Abstract
Background:
The impact of KRAS mutation testing on pancreatic ductal adenocarcinoma (PDAC) samples by endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) for reducing the need to repeat EUS-FNA has been demonstrated. Such testing however is not part of standard practice for endoscopic ultrasound-guided fine-needle biopsy (EUS-FNB).
Objectives:
We aim to analyse the proportion of non-contributive samples by EUS-FNB and to evaluate the impact of KRAS mutation testing on the diagnosis, theranostics and survival.
Design:
In this retrospective study, the impact on diagnosis and survival of KRAS testing for contributive and non-contributive samples by EUS-FNB was analysed.
Methods:
The EUS-FNB samples, combined with KRAS testing using the Idylla® technique on liquid-based cytology from patients with PDAC between February 2019 and May 2023, were retrospectively reviewed. The cytology results were classified according to the guidelines of the World Health Organization System for Reporting Pancreaticobiliary Cytopathology (WHOSRPC).
Results:
A total of 85 EUS-FNB specimens were reviewed. In all, 25 EUS-FNB samples did not lead to a formal diagnosis of PDAC according to the WHOSRPC (30.2%). Out of these 25, 11 (44%) could have been considered positive for a PDAC diagnosis thanks to the KRAS mutation test without carrying out further diagnosis procedures. The sensitivity of KRAS mutation testing using the Idylla technique was 98.6%. According to the available data, survival rates were not statistically different depending on the type of mutation.
Conclusion:
KRAS mutation testing on liquid-based cytology using the Idylla or equivalent technique, combined with the PDAC EUS-FNB sample, should become a standard for diagnosis to avoid delaying treatment by doing another biopsy. Furthermore, knowledge of the KRAS status from treatment initiation could be used to isolate mutations requiring targeted treatments or inclusion in clinical research trials, especially for wild-type KRAS PDAC.
Plain language summary
The echoendoscopic ultrasound diagnostic of pancreatic adenocarcinomas sometimes remains difficult due to the nature of these tumors with a particular microenvironment. For more than 30 years, several authors have underlined the importance of searching for a KRAS mutation on samples taken by echoendoscopic ultrasound to improve diagnostic performance. However, this research is not common practice. Our retrospective study made it possible to review the files of 85 patients with pancreatic adenocarcinoma in whom an echoendoscopic ultrasound biopsy was performed with a search for the KRAS mutation (with second-generation fine needle biopsy). Forty-four percent could have been considered positive for the diagnosis of PDAC thanks to the search for the KRAS mutation without repeating new samples. Furthermore, knowledge of the KRAS mutation type from diagnosis would make it possible to isolate mutations justifying possible targeted treatments.
Keywords
Introduction
On a global scale, the number of new cases of pancreatic ductal adenocarcinomas (PDAC) diagnosed in 2020 and the number of deaths related to this cancer were 490,000 and 460,000, respectively.1,2 The growing incidence of this cancer means it may become the second cause of cancer-related death, with around 800,000 deaths per year in 2030. 3 The poor prognosis (5-year survival rate of 9%, all stages combined) is partly explained by the fact that around 85% of PDAC are diagnosed at locally advanced or metastatic stages due to the lack of specificity and/or late onset of symptoms.4,5 Diagnosis and treatment must also be more rapid, especially as the time between the date of the first consultation suggesting PDAC and that of the imaging to confirm it seems to significantly affect survival.6–9
Endoscopic ultrasound (EUS), with ultrasound-guided aspiration for the cytological examination (fine-needle aspiration) (EUS-FNA) was previously the only key procedure for confirming the diagnosis but collecting contributive samples remains difficult. This is due in particular to the tumour microenvironment comprising a dense desmoplastic stroma and to the often low cell count in the samples collected, especially with EUS-FNA.10,11 Thus, the two main meta-analyses have reported diagnostic values between 85% and 90.4% for sensitivity and 98% for specificity.12,13 Not all studies have good performances. In the work by Lundy et al., 14 reflecting ‘real-world’ results, even if the specificity and positive predictive value remain high (98.1% and 99.5% respectively), diagnostic performances were only 78.6% and 48.6% for sensitivity and negative predictive value (NPV), respectively. Also in this study, 14 the use of repeated procedures was necessary to establish the diagnosis in 18.2% of cases.
The paradigm shift was based on the principle of no longer aspirating but cutting fragments of tissue with a first-generation fine needle to obtain micro-biopsies (FNB reverse-bevel needles), then second generation (fine-needle biopsy) (EUS-FNB) (22-gauge Acquire; Boston Scientific, Burlington, MA, USA, 22-gauge SharkCore; Medtronic, Dublin, Ireland and 20-gauge ProCore; Cook Medical, Bloomington, Ind, USA).15–17 The performances of the Acquire needles and the 22-gauge SharkCore needles appear better in terms of tissue acquisition of all the solid masses of the pancreas. 15
For more than 30 years, several studies have shown the impact of testing the activating mutations of the Kirsten rat sarcoma viral oncogene homolog (KRAS gene) on EUS-FNA samples to differentiate PDAC from benign lesions, such as chronic pancreatitis (CP) or auto-immune pancreatitis (AIP).14,18–30 In 2009, Bournet et al. 20 compared the performances of EUS-FNA alone to those of EUS-FNA combined with KRAS mutation testing for the differential diagnosis between PDAC and pseudotumoral CP. The performances were significantly improved for sensitivity (83–88%), NPV (56–63%) and diagnostic accuracy (86–90%).
The KRAS gene is the locus for the c-k-ras proto-oncogene located on the short arm of chromosome 12 (12q). Its mutation is involved in many cancers, especially in PDAC with a proportion of up to 95%.31,32 Activation of mutation of the KRAS oncogene on codon 12 (exon 2) is the initial event occurring in most cases of PDAC (70–95%), by inducing immunosuppression. 33 Mutations activating the KRAS gene are detected from the intraepithelial neoplasia stage (PanIn)-1A. 34 Mutations can also occur on codons 11, 13, 61 or 146, although they are less common.34,35 When the KRAS oncogene is activated, tumour suppressor genes INK4a-ARF, TP53 and DPC4-SMAD4 are inactivated in most cases of PDAC. 35 The role of KRAS mutation in the initiation, progression and remodelling of the tumour microenvironment in PDAC is in the process of being understood.36–38
The primary objective of this single-centre, retrospective study, carried out in a French Comprehensive Cancer Center over 4 years in PDAC patients, aimed to analyse the proportion of non-contributive samples using the newer-generation FNB needles (second-generation cutting needles) and to evaluate the diagnostic impact of KRAS mutation testing and consequently an evaluation of the sensitivity of KRAS mutation testing on liquid-based cytology. The secondary objective was the theranostic analysis at the time of diagnosis by EUS-FNB and an evaluation of survival according to mutations.
Materials and methods
Study population
This study included retrospectively all consecutive patients in whom PDAC has been diagnosed using new-generation EUS-FNB between February 2019 and May 2023. All EUS-FNB were combined with the Idylla® test for KRAS mutation testing on residual liquid-based cytology. KRAS status was determined in these patients using the next-generation sequencing (NGS) technique, involving a number of methods for sampling by EUS-FNB, 39 by liquid biopsy, surgical biopsy or X-ray biopsy.
EUS-FNB technique
All procedures were performed by the same expert operator (DB) according to recommendations.4,40 The echoendoscopes used were two linear echoendoscopes (Olympus GF-UC140 and GF-UCT180; Olympus Optical, Hamburg, Germany). All the pancreatic lesions were punctured with second-generation cutting needles: 20-gauge side-fenestrated forward-facing bevel needle (ProCore; Cook Medical, Bloomington, IN, USA) or 22-gauge Franseen needle (Acquire; Boston Scientific, Burlington, MA, USA). Two sampling techniques were used: dry-suction technique (after withdrawal of the stylet and application of suction by an attached syringe of 20 or 60 mL of negative pressure, the aspiration needle was moved 20 times within the lesions while also using fanning technique) 17 and the slow-pull technique (sampling was performed by slow removal of the stylet simultaneously creating negative pressure with aspiration by capillarity). 41 In the two cases, the aspirated material was pulled from the echoendoscope and pushed out into a preservative liquid (Cytolyt) by reinsertion of the stylet. Both techniques, aspiration and slow-pull, were associated with improving the analysis of residual liquid-based cytology specimens. 41 All EUS-FNB procedures were performed with Macroscopic On-Site Evaluation. 42 An information leaflet on the nature and risks of EUS-FNB is given to each patient.
Pathological evaluation of cell block
The aspirated material was separated for cell block preparation, cytological evaluation and KRAS mutation analysis. The solid materials were fixed in 4% buffered neutral formalin for a paraffin-embedded cell block.
- Cell block preparation: Cell block sections were examined after haematoxylin and eosin staining for pathological evaluation by one pathologist (cell block diagnosis).
- Liquid-based cytology evaluation: The residual material was treated using the liquid-based cytology method and then immediately evaluated by Papanicolaou staining.
- KRAS mutation testing: The residual liquid-based cytology specimens were stored at 4°C until DNA extraction.
The cell block pathology reports of EUS-FNB were all grouped by our pathologist according to the new guidelines of the World Health Organization (WHO) System for Reporting Pancreaticobiliary Cytopathology (WHOSRPC; Table 1). 43 This new grouping is used to estimate the risk of malignancy and offers clinical treatment options for each category.
The new guidelines of the World Health Organization System for Reporting Pancreaticobiliary Cytopathology. 43
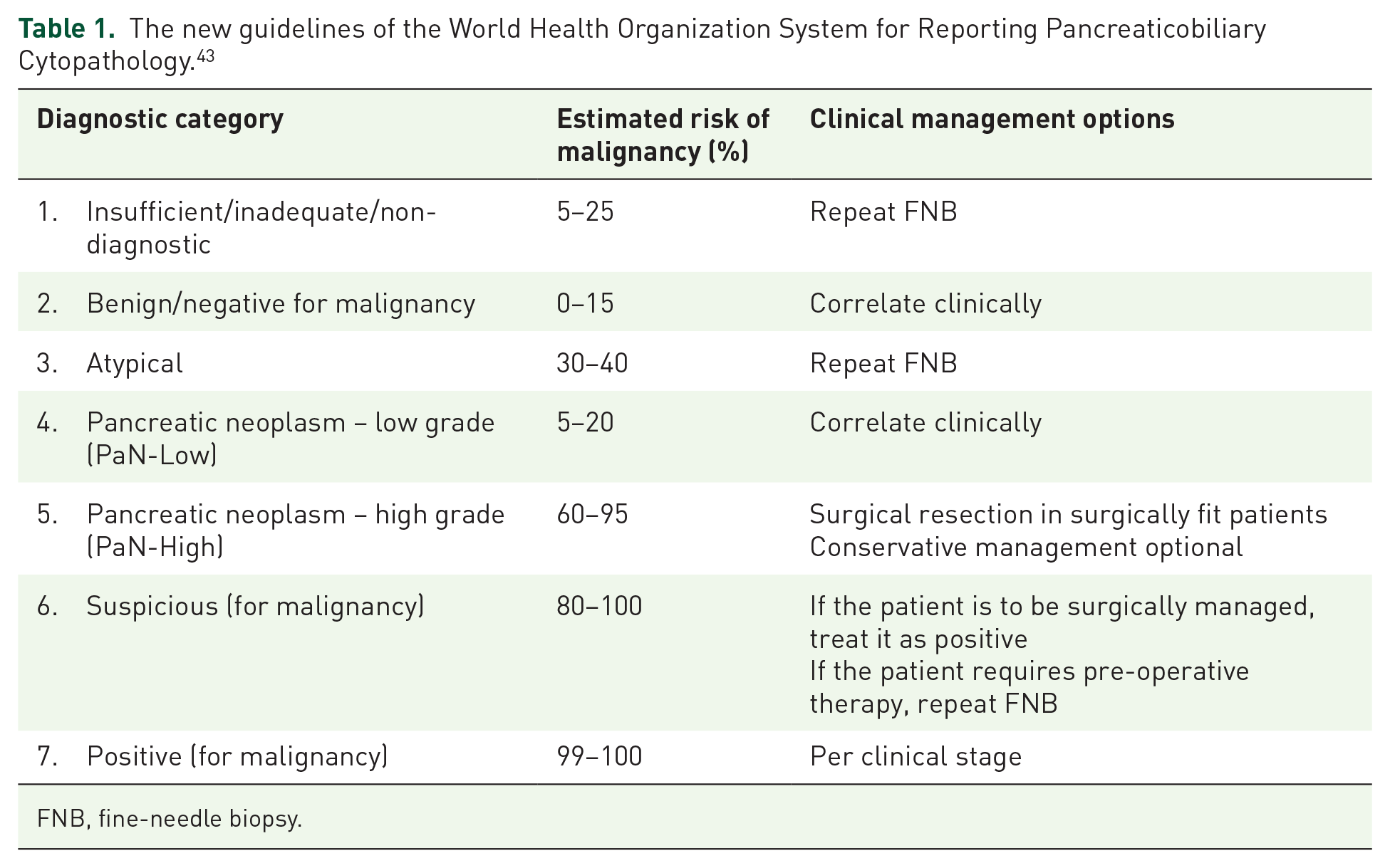
FNB, fine-needle biopsy.
PDAC diagnosis was confirmed if a biopsy review by EUS-FNB formally concluded on category 7. In the other cases, the diagnosis was obtained secondarily by pathological examination of a second EUS-FNB; X-ray or surgical biopsy of the primary tumour or metastasis; or analysis of the surgical specimen.
KRAS mutation extraction methods from EUS-FNB samples for the patients and also by NGS for the sub-group of patients included in the Bergonié Institute Profiling (BIP) protocol
- DNA extraction and quantification for pancreatic EUS-FNB
Samples for molecular biology were not qualified (unknown cellularity). DNA was extracted and eluted in a final volume of 50 µL with the QIAamp DNA Mini Kit according to the manufacturer’s instructions (Qiagen®, Venlo, Netherlands) and quantified with the Qubit kit (Qubit® dsDNA BR Assay Kit; Molecular Probe, Waltham, Massachusetts, USA).
- Idylla KRAS mutation test (Biocartis)
Suitable samples were analysed with Idylla KRAS cartridges loaded with pre-extracted DNA. DNA volume was adapted to concentration so that DNA input was never below 50 ng. The cartridge was then sealed, inserted into the instrument and the analysis started promptly. On completion, data were retrieved via two mechanisms: (1) the CE-IVD (European certification for the distribution of molecular biology reagents) report that can be viewed directly from the Idylla control console and (2) the Idylla Explore utility, which is available via a web browser. The latter allows users to view the raw amplification data in more detail.
- DNA extraction and quantitation for formalin-fixed, paraffin-embedded (FFPE) biopsy samples
DNA was extracted from FFPE tissue after macrodissection to achieve a minimum input of 15% tumour nuclei. DNA samples were extracted with the Maxwell RSC FFPE Plus DNA Kit (Promega®, Charbonnières-les-Bains, France). They were eluted in a final volume of 50 µL; 2 µL of which was used for quantity/quality assessment using QuantiFluor ONE ds DNA System (Promega).
- NGS analysis for DNA extracted from FFPE biopsy samples
This analysis was performed after the beginning of the BIP trial (NCT02534649), an institution-wide permanent screening programme started in our centre in 2014 to identify patients with somatic alterations. Patients are eligible in cases of advanced solid tumours and an ECOG performance status ⩽2. Tumour DNA was isolated from an FFPE archived sample when available or from a fresh tumour biopsy. DNA analysis was performed by NGS sequencing using the FoundationOne CDx panel (Foundation Medicine®, Cambridge, USA) that allows the detection of substitutions, insertion and deletion alterations, and copy number alterations in 324 genes.
Sensitivity evaluation
The criterion standard for the evaluation of the Idylla KRAS mutation test on liquid-based cytology was the NGS analysis performed in all patients (patients included in BIP protocol or performed in routine after the Idylla test for all patients outside the BIP trial). Sensitivity was defined as true positive/true positive + false negative.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 44
Results
Study population
The population analysed retrospectively contained 85 patients (40 women and 45 men), average age of 68 years (extremes: 44–85). In all, 12 cases of PDAC were initially resectable, 5 cases of PDAC were borderline, 32 were locally advanced and 36 were initially metastatic. Table 2 gives an overview of the study population.
Classification of category 7 samples with KRAS mutation testing (58 samples) and of non-category 7 samples (25 samples) according to the WHO System for Reporting Pancreaticobiliary Cytopathology. 43
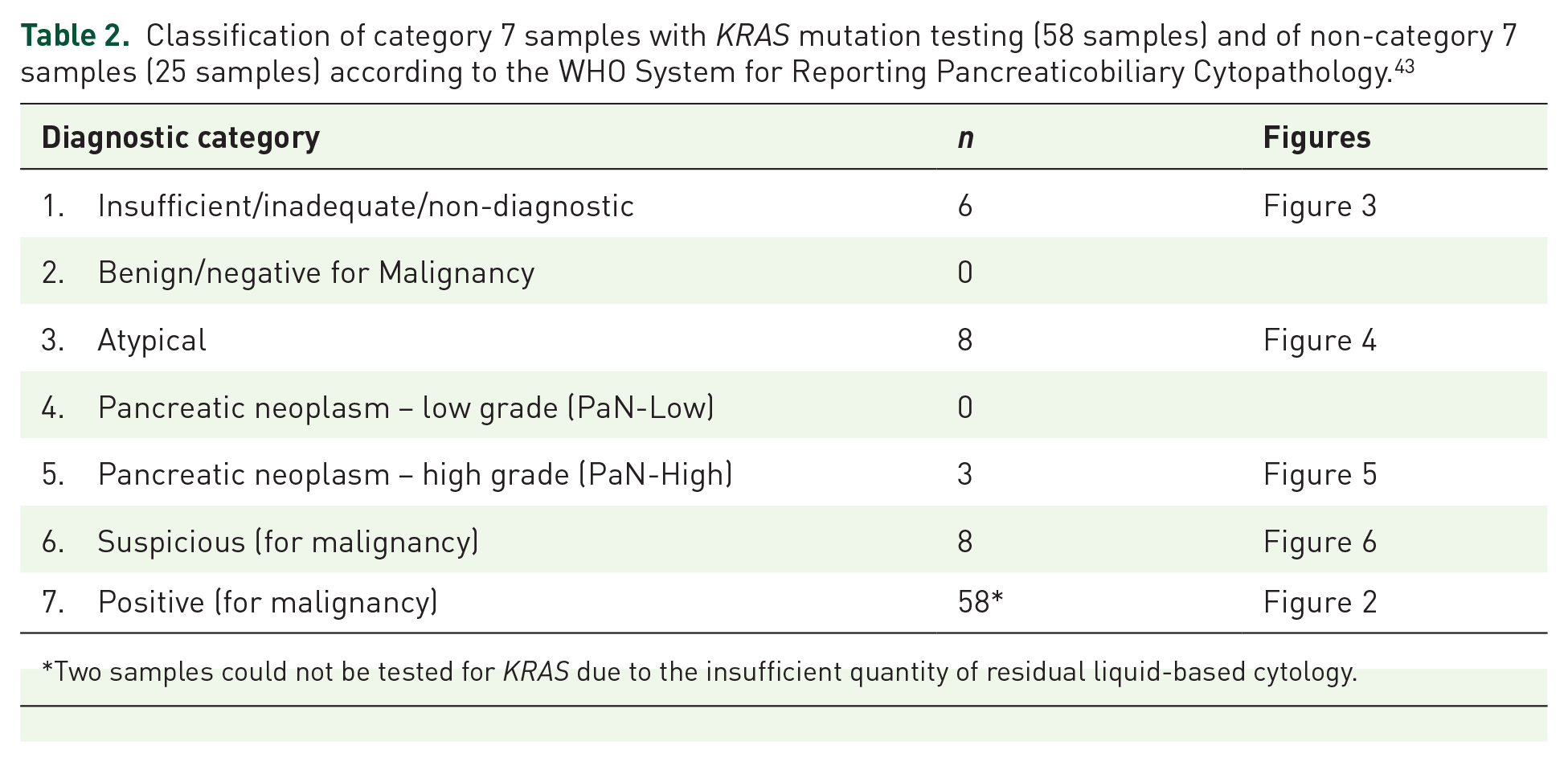
Two samples could not be tested for KRAS due to the insufficient quantity of residual liquid-based cytology.
Analysis of the proportion of non-contributive samples with the EUS-FNB technique and evaluation of the diagnostic impact of KRAS mutation testing
Between February 2019 and May 2023, 85 patients underwent exploration by EUS-FNB combined with KRAS mutation testing using the Idylla method (Figure 1). Two samples could not be tested for KRAS due to the insufficient quantity of residual liquid-based cytology. Among the 83 included patients, the final diagnosis of category 7 (Figure 2), PDAC was confirmed by EUS-FNB in 58 patients (69.8%). No complications with EUS-FNB were observed. Among the 25 patients not diagnosed by EUS-FNB, 4 (16%) were diagnosed with a second EUS-FNB, 17 (68%) with an X-ray or surgical biopsy and 4 with a surgical specimen (16%).
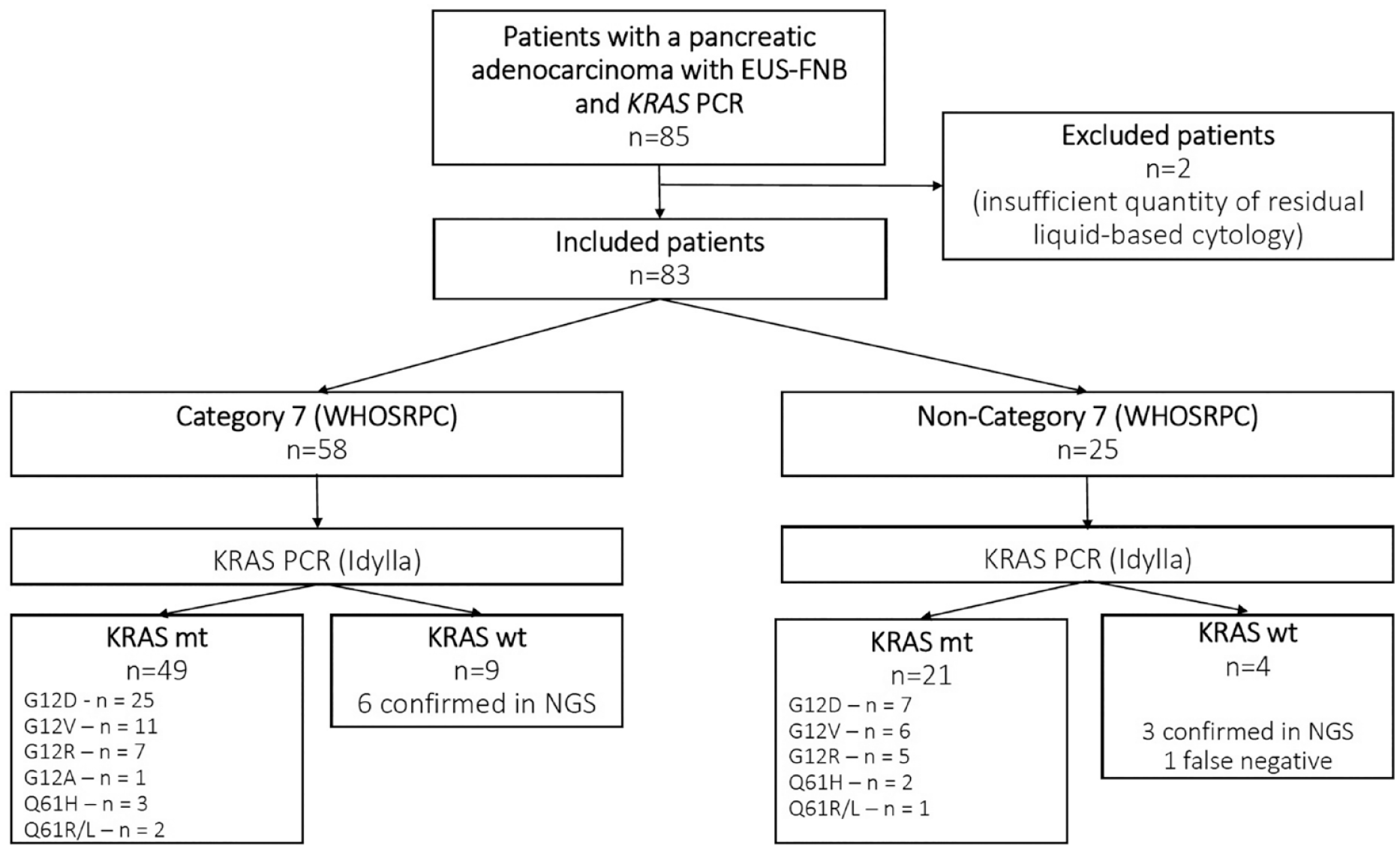
Flow chart of the study participants.

Category 7: Well-differentiated adenocarcinomatous proliferation with pancreato-biliary morphology. Some fragments of a fibrous stroma are visible, containing tumour-like glands, indicating the infiltrating nature.
In total, 25 EUS-FNB samples did not lead to a formal diagnosis of PDAC (30.1%). In this sub-group of non-category 7 samples, KRAS mutation testing by Idylla was positive in 21 patients, therefore in 84% of cases. All non-category 7 samples were reclassified according to the WHOSRPC (Table 1) 43 : six samples were category 1 (Figure 3), eight samples were category 3 (Figure 4), three samples were category 5 (Figure 5) and eight samples were category 6 (Figure 6). Primary surgery was carried out for three of them (two cephalic duodenopancreatectomies and one left splenopancreatectomy). The eight category 6 patients (suspicious for malignancy) were deliberately individualized as this estimated risk of malignancy varies from 80% to 100%. Furthermore, the three category 5 samples were all KRAS mutated for G12V, G12D and G12R, respectively. In total, 11 samples out of the 25 non-category 7 (44%) could have been considered positive for PDAC diagnosis initially thanks to the KRAS mutation test, without carrying out further diagnostic procedures. It should be noted that 10 category 1 and 3 samples were KRAS positive, all confirmed by NGS (4 G12D mutations, 4 G12V mutations and 2 G12R mutations).

Category 1: Haemorrhagic suffusions with fibrous fragments and gastrointestinal contaminants.

Category 3: Epithelial atypia within subacute inflammatory changes for which it is difficult to tell the difference between dystrophy and dysplasia.
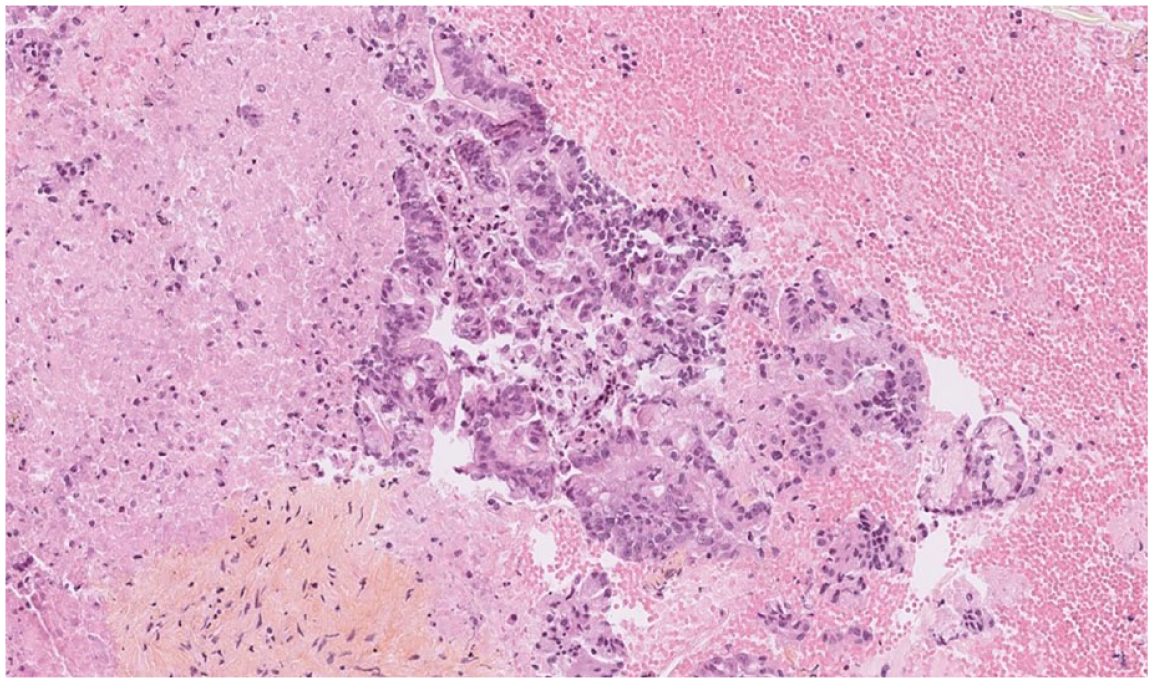
Category 5: Cribriform and adenomatoid structures with mucin-secreting cells or eosinophils with nucleolated atypical nuclei with loss of nuclear polarity.
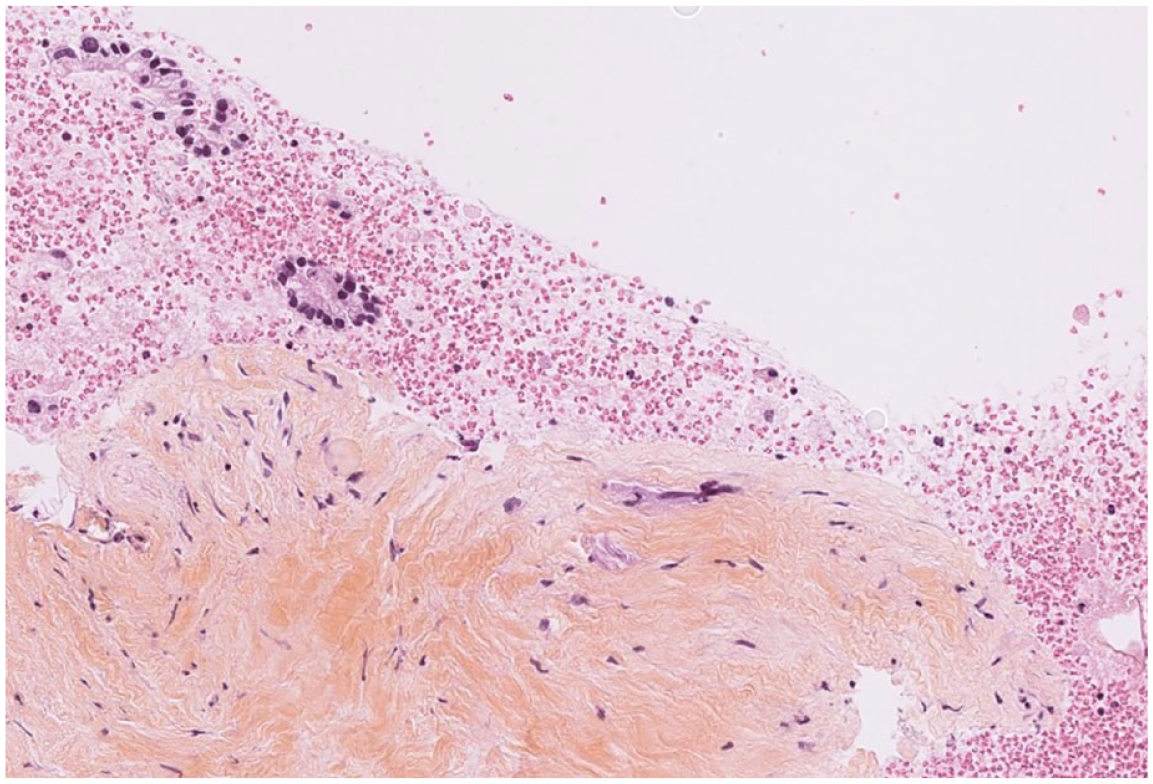
Category 6: Hematic material with a fragment of fibro-hyalin tissue comprising some isolated atypical cells.
Four KRAS tests were negative using Idylla in this group of 25 non-category 7 samples, and the wild-type was confirmed by NGS in two cases as part of the BIP trial. In one patient with a non-category 7 negative Idylla test, the NGS technique carried out outside the BIP trial thus confirmed the wild type. In the fourth patient with a negative Idylla test result by EUS-FNB, a G12D mutation was found thanks to the BIP protocol on another sample taken by NGS. Among the 58 patients whose EUS-FNB sample was category 7, nine presented with a wild-type KRAS tumour, confirmed by NGS in six of them on the EUS-FNB residual liquid-based cytology (n = 4) and by liquid biopsy (n = 2). One of these patients presented with microsatellite instability. In this series, the sensitivity of KRAS mutation testing on liquid-based cytology was 98.6%.
Theranostic analysis
Among the 83 patients, the G12D mutation was the most common (n = 33 – 39.7%), followed by the G12V mutation (n = 17 – 20.4%). The other mutations found were G12R (n = 12 – 14.4%), Q61H (n = 5 – 6%), Q61R/L (n = 3 – 3.6%) and Q12A (n = 1 – 1.2%). No G12C mutation was found. In all, 12 (14.4%) patients were found to have wild-type KRAS which was confirmed by the NGS technique in nine of them.
According to available data and probably due to the low number of the series, overall survival (OS) rates were not statistically different depending on the type of KRAS mutation: 13.3 months for G12V mutation, 11.3 months for G12D mutation, 11 months for G12R mutation and 10 months for KRAS wild type. When collecting data, 26 patients (31.3%) were still alive.
Discussion
The main interest of our study is to confirm the significant diagnostic impact of looking for KRAS mutations in samples of EUS-FNB of PDAC. In all, 11 samples out of the 25 non-category 7 (eight category 6 and three category 5) (44%) could have been considered positive for PDAC diagnosis initially thanks to the KRAS mutation test, without carrying out further diagnostic procedures. If we had determined the 11 KRAS-mutated category 5 and 6 samples as positive for the diagnosis of PDAC, in the end, there would have been only 14 non-contributive samples out of 83 (16.8%), instead of 25 classified non-category 7 based on the pathology data alone. Yet, this 16.8% rate is similar to that of 15–20% of cases for which Lundy et al. 14 or other authors 45 state the need for a second sample, even with second-generation cutting needles.
The appearance of second-generation cutting needles was accompanied by the development of different sampling techniques depending on the structure of PDAC in EUS, as we experimented in our series 17 : modified wet-suction technique (the stylet is removed before using the needle, then the needle is filled with 1–2 mL of saline solution with the application of suction using a 10-mL pre-vacuum syringe) appears superior to the dry suction technique associated with high rates of blood contamination. The slow-pull technique appears to be a valid alternative to wet suction, 41 especially for highly vascularized pancreatic lesions, by reducing the risk of haemorrhagic contamination. 17 There is now good agreement that these newer end-cutting needles avoid using Rapid On-Site Evaluation.15,17,46 Despite all these techniques and unlike other tumour sites, PDAC diagnosis by EUS-FNB remains difficult in at least 15–20% of cases, meaning another sample has to be taken.14,45
The recent guidelines from the WHOSRPC 43 come in addition to the Papanicolaou classification. 47 They have the advantage of offering a clinical approach adapted to the diagnostic category (Table 1). In the two meta-analyses cited above,12,13 the authors had included patients for whom the diagnosis was confirmed to be positive, even if it was only suspected or questionable (categories IV B and V of the Papanicolaou classification), 47 leading to very good diagnostic performances. In our series, category 6 of the WHOSRPC 43 was deliberately determined to be insufficient to confirm the PDAC diagnosis, likely explaining our apparently average diagnostic performances (25 non-category 7 samples, out of 83 EUS-FNB). If patients need to be treated with chemotherapy, classification in category 6 means another sample has to be taken, 43 usually by EUS-FNB, which delays the initiation of the treatment. These treatment options are very useful for category 6 especially. In the same way, classification in category 6 does not appear to us to be sufficient for inclusion in a clinical trial.
In our series, the samples were category 6 in eight patients. Primary surgery was carried out for three of them (two cephalic duodenopancreatectomies and one left splenopancreatectomy). Even if surgical treatment is theoretically allowed for category 6, 43 the indication for pancreatic surgery seems to us to be difficult to set down in this category in which the estimated risk of malignancy varies from 80% to 100%. 43 This may explain why 6–10% of cephalic duodenopancreatectomies carried out in expert centres for suspected clinical-biological, morphological or cytological PDAC corresponds to non-tumoural lesions on the pathological test.48,49 Thirty-five percent of them are AIP. 48 The figures, which are a little old in these two studies,48,49 still apply even in expert centres. We believe that pathological documentation of a solid pancreatic mass is essential, even before surgery, as 15% of pancreatic tumour lesions are not adenocarcinomas. In effect, a non-metastatic solid pancreatic mass is an adenocarcinoma in only 70% of cases. 45
Some US guidelines tend to offer routine KRAS gene testing to improve EUS diagnostic performances. 50 To date in Europe and France, the research for KRAS mutations in EUS-FNB materials from solid pancreatic lesions has not been integrated into current clinical practice which, in our opinion, would improve the diagnostic profitability of EUS-FNB, given the unique nature of PDAC compared to other tumour sites explored by EUS-FNB. To our knowledge, there are no false positives associated with KRAS testing for primary PDAC with tests that are similar to Idylla. A KRAS mutation can be identified during EUS-FNB biopsies of pancreatic metastatic locations, but with a different clinical context, suggestive of echoendoscopic aspects, and a diagnostic orientation that can be given by immunohistochemistry. KRAS mutations can also be identified in a particular sub-group of precancerous lesions such as CP cores or Intraductal Papillary Mucinous Neoplasms of the pancreas. The benefits of searching for KRAS mutations to detect lesions at risk of degeneration seem to warrant repeating a EUS-FNB in the case of category 1, 2 or 3 lesions with positive KRAS mutations.
Other than diagnostic purposes, KRAS mutation testing on the residual liquid-based cytology specimen could also play a role in assessing the prognosis, as shown by Buscail et al. 27 and other authors.22,51,52 These series mainly find G12D mutations with the worst prognosis. A recent series of 5555 samples presented at ASCO 2023 confirms that the G12D mutation is the most common, followed by the G12V and G12R mutations. 53 OS in this series was significantly longer for patients presenting with PDAC with G12R mutation compared to G12D-mutated PDAC (396 versus 311 days, HR 0.81, CI 0.74–0.88, p < 0.0001). 53
In terms of treatment, KRAS status testing by EUS-FNB could be used, for example, to anticipate the response to chemotherapy such as gemcitabine and nab-paclitaxel, or to other treatments.54,55 No G12C mutations were found in our study as they are very rare (1–2%). 54 However, screening for this mutation could allow treatment with KRASG12C inhibitors 54 after a line of chemotherapy. The compilation of a complete molecular profile for PDAC on the samples collected by EUS-FNB is validated by several studies.5,55–58 It could facilitate the inclusion of homogeneous patient populations in clinical research trials. Determination of wild-type KRAS, from the diagnosis, can be used to complete the molecular analysis of the tumour and help select targeted treatments. 59 This sub-group is characterized by a large heterogeneity of molecular anomalies. 59 A recent study showed the benefit of combining the anti-Epithelial Growth Factor Receptor (EGFR) antibody nimotuzumab with gemcitabine in the first-line treatment of patients with non-KRAS mutated, locally advanced or metastatic PDAC. 60 This latter study underlines the usefulness of developing new anti-(EGFR) antibodies in wild-type KRAS PDAC, which is why it is useful to screen early by EUS-FNB. Despite the small number of wild-type KRAS tumours in the series, we believe that confirmation of the wild-type by the NGS technique seems desirable.
The limitations of our analysis are related to the small number in the series and to the retrospective data. One of the significant limitations lies in the non-use of contrast harmonic echo-EUS18,19,61 or DFI 62 which seems to significantly increase the diagnostic accuracy of EUS-FNA and EUS-FNB, in the most complex cases in particular. Furthermore, KRAS testing using the Idylla technique is not available in all centres. Its cost (181.23 Euros per patient), compared to the cost of a new EUS-FNB under general anaesthesia in the event of a non-contributive biopsy, does not seem limiting. These results encourage us to propose a larger, prospective, multicentre, randomized study evaluating a diagnosis of PDAC by EUS-FNB with or without KRAS testing.
In conclusion, our retrospective analysis suggests the usefulness, even with second-generation cutting needles, of KRAS mutation testing on the liquid-based cytology as this technique affirms or confirms the PDAC diagnosis in 44% of cases in which the EUS-FNB sample alone is questionable or suspect. The recent guidelines from the WHOSRPC 43 could be used as a pathological and clinical reference. Our series puts forward that category 6 samples, and certainly category 5 samples positive for KRAS mutations, could be considered sufficient for diagnosing PDAC. Determining the KRAS status also may make it possible to evaluate the prognosis according to the type of mutation. Second-generation cutting needles allow for more complete molecular profiling of PDAC for inclusion in clinical trials. KRAS wild type by EUS-FNB should be confirmed by the NGS technique to offer targeted treatments, especially with new anti-EGFR antibodies. We believe that testing for a KRAS mutation could be part of the diagnostic and theranostic guidelines in light of a solid pancreatic mass in standard practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231224943 – Supplemental material for Echoendoscopic ultrasound pancreatic adenocarcinoma diagnosis and theranostic approach: should KRAS mutation research be recommended in everyday practice?
Supplemental material, sj-docx-1-tag-10.1177_17562848231224943 for Echoendoscopic ultrasound pancreatic adenocarcinoma diagnosis and theranostic approach: should KRAS mutation research be recommended in everyday practice? by Dominique Béchade, Lola-Jade Palmieri, Benjamin Bonhomme, Simon Pernot, Jeanne Léna, Marianne Fonck, Sophie Pesqué, Gautier Boillet, Antoine Italiano and Gilles Roseau in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
