Abstract
Background:
Pancreatic cancers (PCs)—especially pancreatic ductal adenocarcinoma (PDAC)—are among the deadliest digestive system cancers, with a 5 year survival of approximately 13%. Beta blockers (BBs), which inhibit beta-adrenergic receptor-mediated angiogenesis and immunosuppression, are potential candidates for oncological drug repurposing. However, the clinical evidence is inconsistent, and robust subgroup analyses are lacking. This study systematically evaluated the association between BB use and survival in PC patients. Furthermore, subgroup analyses were conducted to clarify differential clinical effects.
Methods:
This study was conducted in accordance with PRISMA guidelines and registered with PROSPERO (CRD420251106076). The PubMed, Embase, Cochrane Library, and Web of Science databases were searched to identify observational studies on all-cause mortality (ACM) and cancer-specific mortality (CSM). Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using RevMan 5.3 and R, and the results were presented in forest plots.
Results:
This analysis included nine retrospective cohort studies involving over 30 000 patients. There were no significant associations between BB use and ACM (HR = 1.07; 95% CI [0.95-1.20]) or CSM (HR = 0.89; 95% CI [0.70-1.14]). However, subgroup analysis revealed that BB use was significantly associated with increased ACM risk in surgical patients (HR = 1.18; 95% CI [1.05-1.31]). Moreover, non-selective BB (NSBB) use significantly reduced CSM risk (HR = 0.81; 95% CI [0.68-0.97]). Both sensitivity and trim-and-fill analyses confirmed the robustness and consistency of these results.
Conclusions:
This meta-analysis presents the first systematic evidence regarding the potential role of NSBBs in mitigating CSM, thus providing support for their potential repurposing. In addition, these findings indicate that perioperative BB use may be associated with increased ACM risk, highlighting the need for careful perioperative risk assessment. To further substantiate these findings, future prospective studies should explore combined approaches, particularly those integrating immune or anti-angiogenic therapies.
Keywords
Introduction
Pancreatic cancers (PCs)—especially pancreatic ductal adenocarcinoma (PDAC)—are among the most lethal malignancies of the digestive system. GLOBOCAN 2022 estimates that there will be 511 000 new cases of PC and 467 000 related deaths worldwide in 2022, making it the sixth-leading cause of cancer-related death and accounting for approximately 5% of all cancer-related fatalities. 1 In the United States, the latest data from the SEER database show that the overall 5 year relative survival rate for patients with PC is just 13%, which is considerably lower than that for patients with other cancers of the digestive system. 2 Owing to its subtle early symptoms and rapid disease progression, more than 50% of patients are diagnosed at advanced stages, either with local spread or distant metastasis. Even with aggressive surgery and standard adjuvant treatments, the 5 year survival rate remains below 20%. 3
The aggressive nature of PCs—particularly PDAC—is strongly influenced by their unique tumor microenvironment. PDAC is frequently characterized by extensive desmoplasia, the activation of fibroblasts associated with the tumor, immune cell infiltration that suppresses the immune response, and abnormal angiogenesis.4,5 Previous studies have demonstrated that the sympathetic nervous system plays a pivotal role in PDAC progression via beta-adrenergic receptors (β2-AR). Specifically, the activation of β2-AR enhances the expression of key factors such as interleukin-6 (IL-6), vascular endothelial growth factor (VEGF), and matrix metalloproteinases (MMPs) through the Src-dependent pathway, thereby facilitating angiogenesis and metastasis.6-8 Moreover, β2-AR signaling contributes to the recruitment of myeloid-derived suppressor cells (MDSCs), thus inhibiting the activation of CD8⁺ T cells and impairing the antitumor immune response.9,10 In addition, both chronic psychological stress and perioperative sympathetic activation have been shown to accelerate the metastasis and recurrence of tumors.11,12
Given the potential to modulate the sympathetic nervous system, beta blockers (BBs) have emerged as promising candidates for repurposing in oncology. Experimental evidence has suggested that BB effectively suppresses stress-induced angiogenesis, inhibits tumor cell proliferation, and enhances the tumor immune microenvironment, thus potentially improving therapeutic outcomes.13,14 Previous epidemiological studies have consistently shown that long-term BB use is associated with improved survival rates across multiple solid tumors, including breast cancer, melanoma, prostate cancer, and ovarian cancer, thus supporting the potential role of these agents in cancer treatment.15-18
The clinical use of BBs to treat PC remains a topic of ongoing debate. While some studies have suggested that BBs, particularly non-selective BBs (NSBBs), may improve survival outcomes by inhibiting sympathetic nervous system activation, reducing angiogenesis, and enhancing the immune microenvironment,19-21 other studies do not support these findings. In fact, some research has even suggested a trend toward worse survival outcomes in patients undergoing radical surgery.22,23 These inconsistencies may be attributed to various factors, such as the specific type of BB used, the timing of administration, the disease stage, and whether BBs are combined with other antitumor therapies. Preclinical studies have underscored the pivotal role of beta-adrenergic signaling in the initiation and progression of PDAC,24-26 although clinical evidence on this matter remains inconsistent.
At present, there is a considerable lack of high-quality systematic reviews and meta-analyses that explore the relationship between BB use and survival outcomes in PC. This gap is particularly noticeable in the absence of subgroup analyses that focus on different BB types, their timing of administration, and associated treatment strategies. To address this gap, the current study synthesizes available clinical data to comprehensively evaluate the associations between BB use and both all-cause mortality (ACM) and cancer-specific mortality (CSM) in PC patients. Furthermore, the current study examines how these associations may vary across different clinical subgroups. Ultimately, the aim of this study was to provide a solid evidence base for the rational use of BBs in PC treatment and to explore their potential to be repurposed as a therapeutic option.
Materials and Methods
This systematic review and meta-analysis was conducted in full accordance with the PRISMA 2020 guidelines 27 and was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO; CRD420251106076). The completed PRISMA 2020 checklist is available in Supplementary File 1. The PubMed, Embase, Cochrane Library, and Web of Science databases were comprehensively searched from inception to July 2, 2025, to identify observational studies published in English. In addition, references from the identified articles were manually reviewed to find any potentially overlooked studies. The search strategy utilized both free-text keywords and MeSH terms, with Boolean operators (AND/OR) to combine terms. The core search terms included “pancreatic cancer,” “pancreatic ductal adenocarcinoma,” “beta-blocker,” and “beta-adrenergic antagonists.” Two independent researchers performed the literature screening process, and any disagreements were resolved by consulting a third researcher.
Inclusion and Exclusion Criteria
The inclusion criteria for this study were as follows: (1) adult patients diagnosed with PC or PDAC; (2) studies comparing BB users and nonusers, including selective BBs (SBBs), NSBBs, and mixed or unspecified types; (3) studies reporting ACM or CSM, with hazard ratios (HRs) and 95% confidence intervals (CIs), or studies where these data can be derived; and (4) studies based on retrospective or prospective cohort designs.
The exclusion criteria were as follows: reviews, meta-analyses, case reports, animal or in vitro studies, or studies in which HRs and 95% CIs could not be extracted or calculated. In the case of duplicate studies, the one with the larger sample size or the most recent publication was included.
Data Extraction
Two researchers independently extracted essential data from the studies, including details such as the first author, publication year, country, and study design. Patient characteristics, including sample size, average age, tumor stage, surgical status, BB definition, follow-up duration, and outcome measures ACM and CSM, were also recorded. In addition, key covariates adjusted for in the multivariate analysis were documented. Any discrepancies between the two researchers were resolved by consulting a third researcher, who reviewed the data and made the final decision.
Quality and Risk of Bias Assessments
The quality of the studies included in this analysis was independently evaluated by two researchers via the Newcastle‒Ottawa Scale (NOS), which has a maximum score of 9 points. Studies scoring ⩾ 6 were categorized as medium to high quality. Any discrepancies in the ratings were reviewed and resolved by consulting a third researcher.
Statistical Analysis
Effect sizes are expressed as HRs with 95% confidence intervals (CIs). When the HR and standard error were unavailable, conversions were made using established methods. Heterogeneity was assessed via the Cochran Q test and I2 statistic. When I2 ⩽ 50%, a fixed effects model was used; when I2 > 50%, a random-effects model was used (DerSimonian and Laird). Subgroup analyses were based on the timing of BB administration, surgical status, and BB type. Sensitivity analysis was performed via the leave-one-out method to assess the impact of each study on the overall effect. Publication bias was evaluated with Begg’s rank correlation and Egger’s regression tests, with correction using the trim-and-fill method when necessary. All analyses were conducted using RevMan 5.3 and R, with statistical significance set at a two-sided P value < .05.
Results
Literature search
A total of 605 articles were retrieved from the following databases: PubMed (n = 118), Embase (n = 379), Cochrane Library (n = 10), and Web of Science (n = 98). After removing duplicates, the titles and abstracts of 535 studies were screened, resulting in the exclusion of 520 irrelevant articles. The full texts of the remaining 15 studies were screened. Ultimately, 9 studies were included in the quantitative meta-analysis. The literature screening process is summarized in Figure 1.

PRISMA flowchart of study selection.
Study characteristics
This study included 9 retrospective cohort studies22,23,28-34 involving a total of 30 044 patients diagnosed with PC or PDAC. The cohort included patients at various disease stages, ranging from early-stage patients undergoing curative surgery (eg, Kirkegård et al 22 and Hüttner et al 28 ) to those with advanced or metastatic disease (eg, Le Bozec et al 23 and Beg et al 31 ). The definitions of BB exposure varied, with some studies focusing on prediagnosis use during specific periods (eg, Støer et al, 29 Yang et al 30 and Udumyan et al 32 ) and others examining use during chemotherapy after diagnosis (eg, Le Bozec et al 23 and Beg et al 31 ). The primary outcomes measured were ACM (7 studies) and CSM (2 studies). Most studies adjusted for potential confounders such as age, sex, tumor stage, comorbidities, and concurrent medication use. The NOS scores ranged from 6 to 9 points, indicating medium to high methodological quality. The study characteristics are summarized in Table 1.
Baseline characteristics of included studies.

ACM, all-cause mortality; BB, beta-blocker; CSM, cancer-specific mortality; NOS, Newcastle–Ottawa Scale; NR, not reported; OS, overall survival; PC, pancreatic cancer; PDAC, pancreatic ductal adenocarcinoma; PFS, progression-free survival.
Quality assessment of the included studies
The quality of the 9 included studies was assessed using the NOS, which provides a score ranging from 6 to 9 points, indicating medium to high overall quality. Most studies achieved full marks in the “selection of study participants” and “outcome assessment” categories. However, several studies received lower scores in the “comparability” category because of insufficient adjustment for potential confounders. The detailed results of the quality assessment are summarized in Table 2.
NOS quality assessment of the included studies.

NOS, Newcastle–Ottawa Scale; ★, score for each domain (maximum: Selection = 4 stars, Comparability = 2 stars, Outcome = 3 stars).
Meta-Analysis Results
ACM (overall analysis)
Seven retrospective cohort studies involving a total of 25 036 patients with PC or PDAC were included to systematically assess the link between BB use and ACM.22,23,28,30,31,33,34 The pooled results revealed no significant association between the use of beta-blockers and ACM risk (HR = 1.07, 95% CI [0.95-1.20], P = .26; I2 = 80%). High heterogeneity and broad confidence intervals suggest substantial differences in population characteristics and definitions of BB exposure across studies (Figure 2).

Forest plot of the overall analysis for ACM.
ACM stratified by the timing of BB use
A total of seven studies performed subgroup analyses on the basis of the timing of BB use.22,23,28,30,31,33,34 The results revealed no statistically significant difference between prediagnostic BB use (HR = 1.10, 95% CI [0.96-1.26], P = .17; I2 = 64%) and postdiagnostic BB use (HR = 1.05, 95% CI [0.82-1.34], P = .69; I2 = 72%). Although not statistically significant, the postdiagnosis group presented slightly lower point estimates, which may suggest that continued BB use could lead to better survival outcomes. However, the difference between the subgroups was nonsignificant (P for subgroup differences = .75; Figure 3).

Forest plot of ACM stratified by the timing of beta-blocker use.
ACM stratified by surgical status
Seven studies performed stratified analyses on the basis of the surgical status of patients.22,23,28,30,31,33,34 The results revealed a significant association between BB use and increased ACM in surgical patients (HR = 1.18, 95% CI [1.05-1.31], P = .004; I2 = 0%). However, no significant correlation was observed in the overall population (HR = 1.02, 95% CI [0.89-1.16], P = .82; I2 = 79%). The low heterogeneity in the surgical subgroup increases the reliability of these findings, suggesting that BBs may have adverse effects on patients undergoing radical surgery. Although the subgroup differences did not reach statistical significance (P for subgroup differences = .10), these results may reflect underlying differences in tumor biology and perioperative stress responses between surgical and nonsurgical patients (Figure 4).
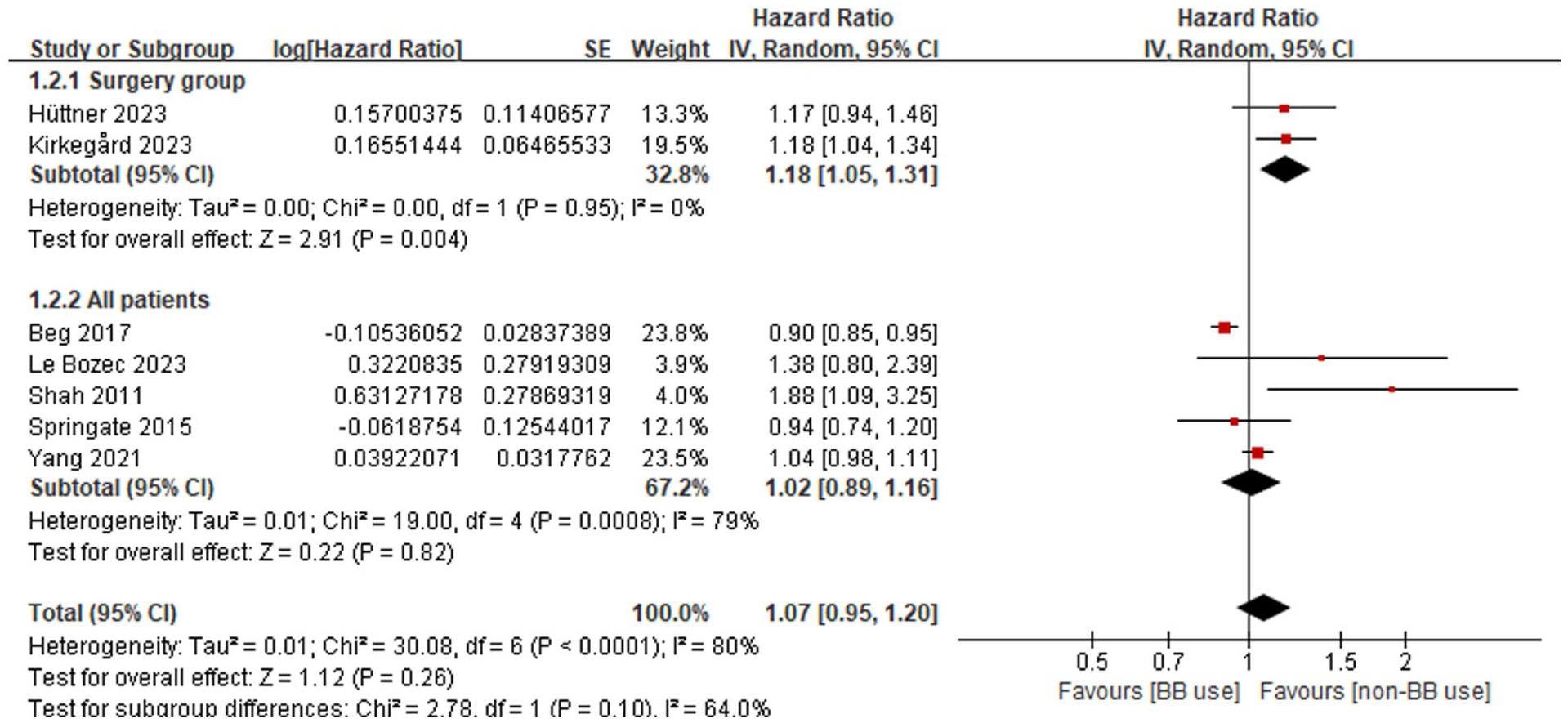
Forest plot of ACM stratified by surgical status.
ACM stratified by BB type
Seven studies conducted subgroup analyses on the basis of the type of BB used.22,23,28,30,31,33,34 The results revealed no significant correlation between NSBB use (HR = 1.07, 95% CI [0.96-1.18], P = .22; I2 = 0%) and mixed/unspecified BB types (HR = 1.09, 95% CI [0.90-1.31], P = .37; I2 = 83%). However, the use of selective β-blockers was not significantly associated with ACM, although a trend toward increased risk was observed (HR = 1.30, 95% CI [0.75-2.25], P = .34; I2 = 84%). Nevertheless, the wide confidence intervals and high heterogeneity suggest limited evidence. No statistically significant differences were observed between the subgroups based on BB type (P for subgroup differences = .78; Figure 5).

Forest plot of ACM stratified by beta-blocker type.
CSM (overall analysis)
Two studies involving 5008 patients assessed the relationship between BB use and CSM.29,32 The combined results revealed no statistically significant association between BB use and CSM (HR = 0.89, 95% CI [0.70-1.14], P = .36; I2 = 86%). The high heterogeneity observed indicates substantial variation in the characteristics and treatment regimens of the study populations (Figure 6).

Forest plot of the overall analysis for CSM.
CSM stratified by BB type
Two studies performed stratified analyses of CSM on the basis of the type of BB used.29,32 NSBB was significantly associated with a reduced risk of CSM (HR = 0.81, 95% CI [0.68-0.97], P = .02; I2 = 0%), suggesting potential survival benefits for patients with pancreatic cancer. However, no relevant data were available for SBBs or mixed/unspecified BB types (Figure 7).

Forest plot of CSM stratified by beta-blocker type.
Sensitivity analysis
To evaluate the robustness of the pooled results, a sensitivity analysis was conducted via the leave-one-out method. After excluding individual studies, the pooled logHR for ACM ranged from 0.04 to 0.11, with no change in the direction of the effect, indicating that the results were stable and reliable. Excluding the study with the largest sample size (Beg et al 31 ) caused the most significant fluctuation, but this did not notably alter the overall conclusion (Figure 8). Since CSM was only assessed in two studies and no sensitivity analysis was performed, the conclusions regarding CSM should be interpreted with caution.
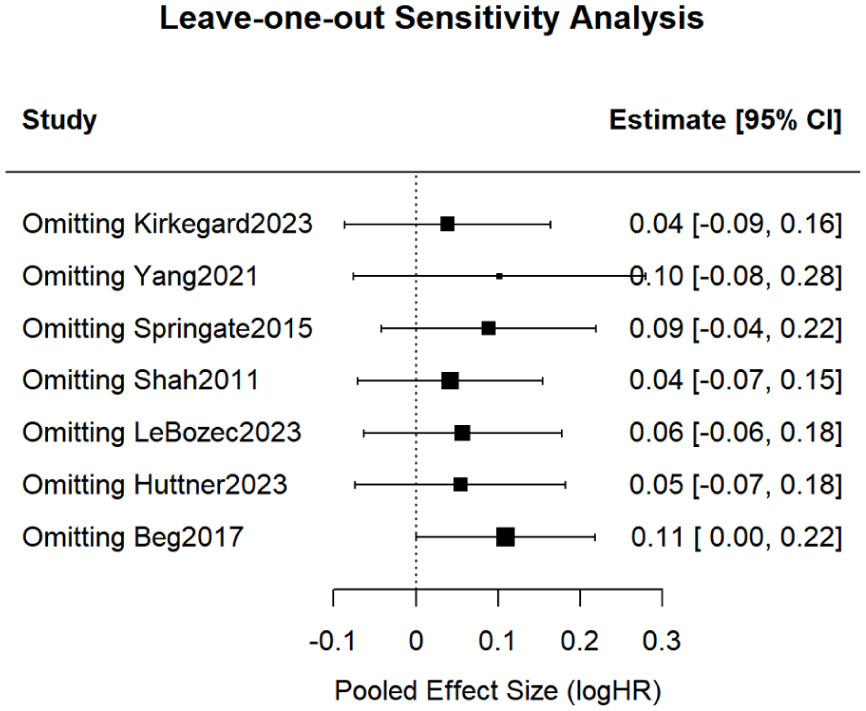
Leave-one-out sensitivity analysis.
Publication bias
Begg’s test indicated no significant publication bias (Kendall’s tau = 1.00, P = .88). Similarly, Egger’s regression test was not statistically significant (intercept = 2.07, P = .13). Trim-and-fill analysis identified two potentially unpublished studies, resulting in a corrected pooled effect size of HR = 1.03 (95% CI [0.92-1.16], P = .59), which was consistent with the precorrection result (HR = 1.07, 95% CI [0.95-1.20]). This finding supports the conclusion that publication bias is unlikely (see Figures 9 and 10). Since CSM was only assessed in two studies and the risk of publication bias was not assessed, the conclusions regarding CSM should be interpreted with caution.

Original funnel plot assessing publication bias.

Funnel plot after trim-and-fill analysis.
Discussion
This study, which represents one of the largest systematic evaluations and meta-analyses conducted to date, examined the association between BB use and survival outcomes in PC patients. The analysis included data from nine retrospective cohort studies involving over 30 000 patients diagnosed with PC or PDAC. The overall results indicated that BB use was not significantly associated with ACM or CSM. However, subgroup analysis revealed a significant association between BB use and increased ACM among patients who underwent radical surgery. In addition, NSBB use was associated with potential survival benefits in terms of a lower risk of CSM. These findings suggest that the effect of BBs on PC outcomes may be influenced by clinical factors such as drug type and treatment strategy.
These findings are consistent with those of large-scale studies by Yang et al 30 and Støer et al, 29 who reported no significant relationship between BB use and survival in PDAC patients. In both animal and clinical studies, Partecke et al 19 reported that propranolol effectively inhibits sympathetic activation and retards the progression of PDAC. Udumyan et al 32 reported that prediagnosis use of NSBBs was linked to a decrease in CSM. Kirkegård et al 22 suggested that perioperative BB use may be associated with poorer postoperative survival, which aligns with the conclusions drawn in the present study. The high heterogeneity in this study (ACM I2 = 80%, CSM I2 = 86%) may be due to differences in the study population (which included both early-stage surgical patients and those with late metastatic disease), varying definitions of BB exposure (long-term use before diagnosis versus short-term use after diagnosis), unequal proportions of drug types, and insufficient control for confounding factors, all of which may influence the stability of the pooled effect. Building upon these sources of statistical heterogeneity, additional variability in patient mix, treatment strategies, and exposure patterns may further influence the robustness of the pooled estimates. Although adjusted hazard ratios were used, residual confounding related to comorbidities, concomitant medications, and differences in treatment strategies cannot be fully excluded. Furthermore, as all included studies were retrospective observational analyses, the associations identified should be interpreted cautiously and cannot be considered causal.
This subgroup difference is likely due to the various biological effects of beta-adrenergic signaling in PDAC. Chronic sympathetic nervous system (SNS) activation has been demonstrated to drive angiogenesis, matrix remodeling, and distant metastasis in PDAC through β2-AR. This process specifically involves the upregulation of VEGF, IL-6, and MMP-9, which promote tumor angiogenesis and metastasis formation;35-37 moreover, SNS activation induces the expression of nerve growth factor (NGF) and its receptor TrkA, which accelerates perineural invasion and early-stage metastasis.38,39 Therefore, NSBBs, which can be used to block both β1- and β2-ARs, are more likely to effectively inhibit this protumorigenic process. In contrast, SBBs may be less effective in improving survival because of their inability to antagonize β2-AR.
In addition, beta-adrenergic signaling is pivotal in remodeling the tumor immune microenvironment. The activation of β2-AR can enhance the immune-suppressive characteristics of MDSCs and tumor-associated macrophages (TAMs) through cAMP/PKA-STAT3 and PI3 K/Akt-dependent fatty acid oxidation (FAO), increasing their secretion of IL-10 and TGF-Beta while suppressing CD8+ T-cell and natural killer cell activation.40-42 Mohammadpour et al 10 demonstrated that β2-AR signaling amplifies the suppressive effects of MDSCs on T cells, thereby facilitating tumor immune evasion. Daher et al 40 reported that beta-blockers notably enhance the initial activation of CD8+ T cells and work synergistically with cancer vaccines to strengthen antitumor immunity. These findings provide a biological rationale for the observed reduction in CSM with NSBBs in this meta-analysis.
This study revealed a notable increase in ACM associated with the use of BBs in patients undergoing radical surgery. This result may be attributed to the hemodynamic instability (eg, hypotension and bradycardia) caused by perioperative BB use, which excessively dampens the body’s stress response, thus impairing NK cell-mediated immune surveillance and increasing the risk of infection and non-tumor-related mortality. Furthermore, patients using perioperative BBs frequently have significant comorbidities, such as cardiovascular disease, thus suggesting that this outcome may partially reflect indication bias. 43 Kirkegård et al 22 further suggested that perioperative BB use could be influenced by selection bias, leading to increased complications in cardiovascular high-risk patients. Moreover, SBB may not adequately block β2-AR-mediated stress signals during the perioperative period, possibly exacerbating postoperative complications due to improper cardiovascular regulation. 44 Therefore, when BBs are prescribed during the perioperative period, it is essential to carefully evaluate the patient’s cardiovascular condition and the specific type of medication administered.
Notably, BB may exhibit synergistic effects when used in conjunction with antiangiogenic agents and immunotherapy. β2-AR inhibition has been demonstrated to reduce hypoxia-induced VEGF expression and enhance tumor vascularization and oxygenation, which in turn increases the effectiveness of chemotherapy and anti-VEGF therapies.13,45 In addition, a study by Partecke et al 19 confirmed that propranolol can work synergistically with bevacizumab to impede tumor angiogenesis and improve tumor perfusion. Moreover, BBs have the potential to enhance the response to immune checkpoint inhibitors and subsequent chemotherapy in patients with tumors by decreasing chronic inflammation and improving immune cell infiltration.46,47 Owing to their safety, affordability, and widespread availability, BBs are anticipated to become a key adjunct to current targeted and immunotherapy strategies.
This study has several limitations. First, the studies included in this analysis were observational, and residual confounding factors remain a potential concern. Second, considerable differences in the type, dosage, timing, and duration of BB use led to significant heterogeneity in the analysis, thus limiting the accurate estimation of drug effects. Third, some studies lack clear definitions for CSM, thus introducing bias in the classification of causes of death. Finally, the number of studies and sample sizes included in the subgroup analyses were limited, particularly for CSM, leading to insufficient statistical power in these results. Future research should focus on large, multicenter prospective randomized controlled trials to identify the optimal timing and target populations for various types of BB. Moreover, standardizing the definition and outcome evaluation of CSM is necessary to reduce heterogeneity. Furthermore, exploring the combined use of BBs with immune checkpoint inhibitors and anti-angiogenic therapy is warranted. In the future, prospective clinical trials and mechanistic studies should aim to clarify the synergistic effects of these agents.
In conclusion, the effects of BBs on survival in PC patients can vary significantly depending on the type of drug and the clinical context in which it is applied. NSBBs may offer long-term survival benefits by inhibiting sympathetic-driven protumor signaling and modifying the immunosuppressive tumor microenvironment, potentially delaying tumor progression. Subgroup analyses indicated a significant increase in ACM among patients receiving BBs during the perioperative period. A non-significant trend toward elevated ACM was also noted with the use of selective SBBs, though the current evidence remains limited and inconclusive. Future studies should focus on large, multicenter prospective randomized controlled trials to determine the optimal timing, target populations, and strategies for combining BBs with immunotherapy or targeted therapies.
Conclusion
This meta-analysis suggests that the general use of BBs is not significantly correlated with the risk of ACM or CSM in patients with PC. NSBBs may be associated with a reduced risk of CSM, but BB use has been linked to higher ACM in patients undergoing curative surgery. These results highlight substantial heterogeneity across subgroups regarding the role of BBs in PC and underscore the importance of considering both drug type and treatment strategy in clinical decision-making. To further clarify the potential therapeutic value of BBs in PC and refine individualized treatment strategies, future high-quality prospective studies and mechanistic investigations are essential.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251411762 – Supplemental material for Beta-Blocker Use and Survival Outcomes in Pancreatic Cancer Patients: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-onc-10.1177_11795549251411762 for Beta-Blocker Use and Survival Outcomes in Pancreatic Cancer Patients: A Systematic Review and Meta-Analysis by Rui Li, Jie Chen, Yingkai Chen, Kai Jin, Yong Chen, Changyu Deng, Xuefen Liu and Yue Li in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
This study did not require institutional review board approval because it is a systematic review and meta-analysis based solely on previously published, publicly available data. No human participants were directly involved, and no individual patient-level information was accessed or analyzed. Therefore, ethical approval and patient consent were not applicable.
Consent to participate
Not applicable. This study is a systematic review and meta-analysis that uses only previously published data and does not involve human participants.
Consent for publication
Not applicable. No individual patient data, images, or identifiable materials were included in this study.
Author Contributions
Rui Li and Jie Chen: Writing the original draft and conceptualization. Kai Jin and Yingkai Chen: Methodology. Yong Chen and Changyu Deng: Supervision and resources. Xuefen Liu and Yue Li: Writing-review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
