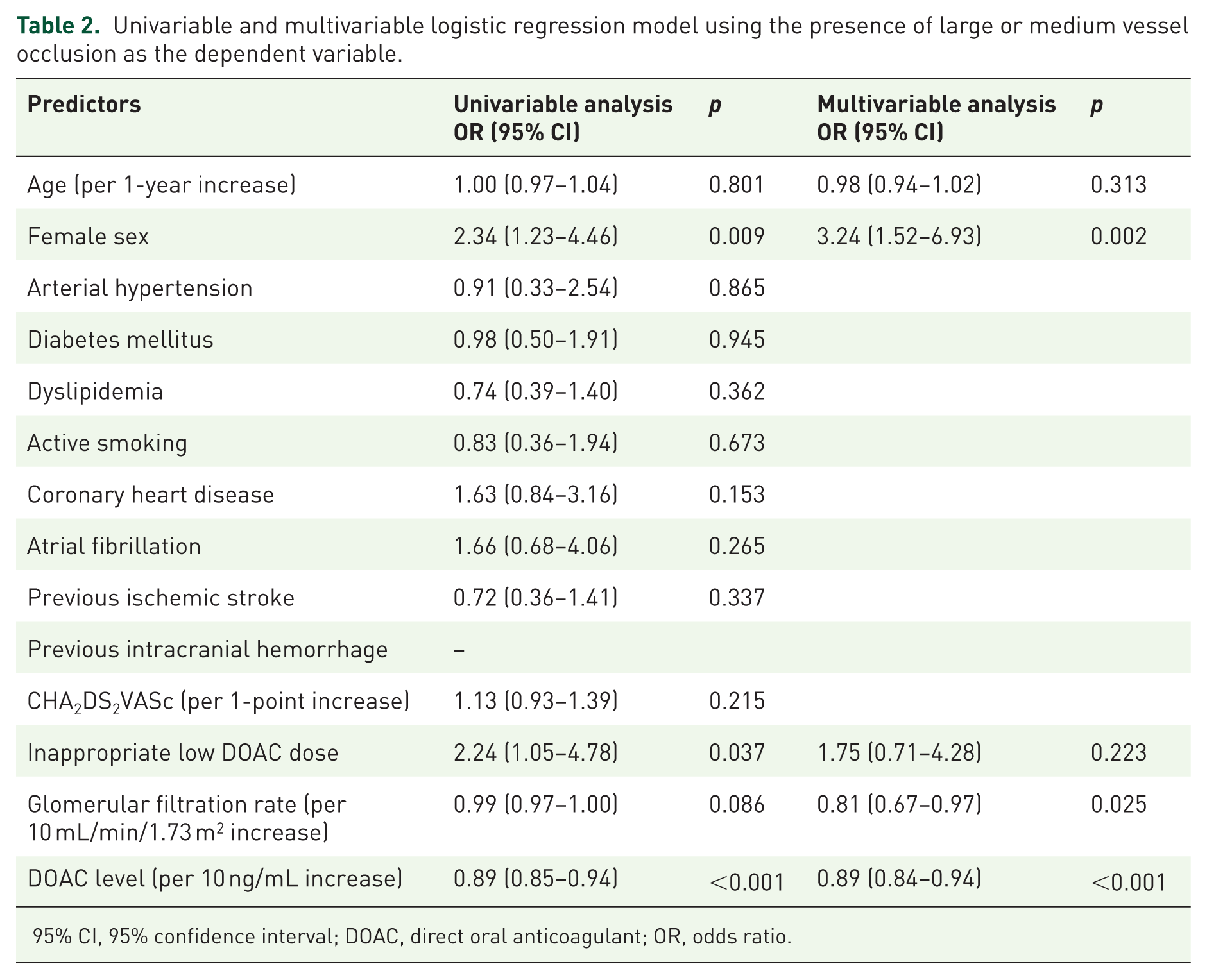
95% CI, 95% confidence interval; DOAC, direct oral anticoagulant; OR, odds ratio.
DOAC plasma levels and stroke/TIA etiology
According to the ASCOD classification, cardiac pathology was the only potentially causal mechanism in 110 patients (67.4%; Table 3). In this subgroup of patients, 20/110 patients had additional changes in other domains for which a causal link with ischemic stroke or TIA was uncertain (atherothrombosis = 19, small vessel disease = 1). Thirty patients had ⩾2 potentially causal mechanisms, the most frequent combination was cardiac pathology and atherothrombosis (n = 18), followed by cardiac pathology and small vessel disease (n = 12; Table 3).
Etiology of ischemic stroke or transient ischemic attack according to the ASCOD stroke phenotype classification.

ASCOD: A = atherothrombosis; S = small vessel disease; C = cardiac pathology; O = other causes; D = dissection. ASCOD causality grades: 0 = not detected; 1 = potentially causal; 2 = causal link is uncertain; 3 = causal link is unlikely but the disease is present.
Patients with low DOAC plasma levels (<50 ng/mL) more frequently had cardiac pathology as the only potentially causal mechanism (76.8% vs 60.6%, p = 0.029). This association did not persist when low DOAC plasma levels were defined as <100 ng/mL (72.8% vs 58.3%, p = 0.057). However, it persisted after exclusion of patients with unknown timepoint of last DOAC intake (76.3% vs 54.6%, p = 0.040) and after exclusion of the 20 patients with cardiac pathology as the only potentially causal mechanism and concomitant pathology for which a causal link was uncertain (65.2% vs 47.9%, p = 0.028). DOAC plasma levels were lower in the group of patients with cardiac pathology as the only potentially causal mechanism (54.2 ng/mL, IQR = 12.8–140.7) in comparison to the rest of the patients (98.8 ng/mL, IQR = 38.2–168.5, p = 0.026).
Discussion
One of the main conclusions of the current study is that, in DOAC-treated patients who experience ischemic stroke or TIA, low DOAC plasma levels (defined as <50 ng/mL) at hospital admission were found in two in every five patients. Two-thirds of patients of the current cohort reported to have taken DOAC longer than 48 h before hospital admission or the last DOAC intake was unknown. One-fifth of the patients were being treated with an inappropriate low DOAC dose. These two findings were more frequent in patients with low DOAC plasma levels and are probably the main reasons for low DOAC plasma levels in this population of patients. In comparison, previous studies have found lower proportions of patients with stroke despite DOAC who presented low DOAC plasma levels (24%–29%),7,8,14 which is probably explained by the specificities of each hospital and each cohort, local protocols for DOAC plasma level measurement, and selection criteria for study inclusion. DOAC adherence in real-world studies was found to be suboptimal in comparison to randomized controlled trials and may influence treatment efficacy. Good therapeutic adherence, defined by proportion of days covered or medication possession ratio ⩾80%, was found in only 66% of patients treated with DOACs in a meta-analysis of 21 studies. 15 Suboptimal adherence to oral anticoagulation with DOAC in patients with AF is known to be associated with increased risk of death and stroke. 16 On the other hand, approximately 10%–15% of patients per year require medically ordered temporary interruption of anticoagulation due to the need of invasive procedures with relevant risk of bleeding, 17 or because of relevant bleeding. Temporary interruption of anticoagulation carries a risk of arterial thromboembolism, which may reach 0.6% during the first 30 days after interruption.18,19 Although results of studies are inconsistent,20,21 inappropriate low DOAC dose may also be theoretically associated with an increased risk of stroke.
Low DOAC plasma levels are, as demonstrated by our study, insufficient to explain the occurrence of ischemic stroke or TIA in the majority of the patients. One-third of patients had multiple concomitant stroke causes, specific non-cardiac stroke causes, or unknown stroke etiology after complete etiological investigation. This finding strengthens the need to carry out a complete etiological investigation in this group of patients because anticoagulation is not sufficient or effective in specific non-cardiac stroke etiologies such as small vessel disease or large vessel disease. This is supported by the finding that the group of patients with multiple simultaneous potentially causal mechanisms, specific non-cardiac potentially causal mechanisms, or with no identified potentially causal mechanisms had significantly higher DOAC plasma levels in comparison to patients with cardiac pathology as the only potentially causal mechanism for the ischemic event. This may, in part, explain why Romoli et al. 22 recently found relevant clinical heterogeneity in a meta-analysis of studies analyzing secondary prevention strategies after breakthrough ischemic stroke in patients treated with DOACs. We think that clinicians should tailor individualized secondary prevention strategies taking into consideration not only concurrent stroke causes, but also objective assessment of therapeutic compliance at hospital admission and hemostaseologic confirmation of effective anticoagulation while on correct DOAC dosing. True breakthrough ischemic stroke or TIA in patients treated with DOAC occurred in 15.3% of our study population. There is no consensus in the literature of how breakthrough ischemic stroke should be defined. 22 A rigorous definition should consider not only if patients or care persons report regular DOAC intake up to the ischemic event, but should also consider the presence of effective anticoagulation in blood tests and the absence of other concomitant major causes for stroke. As an example, the case of a DOAC-treated patient with AF, with high DOAC plasma levels at admission, who has a severe internal carotid artery stenosis and suffers an ipsilateral ischemic stroke should not be classified as a true breakthrough stroke. The definition of breakthrough ischemic stroke or TIA we used in our study takes into account these aspects and reflects true failure of optimal prevention strategies. Future research should focus on this group of patients, which are the ones who could benefit the most from new, more intensive, and more effective anticoagulation treatments and strategies and from interventions such as occlusion of the left atrial appendage.5,23
Our study confirms previous reports showing that DOAC plasma levels are associated with clinical stroke severity and are independent predictors of the presence of LVO or MeVO,7,8,24 which is plausible from the point of view of the pathophysiology of cardioembolic stroke. Accordingly, among our study population, TIA patients had the highest median DOAC plasma levels, followed by patients with ischemic stroke without LVO or MeVO, patients with MeVO, and patients with LVO, who presented the lowest median DOAC plasma levels.
Although there is no recommendation to routinely measure DOAC plasma levels, assessment of DOAC plasma levels in the hyperacute phase of ischemic stroke and TIA at hospital admission may provide valuable information. It may inform decisions concerning intravenous thrombolysis,25,26 it may uncover medication incompliance, it may contribute to understand stroke pathophysiology in individual patients, and it may help to identify true breakthrough stroke, thereby supporting decisions regarding prevention strategies. The finding of high admission DOAC plasma levels in acute ischemic stroke patients with non-valvular AF should trigger a high index of suspicion for concurrent potentially causal pathologies, such as atherothrombosis, small vessel disease, or disorders associated with hypercoagulability. If a careful investigation excludes other concurrent causes, clinicians face the difficult decision of optimizing the antithrombotic strategy. Recent evidence suggests that DOAC to DOAC switch may have benefits in comparison to DOAC to vitamin K antagonist switch or in comparison to the addition of antiplatelets. 22 The benefit of performing left atrial appendage occlusion and continuing DOAC remains uncertain. On the other hand, the finding of low admission DOAC plasma levels should be used as evidence of irregular DOAC intake or interruption of DOAC treatment. Although exclusion of concurrent stroke causes in this group of patients is also important, strategies to improve medication compliance, to minimize drug interactions, and to avoid treatment interruptions should be the focus of the treating physicians.
Relevant limitations of our study include the fact that it is a retrospective monocentric study with a small population size. The small sample size limits the validity of the statistical associations we found and the external validity of our logistic regression models. Logistic regressions and non-parametric correlations may not be the optimal statistical method to analyze DOAC plasma levels, because DOAC plasma levels do not appear to have a linear relationship with thrombin generation inhibition.27,28 The fact that 23% of patients with ischemic stroke or TIA despite anticoagulation with DOAC in our local registry did not receive DOAC plasma levels measurement suggests that there may be a selection bias in the current cohort. The timing of blood sampling related to the last DOAC intake reflects the real-world setting in which this study was performed, and may induce heterogeneity in the results and limit the validity of the conclusions. Likewise, heterogeneity of DOAC assays (namely dabigatran in comparison to apixaban, rivaroxaban, and edoxaban) may also induce relevant bias. The results may not be generalizable to all DOACs, because patients receiving dabigatran and rivaroxaban were underrepresented. Detailed possible reasons for low DOAC plasma levels were not routinely documented in individual records of patients, and the last DOAC intake was unknown for a high proportion of patients.
Conclusion
In conclusion, low DOAC plasma levels were found in two in every five DOAC-treated patients with ischemic stroke or TIA. Stroke etiologies other than isolated cardioembolism were found in one-third of patients. Lower DOAC plasma levels in patients treated with DOAC who experienced ischemic stroke or TIA were associated with more severe neurological deficits and with the presence of medium or LVO. Emergent DOAC plasma level measurement is helpful to support treatment decisions in DOAC-treated patients with ischemic stroke and TIA and should be included in the routine management of these patients.
Supplemental Material
sj-doc-1-tan-10.1177_17562864261415719 – Supplemental material for Acute ischemic stroke or TIA despite DOAC treatment: the contribution of DOAC plasma levels to understand pathophysiological mechanisms
Supplemental material, sj-doc-1-tan-10.1177_17562864261415719 for Acute ischemic stroke or TIA despite DOAC treatment: the contribution of DOAC plasma levels to understand pathophysiological mechanisms by Marcus Rust, Merve Gözalan, Omid Nikoubashman, Martin Wiesmann, Matthias Imöhl, Jörg B. Schulz, Arno Reich and João Pinho in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
