Abstract
Background:
Patient-derived tumor cell cluster (PTC)-based drug sensitivity assays show promise for enabling precision drug selection; however, their predictive value in advanced non-small cell lung cancer (NSCLC) requires further elucidation.
Methods:
To assess the concordance between PTC-based drug sensitivity and clinical outcomes, we conducted a single-center prospective cohort study at Peking University Third Hospital from August 2021 to August 2024. We enrolled 38 consecutive patients, from whom 44 fresh tumor specimens were obtained for PTC-based drug sensitivity assays and compared with paired clinical outcomes of chemotherapy and targeted therapy regimens. Concordance was assessed using the Kappa statistic and receiver operating characteristic curve analysis. A Cox proportional-hazard model was used to identify prognostic factors for progression-free survival (PFS).
Results:
We observed an 81.8% (36/44) concordance between the PTC killing rate (>0% vs 0%) and the best overall clinical response (disease controlled vs disease progression per RECIST v1.1) (Kappa = 0.484, area under the curve (AUC) = 0.740). The concordance with the local response of the biopsied lesions was even higher, at 87.5% (35/40) (Kappa = 0.593, AUC = 0.841). A PTC killing rate >0% was correlated with significantly longer PFS (8.6 vs 2.0 months, P < .001) and emerged as an independent predictor of PFS in multivariate analysis (hazard ratio (HR) 3.507, 95% confidence interval (CI) 1.289-9.536). Exploratory subgroup analysis revealed concordance rates of 73.7% (14/19) for malignant effusion-derived PTCs and 88.0% (22/25) for tumor tissue-derived PTCs; 95.2% (20/21) for first-line therapy and 69.6% (16/23) for later-line therapies; and 80.0% (12/15) for targeted therapy and 85.2% (23/27) for chemotherapy. Notably, the concordance rate reached 100% (14/14) in patients receiving chemotherapy plus immune checkpoint inhibitors.
Conclusions:
These findings validate the predictive value of the PTC-based drug sensitivity assay in guiding personalized treatment for patients with advanced NSCLC and support its clinical translation.
Registration:
Chinese Clinical Trial Registry, Registration No.: ChiCTR2100048791, https://www.chictr.org.cn/showproj.html?proj=129885.
Keywords
Introduction
Advanced non-small cell lung cancer (NSCLC) is the leading cause of cancer incidence and mortality worldwide. 1 Despite survival improvements from targeted therapy and immunotherapy, the overall prognosis remains poor. 2 Optimal therapeutic outcomes depend on stage-specific precision treatment. However, tumors exhibit interpatient, temporal, and spatial heterogeneity, while the diversity of antitumor drugs and combinations necessitates robust methods for personalized selection and efficacy prediction.
In vitro tumor models, including 2-dimensional (2D) cell lines, 3-dimensional (3D) cultures, and patient-derived xenografts (PDXs), are used to guide individualized precision therapies. Each model has limitations. For example, long-term culture can alter the pathological and genetic features of tumor cell lines, and PDXs do not fully recapitulate the tumor microenvironment. 3 Currently, 3D cultures models—such as Matrigel-based patient-derived organoids (PDOs), suspension tumor-like spheroids, organoid-on-a-chip microfluidics system, and air-liquid interface (ALI) organoids—are considered superior for preserving the molecular, genetic, epigenetic, and pathological characteristics of the original tumor.3-6
This study used a 3D suspension culture model of patient-derived tumor cell clusters (PTCs) in low-adhesion plates. In this system, tumor cells, fibroblasts, and immune cells self-assemble into multicellular spheroids (40-300 µm in diameter) that mimic the composition of the parental tumor. The PTC model has been successfully established for various cancers, including gastric, colorectal, breast, lung, gynecological, and urothelial cancer.7,8 It offers high standardization, a short culture time (2-7 days), a high success rate (>85%), and low cost, making it suitable for broad clinical application. A national expert consensus has facilitated the rapid and standardized development of this technique in China. 3
We previously established a drug sensitivity testing system using this lung cancer PTC model (Figure 1). We achieved 81% PTC culture success rate from fresh tumor biopsies and malignant effusions, preserving the histology and genetics of the parental tumors. 8 Drug sensitivity was assessed using a defined panel of anticancer drug concentration, with cell viability measured by an artificial intelligence-based image recognition system. While our preliminary data showed a quantitative concordance with clinical outcomes, 8 predictive accuracy of this in vitro assay requires further clinical validation.

Study flow diagram illustrating lung cancer PTC culture and cell viability assessment.
Therefore, we conducted this single-blind, prospective cohort study to evaluate the predictive value of the PTC-based drug sensitivity assay in patients with advanced NSCLC by assessing the concordance between in vitro responses and clinical outcomes.
Materials and Methods
Study design and participants
This prospective cohort study consecutively enrolled patients with advanced NSCLC treated at Peking University Third Hospital between August 2021 and December 2023, with follow-up concluding on August 1, 2024. The study was reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) Statement. 9
The inclusion criteria were as follows: (1) histologically confirmed locally advanced or metastatic NSCLC; (2) planned to receive chemotherapy or targeted therapy; (3) aged ⩾ 18; (4) an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0-2 with adequate organ function (e.g., bone marrow, hepatic, renal, cardiac, pulmonary). An ECOG PS of 3 was permitted for patients receiving single-agent targeted therapy. Both treatment-naïve and pretreated patients were eligible, if their clinical response was evaluable per Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1). Fresh tumor specimens had to be safely obtained and sufficient for culture.
Exclusion criteria included: (1) insufficient fresh tumor specimens; (2) treatment regimens other than chemotherapy or targeted therapy; (3) uncontrolled comorbidities (e.g. unstable angina, recent myocardial infarction, uncontrolled hypertension, diabetes, or arrhythmias); (4) active infectious diseases; (5) cognitive or psychiatric disorders affecting compliance; or (6) pregnancy or lactation. Criteria for early study termination included voluntary withdrawal, PTC culture failure, or an unevaluable treatment response.
All enrolled patients provided fresh tumor specimens for immediate PTC culture. On successful PTC establishment, in vitro drug sensitivity testing for chemotherapy and/or targeted therapy was promptly performed, corresponding to the patient’s planned clinical regimen. The current in vitro PTC assay cannot evaluate the efficacy of antiangiogenic agents or immune checkpoint inhibitors (ICIs). Tested regimens, including chemotherapy and targeted therapy, were selected by physicians according to clinical guidelines (irrespective of concurrent antiangiogenic agents or ICIs).2,10
Medical information for all eligible participants was collected, including age, sex, clinical stage (American Joint Committee on Cancer [AJCC] 8th edition), genetic alterations, biopsy site, and clinical regimen. Patients were followed up via clinical visit or telephone every 8 weeks until disease progression, death, or the study cutoff date of August 1, 2024. Chest computed tomography (CT) scan, and abdominal ultrasound or CT scan were regularly assessed during each follow-up. In this single-blind study, physicians remained blinded to the PTC results during evaluation, and laboratory staff were blinded to clinical outcomes.
Lung cancer PTC culture and drug sensitivity assay
Fresh tumor samples were collected via surgical resection, endoscopic or needle biopsy, or malignant effusion drainage. Tissue specimens were placed in a preservation solution within 30 minutes of excision, while malignant effusions were collected in sterile drainage bags. All samples were transported at 2°C to 8°C and processed within 24 hours to generate single-cell suspensions. Tissues were mechanically dissociated using a proprietary solution to yield single-cell suspensions. For effusions, cells were pelleted by centrifugation, resuspended in phosphate buffered saline, layered over a lymphocyte separation medium (Ficoll) and centrifuged again. The interphase layer was collected, resuspended in culture medium, filtered through a 100-µm strainer, and centrifuged once more before final resuspension. Detailed methods were described in Supplementary Material Appendix C.
Lung cancer PTC models were established using a patented method (Patent No.: CN113755441B) (Supplementary Material Appendix D). Single-cell suspensions (10⁵ cells/cm2) were seeded into low-adhesion plates. Within 2 to 7 days, cells self-assembled into 3D spheroids (40-300 μm in diameter, regular/irregular morphology, uniform texture, semi-transparent) with ⩾20% tumor cell composition. Cultures that failed to form spheroids within 7 days were considered unsuccessful. Following 3D spheroid formation, cell viability was assessed every 2 to 3 days using the CellTiter-Glo luminescent assay kit (Supplementary Material Appendix E). Drug sensitivity testing required ⩾90% viability and was conducted within 1 week of spheroid formation.
For drug sensitivity testing, spheroids ⩾40 μm were plated (30-50 per well in 96-well low-adhesion plates, in triplicate) and exposed to predetermined antitumor drug concentrations (from prior study) 8 with drug-free wells serving as negative controls. Spheroid morphology was imaged (at ⩾40× magnification) before treatment (t₀) and 7 days after treatment (t₁). An image recognition system calculated the spheroid area ratio (PAi) using the following formula:
SAi = total area per well; n = replicates; t₀ = pre-treatment; t₁ = post-treatment.
The tumor killing rate (TKR) was derived as TKR = 1 – PAi. Cytotoxicity (sensitivity) was defined as a reduction in PTC viable area (0 ⩽ PAi < 1, corresponding to a TKR from >0%), whereas no reduction (PAi ⩾ 1, TKR = 0%) was considered resistance. The test was considered invalid if the control PAi was <0.9 on day 7, indicating a decline in culture viability.
Statistical analysis
Sample size was calculated with the following formula:
n = sample size; α = confidence level; Z = Z-score from the standard normal distribution; p = estimate proportion; δ = margin of error.
Based on prior data showing 82.9% to
The primary endpoints were the PTC-derived TKR, clinical response per RECIST v1.1, and progression-free survival (PFS), defined as the time from treatment initiation to disease progression or death. For measurable lesions, clinical response was categorized as complete remission (CR), partial remission (PR), stable disease (SD), or progressive disease (PD); non-measurable lesions were classified as CR, non-CR/non-PD, or PD.
The primary objective was to evaluate the concordance between PTC drug sensitivity and clinical outcomes. Exploratory subgroup analyses were performed based on treatment modality and PTC source. Statistical analyses included the Kappa statistic, receiver operating characteristic (ROC) analysis, and calculation of the area under the curve (AUC). The secondary objective was to assess the predictive value of PTC drug sensitivity for PFS using Kaplan-Meier analysis, Log-rank tests, and Cox regression analysis. All analyses were performed using SPSS 26.0.
Results
Patient characteristics
From August 2021 to December 2023, 66 consecutive patients were screened, yielding 75 specimens. The PTC culture success rate was 85.3% (64/75), with an average of 8.95 assays (range:

Flow chart of patient enrollment.
The clinical characteristics of 38 patients were as follows: the median age was 63 years (range:
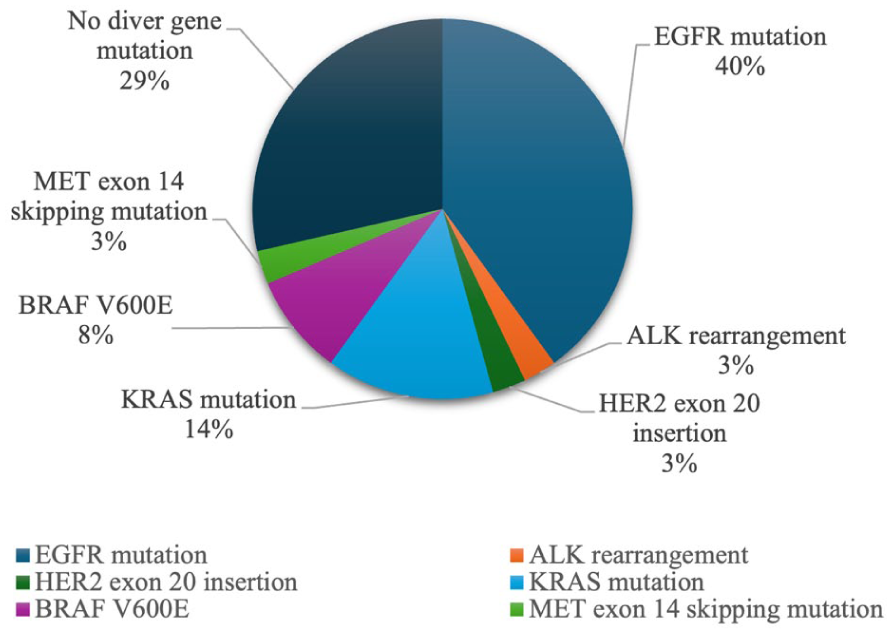
Genetic alterations detected by Next-Generation Sequencing (NGSa).
The PTC specimens were most frequently derived from malignant effusions (43.2%, 19/44), superficial lymph nodes (25.0%, 11/44), and pulmonary lesions (13.6%, 6/44) (Table 1). At the time of specimen collection, 47.7% (21/44) patients planned to receive first-line treatment, 22.7% (10/44) were second-line, and 29.5% (13/44) were third-line or later. Regimens tested in this study included targeted therapy for driver-gene mutations, chemotherapy, and its combinations with antiangiogenic therapy or ICIs (programmed cell death protein 1 [PD-1] or its ligand 1 [PD-L1] inhibitors). Notably, 2 squamous cell lung cancer patients with unknown EGFR status received afatinib per clinical guidelines.2,10 Some patients also received local therapy, such as intracavitary infusion for malignant effusions (22.7%, 10/44), or surgery or radiotherapy for bone metastasis (6.8%, 3/44).
Sites of PTC sampling and anticancer treatment.

ICIs, immune checkpoint inhibitors; PTC, Patient-derived tumor cell cluster.
Concordance between PTC drug sensitivity and clinical response
We first evaluated concordance using a TKR cutoff of ⩾30% to define sensitivity, mirroring the RECIST v1.1 threshold for partial response. This yielded a modest concordance rate of 61.4% (27/44, Kappa = 0.194, AUC = 0.666) between the PTC drug sensitivity assay and overall clinical response (response [CR + PR] vs non-response [SD + PD]) (Figure 4A). However, the optimal Youden index (YI = 0.346) was achieved at a TKR threshold of 2%, which outperformed the 30% cutoff (YI = 0.192).

Prediction ability of the PTC cytotoxicity rate for clinical outcomes. (A) the ROC curve for predicting overall response rate (CR + PR) using a PTC cytotoxicity rate cutoff of ⩾30%, (B) ROC curve for predicting non-PD using a PTC cytotoxicity rate cutoff of >0%, (C) ROC curve for predicting non-PD in local efficacy at the PTC biopsy site using a PTC cytotoxicity rate cutoff of >0% and (D) Kaplan-Meier curves for PFS stratified by PTC drug cytotoxicity rate (>0% vs =0%).
Using this optimized cutoff (TKR > 0% for sensitivity vs TKR = 0% for resistance), the concordance between the PTC drug sensitivity assay and overall clinical response (disease control [CR + PR + SD] vs progression [PD]) improved significantly to 81.8% (36/44), with a Kappa of 0.484 and an AUC of 0.740 (Figure 4B). Among 40 evaluable biopsy sites (excluding 4 sites deemed ineligible due to local therapy or surgery), the concordance between the PTC drug sensitivity assay and the local response of the biopsied lesions was even higher at 87.5% (35/40), with a Kappa of 0.593 and an AUC of 0.841 (Figure 4 C).
Association between PTC drug sensitivity and PFS
A total of 42 paired datasets involving PTC assays and PFS were analyzed. If a patient provided PTC specimens from 2 lesions simultaneously, it was counted as 1 PTC-drug-PFS pair (involving 2 patients); whereas if a patient contributed 2 PTC specimens at different time points before different treatment regimens, these were counted as 2 separate PTC-drug-PFS pairs (4 instances). Patients with a drug-sensitive PTC profile (TKR > 0%) had a significantly longer median PFS than those with a resistant profile (TKR = 0%): 8.6 months (95% CI: 4.
In univariate analysis, both PTC drug sensitivity (hazard ratio (HR) 4.912, 95% CI: 1.
Univariate and multivariate analysis of PFS.

CI, confidence interval; HR, hazard ratio; PFS, progression-free survival; PTC, Patient-derived tumor cell cluster.
Exploratory analysis of specific subgroups
Exploratory analyses showed varying concordance rates across different subgroups (between PTC TKR > 0% and overall response). Concordance was higher for PTCs derived from tumor tissues (88.0%, 22/25) than from malignant effusions (73.7%, 14/19), and for first-line therapy (95.2%, 20/21) compared to later-line therapy (69.6%, 16/23). Concordance was similar for targeted therapy for driver-gene mutations (80.0%, 12/15) and for chemotherapy with or without combinations with antiangiogenic agents or ICIs (85.2%, 23/27). Notably, in patients receiving chemotherapy plus ICI, the concordance rate reached 100% (14/14).
Representative cases
Case 1 (Patient ID: 202403186601): A 75-year-old female with stage IVB (T4NxM1c, AJCC 8th edition) right lung adenocarcinoma harboring an EGFR L858R mutation developed resistance to Osimertinib, presenting with new onset right-sided back pain. Subsequent imaging revealed a chest wall metastasis at the site of the pain. A core needle biopsy of this lesion demonstrated MET amplification (copy number variation [CNV] = 6) alongside the persistent EGFR L858R mutation. The patient was subsequently treated with a combination of almonertinib (a third-generation EGFR-TKI) and crizotinib (a MET inhibitor), which resulted in rapid symptomatic relief of her back pain. A follow-up CT scan at 4 weeks showed significant regression of the chest wall metastasis (Figure 5A, upper panel). This clinical response correlated with the PTC-based drug sensitivity assay, which had shown a 41% TKR for the almonertinib and crizotinib combination (Figure 5A, middle panel).

Representative cases demonstrating concordance between clinical efficacy and PTC drug sensitivity results. The upper panels show pre- and post-treatment computed tomography scans of the target lesions, which were also the sites for PTC biopsy. The middle panels display the corresponding pre- and post-treatment PTC cultures, with drug-free negative control shown in the lower panels. (A) A right chest wall metastasis in a patient with EGFR L858R mutant lung adenocarcinoma. The chest wall metastases, which was resistant to Osimertinib due to secondary MET amplification (CNV = 6), was sensitive to the almonertinib plus crizotinib combination in both the clinical evaluation and the PTC drug sensitivity assay. (B) The primary lung lesion and (C) liver metastases in a patient with squamous cell lung cancer. Both lesions were clinically sensitive to combination therapy with albumin-bound paclitaxel, carboplatin, and a PD-1 inhibitor, and the PTC drug sensitivity assays demonstrated sensitivity to the albumin-bound paclitaxel and carboplatin combination.
Case 2 (Patient ID: 20231121899): A 72-year-old male presented with stage IVB (T2N2M1c, AJCC 8th edition) squamous cell carcinoma of the right lung with hepatic metastases. First-line therapy with albumin-bound paclitaxel, carboplatin, and a PD-1 inhibitor resulted in partial response (PR) after 4 cycles, according to RECIST v1.1 criteria. Concurrent regression was observed in both the primary lung lesion and the hepatic metastases (Figure 5B and C, upper panels). Interestingly, differential drug sensitivity was noted between the 2 lesions. The PTC-based drug sensitivity assays demonstrated a 24% TKR for the lung biopsy specimen and a 4% TKR for the liver biopsy specimen, in response to the albumin-bound paclitaxel and carboplatin combination (Figure 5B and C, middle panels). It is possible that the cytotoxic effect of chemotherapy was enhanced by the addition of the PD-1 inhibitor.
Discussion
This study further validated the concordance between the patient-derived tumor cell cluster (PTC)-based drug sensitivity assay and clinical outcomes in patients with advanced NSCLC, thereby demonstrating its potential clinical utility. The overall success rate of PTC culture and the time required for cultivation from various NSCLC specimens were consistent with previously reported data. On average, each specimen could generate sufficient PTCs for 9 drug sensitivity tests.
The concordance rate between PTC drug sensitivity and disease control (defined as CR, PR, or SD per RECIST v1.1) was 81.8%, while the concordance with local efficacy at the PTC biopsy site was even higher, at 87.5%. A significant difference in PFS was observed between patients with drug-sensitive versus drug-resistant PTC profiles, suggesting the strong predictive and prognostic value of this assay. In addition, our preliminary observations indicated that PTCs derived from solid tumor tissues exhibited higher concordance with clinical efficacy than those derived from malignant effusions. The assay also showed a higher concordance with the efficacy of first-line therapy and was particularly well-correlated with the efficacy of targeted therapy for known driver-gene mutations.
A crucial aspect of this study was determining an appropriate cutoff value for cytotoxicity, as the assay employed fixed drug concentrations. We reviewed a previous validation dataset used for establishing PTC models in NSCLC, which included 82 paired data from 51 patients. 8 In that dataset, a PTC sensitivity cutoff value of ⩾30% was adopted to correspond with the RECIST v1.1-defined “effective” group (CR/PR), yielded a concordance rate of 89%, with most discrepancies occurring in patients with stable disease (SD). However, applying this same standard in the present study yielded a concordance rate of only 61.4%, which remained low (62.5%) even when restricted to patients with measurable disease.
Given that the primary treatment goal in advanced NSCLC is often to delay disease progression, any regimen capable of reducing PTC viability in vitro should be considered beneficial. A high sensitivity cutoff might lead to missing valuable treatment opportunities. Therefore, we ultimately set the PTC cytotoxicity cutoff at >0%, which significantly improved the concordance with clinical disease control (CR/PR/SD) and showed a strong correlation with PFS.
Despite this adjustment, discrepancies remained. Eight patients exhibited discordance between PTC drug sensitivity and clinical efficacy. In 5 of these cases, the PTC drug sensitivity results were also discordant with the local efficacy at the biopsy site. These discrepancies may be partially attributable to the inherent limitation of the PTC drug sensitivity assay in assessing the efficacy of antiangiogenic drugs and ICIs. Nevertheless, clinical guidelines recommend combining these agents with chemotherapy to improve efficacy, and 53.5% (23/43) of patients in our study received such combinations. Furthermore, variations in the route and method of drug administration, such as intracavitary perfusion or metronomic dosing, could also impact efficacy. In addition, tumor heterogeneity across different metastases sites poses a challenge, as a single-lesion biopsy may not accurately reflect the systemic drug response.
Other in vitro 3-dimensional culture models for lung cancer drug sensitivity testing include PDOs and microfluidic organ-on-a-chip systems. Patient-derived organoids require a Matrigel-based extracellular matrix and various supporting scaffolds; however, the lack of standardized protocols, a relatively long culture time (
Several limitations of this study should be noted. First, as a pilot study with a limited sample size, the results require further validation in larger cohorts. Second, the predictive value of PTC drug sensitivity assays for ICIs remains unclear and warrants further investigation. Third, the representativeness of a single-biopsy-derived PTC culture is limited by both temporal and spatial tumor heterogeneity. The use of circulating tumor cells to establish cultures might offer a more comprehensive representation of the disease. 17
Conclusion
To our knowledge, this is the first prospective cohort study to evaluate the clinical predictive value of patient-derived tumor cell cluster (PTC)-based in vitro drug sensitivity testing in advanced NSCLC. We achieved a high culture success rate, irrespective of the sample type, including fresh tumor tissue and malignant effusion samples. The study demonstrated a strong concordance between PTC-based drug sensitivity results and the systemic clinical response to anticancer therapies—including chemotherapy and targeted therapy for driver-gene mutations—with or without the addition of antiangiogenic agents and ICIs. Notably, the PTC-based drug sensitivity assay showed even higher concordance with local tumor response at the biopsy site, underscoring its robust predictive value. These findings warrant further validation of the PTC-based drug sensitivity assay, as a clinically feasible alternative to existing in vitro drug sensitivity models. Coupled with its standardized time-efficient culture procedure, PTC-based drug sensitivity assay has important implications for the development of personalized treatment strategies in NSCLC.
Supplemental Material
sj-pdf-1-onc-10.1177_11795549251405740 – Supplemental material for Predictive Value of Patient-Derived Tumor Cell Cluster–Based Drug Sensitivity Assay in Advanced NSCLC
Supplemental material, sj-pdf-1-onc-10.1177_11795549251405740 for Predictive Value of Patient-Derived Tumor Cell Cluster–Based Drug Sensitivity Assay in Advanced NSCLC by Yangchun Gu, Leilei Yang, Hua Zhang, Shenyi Yin, Lu Yang, Zhentao Liu, Jinyu Yu, Huiying Huang, Juan Li and Baoshan Cao in Clinical Medicine Insights: Oncology
Supplemental Material
sj-pdf-2-onc-10.1177_11795549251405740 – Supplemental material for Predictive Value of Patient-Derived Tumor Cell Cluster–Based Drug Sensitivity Assay in Advanced NSCLC
Supplemental material, sj-pdf-2-onc-10.1177_11795549251405740 for Predictive Value of Patient-Derived Tumor Cell Cluster–Based Drug Sensitivity Assay in Advanced NSCLC by Yangchun Gu, Leilei Yang, Hua Zhang, Shenyi Yin, Lu Yang, Zhentao Liu, Jinyu Yu, Huiying Huang, Juan Li and Baoshan Cao in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
None.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki (2013 revision) and was approved by the Medical Science Research Ethics Committee of Peking University Third Hospital (Project ID: M2021171; Ethics Approval No.: 2021-219-02)
Author contributions
YG contributed to the conceptualization, data curation, and project administration of the study; conducted the validation, formal analysis, and interpretation of the data; wrote the original draft of the manuscript; and developed the figures and tables. LY contributed to the data curation, validation, analysis, and the review of the manuscript. HZ contributed to the conceptualization, methodology, and critical review of the manuscript. SY contributed to the conceptualization, methodology, data curation, project administration, funding acquisition, and the review of the manuscript. LY, ZL, JY, and HH contributed to the data curation, project administration, and the review of the manuscript. JL contributed to the conceptualization, funding acquisition, supervision of the study, and the review of the manuscript. BC contributed to the conceptualization, funding acquisition, investigation, methodology, project administration, and supervision of the study; conducted the validation, analysis, and interpretation of the data; and critically reviewed the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (Grant No: 2020AAA0105203). This study also received support from the Natural Science Foundation of Beijing Municipality (Grant No: 5244030) and the Beijing Municipal Science and Technology Commission, Administrative Commission of Zhongguancun Science Park (Grant No: Z231100007223002).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
De-identified participant data will be made available to scientists 12 months after the publication of this manuscript. Requests should be addressed to
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


