Abstract
Background:
To compare the efficacy and safety of transarterial chemoembolization (TACE) combined with lenvatinib vs TACE alone in intermediate-stage hepatocellular carcinoma (HCC) patients with hypovascular nodules.
Methods:
This retrospective study analyzed the clinical data of intermediate-stage HCC patients with hypovascular nodules who underwent TACE. Patients were categorized into the TACE-Lenv combination group and the TACE monotherapy group according to their receipt of lenvatinib therapy. Overall survival (OS), progression-free survival (PFS), objective response rate (ORR), disease control rate (DCR), progression of hypovascular nodules, and treatment-related adverse events were recorded and analyzed.
Results:
The study enrolled 75 patients, with 40 allocated to the TACE-Lenv group and 35 to the TACE group. The combination therapy group demonstrated significantly higher ORR and DCR (92.5% vs 74.3%, P = .032; 97.5% vs 82.9%, P = .030) compared with TACE monotherapy. The TACE-Lenv group exhibited significantly prolonged median OS and PFS (41.1 vs 19.7 months, P < .001; 20.2 vs 9.9 months, P < .001). In addition, compared with the TACE group, the TACE-Lenv group extended the median time to nodule progression (37.0 vs 16.5 months, P < .001). After propensity score matching, significant differences remained in the aforementioned outcomes between the 2 groups. No significant differences were observed in liver function parameters or the incidence of grade 3 to 4 AEs between the 2 groups after treatment.
Conclusions:
The combination therapy of TACE and lenvatinib demonstrated excellent clinical efficacy in intermediate-stage HCC with hypovascular nodules and may therefore emerge as a preferred treatment option for this specific patient population.
Keywords
Introduction
Transarterial chemoembolization (TACE) is recommended by multiple guidelines as the standard treatment for intermediate-stage hepatocellular carcinoma (HCC).1,2 However, due to the high intratumoral heterogeneity, the efficacy of TACE in some patients with intermediate-stage HCC is not satisfactory, especially for those patients with larger tumors, multiple lesions, or functional liver impairment. 3
Hypovascular nodules are defined as nodules showing no arterial-phase enhancement relative to the surrounding liver on enhanced computed tomography (CT) or magnetic resonance (MR) images and having equivalent or reduced portal perfusion compared with the surrounding liver in the portal venous or equilibrium phase. These nodules are usually over 5 mm and confirmed by at least 2 consecutive image slices. 4 Several studies5,6 have discovered that the presence of hypovascular nodules shown on MR images represents a significant predictive factor for recurrence following radical treatment for early-stage HCC, which includes radiofrequency ablation or hepatic resection. Furthermore, a recent study has demonstrated that intermediate-stage HCC with hypovascular nodules serves as a significant predictive risk factor for TACE refractoriness. 4 This subset of patients may not be appropriate candidates for single TACE therapy. The TACE Plus Sorafenib for Intermediate Stage HCC Trial (TACTICS) indicated that in patients with unresectable HCC, the combination of TACE and sorafenib significantly improved the progression-free survival (PFS) compared with TACE alone. 7 Similarly, the combination of TACE and lenvatinib has also demonstrated superiority in the treatment of unresectable HCC. A retrospective study showed that, compared with TACE alone, TACE combined with lenvatinib prolonged survival in patients with unresectable HCC. 8 In addition, a recent study involving intermediate-stage HCC patients exceeding the up-to-seven criteria reported that combination therapy with TACE and lenvatinib achieved better clinical outcomes. 9 Thus, it is speculated that the combination of TACE and lenvatinib might be more suitable for treating intermediate-stage HCC accompanied by hypovascular nodules. This study aimed to compare the safety and efficacy of TACE in combination with lenvatinib against TACE alone for managing intermediate-stage HCC with hypovascular nodules.
Methods
All procedures performed in the study were in accordance with the Helsinki Declaration of 1975, as revised in 2024. This retrospective study received approval from the Institutional Review Board of the First Affiliated Hospital of Soochow University (Ethics Approval No. 251, 2025), and given its retrospective nature, the requirement for obtaining informed consent was waived. In addition, the reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 10
Patients
Patients with intermediate-stage HCC who received TACE treatment between January 2018 and December 2023 were investigated and screened. All patient data were de-identified. The patients were divided into the TACE alone or TACE-Lenv group according to whether they received lenvatinib orally. The criteria for the final inclusion of patients in the study were as follows: (1) HCC was diagnosed in accordance with the American Association for the Study of Liver Diseases (AASLD) practice guidelines; 11 (2) each patient was accompanied by at least 1 hypovascular nodule; (3) the age of the patients exceeded 18 years; and (4) the Child-Pugh liver function classification 12 was grade A or B and the performance status score was 0. The exclusion criteria were as follows: (1) prior exposure to any form of anti-tumor therapy; (2) co-morbidity with other malignant neoplasms; (3) presence of severe comorbid conditions, such as renal failure and heart failure; and (4) loss to follow-up during the study period.
Procedures
For every patient, the treatment strategy was meticulously determined following comprehensive multidisciplinary deliberations. The TACE procedure was carried out by interventional radiologists with at least 5 years of clinical experience, in accordance with the Chinese clinical practice guidelines for TACE. 13 Superselective catheterization of the tumor-feeding arteries was performed in all patients, and cone-beam computed tomography (CBCT) was used to assist tumor localization and ensure accurate embolization. The TACE treatment was routinely performed via the transfemoral artery approach. A 4F catheter was inserted into the celiac trunk and superior mesenteric artery for angiography to identify the tumor-feeding arteries. Subsequently, a 2.7F coaxial microcatheter was selected and inserted through the outer catheter into the proper hepatic artery, with the tip of the microcatheter surpassing the ostium of the gastroduodenal artery. Doxorubicin (30-50 mg) and oxaliplatin (100-200 mg) were slowly injected into the hepatic artery through the microcatheter (infusion time ⩾20 min). Then, the microcatheter was superselectively advanced into the tumor-supplying artery, and embolization was carried out using an emulsion mixture of doxorubicin (10 mg) and lipiodol (10-20 mL, Guerbet, Villepinte, France). After that, gelatin sponge particles (Alicon, Hangzhou, China) were injected to completely embolize the tumor-supplying artery.
Lenvatinib was orally administered to patients in the TACE-Lenv group within 1 week after the initial TACE procedure. The dosage of lenvatinib was adjusted according to the patient’s body weight (12 mg/day for patients weighing ⩾60 kg and 8 mg/day for those weighing <60 kg). It was permissible to temporarily suspend the drug administration in cases where transient liver function impairment occurred subsequent to the TACE procedure. In the presence of drug-related adverse events (AEs), appropriate actions, including dose modification, temporary suspension, or permanent discontinuation of the drug, were implemented in strict accordance with the established adverse reaction management protocols. 14
Follow-up and Assessment
The baseline laboratory data, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), serum bilirubin (BIL), albumin, blood cell count, and alpha-fetoprotein (AFP), were obtained within 1 week before treatment. Re-examination was carried out 1 week after treatment and then repeated every 3 months thereafter. The occurrence of fever, emesis, abdominal pain, diarrhea, hand-foot syndrome, proteinuria, and hypertension in patients after treatment was recorded. Safety and toxicity were evaluated by comparing changes in laboratory values and the incidence of the aforementioned AEs. The AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE, Version 5.0). 15
Baseline imaging examinations (contrast-enhanced CT or magnetic resonance imaging [MRI]) were completed within 1 month prior to treatment initiation. Follow-up examinations were conducted every 6 to 8 weeks thereafter, and tumor therapeutic response was evaluated according to the Modified Response Evaluation Criteria in Solid Tumors (mRECIST). The objective response rate (ORR) was defined as the percentage of patients achieving complete response (CR) or partial response (PR) sustained for at least 4 weeks. The disease control rate (DCR) was defined as the proportion of patients without disease progression. Overall survival (OS) was calculated from treatment initiation to death or the last follow-up, while PFS was measured from treatment start to tumor progression or death. In addition, progression of hypovascular nodules during follow-up was documented. A hypovascular nodule was considered progressive if its diameter increased by ⩾25% or if it developed enhanced vascularity with imaging features typical of HCC. Time to nodule progression (TTNP) was defined as the interval from treatment initiation to hypovascular nodule progression. Follow-up continued through December 31, 2024.
Statistical Analysis
Categorical variables were expressed as numbers and percentages, with between-group differences analyzed using the chi-square test or the Fisher exact test. Normally distributed continuous variables were presented as mean ± standard deviation, and between-group differences were compared using the independent samples t-test. Non-normally distributed continuous variables were reported as median (quartile), and between-group differences were assessed using the non-parametric Mann-Whitney U-test. The propensity score matching (PSM) method was employed to address potential selection bias. The variables used for matching included age, gender, hepatitis history, AFP, tumor number, Child-Pugh class, tumor diameter, and hypovascular nodule diameter. The 2 groups were matched 1:1 according to the treatment regimen, with a caliper width equal to 0.2. The Kaplan-Meier analysis was used to estimate TTNP, OS, and PFS, with between-group differences evaluated by the log-rank test. The Cox proportional hazards regression models were applied for univariate and multivariate analyses of OS to assess potential prognostic factors. A P-value < .05 was considered statistically significant. All statistical analyses were performed using SPSS version 26.0 and R version 4.1.2.
Results
Patient Characteristics
After strict screening of 672 intermediate-stage HCC patients who underwent TACE treatment, 75 patients with hypovascular nodules were ultimately included in the study cohort, comprising 40 cases treated with TACE combined with lenvatinib and 35 cases receiving TACE monotherapy. The complete screening flowchart is shown in Figure 1. Prior to PSM, the cohort comprised 62 (82.7%) male and 13 (17.3%) female patients, with a mean age of 63.40 ± 10.84 years. The median maximum intrahepatic tumor diameter was 54 mm (range = 16-140 mm), and the median diameter of hypovascular nodules was 12 mm (range = 6-33 mm). The baseline characteristics of the enrolled patients before and after PSM are presented in Table 1. After PSM, 25 patients were screened in both the 2 groups, with covariates showing improved balance, especially in gender.
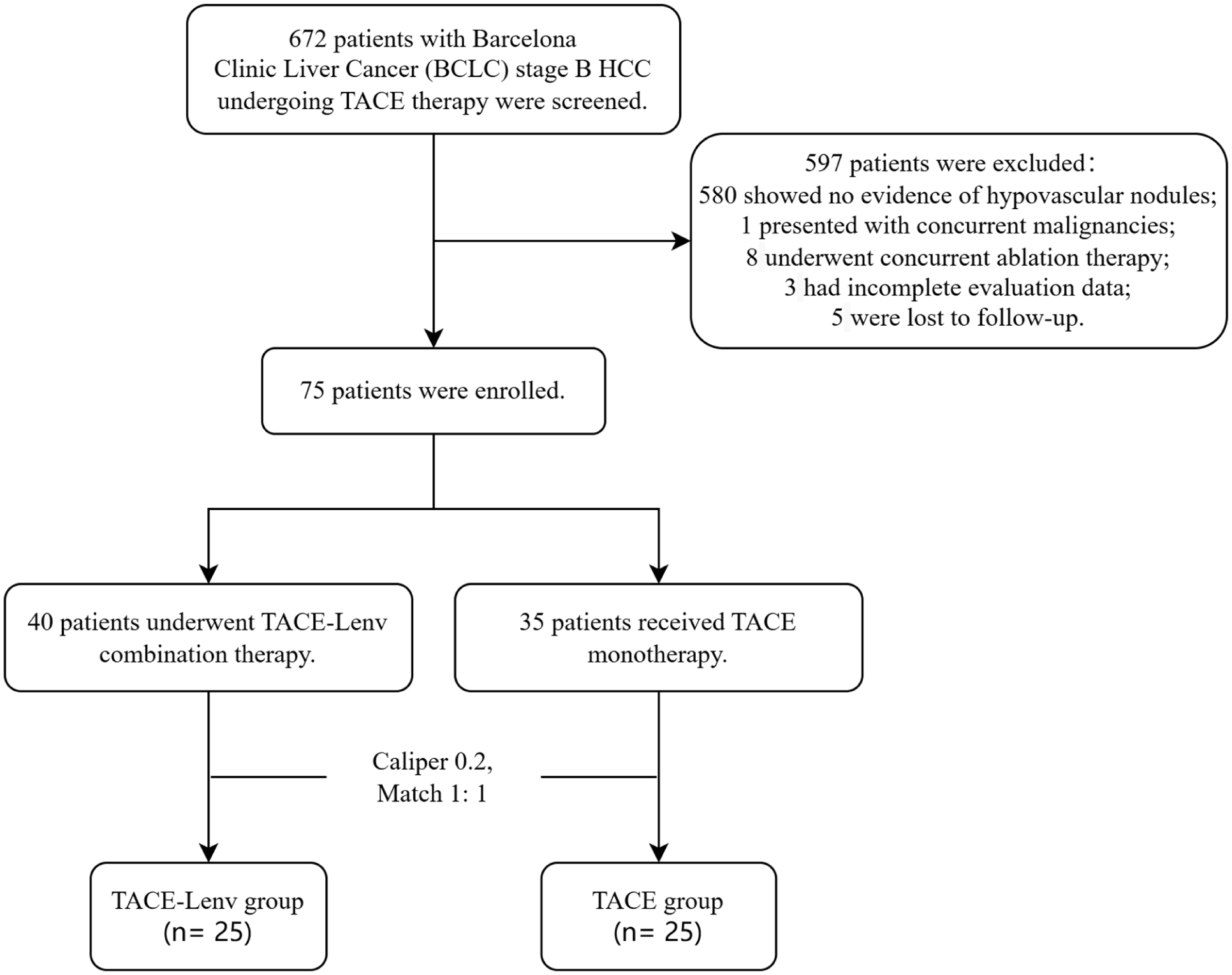
Flowchart of patients’ screening and inclusion in the study.
Clinical Baseline Characteristics of the 2 Groups.
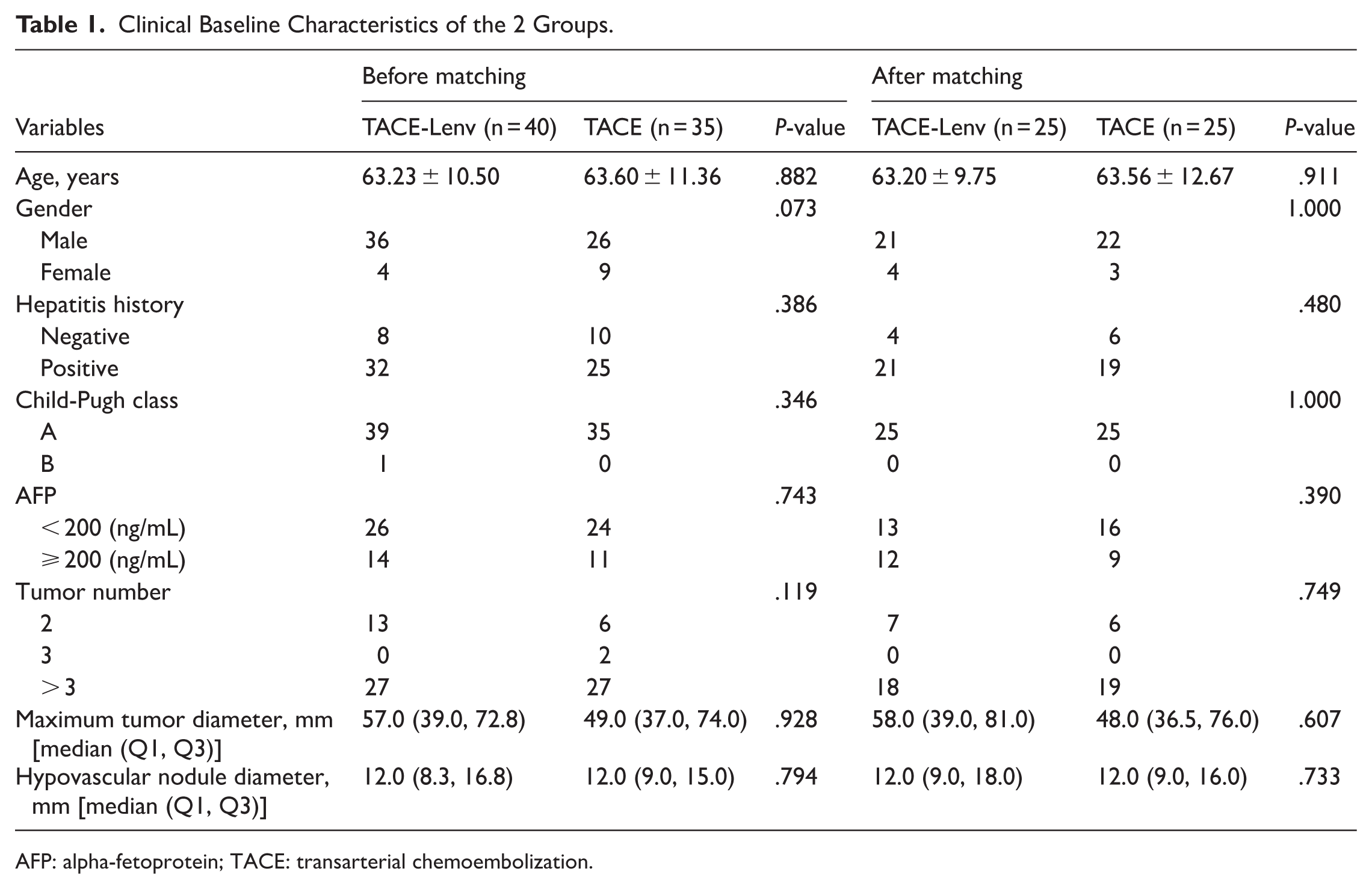
AFP: alpha-fetoprotein; TACE: transarterial chemoembolization.
Efficacy
The tumor response outcomes of the 2 groups are shown in Table 2. Prior to PSM, the TACE-Lenv group exhibited a significantly lower progressive disease (PD) rate compared with the TACE group (2.5% vs 17.1%, P = .030). In addition, the TACE-Lenv group demonstrated significantly higher ORR and DCR than the TACE group (ORR = 92.5% vs 74.3%, P = .032; DCR: 97.5% vs 82.9%, P = .030). After matching, the PR, ORR, and DCR rates in the TACE-Lenv group were significantly higher than those in the TACE group (P < .05).
Objective Response Rate of the 2 Groups, n (%).

CR: complete response; DCR: disease control rate; ORR: objective response rate; PD: progressive disease; PR: partial response; SD: stable disease; TACE: transarterial chemoembolization.
P < .05.
In the TACE-Lenv group, the median OS was 41.1 months (95% CI = 30.2-52.7 months), compared with 19.7 months (95% CI = 17.0-22.4 months) in the TACE group, with the combination group demonstrating a significantly longer median OS than the TACE group (P < .001, Figure 2A). The TACE-Lenv group also exhibited a prolonged median PFS of 20.2 months (95% CI = 15.0-25.4 months) vs 9.9 months (95% CI = 3.4-16.4 months) in the TACE group (P < .001, Figure 2B). After matching, the median OS and PFS in the TACE-Lenv group remained significantly longer than those in the TACE group (41.4 vs 19.7 months, P = .005, Figure 2C; 21.6 vs 9.9 months, P = .001, Figure 2D). In addition, multivariate analysis identified TACE combined with lenvatinib (hazard ratio [HR] = 0.300; 95% CI = 0.158-0.571; P < .001) and tumor diameter (HR = 1.014; 95% CI = 1.003-1.025; P = .013) as independent prognostic factors for OS (Table 3).

(A) Kaplan-Meier survival curves for overall survival in the 2 groups before PSM. (B) Kaplan-Meier survival curves for progression-free survival in the 2 groups before PSM. (C) Kaplan-Meier survival curves for overall survival in the 2 groups after PSM. (D) Kaplan-Meier survival curves for progression-free survival in the 2 groups after PSM.
Univariate and Multivariate Analyses of Prognostic Factors for Overall Survival.

AFP: alpha-fetoprotein; TACE: transarterial chemoembolization.
P < .05.
In the TACE-Lenv group, 18 patients (45.0%) exhibited progression of hypovascular nodules, which was significantly lower than the 24 patients (68.6%) observed in the TACE group (P = .040). Furthermore, the median TTNP in the TACE-Lenv group was 37.0 months (95% CI = 28.1-46.0 months), compared with 16.5 months (95% CI = 8.2-24.8 months) in the TACE group, demonstrating a significantly prolonged median TTNP in the combination group vs the monotherapy group (P < .001, Figure 3A). After matching, the median TTNP in the TACE-Lenv group remained significantly longer than that in the TACE group (33.2 vs 16.5 months, P = .005, Figure 3B).
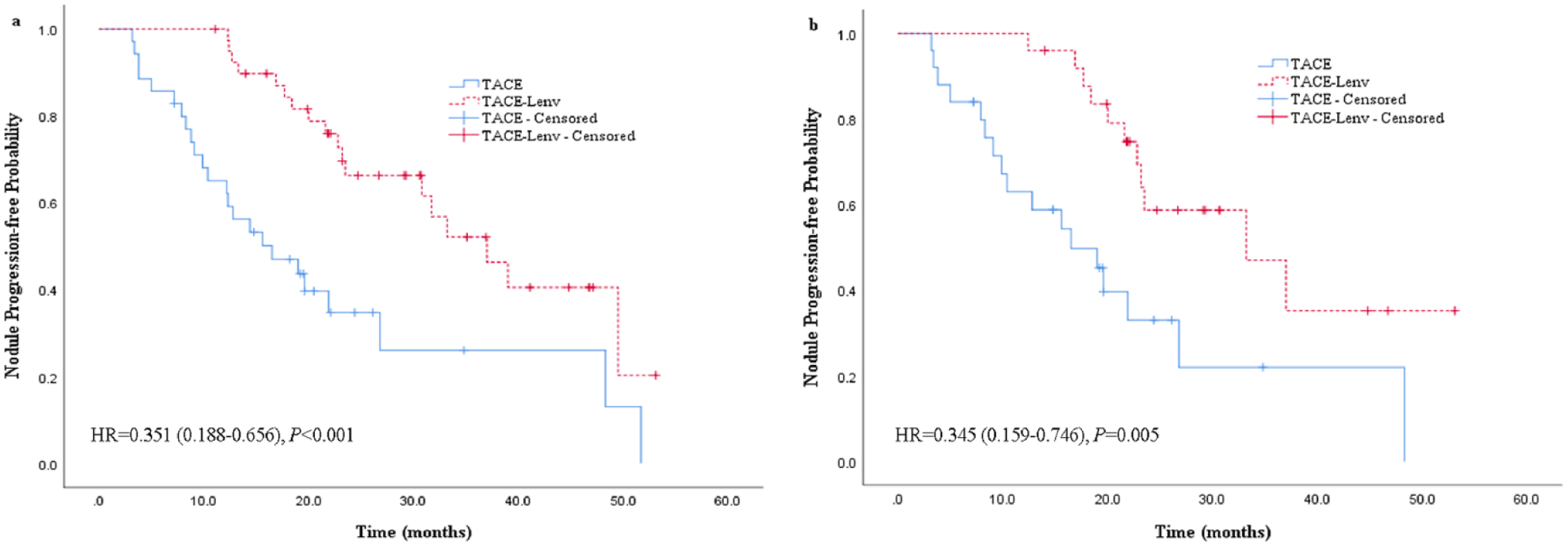
(A) Kaplan-Meier survival curves for time to progression of hypovascular nodules in the 2 groups before PSM. (B) Kaplan-Meier survival curves for time to progression of hypovascular nodules in the 2 groups after PSM.
Safety and Adverse Events
No treatment-related deaths occurred in either group. Table 4 summarizes the AEs observed in both groups. Thrombocytopenia was observed in 7 patients in the TACE-Lenv group and 3 patients in the TACE group (P = .472). No significant differences were found in the proportions of elevated liver function indicators between the 2 groups, including ALT, AST, and BIL. In addition, the incidence rates of fever, emesis, abdominal pain, and diarrhea were comparable between the groups. Notably, some patients in the TACE-Lenv group experienced AEs such as hypertension, proteinuria, and hand-foot syndrome, with the incidence of proteinuria being significantly higher than that in the TACE group (P = .028). Severe AEs (grade 3-4) primarily occurred in the TACE-Lenv group, including 1 case of myelosuppression, 4 cases of elevated ALT, 2 cases of proteinuria, 1 case of hand-foot syndrome, 1 case of emesis, and 1 case of diarrhea. In the TACE group, 2 patients developed severe ALT elevation. No significant difference was observed in the incidence of severe AEs between the 2 groups.
Adverse Events, n (%).

ALT: alanine aminotransferase; AST: aspartate aminotransferase; BIL: serum bilirubin; TACE: transarterial chemoembolization.
P < .05.
Discussion
This study demonstrated that in patients with intermediate-stage HCC accompanied by hypovascular nodules, the combination therapy of TACE and lenvatinib significantly improved OS and PFS compared with TACE monotherapy. Furthermore, the combined regimen reduced the progression rate of hypovascular nodules and prolonged the time to their progression. Given that intermediate-stage HCC is characterized by high heterogeneity, multiple prognostic factors including tumor size, tumor number, and liver function should be considered in survival analysis. 16 Although previous studies9,17 have explored combination therapy (TACE and tyrosine kinase inhibitors) for intermediate-stage HCC patients with various clinical features, none have specifically addressed its efficacy in patients with coexisting hypovascular nodules. This study provides novel insights into therapeutic strategy optimization for this distinct subgroup.
The TACE is recognized as the standard treatment for intermediate-stage HCC. 18 However, a subset of patients exhibits suboptimal or even no response to TACE therapy.19,20 Repeated TACE procedures in these patients may reduce treatment sensitivity, ultimately leading to TACE refractoriness. 21 A recent study has demonstrated that hypovascular hepatic nodules carry a high risk of progression to HCC, serving as a significant predictive risk factor for TACE refractoriness. 4 The mechanisms underlying TACE refractoriness may be associated with hypoxia-induced elevation of vascular endothelial growth factor (VEGF) levels and subsequent neovascularization following TACE treatment. 22 For such patients, combination therapy with lenvatinib might delay post-TACE tumor revascularization and recurrence by inhibiting angiogenesis and tumor proliferation. 23 These effects could potentially modify the immunosuppressive tumor microenvironment, enhance anti-tumor immune responses, improve synergistic therapeutic efficacy, reduce or even reverse TACE refractoriness, and ultimately prolong patient survival. 24
A recent review 25 indicated that the median OS of intermediate-stage HCC treated with conventional TACE ranged from 20.0 to 28.6 months, which is longer than the median OS of 19.7 months observed in this study for TACE monotherapy. The discrepancy may be related to the inclusion of patients with hypovascular nodules in this study, as hypovascular nodules have been reported by Toyoda et al 26 as an adverse prognostic factor in HCC patients. A previous study 9 investigating TACE combined with lenvatinib in intermediate-stage HCC patients exceeding the up-to-seven criteria reported median OS and PFS of 28 months and 8.2 months, respectively. In contrast, this study demonstrated superior outcomes with TACE combined with lenvatinib, showing median OS of 41.1 months and PFS of 20.2 months. This difference might be explained by the larger tumor size in the previous study, as tumor diameter is an independent prognostic factor for HCC survival. 27 This finding is further supported by the multivariate analysis, which identified tumor size as an independent predictor of OS.
Hypovascular nodules are considered borderline hepatocellular lesions pathologically categorized as high-grade dysplastic nodules or early HCCs, with potential progression to typical hypervascular HCC during follow-up. 28 The previous study 6 demonstrated that 27.5% of non-hypervascular nodules progressed to typical hypervascular HCC during follow-up, which was lower than the progression rate of hypovascular nodules observed in this study. The potential reasons may include the following 2 aspects: First, the former study exclusively enrolled patients with early-stage HCC, where nodules were detected at an earlier phase, requiring an extended duration to progress to typical HCC. In contrast, this study focused on intermediate-stage HCC cases, predominantly characterized by multicentric lesions, suggesting that hypovascular nodules might have already been in progression at the time of detection. Second, the former investigation only documented patients exhibiting enhanced arterial blood supply in nodules, whereas in this study, disease progression was defined as either a ⩾25% increase in nodule diameter or augmented arterial blood supply. It is noteworthy that combination therapy significantly extended the progression-free interval of hypovascular nodules in this study. We speculate that the underlying mechanism may be as follows: Lenvatinib, as a multi-target tyrosine kinase inhibitor that mainly targets VEGF receptors, 29 can inhibit the proliferation and migration of vascular endothelial cells, reduce the formation of collateral vessels, and regulate the tumor microenvironment of hypovascular nodules, thereby further inhibiting the progression of hypovascular nodules.
The incidence of AEs in this study was consistent with previous literature reports, 24 with the majority being grade 1 to 2 events. Intergroup differences were primarily observed in the occurrence rates of hypertension, proteinuria, and hand-foot syndrome, particularly the higher incidence of proteinuria, which is mechanistically associated with the pharmacological characteristics of lenvatinib. No significant difference was identified in the incidence of severe AEs between groups, and all complications were effectively managed through dosage adjustments or symptomatic treatment. These findings collectively demonstrated that the combination therapy of TACE and lenvatinib maintained an acceptable safety profile for intermediate-stage HCCs with hypovascular nodules.
Several limitations of this study should be acknowledged. First, the relatively small sample size in this study may limit the generalizability of the findings and reduce statistical power to detect subtle associations. Second, the retrospective study design inherently carries susceptibility to selection bias. In addition, the progression of hypovascular nodules was evaluated solely based on imaging characteristics without histopathological verification. Finally, post-progression therapies were not standardized in this study. The heterogeneous subsequent treatments administered to patients with progressive disease may have affected OS outcomes, which should be taken into consideration when interpreting the survival data.
Conclusions
In conclusion, compared with TACE monotherapy, the combination of TACE with lenvatinib demonstrates superior clinical outcomes in intermediate-stage HCCs with hypovascular nodules. This combined approach yields significant improvements in ORR, DCR, OS, and PFS while effectively delaying the progression of hypovascular nodules. The therapeutic regimen maintains an acceptable safety profile, suggesting that combination therapy may represent a preferred treatment strategy for this specific patient subgroup characterized by coexisting hypovascular nodules.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study has obtained IRB approval from the First Affiliated Hospital of Soochow University (Ethics Approval No. 251, 2025) on March 24, 2025.
Consent to Participate
The requirement for informed consent was waived due to the retrospective design of this study.
Consent for Publication
Not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research: This work was supported by the National Natural Science Foundation of China Project (grant number 82272094).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
Data produced or analyzed for this research can be obtained from the lead author upon reasonable request.
