Abstract
Background:
To retrospectively investigate the influence of pretreatment anemia and hemoglobin levels on the survival of nasopharyngeal carcinoma patients treated with concurrent chemoradiotherapy (C-CRT).
Methods:
A total of 149 nasopharyngeal carcinoma patients who received C-CRT were included. All patients had received 70 Gy to the primary tumor plus the involved lymph nodes, and 59.4 Gy and 54 Gy to the intermediate- and low-risk neck regions concurrent with 1–3 cycles of cisplatin. Patients were dichotomized into non-anemic and anemic (hemoglobin <12 g/dL (women) or <13 g/dL (men)) groups according to their pre-treatment hemoglobin measures. Receiver operating characteristic (ROC) curve analysis was utilized for accessibility of a pre-treatment hemoglobin cut-off that impacts outcomes. Potential interactions between baseline anemia status and hemoglobin measures and overall survival, locoregional progression-free survival (LRPFS), and progression-free survival were assessed.
Results:
Anemia was evident in 36 patients (24.1%), which was related to significantly shorter overall survival (P=0.007), LRPFS (P<0.021), and progression-free survival (P=0.003) times; all three endpoints retained significance in multivariate analyses (P<0.05, for each). A baseline hemoglobin value of 11.0 g/dL exhibited significant association with outcomes in ROC curve analysis: hemoglobin <11.0 g/dL (N=26) was linked with shorter median overall survival (P<0.001), LRPFS (P=0.004), and progression-free survival (P<0.001) times, which also retained significance for all three endpoints in multivariate analyses and suggested a stronger prognostic worth for the hemoglobin <11.0 g/dL cut-off value than the anemia status.
Conclusion:
Pre-C-CRT hemoglobin <11.0 g/dL has a stronger prognostic worth than the anemia status with regard to LRPFS, progression-free survival, and overall survival for nasopharyngeal carcinoma patients.
Introduction
Radical concurrent chemoradiotherapy (C-CRT) is the current standard treatment modality for non-metastatic nasopharyngeal carcinoma (NPC), with radiotherapy (RT) alone being reserved just for T1N0M0 disease stage.1,2 Notwithstanding the remarkable variations among the T and N definitions of the available systems, the tumor-node-metastasis (TNM) staging still stays to be the gold standard for prognostic stratification and treatment guidance of the NPC patients. 3 However, significantly different outcomes among the similarly treated same disease stages underscore the need for identification of more relevant novel prognosticators.3-5 Therefore, to complement the TNM, ongoing research has focused on the pursuit of highly practical and precise prognostic factors such as the biochemical and hematological parameters.6-9
Tumor hypoxia is a surrogate marker of radio- and chemo-resistance for many tumors including the NPC. Hypoxia stimulates the secretion of hypoxia-inducible factors and resultant increased rates of neo-angiogenesis and genetic mutations, which favor the development of a new aggressive tumor genotype that is highly resistant to free-radical-induced apoptosis.10-12 Granting this fundamental knowledge, various authors have investigated the availability of hypoxia-related prognostic factors in NPC patients.13-15 Low hemoglobin (Hgb) measure is one such factor, and therefore anemia, which is an indirect indicator of tumor hypoxia. 11 Evidence is growing for NPC, but still the exact influence of pre-treatment Hgb levels on tumor control rates and survival outcomes is controversial due to the inclusion of heterogeneously treated NPC patients rather than a homogenous C-CRT protocol, and the use of non-standard anemia definitions, such as the use of the same cut-off for both genders as opposed to the World Health Organization’s (WHO) sex-based criteria. Therefore, this retrospective research was conducted to assess the impact of pre-treatment anemia status on the clinical outcomes of newly diagnosed NPC patients treated with definitive C-CRT, and the accessibility of a specific Hgb cut-off that is associated with patients’ prognoses.
Methods
Study population
The database maintained by the Baskent University Faculty of Medicine, Department of Radiation Oncology Head and Neck Cancers Group was retrospectively searched to identify the NPC patients treated with C-CRT between January 2007 and December 2015. The key eligibility criteria were: (a) age 18–80 years; (b) Karnofsky Performance Score ⩾70; (c) histologically proven squamous cell carcinoma (SCC); (d) clinical/radiological proof of T2-4N0-3M0 or T1-4N1-3M0 disease stage according to the TNM staging system (7th edition); (e) body mass index ⩾20 kg/m2; (f) no prior RT or chemotherapy history; (g) received platinum-based C-CRT, available fluorodeoxyglucose-positron emission tomography-CT (PET-CT) and chest CT scans; (h) no evidence of brain metastasis on magnetic resonance imaging (MRI) scans obtained within 1 month of treatment; (i) available RT and chemotherapy charts; (j) pre-treatment complete blood count and biochemistry tests; (k) pre-treatment and follow-up head and neck clinical examinations; and (l) follow-up MRI and PET-CT scans.
Concurrent chemoradiotherapy
All patients received definitive C-CRT. As detailed in Table 1, the technique was 3-dimensional conformal RT between January 2007 to June 2011, and intensity-modulated RT thereafter. Regardless of the utilized technique, RT was administered in a daily fractionation basis: 5 days/week, for 7 weeks. Chemotherapy comprised cisplatin 75–80 mg/m2 (every 3 weeks) for the concurrent and the two cycles of cisplatin-based regimens (every 3 weeks) for the adjuvant treatment phases, respectively.
Details of radiotherapy.

CTV: clinical target volume; CT: computerized tomography; GTV: gross tumor volume; Gy: Gray; LN: lymph node; MRI: magnetic resonance imaging; PET-CT: positron emission tomography-computerized tomography; PTV: planning target volume.
Hemoglobin measurements
Anemia was defined as any pretreatment Hgb <12 g/dL in females and Hgb<13 g/dL in males according to the WHO criteria by utilizing the baseline complete blood count tests obtained at the first day of C-CRT. 22
Toxicity assessments
During the C-CRT, toxic events were assessed weekly or more frequently, if necessary. Both acute and late toxicities were reported according to the Common Terminology Criteria for Adverse Events (v3), with scores mirroring the worst grade ascertained. After the completion of C-CRT, patients received regular examinations every 3 months for the first 2 years, every 6 months between the third to fifth years, and annually (or more often) thereafter.
Assessment of treatment response
Treatment response was surveyed in the same intervals with the toxicity evaluations as specified above. Salvage interventions, such as neck dissection, systemic chemotherapy, re-irradiation, or their combinations, were provided to patients with confirmed local and/or regional relapses or distant metastatic events, as indicated. For each evaluation, all patients underwent detailed endoscopic examinations for the index NPC and other head and neck regions in order to ascertain any local/regional recurrences and their extent. Although the study design was retrospective, the treatment response was assessed prospectively, and first radiological evaluations were performed at the 90-day follow-up visit utilizing restaging PET-CT scans, and were scored according to the EORTC-1999 guidelines (the PET Response Criteria in Solid Tumors (PERCIST) for patients evaluated after 2009). Head and neck CT and/or MRI replaced the PET-CT scans at any timepoint when complete metabolic response was reported. Additional neck/abdominal ultrasonography or abdominal CT, chest CT, brain MRI, and bone scintigraphy were used for disease restaging as indicated.
Statistical analysis
The primary endpoint was the association between pre-C-CRT Hgb values and overall survival (OS): the interval between the onset of C-CRT and death/last follow-up. Secondary endpoints included the associations between pre-C-CRT Hgb values and progression-free survival (PFS) or locoregional PFS (LR-PFS): the interval between the onset of C-CRT and any type of disease progression or last follow-up/death (for PFS), or progression/recurrence at the nasopharynx and/or ipsilateral/contralateral neck or death (for LRPFS), respectively.
The Hgb variables that were assessed incorporated the pre-C-CRT Hgb measures and anemia status according to the WHO criteria: Hgb<12.0 g/dL for women and <13.0 g/dL for men. 22 Receiver operating characteristic (ROC) curve analysis was utilized for testing the ability of pre-C-CRT Hgb levels to discriminate outcomes. The curve fitting analysis method was utilized to test the validity of the ROC analysis defined cut-offs, where necessary. Frequency distributions were utilized for categorical variables, while means, medians, and ranges were used to describe continuous variables. Frequency distributions among different groups and their correlations were compared by utilizing Chi-square tests, Student’s t-tests, Pearson’s exact test, or Spearman correlations as appropriate. The impact of potential risk factors on OS, LRPFS, and PFS was assessed with Kaplan–Meier estimates and log-rank tests. The Cox proportional hazards model was utilized for multivariate analysis and incorporated only the factors exhibiting significance in the univariate analysis. A two-sided P value <0.05 was considered significant. In order to limit the chance related false positive discoveries, the noteworthiness of within-subgroup treatment effects was adjusted for multiplicity by utilizing the Bonferroni correction for comparisons between three or more subgroups.
Results
Our database search revealed 197 NPC patients, but 48 of them were excluded from the analyses for receiving upfront induction chemotherapy (n=28) and C-CRT with weekly cisplatin (n=20), respectively, leaving 149 patients eligible for analysis. Patient characteristics are as shown in Table 2, with no notable differences between the non-anemic (n=113) and anemic (n=36) cohorts (P=0.36-0.96). Treatment protocol was relatively well tolerated with only 1 (0.7%) treatment-related death due to intractable necrotic nodal-cutaneous fistula at the fifth month of follow-up. Grades 3 (n=78; 52.3%) and 4 (n=28; 18.8%) acute toxicities were reported in 106 (71.1%) patients. During the C-CRT, 105 (70.5%) patients were able to receive three courses of prescribed chemotherapy, while 76 (51.0%) were additionally able to receive one (n17; 11.4%) or two (n=59; 39.6%) courses of adjuvant chemotherapy.
Baseline disease and patient characteristics.
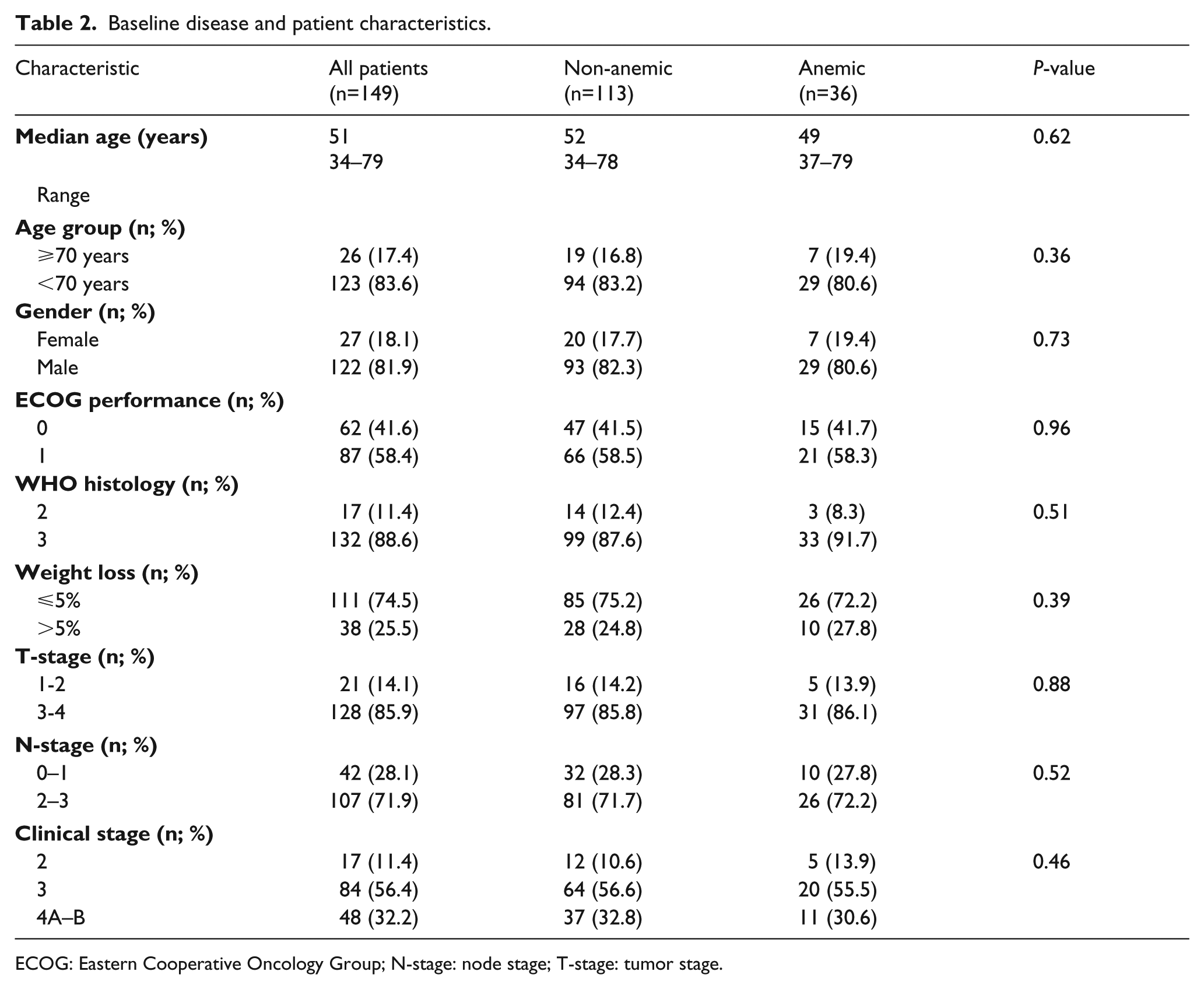
ECOG: Eastern Cooperative Oncology Group; N-stage: node stage; T-stage: tumor stage.
At a median follow-up of 54.3 months (range 5.0–80.130), 110 patients (73.8%) were alive and 81 of them (54.4%) were free of disease progression. For the entire population, the median OS and LRPFS was not yet reached during this final analysis with a median PFS time of 96.0 months (95% confidence interval (CI) 56.4, 135.6), while the 10-year OS, LRPFS, and PFS rates were 66.4%, 53.7%, and 44.3%, respectively.
The median baseline Hgb level was 13.7 g/dL (range 7.5–16.7) and 23/149 (15.4%) patients were anemic according to the WHO criteria. Survival analysis after dichotomization of patients into non-anemic versus anemic groups revealed that the median OS (not reached yet in both groups; P=0.007), LRPFS (not reached yet vs. 33 months; P=0.021), and PFS (98 vs. 19 months; P=0.003) were all significantly superior in the non-anemic cohort (Figure 1). Likewise, the 10-year survival estimates also favored the non-anemic cohort: (68.3% vs. 54.7%) for OS, (54.7% vs. 50.4%) for LRPFS, and (%44.5 vs. 38.6%) for PFS, respectively.
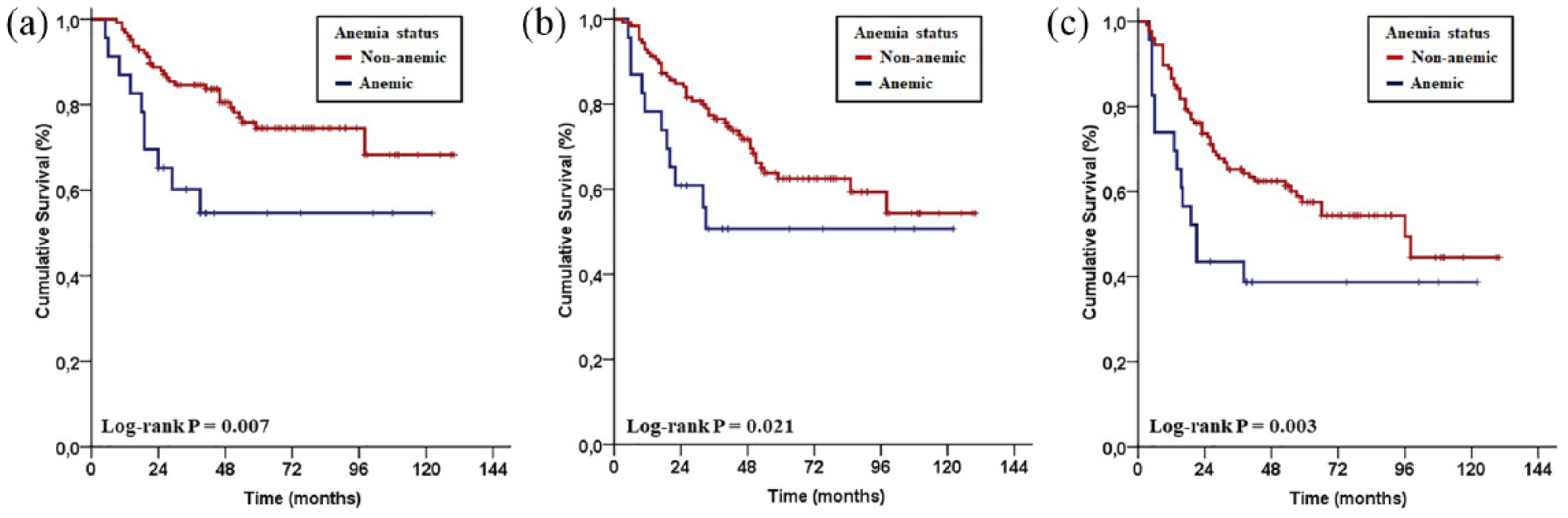
Survival according to pre-treatment anemia status. (a) Overall survival. (b) Locoregional progression-free survival. (c) Progression-free survival. Red lines indicate non-anemic patients; blue lines, anemic patients.
We also searched for accessibility of a potential cut-off Hgb value that may interact with outcomes. In the ROC curve analysis, baseline Hgb values of 11.0 g/dL (area under the curve (AUC) 86.6%, sensitivity 81.4%, and specificity 76.3%), 10.9 g/dL (AUC 78.6%, sensitivity 73.3%, and specificity 71.1%), and 11.1 g/dL (AUC 80.4%, sensitivity 78.5%, and specificity 72.7%) emerged to be the cut-offs to demonstrate significant association with median OS, LRPFS, and PFS durations, respectively (Figure 2). Considering the fact that the three cut-off values were very close to 11.0 g/dL of LRPFS, we utilized the rounded 11.0 g/dL as the common cut-off for dichotomizing patients into two groups for further analyses: Group 1: Hgb<11.0 g/dL (n=26) and Group 2: Hgb⩾11.0 g/dL (n=123). The curve fitting analyses also confirmed the ROC analysis defined rounded Hgb⩾11.0 g/dL cut-off point with the y=0.0176x + 10.168 (R2= 0.3274) equation where y and x represented the OS times and Hgb measures, respectively. Comparative analyses revealed that the Hgb⩾11.0 g/dL group had significantly longer OS (not reached yet in both groups; P<0.001), LRPFS (not reached yet in both groups; P=0.004), and PFS (96 vs. 21 months; P<0.001) durations than the Hgb<11.0 g/dL group (Figure 2). Similarly, the 10-year OS (71.3% vs. 56.9%), LRPFS (57.7% vs. 48.2%), PFS (49.0% vs. 35.6%) estimates also favored the Hgb<11.0 g/dL group.
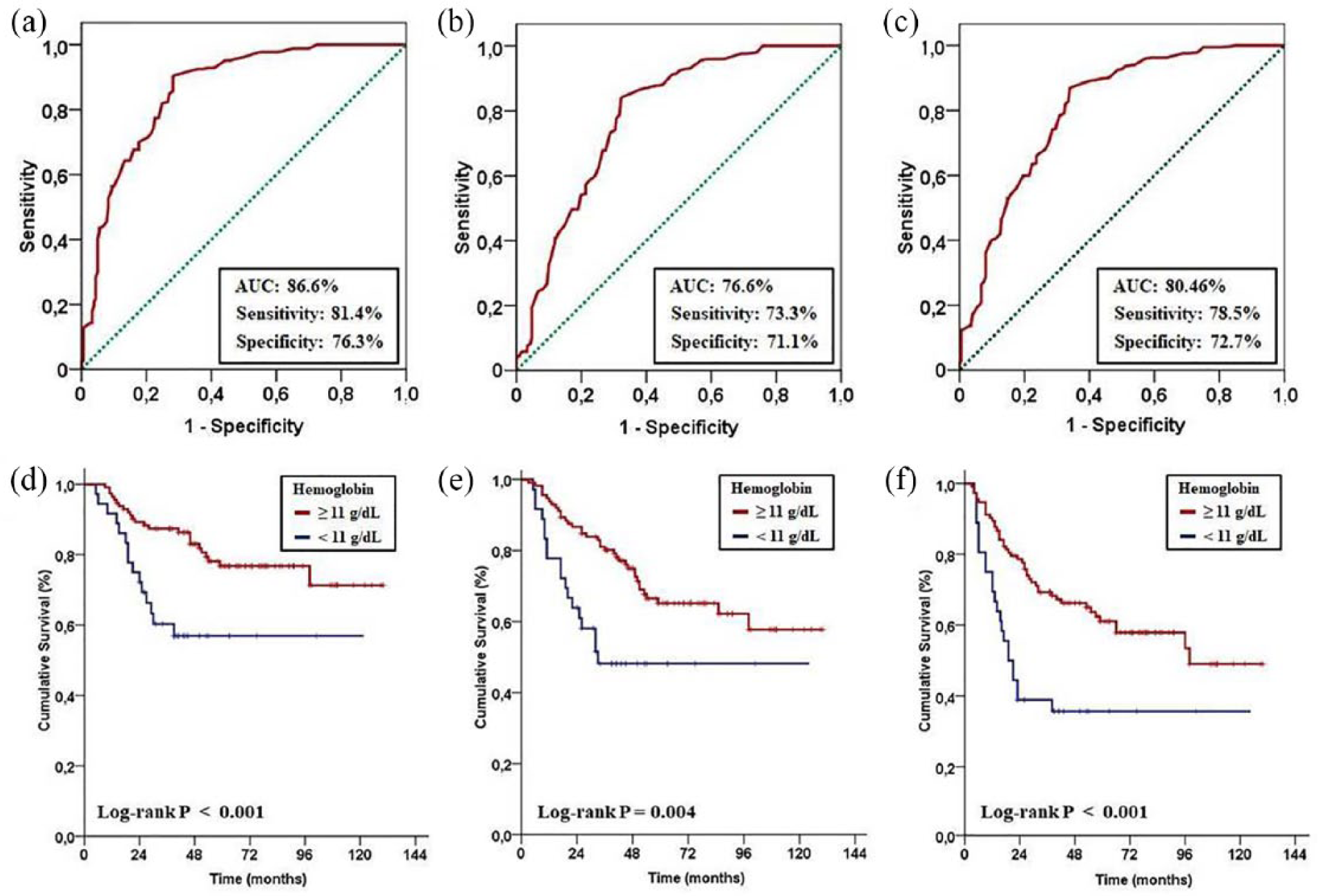
Receiver operating characteristic curve analysis outcomes: (a) Overall survival. (b) Locoregional progression-free survival. (c) Progression-free survival; and survival according to pre-treatment Hgb value. (d) Overall survival. (e) Locoregional progression-free survival. (f) Progression-free survival. Red lines indicate patients with Hgb levels ⩾11 g/dL; blue lines, patients with Hgb levels <11 g/dL.
On univariate analyses, lower T-stage (2–3 vs. 4), lower N-stage (0–1 vs. 2–3), earlier overall disease stage (2–3 vs. 4), lesser weight loss (WL) over past 6 months (⩽5% vs. >5%), normal Hgb levels (non-anemic vs. anemic), and higher Hgb levels (⩾11.0 vs. <11.0 g/dL) were found to be associated with favorable OS, LRPFS, and PFS (Table 3), and each variable retained independent significance in multivariate analyses (P < 0.05 for each), except for the T-stage (Table 3). As the pretreatment Hgb levels (⩾11.0 vs. <11.0 g/dL) and WL status (⩽5% vs. >5%) appeared to be the strongest factors affecting survival outcomes, we additionally searched for a possible correlation between these two factors, which might have influenced clinical outcomes in an unpredictable manner. However, suggesting that both factors had an independent prognostic worth on outcomes in their own way, the results of Spearman’s correlation analysis revealed no significant association between these two factors (r2= −0.12; P=0.34).
Results of uni- and multivariate analysis.
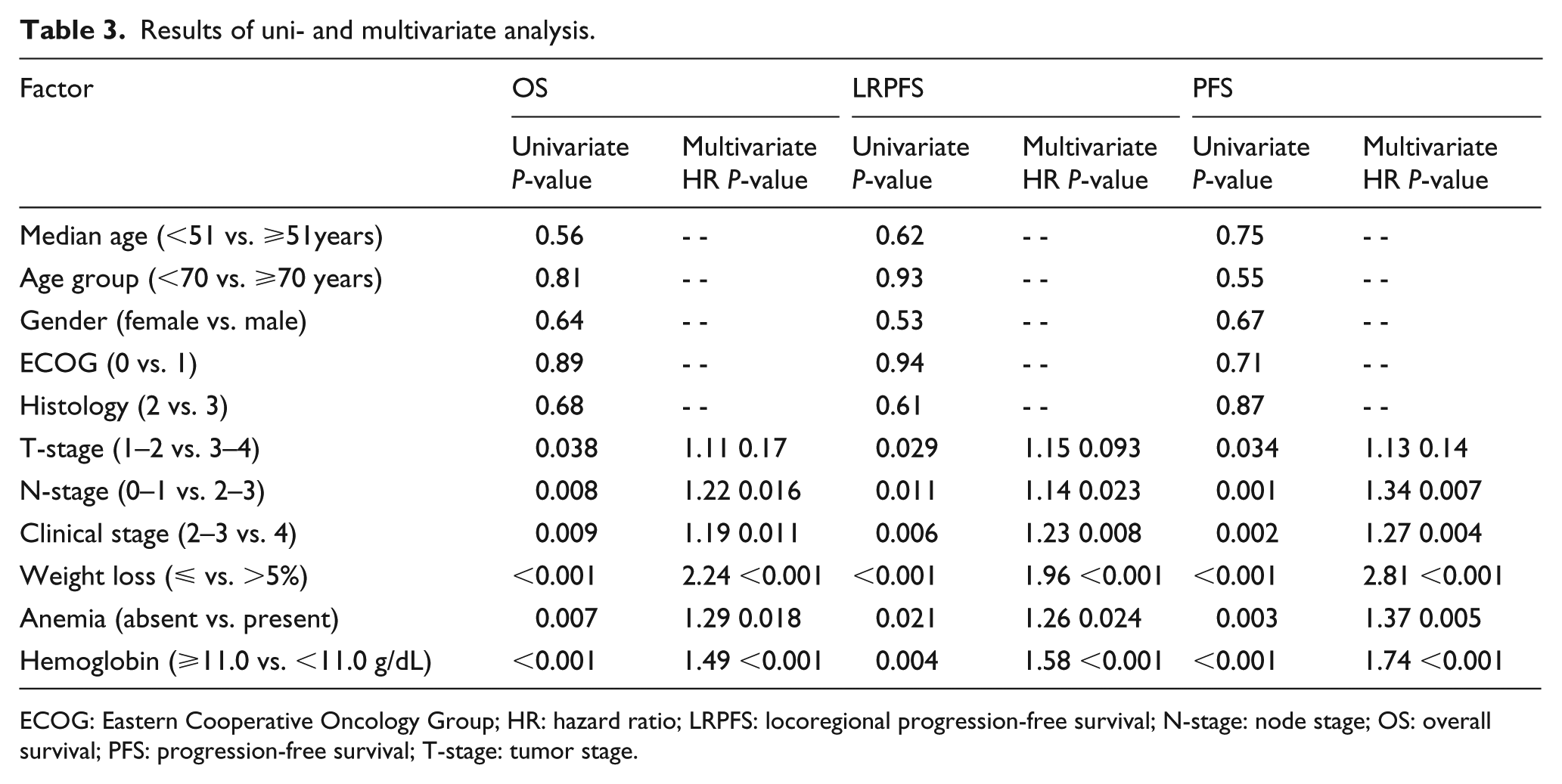
ECOG: Eastern Cooperative Oncology Group; HR: hazard ratio; LRPFS: locoregional progression-free survival; N-stage: node stage; OS: overall survival; PFS: progression-free survival; T-stage: tumor stage.
Discussion
The present outcomes of 149 NPC patients treated with C-CRT showed that the pretreatment anemia was associated with significantly poorer survival outcomes in terms of LRPFS, PFS, and OS times. Furthermore, compared with the anemia status, we also demonstrated that pre-treatment Hgb<11.0 g/dL was more strongly related with significantly shorter LRPFS, PFS, and OS outcomes.
Indicating radio- and chemoresistance, more than 50% of all solid tumors exhibit hypoxic areas.23-27 Therapeutic radiation ionizes H2O and forms reactive oxygen species, which react with DNA and form DNA radicals. 28 If available in satisfactory amounts, O2 reacts with the DNA radicals and fixes the radiation-induced DNA damage. Therefore, the tumor cell killing efficiency of ionizing radiation is fortified by O2 up to 2.5 to 3 times; namely the oxygen enhancement ratio. Strong evidence suggests that the critically lower levels of Hgb are correlated with a detrimental tumor oxygenation status, 29 and, therefore, diminished tumor control rates and survival outcomes.30-32 Even though our current information on the factual rates of hypoxia incidence and its intratumoral extent in NPC is limited to a few patients incorporated in other head and neck cancer studies, the available proof has clearly shown the presence of severely hypoxic tumor zones in this patient group.33-36 Therefore, various studies investigated the impact of anemia on outcomes of NPC patients (i.e. anemia is either present at the pre-treatment period or emerges de novo during the treatment course) and searched for a particular cut-off Hgb value that may be useful in the further prognostic stratification of such patients.14-21 However, forming the basis for the present research, the reported results conflicted with the proposed Hgb cut-offs and the true influence of anemia on patients’ outcomes, which may have been affected by the significant variations in anemia definitions.14-21
Our data showed that pre-C-CRT anemia adversely affected the OS (P=0.007), LRPFS (P=0.021), and PFS (P=0.003) outcomes of NPC patients. This finding is consistent with previous reports in the head and neck SCCs 37 and some of the NPC literature proposing a poor prognostic role for pre-C-CRT anemia.14,21,38 Agreeing with our outcomes, Mai et al., 38 Zhang et al., 14 and Zhang et al. 21 had previously shown that the pre-treatment anemia was an independent surrogate of worse tumor control rates and survival results. Likewise, combined scoring models also incorporated pre-treatment anemia as an indispensable poor prognostic factor in NPC patients.3,39-41 However, in other studies mid- rather than the pre-treatment anemia has been noted to display a prognostic worth.16,20,42 Although it is difficult to disclose these conflicting outcomes with solid reasons, some reasonable assumptions can be claimed. First, rendering it almost impossible to identify a thoroughly anemic patient population—and therefore their relative outcomes—the pre-treatment anemia was defined at notably higher Hgb cut-offs compared to the WHO and the Chinese Society of Clinical Oncology definitions in some such studies.22,43 For instance, Li et al. 41 set the Hgb cut-off at 13.9 g/dL in their prognostic nomogram. Patient stratification with such types of methodologies may cause fallacious increments in the relative percentages of anemic subgroups with resultant false negative outcomes. Second, as discussed in the next paragraphs, such use of non-standard high Hgb cut-offs may overestimate but not reflect the real estimates of NPC patients with truly hypoxic tumors, and therefore their outcomes. Regarding the proposed strong relationship between the Hgb levels and the extent of hypoxic fractions—which is a surrogate marker of radio- and chemoresistance—we kindly recommend the use of well-established universal Hgb cut-offs for anemia definition in order to prevent meaningless discrepancies among the outcomes of different studies.
Another vital finding of the present investigation was the rise of Hgb<11.0 g/dL as the cut-off that stratified the study population into two significantly distinct survival groups in a stronger way than the pre-C-CRT anemia status regarding the OS (P<0.001 vs. 0.007), the LRPFS (P=0.004 vs. 0.021), and the PFS (P<0.001 vs. 0.003). Previously, Chua et al. 16 observed significantly poorer 5-year local recurrence-free (60% vs. 80%; P=0.0059) and disease-specific survival rates (51% vs. 68%; P=0.001) in NPC patients with a midradiation Hgb⩽11.0 g/dL than their Hgb>11.0 g/dL partners. Chua’s finding lends support to the relevance of our Hgb<11.0 g/dL cut-off, but Chua’s observation was viable only for midradiation as opposed to the pre-C-CRT period proposed by our results. This discrepancy might be related to the methodologic differences between the two studies: being far above the recommended anemia definitions, Chua et al. 16 utilized mean pre-treatment Hgb values of 13.7 g/dL for RT alone and 13.6 g/dL for induction chemotherapy plus RT instead of using the statistically more reliable one proposed by ROC curve analysis. Thus, it is reasonable to anticipate that the authors might have missed the chance to detect a relevant pre-treatment Hgb cut-off at a lower value. Finally, the Hgb ⩽11.0 g/dL cut-off that emerged here was directly supported by a more recent NPC study, 40 where pre-treatment Hgb⩽11.0 g/dL was reported among the factors predicting distant failures following C-CRT.
The underlying exact mechanism how Hgb<11.0 g/dL relates to outcomes of NPC patients is not clear yet. Nevertheless, several experimental studies undoubtedly proved that tumor hypoxia was more frequent in anemic than the non-anemic animals, with further clear proof for improved tumor oxygenation status after the amendment of anemia. 29 Mathematical models and animal studies support our Hgb cut-off of 11.0 g/dL.44,45 A study modelling the influence of anemia on tumor blood flow clearly showed that the tumoral O2 delivery was reaching its peak at Hgb values around 11.0 g/dL and was decreasing at lower Hgb measures, 44 which was confirmed by another blood flow and O2 delivery study in subcutaneous tumors of rats made anemic by hemodilution. 45 The outcomes of clinical studies in head and neck SCCs further confirm the relevance of our Hgb cut-off value.46,47 Rudat et al. 46 reported significantly larger hypoxic fractions in tumors of patients presenting with Hgb⩽11 g/dL than their Hb>11 g/dL counterparts (33.9% vs. 22.6%; P=0.004). Similarly, in the Clavo et al. 47 study, the hypoxic fraction was 34% in tumors of non-anemic patients compared with 47% in anemic patients, and 69% in the most anemic patients (Hgb <11.5 g/dL; P=0.032). Although future larger prospective studies may prove beneficial, considering the aforementioned basic and clinical studies together with our present findings, accessible evidence suggests an Hgb value around 11 g/dL to be the optimal cut-off for the prediction of distinctive outcomes of NPCs.
Our study is empowered by two factors. First, we consistently used PET-CT for disease staging and RT planning, and treated all patients in a similar design respecting the C-CRT and adjuvant chemotherapy conventions. Second, we utilized the gender-based anemia definition recommended by the WHO, instead of utilizing the 11.0 g/dL or 12.0 g/dL as the regular cut-off for both genders. However, the present research also had at least two major drawbacks. First, it was a single-institutional retrospective analysis with a relatively small cohort size. Second, we constrained our investigation to baseline anemia status and Hgb levels, which dispossessed the elucidation of potential influences of the de novo anemia or further lowered Hgb levels throughout the C-CRT and adjuvant chemotherapy periods. Such impacts may well have altered the actual rates of both the anemic patients and the long-term reduced Hgb levels below the cut-off of 11.0 g/dL, and, hence, the related clinical results displayed here.
Conclusion
The outcomes of the present study indicate that both the pre-C-CRT anemia and, more strongly, the baseline Hgb<11.0 g/dL are independent associates of significantly poorer LRPFS, PFS, and OS outcomes in NPC patients. These discoveries unequivocally underline the requirement for correction of anemia, and therefore hypoxia, with the future accessibility of agents with no tumor growth stimulatory effects.
Footnotes
Author contributions
Berna A. Yildirim, Nur Y. Ekici, Ali A. Besen, Huseyin Mertsoylu collected and assembled the data; Erkan Topkan, Ugur Selek, Yurday Ozdemir conceived and designed the study; and Erkan Topkan, Ugur Selek, Yurday Ozdemir, Ali A. Besen, Berna A. Yildirim, Huseyin Mertsoylu performed the data analysis and interpretation; and Erkan Topkan, Nur Y. Ekici, Ugur Selek wrote the manuscript. All authors gave their final approval of the manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
