Abstract
Background:
Myelosuppression is a frequent complication in patients with nasopharyngeal carcinoma (NPC) undergoing chemoradiotherapy. Current clinical practice relies predominantly on treatment-phase monitoring for myelosuppression risk assessment, while effective pretreatment prediction tools are lacking. This study developed a predictive model based on pretreatment clinical indicators to facilitate early identification of high-risk patients and support clinical decision-making.
Methods:
We conducted a retrospective cohort study using electronic medical records of 210 patients with NPC who received chemoradiotherapy at the First Affiliated Hospital of Bengbu Medical University between May 2016 and December 2021. Using R software, patients were randomly allocated into a training set (n = 150) and an internal validation set (n = 60) at a 7:3 ratio. Variable selection was performed using Least Absolute Shrinkage and Selection Operator regression, followed by univariable and multivariable logistic regression analyses to identify potential predictors. Following categorization of these identified potential predictors, Firth penalized-likelihood regression was employed to correct for small-sample bias, while multicollinearity was rigorously assessed using variance inflation factors (VIFs). A predictive nomogram was subsequently constructed. Model performance was evaluated through multiple validation metrics, including the concordance index (C-index), receiver operating characteristic curve analysis, clinical decision curve analysis, and calibration curve.
Results:
Multivariable logistic regression analysis identified 3 potential predictors of myelosuppression: pretreatment plateletcrit (PCT), direct bilirubin (DBIL), and sodium ions (Na+) (all P < .05). All these potential predictors met strict stability criteria after conversion to categorical variables (all VIF < 2.1, with a predefined threshold of VIF < 5). Model evaluation demonstrates that the developed nomogram exhibits favorable predictive performance.
Conclusion:
Pretreatment PCT, DBIL, and Na+ may serve as potential predictors of myelosuppression in patients with NPC undergoing chemoradiotherapy. This nomogram could serve as a risk stratification tool to identify high-risk patients before treatment, enabling early interventions for myelosuppression prevention.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a kind of cancer of squamous cells that emerges in the nasopharynx’s epithelial tissues. 1 Nasopharyngeal carcinoma is somewhat uncommon in comparison with other cancer types, but it exhibits distinct geographic distribution patterns, with East and Southeast Asia accounting for more than 70% of newly reported cases. 2 Due to its insidious onset and aggressive biological behavior, approximately 70% of patients with NPC are diagnosed with advanced-stage disease at initial presentation. Nevertheless, contemporary chemoradiotherapy regimens have remarkably improved outcomes, achieving 5-year survival rates approaching 80% even in locally advanced cases. 3
Despite these therapeutic advances through intensity-modulated radiotherapy and optimized chemotherapy, 4 treatment-related myelosuppression remains a major clinical challenge. Defined as hematologic toxicity resulting from impaired bone marrow function, myelosuppression manifests as reductions in erythrocytes, leukocytes, and thrombocytes. 5 As the common hematologic complication of chemoradiotherapy, myelosuppression not only causes immunosuppression but often requires treatment interruptions or dose reductions, significantly compromising therapeutic efficacy. 6 Consequently, developing a risk prediction model for myelosuppression holds significant clinical importance for optimizing treatment strategies in patients with NPC.
Currently, although few studies have specifically examined correlative factors for chemoradiotherapy-induced myelosuppression in patients with NPC, evidence from other malignancies receiving similar regimens provides insights. A recent study identified multiple risk factors for chemotherapy-induced myelosuppression in esophageal cancer, including treatment regimen, concomitant medications, demographic characteristics (sex, age), hematological parameters (hemoglobin [Hb], platelet count [PLT], and red blood cell [RBC] counts), renal function markers (Cr, blood urea nitrogen, and urinary protein), and heart function indicators (CK). 7 Pelvic irradiation doses of 10 to 20 Gy have been identified as a significant risk factor for myelosuppression and subsequent hematologic toxicity in patients with cervical cancer undergoing radiotherapy or chemoradiotherapy. 8 Although these tumors differ anatomically from NPC, the findings still provide valuable reference insights. In NPC specifically, PD-1 blockade response and Linc00312 single-nucleotide polymorphism serve as distinct biomarkers for therapeutic efficacy and chemoradiotherapy-induced hematological adverse events, respectively, in patients with NPC undergoing chemoradiotherapy.9,10 However, the risk prediction model for chemoradiotherapy-induced myelosuppression in patients with NPC remains undefined, and a validated risk assessment tool for this population is lacking. Therefore, exploring potential predictors based on clinical data may facilitate the development of more effective risk prediction models for chemoradiotherapy-induced myelosuppression in patients with NPC.
Risk prediction models, which integrate multiple variables to predict individual outcomes or risks, are valuable tools in clinical practice. 11 Currently, most existing risk prediction models for NPC are primarily based on radiomic features or biomarkers, focusing on diagnostic and prognostic applications.12-14 Existing models demonstrate limited capacity to facilitate timely treatment strategy modifications for patients with NPC in clinical practice. Therefore, developing and validating a clinically applicable prediction model for chemoradiotherapy-induced myelosuppression in patients with NPC is imperative.
This study developed and validated a comprehensive risk prediction model to identify patients with NPC at high risk of developing chemoradiotherapy-induced myelosuppression. This model may facilitate timely prophylactic interventions and potentially reduce the incidence of treatment-related myelosuppression.
Materials and Methods
Study design and population
This study aimed to develop and validate a nomogram for assessing the risk of myelosuppression in patients with NPC undergoing chemoradiotherapy. This retrospective cohort study collected electronic medical records of 574 patients with NPC diagnosed at the First Affiliated Hospital of Bengbu Medical University between May 2016 and December 2021. Inclusion criteria: (1) individuals who have been pathologically diagnosed with NPC for the first time, (2) patients with no other malignancies in addition to NPC, (3) patients who underwent radiotherapy and chemotherapy, (4) patients with a complete clinical electronic medical record, and (5) completion of hospital database records and follow-up data. Exclusion criteria: (1) presence of other severe underlying diseases, (2) lack of pretreatment data for NPC, (3) significant missing data in the pretreatment clinical records, and (4) presence of severe infectious diseases. Based on these inclusion/exclusion criteria, 210 patients with NPC receiving chemoradiotherapy were ultimately enrolled in the study. The study flowchart is presented in Figure 1. The study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024. This study follows the STROBE guidelines 15 (Supplementary File).

Flow chart of this study.
Data collation
The clinical data were systematically compiled into a standardized Excel database. Samples exceeding a 15% threshold for missing data were excluded from subsequent analyses to ensure data quality. For continuous variables with limited missing values (⩽15%), missing data were imputed using the mean value of the respective variable to maintain dataset integrity. Based on pre-established inclusion and exclusion criteria, we performed a comprehensive retrospective analysis of clinical data from 210 patients with NPC who underwent chemoradiotherapy. The included clinical indicators encompassed gender, age, pretreatment hematological profiles (complete blood count [CBC]), biochemical indicators, and systemic inflammatory markers, such as neutrophil-to-lymphocyte ratio (NLR). Categorical variables were assigned numerical codes according to predefined classification schemes: gender was binary-coded with male = 0 and female = 1.
Variable definitions, measurement methods, and diagnostic criteria
All clinical indicators were evaluated in accordance with World Health Organization (WHO) standards. Demographic variables included gender (categorized as male or female based on medical records) and age (recorded as a continuous variable in years at diagnosis). Pretreatment hematological profiles were obtained from CBC analysis performed within 7 days prior to treatment initiation using automated hematology analyzers. Biochemical indicators were measured from venous blood samples collected during the same period, with serum separated by centrifugation and analyzed using clinical chemistry analyzers following International Federation of Clinical Chemistry (IFCC) standard methods. All blood samples were processed within 2 hours of collection. Systemic inflammatory markers such as NLR were calculated as the absolute neutrophil count divided by the absolute lymphocyte count from pretreatment CBC results. All laboratory testing was performed in our institutional clinical laboratory following standardized operating procedures.
Furthermore, during chemoradiotherapy for patients with NPC, the diagnosis of myelosuppression strictly adhered to the WHO grading criteria, as detailed in Table 1.
Grading criteria for myelosuppression.

Abbreviations: HGB, hemoglobin; NEUT, neutrophil count; PLT, platelet count; WBC, white blood cell count.
Randomization and feature selection using least absolute shrinkage and selection operator regression
Using R statistical software (version 4.3.3; R Foundation for Statistical Computing, Vienna, Austria), the cohort of 210 patients with NPC undergoing chemoradiotherapy was randomly partitioned into a training set (n = 150) and an internal validation set (n = 60) at a 7:3 ratio. A balance test was performed to assess the comparability between the 2 groups.
Feature selection was performed on the training set using the Least Absolute Shrinkage and Selection Operator (LASSO), a robust machine learning method that simultaneously performs variable selection and regularization to enhance model generalizability while preventing overfitting.
Univariable and multivariable logistic regression for predictive modeling
In the training cohort (n = 150), features selected by LASSO regression were analyzed using univariable logistic regression. Statistically significant variables (P < .05) were included in multivariable logistic regression to identify independent predictors of myelosuppression (retained at P < .05). To enhance the interpretability of the model, these predictors were converted into categorical variables for constructing a clinical nomogram to facilitate individualized risk stratification.
Model optimization and diagnostics
These continuous variables used for modeling were converted into categorical variables and encoded as dummy variables, with the lowest-risk group (eg, PCT ⩽ 0.20) designated as the reference group. All dummy variables for the reference group were coded as 0; if a patient belonged to a nonreference group, the corresponding dummy variable was assigned a value of 1, whereas others were set to 0. All categories were retained in the model to maintain structural integrity. To address small-sample bias and data separation, we implemented Firth penalized-likelihood regression for robust parameter estimation. In addition, multicollinearity was rigorously assessed using variance inflation factors (VIF).
Model performance evaluation and internal validation
The model’s predictive performance was comprehensively assessed through multiple validation approaches. Discriminative ability was evaluated using the concordance index (C-index), while predictive accuracy was evaluated through receiver operating characteristic (ROC) curve analysis. Clinical effectiveness was estimated using decision curve analysis (DCA), while the calibration of the model was examined using calibration curve (CC). In addition, internal validation was performed using the validation cohort to further evaluate the robustness of the model.
Statistical methods
Statistical analyses were performed using SPSS software (version 27.0; IBM Corporation, Armonk, New York). All continuous variables were tested for normality and homogeneity of variance. For continuous variables exhibiting normal distribution and homogeneity of variance, between-group comparisons were conducted using independent samples t-tests, with outcomes reported as mean ± standard deviation (SD). For non-normally distributed variables, nonparametric analyses were performed using the Wilcoxon rank-sum test, with results expressed as median (P25, P75). Categorical variables were analyzed using Pearson χ2 test. Univariable and multivariable logistic regression analyses were performed on clinical predictors to identify independent predictors associated with myelosuppression in patients with NPC undergoing chemoradiotherapy. LASSO regression, Firth penalized-likelihood regression, VIF calculation, risk prediction nomogram construction, and model evaluation were conducted using R 4.3.3. The model was assessed using 4 methods: C-index, ROC, DCA, and CC. Internal validation was also performed. A statistically significant difference is shown by a P value of <.05.
Results
Clinical characteristics of the patients
We retrospectively collected electronic medical records of 574 patients with NPC, and finally enrolled 210 eligible cases based on the predefined inclusion and exclusion criteria. All 210 enrolled patients with NPC received chemoradiotherapy. During the treatment, 115 cases (54.76%) developed myelosuppression, whereas 95 cases (45.24%) did not. The cohort included 157 males (74.76%) and 53 females (25.24%), with a median age of 52.42 years (range: 47-60.75 years). Table 2 displays the baseline demographic and pretreatment clinical characteristics of the 210 patients with NPC who underwent chemoradiotherapy.
Baseline data for NPC chemoradiotherapy patients.

Abbreviations: AG, anion gap; AGR, albumin globulin ratio; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; Apo(a), apolipoprotein(a); Apo(b), apolipoprotein(b); AST, aspartate transferase; BASO, basophil count; BASO%, basophil percentage; BUN, blood urea nitrogen; Ca, total calcium ions; Ca2+, ionized calcium; CAR, c-reactive protein to albumin ratio; ChE, cholinesterase; Cl−, chloride ions; Cr, creatinine; CRP, C reactive protein; Cys-C, cystatin C; DBIL, direct bilirubin; EOS, eosinophil count; EOS%, eosimophil percentage; GLOB, globulin; GLU, glucose; HCO3, bicarbonate; HCT, hematocrit; HDL, high density lipoprotein; HGB, hemoglobin; IBIL, indirect bilirubin; K+, potassium ions; LDL, low density lipoprotein; LMR, lymphocyte-monocyte ratio; Lp(a), lipoprotein(a); LYC, lymphocyte count; LYM%, Lymphocyte percentage; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MONO, monocyte count; MONO%, monocyte percentage; MPV, mean platelet volume; Na+, sodium ions; NEUT, neutrophil count; NEUT%, neutrophil percentage; NLR, neutrophil lymphocyte ratio; OP, osmotic pressure; P, inorganic phosphorus; PA, prealbumin; PAR, platelet-albumin ratio; PCT, plateletcrit; PDW, platelet distribution width; P-LCR, platelet-large cell ratio; PLR, platelet-lymphocyte ratio; PLT, platelet count; RBC, red blood cell; RBP, retinol binding protein; RDW-CV, red cell distribution width-coefficient of variation; RDW-SD, red cell distribution width-standard deviation; RET, reticulocyte; RETP, reticulocyte percentage; SOD, superoxide dismutase; TBA, total bile acid; TBIL, total bilirubin; TC, total cholesterol; TG, triglyceride; TP, total protein; UA, uric acid; WBC, white blood cell count; γ-GT, γ-glutamyl transferase.
Balance test and LASSO regression-based feature screening
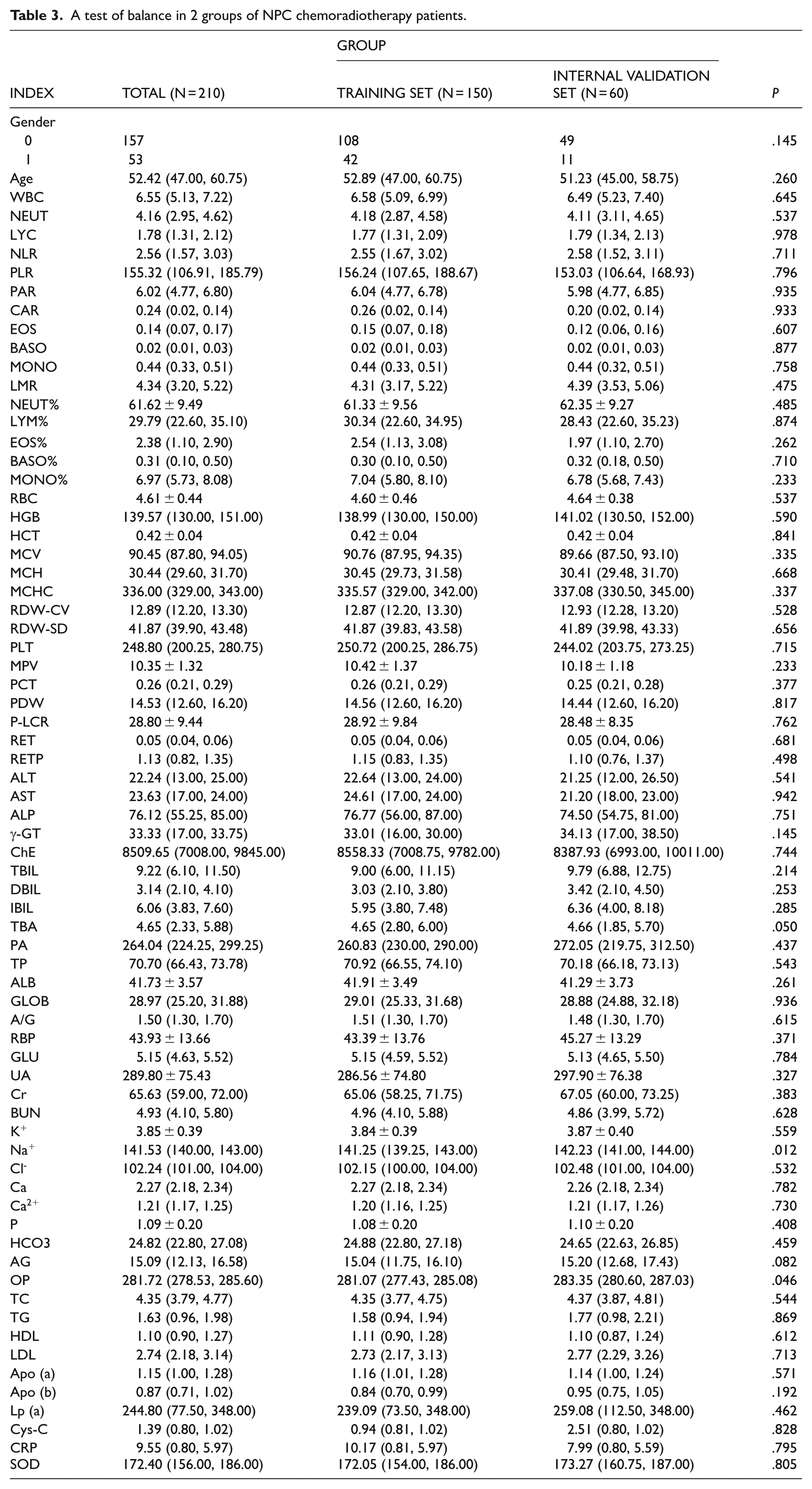
The 210 patients with NPC receiving chemoradiotherapy were randomly allocated into a training set (n = 150) and an internal validation set (n = 60) at a 7:3 ratio. Balance testing of baseline characteristics (all P > .05) confirmed comparability between the 2 cohorts (Table 3).
A test of balance in 2 groups of NPC chemoradiotherapy patients.
To identify the most relevant predictive features, LASSO regression was implemented for initial feature selection across all variables in the training dataset. The analysis demonstrated 2 distinct optimal regularization parameters: Lambda.min and Lambda.lse, each corresponding to different sets of predictive variables. Specifically, Lambda.min identified 8 significant predictive variables, whereas Lambda.lse identified 3 more concise predictor variables (Figure 2A and B). Subsequent logistic regression analysis was performed using the 8 significant predictive variables selected at Lambda.min, comprising: pretreatment RBC count, plateletcrit (PCT), direct bilirubin (DBIL), monocyte percentage (MONO%), basophil count (BASO), sodium ions (Na+), calcium ions (Ca2+), and lipoprotein(a) (Lp(a)).

LASSO regression. (A) LASSO regression model coefficient compression path graph for all predictor variables; (B) LASSO regression cross validation results graph. Eight predictor variables were screened, which were pretreatment RBC, PCT, DBIL, MONO%, BASO, Na+, Ca2+, and Lp(a).
Univariable logistic regression analysis of clinical predictors
Univariable logistic regression was performed on the training set (n = 150) to screen potential myelosuppression predictors. The analysis incorporated myelosuppression occurrence as the dependent variable and the 8 LASSO-selected predictors as independent variables. Our findings revealed that 7 clinical predictors showed statistically significant associations (all P < .05) with myelosuppression risk in chemoradiotherapy-treated patients with NPC: pretreatment RBC (OR = 0.473, 95% CI = [0.227-0.989], P = .047), PCT (OR = 0.008, 95% CI = [0.000-0.843], P = .042), DBIL (OR = 0.648, 95% CI = [0.505-0.831], P = .001), MONO% (OR = 1.305, 95% CI = [1.072-1.589], P = .008), BASO (OR = 0, 95% CI = [0.000-0.000], P = .005), Na+ (OR = 0.885, 95% CI = [0.791-0.989], P = .032), and Lp(a) (OR = 1.003, 95% CI = [1.001-1.005], P = .007) (Table 4).
Univariable logistic regression analysis of myelosuppression in patients with NPC with chemoradiotherapy.

Abbreviations: CI, confidence interval; OR, odds ratio; SE, standard error.
Multivariable logistic regression analysis of clinical predictors
Building upon the univariable analysis findings, we performed multivariable logistic regression analysis on the training cohort comprising 150 patients to identify independent predictors for myelosuppression. The dependent variable was defined as myelosuppression occurrence, while the 7 clinically significant predictors (all P < .05) from the univariable analysis served as independent variables. Multivariable logistic regression analysis revealed 3 significant independent factors predicting myelosuppression risk in this NPC population receiving chemoradiotherapy: pretreatment PCT (OR = 0.001, 95% CI = [0.000-0.243], P = .015), DBIL (OR = 0.728, 95% CI = [0.562-0.944], P = .017), and Na+ (OR = 0.879, 95% CI = [0.774-0.999], P = .048) (Table 5).
Multivariable logistic regression analysis of myelosuppression in patients with NPC with chemoradiotherapy.

Firth penalized-likelihood regression and collinearity diagnostics
Multivariable logistic regression identified 3 potential predictors of myelosuppression in patients with NPC undergoing chemoradiotherapy (all P < .05). After converting these 3 potential predictors into categorical variables and specifying reference groups, Firth penalized-likelihood regression was performed. Furthermore, multicollinearity was strictly assessed using the VIF (Table 6). All variables met the stringent stability criteria (measured VIF values < 2.1; threshold set at VIF < 5).
Results of Firth penalized-likelihood regression and VIF after conversion to categorical variables.

Abbreviation: Ref, reference category.
Development of a risk prediction nomogram model
Based on multivariable logistic regression analysis of predictors in the training cohort of patients with NPC, we identified pretreatment PCT, DBIL, and Na+ as potentially independent predictors, which were subsequently converted to categorical variables and integrated into a risk prediction nomogram (Figure 3). This clinically applicable model was specifically developed to estimate the probability of myelosuppression occurrence in patients with NPC receiving chemoradiotherapy. The formula for this prediction model is as follows:

where P represents probability of myelosuppression occurrence;

The nomogram model for predicting the risk of myelosuppression in patients with NPC with chemoradiotherapy.
Internal evaluation and validation of the risk prediction nomogram model
The model demonstrated robust discriminative ability, with concordance indices (C-indices) of 0.763 and 0.811 for internal evaluation and validation, respectively. ROC curve analysis revealed areas under the curve (AUCs) of 0.7359 and 0.7435 for the training and validation sets, respectively (Figure 4A and B), confirming the model’s excellent predictive accuracy. Decision curve analysis indicated substantial clinical utility, with net benefit ranges of 40% to 90%, 19% to 74%, and 76% to 78% across different probability thresholds (Figure 5A and B). Furthermore, the CCs exhibited good agreement between the CC and the ideal curve (Figure 6A and B).

Comparing the internal evaluation and internal verification area under the ROC curve. (A) Internal evaluation (AUC = 0.7359); (B) internal verification (AUC = 0.7435).
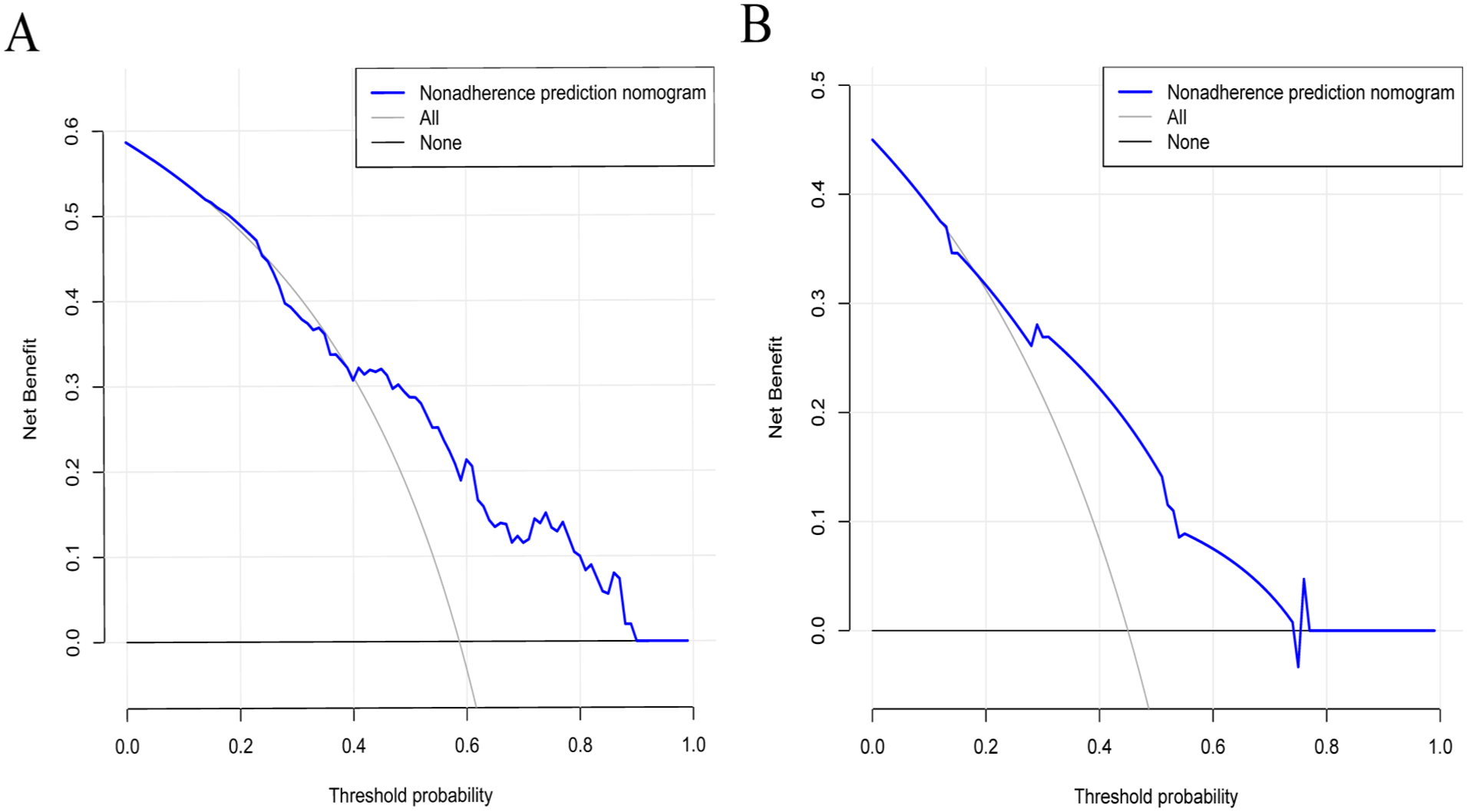
DCA curve of internal evaluation and internal verification. (A) Internal evaluation clinical net benefits of 40% to 90%; (B) internal verification clinical net benefits of 19% to 74% and 76% to 78%.

Calibration curve of internal evaluation and internal verification. (A) The calibration curve from internal evaluation show good agreement with the ideal curve; (B) the calibration curve from internal verification has good agreement with the ideal curve.
Discussion
In recent years, the incidence of NPC has continued to rise due to factors such as tobacco use, dietary patterns, genetic predisposition, and Epstein-Barr virus infection.16-19 With the advancement of medical and healthcare technologies, novel targeted therapies and immunotherapies have provided more options for cancer treatment. However, due to the high costs of these treatments and individual differences among patients, the majority of people still opt for chemoradiotherapy as their treatment regimen. Induction chemotherapy combined with concurrent radiotherapy has been established as the standard treatment for locally advanced NPC, demonstrating high rates of local control and overall survival. 20 Although chemoradiotherapy effectively combat tumor cells, they invariably produce adverse effects as an unavoidable consequence.21-23 Myelosuppression is one of the most common adverse reactions to chemoradiotherapy, which increases risks of bleeding and infection. When myelosuppression occurs during treatment, the standard management approach involves dose reduction or treatment delays—though these interventions may potentially compromise therapeutic efficacy. 24
Currently, the management of myelosuppression primarily relies on monitoring hematologic parameters during treatment. If the risk of myelosuppression could be predicted prior to chemoradiotherapy, it would significantly reduce patient suffering. However, few studies have investigated risk prediction of chemoradiotherapy-induced myelosuppression in patients with NPC. Accordingly, we developed a predictive model to assess the risk of myelosuppression in patients with NPC undergoing chemoradiotherapy, which may facilitate early clinical intervention and timely treatment adjustment to reduce patient suffering.
Previous studies have suggested that myelosuppression may be associated with thrombocytopenia.25,26 However, studies specifically focusing on the role of PCT in predicting myelosuppression are limited. Plateletcrit, calculated as the product of mean platelet volume (MPV) and PLT, serves as a composite index reflecting both quantitative (PLT) and qualitative (MPV) platelet characteristics. Our analysis using Firth penalized-likelihood regression revealed that higher pretreatment PCT levels (groups 3 and 4) were significantly associated with reduced risk of chemoradiotherapy-induced myelosuppression (OR = 0.274 and 0.312, respectively), whereas intermediate PCT levels (group 2) showed no significant effect. This inverse relationship suggests that PCT may serve as a protective marker, potentially reflecting better-preserved hematopoietic reserve prior to treatment. The interdependent relationship between MPV and PLT indicates that PCT offers a more comprehensive assessment of platelet quality than either parameter alone. Therefore, PCT may hold greater value for evaluating hematopoietic function in high-risk patients. The observed protective effect of higher PCT could stem from its dual representation of platelet quantity and size, which might collectively mitigate treatment-related bone marrow damage. One study 27 indicated that thrombopoietin receptor agonists could prevent thrombocytopenia in patients with cancer, with minimal risk when used appropriately. 28 In addition, pretreatment platelet parameters have prognostic value in other cancers. 29 While these findings support the role of platelets in treatment outcomes, our study is the first to identify pretreatment PCT as a specific predictor of myelosuppression in patients with NPC undergoing chemoradiotherapy. The nonlinear association (with significance only at higher PCT ranges) underscores the need for further research to clarify optimal PCT thresholds for clinical decision-making.
Pretreatment DBIL was identified as another potential predictor of myelosuppression in patients with NPC undergoing chemoradiotherapy. Our Firth penalized-likelihood regression analysis revealed a striking dose-response relationship: compared with the reference group (group 1), patients with elevated DBIL levels showed progressively greater protection against myelosuppression—group 3 (OR = 0.241, P = .007), group 4 (OR = 0.276, P = .029), and most markedly in group 5 (OR = 0.102, P < .001). This graded protective association suggests DBIL may serve as a novel biomarker for hematopoietic reserve. Abnormal serum bilirubin levels are recognized as markers of hematological disorders and are linked to the prognosis of numerous malignant tumors. The observed inverse relationship between DBIL levels and myelosuppression risk aligns with existing evidence that DBIL functions as an independent prognostic factor. 30 Our findings extend this understanding by demonstrating its predictive value specifically for chemoradiotherapy toxicity. Direct bilirubin levels are known to relate to tumor progression and response to platinum-based chemotherapy, and nomograms incorporating DBIL outperform TNM staging in outcome prediction. 31 While Zhu et al 32 reported that DBIL > 8.1 μmol/L predicts thrombocytopenia risk, our results identify much lower thresholds (2.7-3.6 μmol/L) as clinically significant for myelosuppression prediction. This discrepancy may reflect differences in study populations or treatment regimens, but consistently supports DBIL’s role in hematological toxicity. Notably, since patients developing myelosuppression often progress to thrombocytopenia, 33 our findings suggest pretreatment DBIL assessment could help identify high-risk patients for closer monitoring or preventive strategies. To our knowledge, this study is the first to identify pretreatment DBIL as a potential predictor for myelosuppression in patients with NPC receiving chemoradiotherapy. The exact role of DBIL in the pathogenesis of myelosuppression requires further investigation.
Moreover, this study found that pretreatment Na+ may serve as a potential predictor of myelosuppression in patients with NPC undergoing chemoradiotherapy. In the stratified analysis, group 3 showed a significant association with reduced risk (OR = 0.313, P = .016), suggesting a protective effect. However, no significant effects were observed in other groups (all P > .05), which might be attributed to insufficient sample size or a true threshold effect. The potential biological plausibility of this association is supported by several lines of evidence. First, Li et al 34 found that sodium-dependent glucose transporters 2 inhibitors (SGLT-2i) may induce pancytopenia and cause myelosuppression. This suggests that Na+ may have a biological association with myelosuppression. Furthermore, previous studies35-37 have established correlations between Na+ levels and both tumor aggressiveness and treatment response across various malignancies. Although the role of Na+ in NPC treatment toxicity has been underexplored, these collective findings warrant further investigation into its potential as a predictive biomarker.
Despite the clinical significance of myelosuppression in patients with NPC receiving chemoradiotherapy, the current literature reveals a paucity of studies investigating risk factors and developing predictive models for this complication. This significantly hinders effective clinical prevention strategies. In this study, using real-world clinical data from the hospital cohort, we identified potential predictors of myelosuppression in patients with NPC undergoing chemoradiotherapy by combining LASSO regression with univariable and multivariable logistic regression analyses. Subsequently, these predictors were transformed into categorical variables to construct a risk prediction nomogram. The model, incorporating pretreatment PCT, DBIL, and Na+ as key predictors, demonstrated excellent predictive performance during internal validation, offering a valuable tool for individualized risk stratification and treatment optimization in this patient population.
However, several limitations should be acknowledged. First, the study may be constrained by the relatively limited sample size and single-institution data source, which precluded the assessment of additional potential predictors and external validation of the model. Second, and perhaps more critically, the retrospective design led to the unavailability of certain important patient information. As evidenced by previous studies in other cancers, treatment regimens (such as drug combinations and cycle intervals) 38 and concomitant medications (particularly supportive care drugs like granulocyte colony-stimulating factors or antimicrobial agents) 39 are also significant predictors of myelosuppression. Unfortunately, detailed data on these variables were not consistently accessible in our retrospective cohort. The absence of these key confounding factors undoubtedly limits the model’s performance ceiling and may have led to an overestimation or underestimation of the risk associated with the variables we did include. We acknowledge that their incorporation could substantially improve the model’s discriminative ability and calibration performance. Third, the sample size of this study was determined by the number of eligible cases in the retrospective cohort, providing a preliminary exploration into the risk of hematologic toxicity in patients with NPC undergoing chemoradiotherapy. No formal sample size calculation was performed a priori, and the limited sample size may compromise the statistical power of our findings. In addition, although some categories showed no statistical significance (P > .05), all were retained in the final model to maintain structural completeness and ensure clinical interpretability. Despite these limitations, our study provides a foundational model based on readily available hematological parameters. Future work will involve prospective, multicenter cohort studies to systematically collect more comprehensive clinical data, incorporate additional predictive variables, and conduct external validation, thereby enabling the development of a more robust and clinically applicable prediction model.
Conclusion
In conclusion, we have successfully developed and validated a risk prediction model for chemoradiotherapy-induced myelosuppression in patients with NPC. This clinically applicable model may provide valuable predictive insights that may enable clinicians to implement proactive preventive strategies and optimize treatment regimens, thereby potentially reducing the incidence and severity of treatment-related myelosuppression. To further strengthen the clinical utility and generalizability of our findings, we strongly recommend conducting large-scale, multicenter prospective cohort studies with diverse patient populations. Such validation efforts would not only confirm the robustness of our model but also facilitate its integration into routine clinical practice for improved patient management.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251381678 – Supplemental material for Development of a Predictive Model for the Risk of Myelosuppression in Patients With Nasopharyngeal Carcinoma Undergoing Chemoradiotherapy
Supplemental material, sj-docx-1-onc-10.1177_11795549251381678 for Development of a Predictive Model for the Risk of Myelosuppression in Patients With Nasopharyngeal Carcinoma Undergoing Chemoradiotherapy by Ruo-han Wang, De-yue Jiang, Jin Lu, Li-xue Xun, Fan Wang, Qian-qian Shao and Hao-xuan Zhang in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
We thank the Ethics Committee of Bengbu Medical University for supporting this study.
Ethical Considerations
The study was conducted in accordance with the principles of the Declaration of Helsinki. The study was conducted in accordance with local regulations and institutional requirements and was approved by the Ethics Committee of Bengbu Medical University (approval number: [2023] 351; Date of approval: May 26, 2023). The requirement for informed consent from patients/guardians/participants was waived by the Ethics Committee due to the retrospective nature of this study.
Consent to Participate
This was a retrospective study and the Ethics Committee waived the requirement of informed consent for patients/guardians/participants. Therefore, informed consent of patients/guardians/participants was not applicable to this study.
Consent for Publication
The Ethics Committee waived the requirement for informed consent for patients/guardians/participants. Therefore, patients’/guardians’/participants’ consent for publication was not applicable to this study.
Author Contributions
Conception and design of this study: H-XZ, JL, and R-HW. Data collection, analysis, and interpretation: L-XX, FW, R-HW, and D-YJ. Drafting of the article: R-HW. Critical revision of the important intellectual content of this article: Q-QS, R-HW, JL, and H-XZ. All authors have read, revised and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Digital Medicine and Smart Health Key Laboratory of Anhui Province (Bengbu Medical University) open project key project (grant no. AHCM2024Z006), Open Research Project of the Provincial Key Scientific Research Platform of Anhui Province, Bengbu Medical University (grant no. AHCM2023Z003) and Key Research Project of Natural Science in colleges and universities of Anhui Provincial Department of Education (grant no. 2023AH051976).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the results of this study are not publicly available as they relate to the privacy of the participating researchers, but the corresponding author can be contacted upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
