Abstract
Background:
The Kirsten rat sarcoma viral oncogene homolog (KRAS) gene, specifically the cysteine residue mutation KRAS (G12C), has garnered significant attention as a therapeutic target for solid cancer patients with KRAS mutations. Despite this interest, the efficacy and safety profiles of KRAS G12C inhibitors remain incompletely understood. In this study, we comprehensively evaluate the effectiveness and toxicity of relevant KRAS G12C inhibitors (Sotorasib, Adagrasib, Garsorasib, and Divarasib) in patients with colorectal cancer (CRC), non-small-cell lung cancer (NSCLC), and pancreatic ductal adenocarcinomas (PDAC).
Methods:
Our systematic review is guided by Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. We review the available clinical trials data on KRAS G12C inhibitors in KRAS G12C-mutated solid tumors. We searched PubMed, EMBASE, Cochrane Library, and major international conferences for clinical trials from January 2020 until August 2023.
Results:
A total of 17 eligible studies were included. KRAS G12C inhibitions with Sotorasib (41.2%) and Adagrasib (41.2%) each of them were reported in 7 studies. Divarasib was reported in 2 studies (11.8%) and Garsorasib was reported in 1 study (6.7%). Sotorasib showed a significant clinical benefit in terms of objective response rate (ORR) (7.1%-47%), progression-free survival (PFS) (4-6.8 months), and overall survival (OS) (4-24 months); it is more efficient in NSCLC patients with an OS of 2 years, PFS of 6.3 months, and an ORR of 41%. Adagrasib also showed significant clinical activity with an ORR (19%-53%), PFS (3.3-11.1 months), and OS (10.5-23.4 months), with more effectiveness in NSCLC patients with an OS of 23.4 months, PFS of 11.1 months, and an ORR of 53.3%. Adagrasib is more efficient with an ORR of 35.1%, PFS of 7.4 months, and an OS of 14 months in patients with PDAC, than Sotorasib which showed an ORR of 21%, PFS of 4 months, and an OS of 6.9 months. However, Adagrasib and Sotorasib are moderately efficient in CRC clinical trials.
Conclusion:
This study confirms that patients treated with these KRAS G12C inhibitors, exclusively or combined with conventional therapies, achieve better treatment responses and modulate the progressions of these solid tumors.
Introduction
Globally, there were approximately 20 million new cases of cancer and nearly 9.7 million cancer-related deaths in 2022. 1 Solid tumors account for roughly 90% of both human cancer cases and cancer-related deaths. 2
The Kirsten rat sarcoma viral oncogene homolog (KRAS) is one of the biomarkers that can be used in clinical practice prognosis and targeted therapies for cancer. The panel of KRAS genetic alterations is helpful in defining the therapeutic strategy for a better management of patients. 3 KRAS is a key mediator of the RAS/RAF/MAPK signaling cascade and promotes cellular growth and proliferation. 4 KRAS is a proto-oncogene that encodes a small GTPase transductor protein. KRAS belongs to a group of small guanosine5′-triphosphate (GTP) binding proteins, known as RAS superfamily or RAS-like GTPases. 5 KRAS protein cycles between active GTP-bound and inactive guanosine-5′-diphosphate (GDP)-bound, respectively. 6 KRAS gene mutations are one of the most commonly identified driver mutations, accounting for 86% of RAS mutations in various human malignancies. 7 KRAS gene mutations are detected in 45% of colorectal cancer (CRC), 8 15% to 30% of lung cancers patients, mostly in non-small-cell lung cancer (NSCLC),6,9 and in approximately 90% of pancreatic ductal adenocarcinomas (PDAC) tumors. 10
Associated with poor prognosis and resistance to treatment, KRAS G12C represents a significant clinical relevance. KRAS G12C mutation has a causal role in 14% of NSCLC (∼14 000 new US cases annually), 11 in 5% of CRC (∼5000 new US cases annually),11-13 and in 1% to 2% of PDAC.14,15
The KRAS G12C is a single point mutation with a glycine-to-cysteine substitution at codon 12 (KRAS c.34G > T) near a narrow pocket in the KRAS protein. This substitution favors the activated state of KRAS, amplifying signaling pathways that lead to oncogenesis. 16 Advances in understanding the protein structure have led to the investigation of inhibitors that target the pocket, potentially locking the KRAS G12C mutant protein in the inactive state. However crystallographic studies reveal the formation of a new pocket in the switch II effector region of a mutant allele, KRAS G12C. 17 The mutated cysteine binds next to the pocket (P2) of the switch II region. The P2 is present only in the inactive GDP-bound conformation of KRAS and has been exploited to establish covalent inhibitors of KRAS G12C.17-21
Therefore, the KRAS G12C mutations are newly under investigation for treatment regimens. Recently, a breakthrough reported the development of small molecules that selectively and irreversibly bind to KRAS G12C, and so far, early results of phase 2 studies of the 2 KRAS G12C inhibitors, Adagrasib (MRTX849) and Sotorasib (AMG510), have been reported. 22 Clinical trials of Adagrasib and Sotorasib have shown promising activity in cancers harboring KRAS G12C, 23 including CRC and NSCLC.16,24,25 Currently, the US Food and Drug Administration (FDA) granted accelerated approval to Adagrasib and Sotorasib having marked clinical responses across patients with KRAS G12C-mutated advanced CRC whose cancer has progressed following prior treatment with chemotherapy and an anti-vascular endothelial growth factor therapy (anti-VEGF),26,27 and NSCLC who have received at least one prior systemic therapy.28-30
We conducted this systematic review of studies providing updated data about the current targeted therapy strategy by newly discovered molecules of KRAS G12C inhibition with Sotorasib, Adagrasib, Garsorasib, and Divarasib in patients with NSCLC, CRC, and PDAC to evaluate the efficacy and safety of these KRAS G12C inhibitors in patients with these solid tumors. Our finding highlights the effectiveness and safety of the use of KRAS G12C inhibitors for the treatment of patients with solid tumors.
Methods of research
Search strategy and data extraction
This systematic review provides a comprehensive evaluation of the efficacy of newly developed KRAS G12C inhibitors in treating solid malignancies, including CRC, NSCLC, and PDAC. To achieve this, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 31 We systematically searched the following databases from January 2020 to August 2023: PubMed, ScienceDirect, Embase, Scopus, Google Scholar, and Web of Science and the Cochrane Library. These databases were selected to ensure comprehensive coverage of relevant clinical research and systematic reviews. The retrieval period was selected to encompass the most relevant and recent advancements in KRAS G12C inhibitor research and clinical trials. While KRAS mutations have been known for decades, the development of effective inhibitors targeting the KRAS G12C mutation is a relatively recent advancement, with significant breakthroughs occurring after 2020. This timeframe allows for the inclusion of studies that reflect the current clinical landscape of KRAS G12C-targeted therapies.
Only articles published in peer-reviewed journals were included. To confirm the peer-reviewed status of the journals, we verified each journal’s indexing in reputable databases such as PubMed, Embase, Scopus, and the Cochrane Library. In addition, we checked the editorial policies of the journals to ensure that they follow a peer-review process. This verification process ensured that all included studies met our criteria for scientific rigor and quality.
To evaluate the quality and reliability of the included studies, we conducted a risk of bias assessment using the Cochrane Risk of Bias tool for randomized controlled trials and the Newcastle-Ottawa Scale for observational studies. Each study was independently assessed by 2 reviewers, and discrepancies were resolved through discussion. Identified risks of bias were discussed in the results and recommendations sections to provide a transparent evaluation of the potential impact on the review’s findings.
Figure 1 illustrates the PRISMA flowchart detailing the review process. Our search strategy incorporated a range of terms: “colorectal cancer,” “CRC,” “CRC tumor,” “metastatic colorectal cancer,” “mCRC,” “lung cancer,” “lung tumor,” “non-small cell lung cancer,” “NSCLC,” “pancreatic cancer,” “pancreatic ductal adenocarcinoma,” “PDAC,” “KRAS,” “KRAS mutation,” “KRAS G12C,” “KRAS G12C mutation,” “G12C,” “G12C mutation,” “KRAS G12C inhibitors,” “KRAS G12C-targeted treatments,” “Sotorasib,” “Adagrasib,” “Garsorasib (D-1553),” and “Divarasib (GDC6036).”
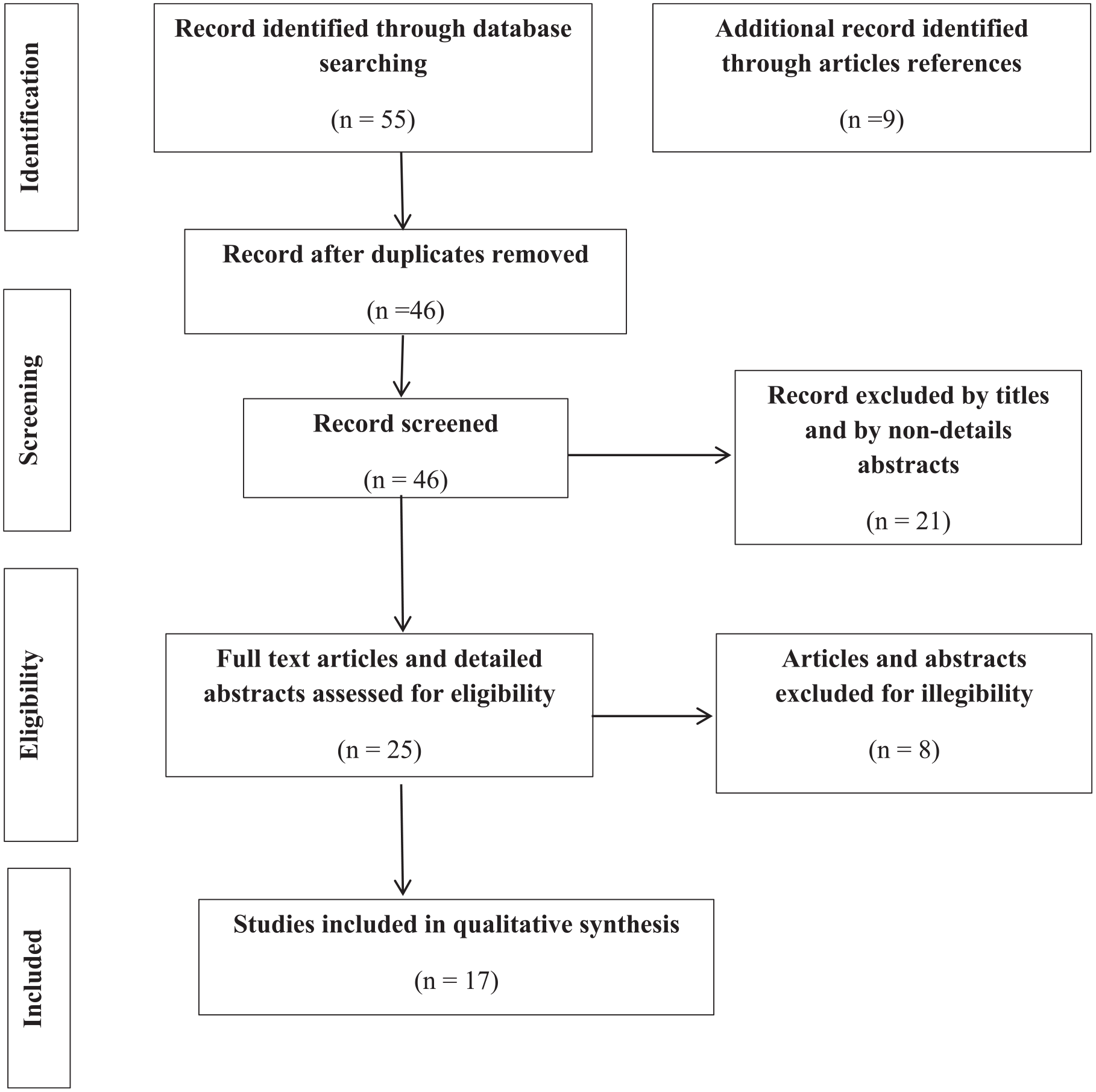
Flowchart for included and excluded studies in this review according to the PRISMA guidelines.
In addition to database searches, we performed a manual review of the bibliographies of the included studies and relevant review articles to identify additional eligible studies. We also searched for pertinent abstracts from leading multidisciplinary medical oncology associations, including the American Society of Clinical Oncology and the European Society for Medical Oncology, to uncover the latest research and newly published studies.
To address potential duplication in the data, studies with overlapping datasets were carefully evaluated. Preference was given to the most comprehensive and recent publications, prioritizing studies based on their dataset completeness, publication date, and relevance. This approach ensured that duplicate data did not bias the findings.
To further mitigate the risk of duplication, we used The National Clinical Trial numbers (NCT) to identify publications arising from the same clinical trials. In cases where multiple publications with the same NCT number were identified, these publications were grouped, and their datasets were compared with identify overlaps. Data from the most comprehensive and recent publication were included to avoid redundancy and bias. Distinct analyses or non-overlapping data reported across publications were considered separately and appropriately documented.
Duplicate titles were first eliminated. We then conducted a title check to assess relevance. The remaining publications’ abstracts were examined, and irrelevant ones were removed. For the remaining articles, full-text manuscripts and/or conference posters or presentations were reviewed. When multiple versions of the same data were available, preference was given to the most comprehensive and recent updates.
Inclusion criteria
We designed precise inclusion criteria to ensure the quality and pertinence of the studies incorporated in this review and to avoid risk of bias. Particularly, studies must have involved the clinical activity of KRAS G12C inhibitors (either as monotherapy or combined with chemotherapy) in pretreated patients with mCRC, NSCLC and PDAC harboring KRAS G12C mutation; study participants genotyped for KRAS exon 2 (codons 12) specifically G12C mutation. Information was carefully extracted from all eligible studies. The following data were collected from each study: first author’s name, year of publication, cancer type, number of patients with KRAS G12C mutations, median age, previous treatment protocols, study treatment protocols, dose of treatment, response criteria, complete response (CR) or partial response (PR), confirmed objective response rate (ORR), median follow-up, disease control rate (DCR), duration of response (DOR), median progression-free survival (PFS), and overall survival (OS). Letters, comments, and review articles or meta-analysis without original data were excluded from analysis.
Results
Literature research
The literature search was conducted following PRISMA guidelines and is visually represented in Figure 1. After an initial search across designated databases, 55 publications were identified. Following the removal of unrelated and duplicate records, 46 articles remained for further screening. These were assessed based on their titles and abstracts, leading to the exclusion of 21 articles that did not meet the inclusion criteria. The full text of the remaining 25 articles was then thoroughly reviewed. Eight additional studies were excluded due to factors such as non-detailed abstracts or insufficient inclusion criteria, as outlined in Figure 1. Key details such as publication dates, authorship, and targeted inhibition molecules were extracted from the eligible studies and organized using Microsoft Excel for further analysis. These publications were assessed for eligibility and 17 studies were selected for this review: 10 (58.8%) are full text articles,15,32-40 and 7 (41.2%) are detailed abstracts.41-47
Results of risk of bias assessment
The risk of bias for the included studies was assessed using the Cochrane Risk of Bias tool for randomized controlled trials (RCTs) and the Newcastle-Ottawa Scale (NOS) for observational studies (see Table 1). Most RCTs demonstrated a low to moderate risk of bias, with concerns primarily related to blinding due to the open-label design of some studies. Randomization and allocation concealment were adequately reported in most RCTs, contributing to a low risk of selection bias. However, performance bias was noted in a few studies due to the lack of blinding, which could influence the reporting of subjective outcomes. Detection bias was generally low, as objective measures such as ORR and PFS were used for outcome assessment. For observational studies, the NOS scores indicated moderate to high quality, with most studies performing well in the selection and outcome domains. However, some studies had incomplete outcome data, which was addressed through sensitivity analyses to ensure the robustness of the findings. Overall, the risk of bias across the included studies was considered acceptable, with no significant concerns undermining the validity of the conclusions. These findings suggest that the included studies are of sufficient quality to support the conclusions of this review.
Risk of bias assessment of included studies.

Abbreviation: NI, not identified.
Studies characteristics
Seventeen studies met our inclusion criteria,15,32-47 comprising 10 studies on NSCLC patients,32-36,39,40,43,45,46 8 on CRC patients,32,33,36-38,42,44,47 2 on PDAC patients,15,41 and 3 studies that included both CRC and NSCLC patients.32,33,36 The studies included a mix of phase I, phase II, and 1 phase III trial, 40 with eligibility criteria generally including patients aged 18 years or older with KRAS G12C mutations. These patients often had prior exposure to platinum-based chemotherapy, systemic therapy, and inhibitors of programmed death 1 (PD-1) or programmed death ligand 1 (PD-L1), reflecting a heavily pretreated population. Across the 17 studies, a total of 1336 patients with KRAS G12C mutations were included, with the majority being NSCLC patients 890/1336 (66.6%), followed by CRC 351/1336 (26.3%) and PDAC 95/1336 (7.1%) patients. Treatment regimens involved KRAS G12C inhibitors such as Sotorasib, Adagrasib, Garsorasib (D-1553), and Divarasib (GDC6036) at varying doses ranging from 400 mg to 960 mg daily, depending on the specific study protocol.
Of these studies, 7 (41.2%) focused on Sotorasib, with 685 patients receiving a standard dose of 960 mg orally once daily.15,32,35,38-40,43 Similarly, 7 studies (41.2%) reported on Adagrasib, which involved 407 patients who were treated with a 600 mg dose twice daily.33,34,37,41,44,46,47 The 2 studies on Divarasib (GDC6036) included 115 patients receiving doses between 50 and 400 mg once daily,36,42 while one study (6.7%) on Garsorasib (D-1553) involved 74 patients receiving 600 mg orally once daily. 45 The study characteristics and patient demographics are summarized in Table 2, highlighting the diversity in study design, population, and dosing strategies across the included studies. In addition, a total of 125 (9.4%) patients with CRC analyzed in Table 3, 3 studies evaluated patients who received Adagrasib combined with Cetuximab,37,44,47 and 1 study evaluated patients who received Divarasib (GDC-6036) combined with Cetuximab. 48
The main characteristics of the included studies.

Abbreviations: CRC, colorectal cancer; F, female; M, male; NI, not identified; NSCLC, non-small cell lung cancer; PD-1, programmed death 1; PDAC, pancreatic ductal adenocarcinoma; PD-L1, programmed death ligand 1.
The main characteristics of the CRC patients treated with KRAS G12C inhibitors combined with Cetuximab.

Abbreviations: CRC, colorectal cancer; F, female; M, male; NI, not identified; NSCLC, non-small cell lung cancer; PDAC, pancreatic ductal adenocarcinoma.
Efficacy of KRAS G12C inhibitor molecules as monotherapy in NSCLC, CRC, and PDAC
KRAS G12C inhibition in patients with NSCLC
Sotorasib
The ORR for Sotorasib varied significantly across studies, ranging from 28.1% to 47%, median follow-up was 2.8 to 24 months, DCR was 82.5% to 88.1%, DOR was 3.9 to 12.3 months, and the median PFS ranged from 5.5 to 6.8 months, while OS was ranging from 4 to 23.4 months. These differences may be attributed to variations in patient populations and study designs. Notably, Sotorasib was tested in a larger cohort of NSCLC (543 patients), possibly contributing to the wider range of outcomes.32,35,39,40,43
Adagrasib
Adagrasib was tested in 213 patients with NSCLC33,34,46 which demonstrated a slightly more consistent ORR of 42.9% to 45%, median follow-up was ranged between 12.9 and 19.6 months, DCR was 79.5%, DOR was about 8.5 to 16.4 months, PFS ranged from 6.5 to 11.1 months, and OS was 12.6 months to 23.4 months, suggesting that Adagrasib and Sotorasib might offer prolonged disease control in patients with NSCLC.
Garsorasib and Divarasib
Garsorasib achieved an ORR of 40.5%, 45 and Divarasib showed a notably higher ORR of 53.4%, 36 suggesting that Divarasib may induce a stronger initial response. However, data on other efficacy measures such as DOR and OS for these newer inhibitors is lacking, which limits the ability to draw definitive conclusions.
Clinical implications
While Sotorasib and Adagrasib are the most well-established KRAS G12C inhibitors in NSCLC, newer molecules like Divarasib may offer higher response rates. However, the lack of long-term data means the choice of therapy should be tailored to the patient’s clinical needs, carefully considering both short-term efficacy and long-term outcomes.
KRAS G12C inhibition in patients with CRC
Sotorasib
Sotorasib demonstrated an ORR of 7.1% to 9.7%, with a DCR ranging from 73.8% to 82.3%. The median PFS was consistently around 4 months across studies.32,38 These findings suggest that Sotorasib provides stable disease control, though the response rate remains modest.
Adagrasib
Adagrasib achieved a higher ORR of 19.6% to 50%, with a DCR of 86% to 87%. PFS ranged from 3.3 to 5.6 months. Adagrasib may offer better disease control, but its impact on progression-free survival varies more across studies.33,37,44,47
Divarasib
Divarasib achieved an ORR of 24% to 29.1%, and the PFS was 5.6 months,36,42 outperforming both Sotorasib and Adagrasib in terms of tumor shrinkage. However, data on long-term outcomes such as OS, DCR, and DOR are currently unavailable, which necessitates further study.
Clinical implications
For patients with CRC, Divarasib appears to offer the most promising ORR, while Sotorasib provides more consistent long-term control. The choice between these inhibitors may depend on whether immediate tumor reduction or sustained disease control is prioritized in a patient’s treatment plan.
KRAS G12C inhibition in patients with PDAC
Sotorasib
Sotorasib showed an ORR of 21%, a PFS of 4 months, and DCR was about 84% in patients with PDAC. 15 These results suggest that Sotorasib can provide disease control, though its efficacy in PDAC appears lower than in NSCLC or CRC.
Adagrasib
Adagrasib reported an ORR of 35.1%, with a median PFS of 7.4 months, and OS was 14 months. 41 These results indicate that Adagrasib might be more effective in patients with PDAC compared with Sotorasib, particularly in prolonging progression-free survival and the OS.
Clinical implications
In patients with PDAC, Adagrasib appears to outperform Sotorasib in both ORR and PFS. Given the aggressive nature of PDAC, Adagrasib may offer more meaningful clinical benefits for these patients.
The numerical detail of the effectiveness of each molecule across the tumors types studied is represented in Table 4.
KRAS G12C inhibitors potency in patients with NSCLC, CRC, and PDAC.

Abbreviations: CRC, colorectal cancer; DCR, disease control rate; DOR, duration of response; NI, not identified; NSCLC, non-small cell lung cancer; ORR, objective response rate; OS, overall survival; PDAC, pancreatic ductal adenocarcinoma; PFS, progression-free survival.
Efficacy of KRAS G12C inhibition in combination with cetuximab in patients with CRC
Adagrasib + Cetuximab
In combination with Cetuximab, Adagrasib achieved the ORR of 43% to 46%, a PFS of 6.9 months, and an impressive 100% of DCR (Table 5),37,44,47 indicating excellent disease control in patients with CRC. These results suggest that the combination therapy might be highly effective for patients who do not respond well to monotherapy.
Clinical activity of KRAS G12C inhibitors combined with Cetuximab in patients with CRC.

Abbreviations: CRC, colorectal cancer; DCR, disease control rate; DOR, duration of response; NI, not identified; NSCLC, non-small cell lung cancer; ORR, objective response rate; OS, overall survival; PDAC, pancreatic ductal adenocarcinoma; PFS, progression-free survival.
Divarasib + Cetuximab
The combination of Divarasib and Cetuximab showed an ORR of 62% (Table 5), 48 which is slightly higher than Adagrasib + Cetuximab. However, the limited sample size in this study warrants further investigation to confirm these findings.
Clinical implications
Combination therapy with Adagrasib and Cetuximab might be more effective in CRC, particularly for patients who have limited response to KRAS G12C monotherapy. However, Divarasib + Cetuximab also present a promising alternative that requires further exploration.
Safety profiles of KRAS G12C inhibitors in the solid tumors studied
Adagrasib was associated with higher rates of nausea and diarrhea, while Sotorasib had more frequent elevations in ALT and AST, indicating a higher risk of liver toxicity. Divarasib and Garsorasib, on the other hand, exhibited a higher rate of grade 3 TRAEs, which might limit their clinical use despite their efficacy. The details about TRAEs are represented in Table 6.
Summary of treatment-related adverse events of KRAS G12C inhibitors.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; NI, not identified.
Clinical implications and management strategies
The choice of KRAS G12C inhibitors should consider the patient’s comorbidities. Patients with a history of liver disease might benefit more from Adagrasib, while those prone to gastrointestinal issues might prefer Sotorasib. Divarasib and Garsorasib’s higher rates of severe adverse events require careful monitoring in clinical practice. Consistent surveillance and proactive control are crucial for optimizing the therapeutic benefit of KRAS G12C inhibitors while reducing the impact of adverse events on patients’ quality of life.
Discussion
Conventional treatment with chemotherapy, radiotherapy, or surgery is the therapy of choice for most cancer patients.49-51 In the last decade, various targeted treatments have considerably evolved owing to increasing knowledge in cancer therapy and in immuno-oncology, allowing the development of precision medicine as a more specific and less toxic way to manage cancer. 52 Therapeutic drugs have always been the dominant strategy for unresectable locally advanced or metastatic solid tumors. Both the number of solid tumor drugs and their proportion among all FDA-approved drugs have increased during the past 31 years, especially in the most recent decade. More importantly, cytotoxic drugs have evolved into drugs with more precise targeting effects, including small-molecule targeted drugs, monoclonal antibodies (mAbs), and antibody–drug conjugates (ADCs). 53
Despite these recent advances in treatment for solid tumors, the OS is still relatively poor, and there is a continuous need for more effective therapies. The use of tyrosine kinase inhibitors (TKIs) has acquired an essential role in the treatment of relapsed or refractory tumors and its use has increased, being currently a common approach in solid cancer treatment.54-56 The KRAS G12C inhibition is a potential therapeutic strategy for advanced-stage KRAS G12C-mutant solid tumors. Recently, several inhibitors targeting KRAS G12C with similar covalent binding mechanisms have been investigated in clinical trials being Sotorasib, Adagrasib, Garsorasib, and Divarasib.
The findings of this review support the treatment by KRAS G12C inhibitions using Sotorasib, Adagrasib, Garsorasib, and Divarasib for patients with CRC, NSCLC, and PDAC. This wide variety of KRAS G12C inhibitors was described in the 17 studies analyzed in our review. To facilitate the discussion, only the 2 inhibitors Sotorasib and Adagrasib that were used most frequently (41.2%) will be discussed in depth.
Sotorasib and Adagrasib were evaluated mostly in phase I, and II trials. Sotorasib showed a significant clinical benefits in terms of ORR (7.1%-47%), PFS (4-6.8 months), and OS (4-24 months), more efficient in NSCLC patients with a maximum of an OS of up to 2 years, PFS of up to 6.3 months, and with an ORR of 41%. For Adagrasib, clinical trials also showed a significant clinical activity with an ORR (19%-53%), PFS (3.3-11.1 months), and OS (10.5-23.4 months), also more effectiveness in NSCLC patients with a maximum of an OS up to 23.4 months, PFS of up to 11.1 months, and with an ORR of 53.3%. In patients with PDAC, Adagrasib appeared more efficient than Sotorasib, showing an ORR of 35.1%, a PFS of about 7.4 months, and an OS of up to 14 months, compared with Sotorasib’s ORR of 21%, a PFS of about 4 months, and an OS of up to 6.9 months. In CRC clinical trials, Adagrasib and Sotorasib showed some antitumor activity, but their overall efficacy was limited compared with their performance in NSCLC and PDAC. For Adagrasib, the observed ORR ranged from 19%-50% with a PFS of up to 5.6 months, while Sotorasib had a lower ORR ranging from 7.1%-9.7% and a PFS of up to 4 months. These results indicate that while some patients with CRC respond to these treatments, the benefits are less pronounced compared with other cancer types.
Across a variety of tumor types, clinical evidence continues to be generated. In preclinical analyses, treatment with Sotorasib led to the regression of KRAS G12C tumors and improved the anti-tumor efficacy of chemotherapy and targeted agents. 25 The CodeBreaK 200 trial showed that Sotorasib led to a 34% decrease in relative risk of disease progression or death compared with docetaxel but yielded no improvement in overall survival. 57
Sotorasib is the only FDA-approved KRAS G12C inhibitor and has shown satisfying results in real-world studies. 58 Garsorasib and Divarasib also represent a promising therapeutic option for patients with KRAS G12C-mutated NSCLC and CRC with a well-tolerated safety profile and encouraging antitumor activity; however, more studies are needed to develop that the results are still inconclusive, partially because of the relatively small sample size of each study.
Studies have shown that while Sotorasib and Adagrasib exhibit significant antitumor efficacy as monotherapies, their combination with conventional therapies can enhance clinical outcomes. For instance, in patients with NSCLC, Sotorasib combined with chemotherapy demonstrated improved anti-tumor efficacy compared with monotherapy. 59 Similarly, in patients with CRC, combining Adagrasib with the anti-EGFR drug Cetuximab resulted in a higher ORR (43%-46%) compared with Adagrasib alone (19%-50%). These findings suggest that combination therapies may offer a more robust clinical benefit, particularly in cancer types where monotherapy shows limited efficacy. In addition, while the safety profiles of these combinations are generally manageable, there is an increased risk of severe side effects, such as hepatotoxicity when Sotorasib is used with anti-PD-(L)1 therapy. Therefore, further extensive trials are needed to confirm these findings and optimize treatment protocols. 36
Our systematic review revealed that Sotorasib and Adagrasib exhibited significant antitumor efficacy in NSCLC, and PDAC patients with KRAS G12C mutation primarily as monotherapy.
In patients with CRC, while there were some responses observed, the overall efficacy was less significant compared with NSCLC and PDAC. The researchers state that although initial data from a study of Adagrasib monotherapy are promising in patients with KRAS G12C-mutated CRC, reactivation may occur in the RAS–MAPK signaling pathway due to epidermal growth factor receptor (EGFR)-mediated adaptive feedback. Preclinical studies have shown that the EGFR blockade may sensitize tumors to KRAS G12C inhibition and the combination of an anti-EGFR antibody (Cetuximab) with a KRAS G12C inhibitor showed greater anti-tumor activity than single-agent KRAS G12C inhibition. 60 New researches suggest that combining KRAS inhibitors with anti-EGFR drugs in CRC may lead to better clinical outcomes. The combination of Adagrasib plus Cetuximab demonstrated a meaningful improvement over Adagrasib monotherapy in a heavily pretreated patient population with KRAS G12C-mutated CRC, resulting in a 43% ORR vs an ORR of 22% for the monotherapy. 8 Our findings confirm the efficacy of this combination revealing that the ORR of 43% to 46% vs an ORR of 19% to 50% for the monotherapy. Hence, combining Cetuximab with Adagrasib may enhance the inhibition of KRAS-dependent signaling or overcome adaptive feedback to delay resistance and improve outcomes. In addition, while Divarasib was not explicitly covered in our results section due to limited data from the selected studies, other studies have shown that Divarasib in combination with Cetuximab demonstrated also a manageable safety profile and promising clinical activity.37,44,47,48 Our systematic review support that the addition of Cetuximab to Adagrasib or Divarasib may lead to robust clinical benefit in patients with CRC KRAS G12C-positive.
The evaluation of the TRAEs resulting from the daily administration of Sotorasib, Adagrasib, Garsorasib, and Divarasib on the quality of life of patients with solid tumors is crucial. In addition, the profile of TRAEs allows for early identification and proactive management, preventing or minimizing potential complications by tailoring treatment decisions to individual patients and promoting a more personalized and targeted approach. Our systematic review showed that TRAEs were similar across all solid cancer types studied. Most patients in the selected clinical trials were experienced TRAEs with grade 1, 2, 3, and rarely with grade 4. The most common TRAEs were nausea, diarrhea, fatigue, vomiting, anemia, ALT, creatinine, and AST increase. Overall, most of the AEs were manageable and the patients tolerated the study treatment well. Even though, another study mentioned that KRAS G12C inhibitor Sotorasib may trigger severe immune-mediated hepatotoxicity when used in sequence or in combination with anti-PD-(L)1 in NSCLC, with a significantly increased risk of severe Sotorasib-related hepatotoxicity and severe non-liver AEs. So it is suggested to avoid starting Sotorasib within 30 days from the last anti-PD-(L)1 infusion. 61 The ongoing assessment of TRAEs contributes to a continuous learning process, leading to the refinement of the treatment protocol and improved patient outcomes over time. Their declaration during clinical trials is important before drug approval and post-marketing surveillance.
The strength of this systematic review is its comprehensiveness; this is the most comprehensive review to date of the studies using Sotorasib, Adagrasib, Garsorasib, and Divarasib as monotherapy, and included also the studies of Adagrasib and Divarasib used in combination with Cetuximab.
The main limitation of the current review is the high proportion of abstracts, in which information is often incomplete and for which peer review may have been less stringent. However, the inclusion of Congress materials allowed us to capture the most up-to-date findings, even if full results have not yet been published, which is important for the fast-moving field of oncology. Furthermore, due to the small sample size of those clinical trials, few studies enroll sufficient patients of a specific age or gender to allow assessment of drug efficacy among these particular subsets of patients. Pooling data from many trials could provide the statistical weight to overcome these limitations and to better inform treatment in the clinic. Finally, it would be advantageous to pool the Disease control, the duration of response, and the overall survival details, to enable further analyses of the outcome.
Conclusions
In conclusion, this extensive systematic review has demonstrated the efficacy and safety of KRAS G12C inhibitors Sotorasib, Adagrasib, Garsorasib, and Divarasib primarily as monotherapy for the treatment of solid tumors, including NSCLC, CRC, and PDAC. The clinical trials analyzed provide robust evidence that Sotorasib and Adagrasib are particularly effective in NSCLC, with consistent real-world data validating their use in clinical settings. For CRC and PDAC, the initial clinical data are promising, showing that these inhibitors have the potential to offer significant therapeutic benefits.
These findings also underscore the importance of personalized treatment strategies, as the efficacy of KRAS G12C inhibitors appears to be influenced by tumor type and previous treatments. This highlights the necessity for ongoing research to optimize dosing, combination strategies, and patient selection criteria to maximize therapeutic outcomes.
In light of these findings, future research should focus on expanding the evidence base for the use of KRAS G12C inhibitors in other tumor types, exploring potential resistance mechanisms, and refining combination therapies to further enhance efficacy. Clinically, this review supports the integration of these inhibitors into treatment protocols, reinforcing their role in improving patient prognosis and quality of life in solid tumors.
Footnotes
Acknowledgements
The authors thank the reviewers and editors for their constructive comments that significantly enhanced the manuscript.
Ethical considerations
Not applicable as this study is a systematic review.
Consent to participate
Not applicable as this study is a systematic review.
Consent for publication
Not applicable as this study is a systematic review.
Author contributions
SEZ and AL conceived the study, exploited data, and coordinated and drafted the paper. SEZ, YB, SB, HEA, RA, MRT, and SEK, participated in the study design. SEZ, EAB, and ILA were involved in data analyses. SEZ, RAEH, and KE reviewed the manuscript. All authors have read and agreed to the version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable as this study is a systematic review.
