Abstract
Objective
This study aimed to identify risk factors for the occurrence and growth of thyroid nodules (TNs) in euthyroid individuals, and to develop a machine learning-assisted predictive model (least absolute shrinkage and selection operator (LASSO) Cox regression) for preliminary individualized risk stratification.
Methods
A nine-year retrospective cohort of 1140 participants with normal thyroid function (6444 data points) was analyzed. First, LASSO regression—a classic regularization algorithm in machine learning—was applied to screen key predictors from 17 candidate variables. The selected variables were then incorporated into multivariable Cox proportional hazards models to construct a LASSO-Cox predictive model. A nomogram was developed, and model performance was assessed using the area under the curve (AUC), bootstrap-corrected concordance index, calibration plots, and decision curve analysis (DCA).
Results
Machine learning-based LASSO regression screened 10 variables for TN occurrence (age, sex, systolic blood pressure (SBP), diastolic blood pressure (DBP), waist circumference (WC), body mass index (BMI), fasting plasma glucose (FPG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), metabolic syndrome (MetS)) and 10 variables for TN enlargement (age, sex, SBP, BMI, hemoglobin A1c (HbA1c), TC, triglycerides, HDL-C, uric acid, MetS). Cox analysis further confirmed that sex, SBP, DBP, WC, BMI, FPG, HDL-C, and MetS were predictors of TN occurrence, while sex, HbA1c, HDL-C, and MetS were associated with TN enlargement. The LASSO-Cox nomogram achieved AUCs of 0.65 for occurrence and 0.64 for enlargement, with bootstrap-corrected C-indices of 0.62 and 0.64. Calibration showed good agreement between predicted and observed risks, and DCA demonstrated consistent net benefit across threshold probabilities of 0.05–0.6.
Conclusion
Sex, HDL-C, and MetS are key predictors of TN occurrence and enlargement. The machine learning-assisted LASSO-Cox nomogram, based on routine clinical indicators, shows moderate discrimination and good calibration. This preliminary tool may assist in population-level risk stratification; however, its modest discrimination limits its immediate clinical implementation.
Keywords
Introduction
Thyroid nodules (TNs) are a common endocrine disorder, with reported prevalence ranging from 34% to 68% across populations and diagnostic methods.1,2 The widespread use of ultrasound has substantially increased detection rates due to its non-invasive nature 3 and recent clinical guidelines emphasize the need for integrated risk-stratification tools that combine metabolic and imaging data for precision assessment. 4 In high-income countries, rising detection is primarily driven by advanced imaging and screening programs, whereas in low- and middle-income settings, environmental exposures and iodine status play a more prominent role. A nationwide survey in China (2018) reported an age- and sex-standardized prevalence of 37.1% among adults—44.7% in females and 29.8% in males. 5 More recently, a 2024 study in Zhejiang Province involving over 10,000 adults found a detection rate of 50.98%, 6 underscoring the growing public health relevance of this condition. Although most nodules are benign, approximately 7%–15% may harbor malignancy, accounting for 2.1% of global cancer cases.7,8
Studies have identified various metabolic and physiological factors associated with the occurrence of thyroid nodules. Factors such as female gender, advanced age, central obesity, hypertension, and diabetes have been established as independent risk factors. 9 Conversely, normal triglyceride levels are considered a protective factor. 10 Both male and female patients with metabolic syndrome show an increased risk of thyroid nodules, which appears to be independent of age. 11 –13 These factors may influence the formation and progression of thyroid nodules through different mechanisms, including insulin resistance-mediated cell proliferation, chronic inflammation, and lipid metabolism disorders.
However, many existing studies have not excluded individuals with thyroid dysfunction. Given that conditions like hypothyroidism can directly stimulate thyroid tissue growth via elevated thyroid-stimulating hormone (TSH) or interact with metabolic pathways, they may confound the observed relationships between metabolic markers and nodule formation. Consequently, the independent contribution of metabolic factors to both the occurrence and long-term enlargement of TNs in euthyroid individuals remains poorly characterized.
Furthermore, prior prediction models have primarily focused on cross-sectional screening or on the risk of malignant transformation,14,15 with limited attention to the natural history of benign nodules in metabolically diverse, thyroid-function-normal populations. To address these gaps, we conducted a nine-year longitudinal cohort study in a large sample of euthyroid adults to systematically evaluate metabolic and clinical determinants of incident TNs and their subsequent growth.
Materials and methods
Ethical approval and data protection
This study adhered to the ethical standards of the Declaration of Helsinki (2013 revision) and was approved by the Sichuan Provincial People's Hospital Institutional Review Board (IRB) (Approval No. 487, 2024), which explicitly granted permission for secondary use of anonymized clinical data for research purposes. Written informed consent was obtained from each human participant prior to their inclusion in the study: For participants with full cognitive capacity, consent was documented via signed written forms; for participants with physical or cognitive limitations preventing written signature, verbal informed consent was obtained in the presence of an independent witness, and the witness completed a consent verification form to confirm the process. All consent documents were stored in the hospital's secure research archive.
All data were anonymized (via de-identification of personal identifiers such as names, ID numbers, and contact information) to protect the privacy of the participants, and the authors had no access to information that could identify individual participants during or after data collection. The data used in this study were accessed for research purposes on 14/08/2024.
Design and sample
This retrospective study utilized research data extracted from the health management system of Sichuan Provincial People's Hospital, covering the period from 01/01/2015 to 31/12/2023. The subjects included individuals who underwent at least 3 out of 9 consecutive thyroid ultrasound examinations at our center (to ensure longitudinal data density while balancing feasibility). With the assistance of the Information Department, basic information, thyroid ultrasound data, and biochemical indicators of the examinees were exported using Excel.
Inclusion criteria
Participants were required to undergo at least three comprehensive examinations over nine years to minimize measurement bias and ensure longitudinal data reliability, with all of the following information available: Age, sex, body mass index (BMI), fasting plasma glucose (FPG), systolic blood pressure (SBP), diastolic blood pressure (DBP), triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), uric acid (UA), Low-Density Lipoprotein Cholesterol (LDL-C), TSH, free thyroid hormones (FT4), glycated hemoglobin (HbA1c), waist circumference (WC), past medical history, current medical history, and surgical history. No history of thyroid-related diseases, current thyroid conditions, or thyroid surgeries.
Exclusion criteria
Individuals with a history of thyroid-related diseases or current thyroid conditions, as well as those with a history of thyroid surgeries. Additionally, individuals with abnormal results for TSH and FT4 in their examination data were excluded. Incomplete data: Records with missing examination dates or missing outcome information were excluded. For baseline covariates, missingness was assessed for each variable. Given the low proportion of missing values (<5% per variable), participants with partially missing baseline data were retained, and missing values were handled using multiple imputation, as described in the Statistical Analysis section. The inclusion–exclusion process is visualized in Figure 1, with detailed sample size changes at each step. Final inclusion: 1140 individuals (6444 data points), with euthyroid status confirmed and acceptable completeness of baseline data.

Flowchart of participant inclusion and exclusion data preparation.
General information and laboratory tests
General examinations
Height and weight of the participants were measured using a standardized HNH-318 body scale (Shenzhen Shuangjia Electronics Co., Ltd), and BMI was calculated as kg/m2. WC was measured manually with a tape measure. Blood pressure measurements, including SBP and DBP, were taken using a standardized Omron HPB-9020 mercury sphygmomanometer. The average of two measurements was recorded as the final value. All measurement instruments (e.g., HNH-318 body scale, Omron HPB-9020 sphygmomanometer) were calibrated annually according to hospital quality control protocols. Calibration records were archived and verified to ensure data consistency throughout the study. Additionally, health management center nurses, who received specialized training, collected general and health-related information from the participants using a standardized script.
Laboratory tests
Blood samples (5–10 ml) were collected from fasting individuals (fasting for at least 8 h) by phlebotomists at the hospital's health examination center and laboratory department. The samples were then sent to the hospital's laboratory for analysis, which included measurements of HbA1c, FPG, TG, TC, HDL-C, UA, LDL-C, TSH, and FT4. Serum TSH and free T4 levels were measured using chemiluminescence immunoassay following the standards of Sichuan Provincial People's Hospital, with consistent participation in an External Quality Assessment Program to ensure accuracy.
Ultrasound examination of the thyroid
Thyroid examinations were performed using a color Doppler ultrasound system with a 7.5 MHz probe, conducted by experienced thyroid ultrasound specialists. The participants were positioned supine with their necks fully exposed. The examination recorded the size, echogenicity, presence of nodules, number of nodules, size, margins, echogenicity, and any calcifications of the thyroid. TNs were diagnosed according to the guidelines published by the Endocrinology Branch of the Chinese Medical Association in 2023. 16 Participants with TNs detected on ultrasound were included in the TN group (n = 3000). Those with no TNs during the same examination period were included in the non-TN group (n = 3444).
Diagnostic assessment criteria
According to the guidelines from the Chinese Diabetes Society, 17 the diagnostic criteria for metabolic syndrome (MetS) require the presence of at least three of the following five components: (1) Abdominal obesity: WC ≥90 cm in men and ≥85 cm in women; (2) FPG ≥ 6.1 mmol/L and/or 2-h postprandial glucose ≥ 7.8 mmol/L, or a diagnosis of diabetes with ongoing treatment; (3) Blood pressure ≥130/85 mmHg, or a diagnosis of hypertension with ongoing treatment; (4) TG ≥ 1.7 mmol/L; (5) Fasting HDL-C < 1.04 mmol/L.
Definition of TN outcomes
Occurrence of TNs: Defined as the first detection of one or more TNs on ultrasound during follow-up in participants with no nodules at baseline.
Growth of TNs: Defined as the first occurrence during follow-up of either (1) an increase in maximum diameter of ≥2 mm in any existing nodule compared to baseline, or (2) emergence of new nodules in participants with pre-existing nodules. The event time was recorded as the date of the ultrasound examination when growth was first detected. Although nodule growth is inherently a continuous biological process, we operationalized enlargement as a discrete time-to-event outcome to enable survival analysis and to capture the timing of clinically detectable progression in routine follow-up practice.
According to the 2019 guidelines for the management of hyperthyroidism by the Chinese Society of Endocrinology, 18 patients with clinical hyperthyroidism, subclinical hyperthyroidism, or non-thyrotoxic thyroid conditions exhibit TSH levels below the normal range. Thus, individuals with TSH <0.1 μU/mL are classified as having hyperthyroidism and were excluded from the study.
According to the 2019 guidelines for the management of hypothyroidism by the Chinese Society of Endocrinology, 19 serum TSH and FT4 are the primary indicators for diagnosing primary hypothyroidism. Therefore, individuals with TSH >4.2 μU/mL and FT4 < 9.0 pmol/L are classified as having hypothyroidism and were excluded from the study.
Statistical analysis
All analyses were conducted using R version 4.0.5. Overall, the proportion of missing values was low (<5% for individual variables). Missing values in baseline covariates were handled using multiple imputation to minimize potential bias and preserve statistical power, and results from the imputed datasets were combined for subsequent analyses. Continuous variables were assessed for normality using the Shapiro–Wilk test. Non-normally distributed continuous variables are presented as median and interquartile range [M(P25–P75)] and compared between groups using the Mann–Whitney U test. Categorical variables are expressed as frequencies and percentages [n (%)], with between-group differences evaluated by the chi-squared (χ2) test. To account for potential confounding in baseline comparisons, multivariable logistic regression models adjusting for age, sex, and MetS status were performed. The origin of the time scale was the date of the baseline health examination, and participants were censored at the date of their last follow-up if the event had not occurred.
To identify robust predictors, least absolute shrinkage and selection operator (LASSO) Cox regression was performed. All continuous predictors were standardized before analysis. Ten-fold cross-validation was used to select the optimal regularization parameter (λ) corresponding to the minimum cross-validated partial likelihood deviance. LASSO variable selection and subsequent Cox proportional hazards modeling were conducted within each imputed dataset, and pooled regression coefficients and standard errors were reported. The variables retained by LASSO were then entered into multivariable Cox proportional hazards models fitted with the survival package. The proportional hazards assumption was verified for all covariates using Schoenfeld residuals, with no significant violations detected (all p > 0.05).
A clinical risk prediction nomogram was developed based on the final Cox models. Model performance was evaluated using three complementary metrics. Discrimination was assessed using time-dependent area under the receiver operating characteristic curve (AUC) at 5 and 9 years, and the concordance index (C-index), with internal validation via 1000 bootstrap resamples to obtain bias-corrected estimates. Calibration was evaluated using calibration curves comparing predicted and observed event probabilities across deciles of risk, with 95% confidence intervals derived from 1000 bootstrap replicates. Clinical utility was assessed using decision curve analysis (DCA), which quantified net benefit across threshold probabilities ranging from 0.05 to 0.60, compared with default strategies of “treat all” or “treat none.”
All statistical tests were two-sided, and p-values <0.05 were considered statistically significant. All predictor variables were derived from baseline measurements; follow-up assessments were not incorporated as time-varying covariates due to irregular examination intervals.
Results
The univariate analysis of the TN group and non-TN group
The overall prevalence of TNs was 46.6%, with 22.5% among females and 24.0% among males. In univariate comparisons, participants with TNs were significantly more likely to be female and older, and had higher levels of systolic and DBP, HbA1c, and FPG compared to those without nodules (all P < 0.05). Although BMI and WC did not differ significantly in the overall population, gender-stratified analyses revealed significantly higher values in nodule-positive individuals within both sexes (P < 0.01). Additionally, no significant difference in LDL-C levels was observed between groups among females (see Table 1).
Comparison of baseline characteristics between thyroid nodule group and non-thyroid nodule group.

Note: “–” indicates no data. Units are as indicated in the table.
SBP: systolic blood pressure; DBP: diastolic blood pressure; WC: waist circumference; HDL-C: high-density lipoprotein cholesterol; HbA1c: glycated hemoglobin A1c; FPG: fasting plasma glucose; TG: triglyceride; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; UA: uric acid; MetS: metabolic syndrome; BMI: body mass index.
LASSO regression analysis of risk factors
LASSO regression identified a parsimonious set of predictors for TN outcomes from the full baseline variable pool. For incident TNs, selected variables included age, sex, blood pressure indices, adiposity measures (BMI and WC), glycemic and lipid parameters (FPG, TC, HDL-C), and MetS status. For nodule enlargement, the model retained age, sex, SBP, BMI, HbA1c, lipid profile (TC, TG, HDL-C), UA, and MetS. These variables were carried forward into multivariable survival modeling (see Figure 2).

Least absolute shrinkage and selection operator (LASSO) regression coefficient path diagrams for risk prediction of thyroid nodule occurrence (A–B) and enlargement (C–D). (Left) Coefficient profiles of variables as a function of log(λ). (Right) Ten-fold cross-validation for selection of the optimal λ value. As λ increases, variable coefficients shrink toward zero, and those with non-zero coefficients are retained, providing the basis for subsequent Cox regression analysis.
Multivariable Cox regression analysis
Multivariable Cox regression identified several independent predictors of TN outcomes. For incident TNs, female sex, elevated DBP, higher BMI, presence of MetS, and lower HDL-C were associated with increased risk. In contrast, higher SBP, WC, and FPG were associated with reduced risk. These inverse associations should be interpreted cautiously, as they may reflect detection bias, residual confounding, or non-linear metabolic effects in euthyroid populations rather than actual protective biological mechanisms. For nodule enlargement during follow-up, female sex and lower HDL-C levels remained significant risk factors; however, both HbA1c and MetS were inversely associated with enlargement (i.e., higher levels were linked to a lower risk). Notably, low HDL-C was the only factor consistently associated with higher risk for both nodule development and progression, underscoring its potential role as a central metabolic marker in TN pathogenesis (see Table 2).
Multivariable cox regression model results and hazard ratios for thyroid nodules (occurrence and enlargement).

Note: “–” indicates no data. Units are as indicated in the table.
SBP: systolic blood pressure; DBP: diastolic blood pressure; WC: waist circumference; BMI: body mass index; FPG: fasting plasma glucose; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; MetS: metabolic syndrome; HbA1c: hemoglobin A1c; TG: triglycerides.
Nomogram construction and predictive performance
Based on Cox regression results, nomograms were constructed to quantify the contribution of each predictor to TN occurrence and enlargement. Predicted risk points for individual variables are summarized in Table 3. The model showed moderate discrimination, with AUCs of 0.65 (95% CI: 0.64–0.66) for TN occurrence and 0.64 (95% CI: 0.63–0.66) for TN enlargement. Bootstrap-validated concordance indices were 0.62 (95% CI: 0.60–0.63) and 0.64 (95% CI: 0.62–0.66), respectively, indicating stable predictive performance (see Figure 3).
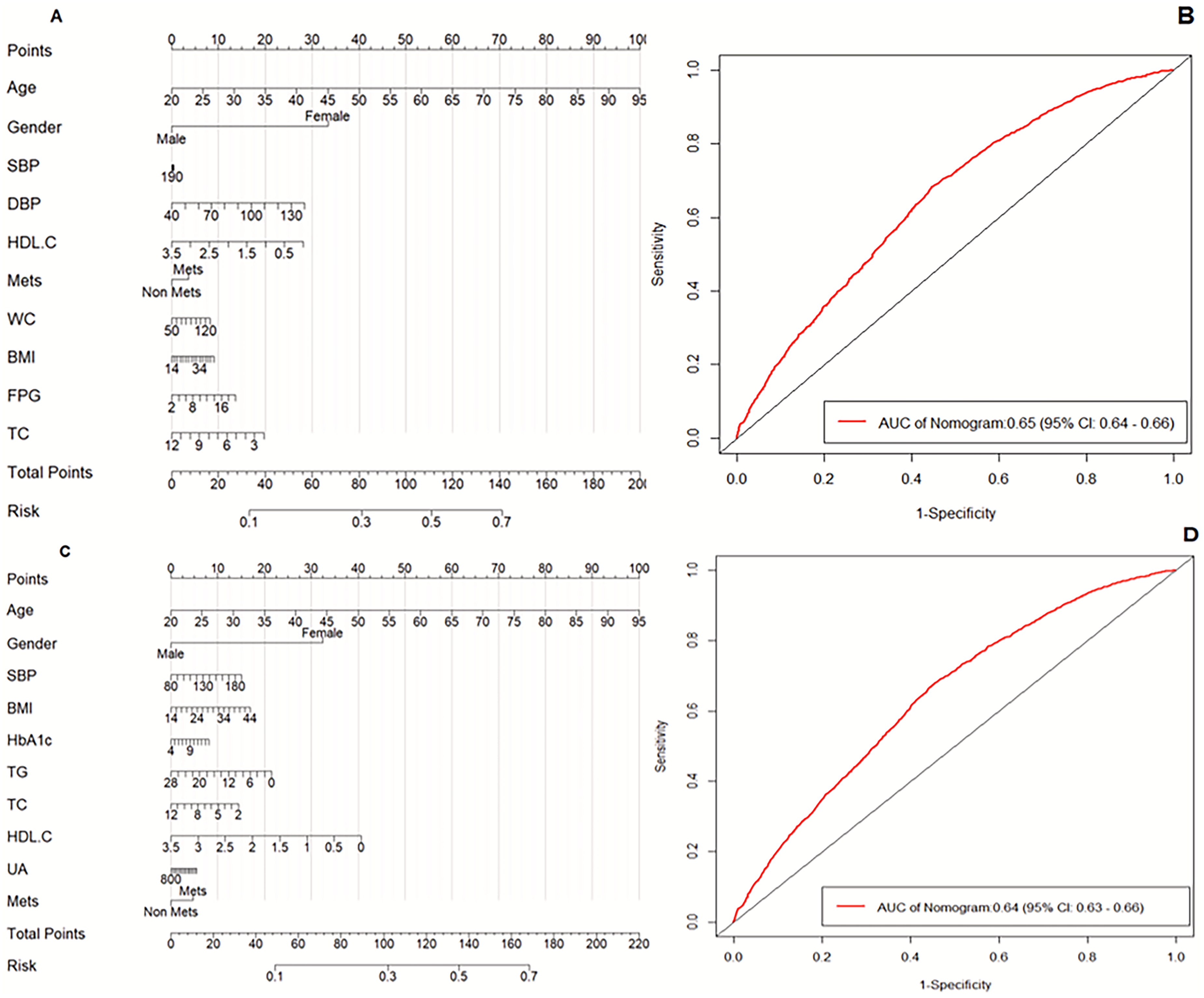
Nomogram and predictive performance evaluation for thyroid nodule occurrence and enlargement risk. (A and C) Nomograms for occurrence and enlargement. (B and D) ROC curves for the two models.
Variable-points correspondence from nomogram for thyroid nodule occurrence and enlargement risks.
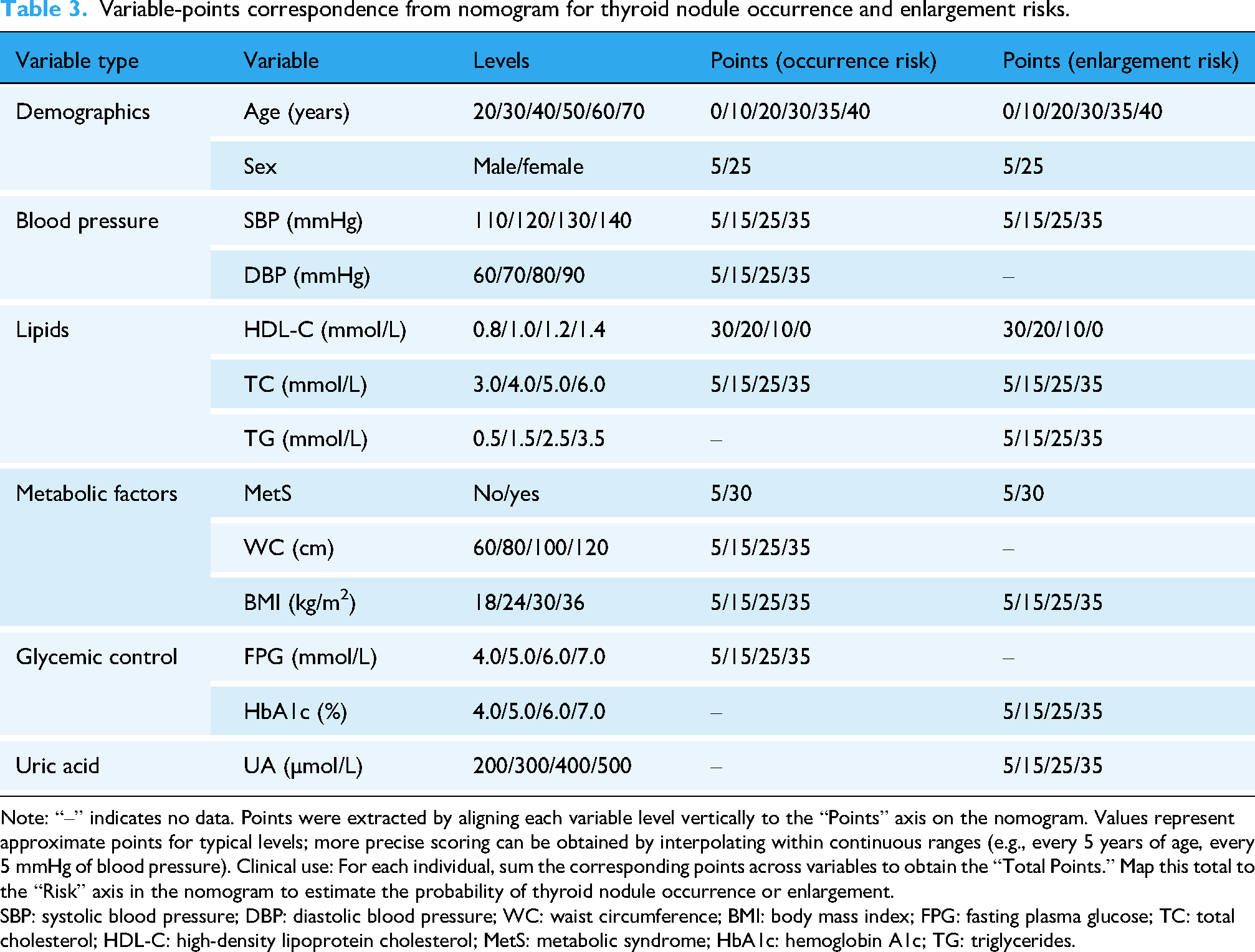
Note: “–” indicates no data. Points were extracted by aligning each variable level vertically to the “Points” axis on the nomogram. Values represent approximate points for typical levels; more precise scoring can be obtained by interpolating within continuous ranges (e.g., every 5 years of age, every 5 mmHg of blood pressure). Clinical use: For each individual, sum the corresponding points across variables to obtain the “Total Points.” Map this total to the “Risk” axis in the nomogram to estimate the probability of thyroid nodule occurrence or enlargement.
SBP: systolic blood pressure; DBP: diastolic blood pressure; WC: waist circumference; BMI: body mass index; FPG: fasting plasma glucose; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; MetS: metabolic syndrome; HbA1c: hemoglobin A1c; TG: triglycerides.
Calibration and clinical utility
Calibration curves with bootstrap correction demonstrated good agreement between predicted and observed risks for both TN occurrence and enlargement, particularly within the clinically relevant probability range (0.2–0.8), confirming reliable absolute risk estimation. DCA showed that the nomogram provided higher net benefit than “treat-all” or “treat-none” strategies across threshold probabilities of 0.05–0.6, supporting its potential clinical utility for guiding screening and intervention strategies (see Figure 4).

Calibration and decision curve analyses for predicting thyroid nodule and its enlargement. Calibration and decision curve analyses of thyroid nodule risk models. (A and B) Calibration plot and decision curve for the occurrence model. (C and D) Calibration plot and decision curve for the enlargement model. Calibration plots compare predicted versus observed probabilities, with the diagonal line indicating perfect calibration. Decision curves illustrate the net clinical benefit across threshold probabilities, showing that the developed models provide higher net benefit than the “treat none” or “treat all” strategies.
Discussion
This study compared baseline characteristics between individuals with and without TNs in a large cohort of euthyroid adults, thereby minimizing confounding by thyroid dysfunction. Consistent with prior reports, we observed a higher prevalence among females and significant differences across multiple metabolic parameters. Although BMI and WC did not differ significantly in the overall population, gender-stratified analyses revealed elevated adiposity measures in nodule-positive participants within both sexes. This gender-specific pattern in anthropometric indices may be explained by sex hormone-mediated differences in adipokine secretion and central metabolic sensitivity, as recent work demonstrates that estradiol regulates leptin signaling via hypothalamic Cited1 to modulate energy homeostasis. 20 These findings reinforce the intimate link between TN development and metabolic dysregulation. Elevated fasting glucose, 21 hypertension, diabetes, and dyslipidemia—particularly in men 22 —have been consistently associated with TNs, as also observed in a prior cross-sectional analysis of this cohort. 23 While low HDL-C and hypercholesterolemia have been linked to malignant thyroid disease,24,25 our results extend these associations to benign nodules in euthyroid individuals.
Using LASSO regression—a well-established machine learning technique for high-dimensional variable selection—to reduce model complexity and mitigate multicollinearity, we identified a core set of metabolic and anthropometric predictors for both incident TNs and their enlargement. Notably, although several factors overlapped between the two outcomes, MetS showed divergent associations: It was positively associated with nodule occurrence but inversely related to subsequent enlargement. This unexpected directionality suggests that MetS may primarily facilitate initial nodule formation—possibly through insulin resistance, chronic low-grade inflammation, and altered lipid metabolism 26 —without necessarily driving later growth. In contrast, HDL-C emerged as a consistent protective factor across both endpoints, plausibly reflecting its anti-inflammatory and antioxidant properties. 27 The inverse associations of HbA1c and FPG with nodule outcomes, although counterintuitive, may reflect detection bias, non-linear dose–response relationships, or biological heterogeneity among euthyroid individuals, and should therefore be interpreted with caution.
Our findings extend prior work by demonstrating that routinely available metabolic markers can inform longitudinal risk assessment even in the absence of thyroid dysfunction. Previous studies have linked MetS to TN prevalence,28,29 suggested WC may outperform BMI in risk prediction, 30 identified DBP as a risk factor in younger women, 31 and confirmed elevated risk among females, older adults, and individuals with central obesity or high fasting glucose in a large Southwest Chinese cohort. 32 For example, Li et al. 33 reported TG and UA as predictors of nodule enlargement, implicating oxidative stress-related mechanisms, while Zhao et al. 34 observed a stronger association between hyperuricemia and TNs in females. Unlike cross-sectional models such as that of Liu et al. 35 (AUC = 0.68), which focuses on prevalent nodules, our longitudinal approach predicts both incident nodule formation and subsequent growth using time-to-event modeling, enabling proactive risk stratification. To the best of our knowledge, this is the first longitudinal model to simultaneously address both outcomes using only routinely available clinical data in a euthyroid population.
The nomograms developed here translate the LASSO-Cox hybrid model into a practical tool for individualized risk communication. While LASSO handled variable selection, the Cox framework appropriately modeled time-to-event dynamics for nodule occurrence; for enlargement, we treated the first detection of growth as an event, acknowledging this as a pragmatic approximation of a continuous process. Although our model demonstrates modest discriminative performance, it is comparable to other metabolic risk scores in endocrine research. 36 Notably, its AUC is lower than that of image-based deep learning models—such as Weng et al., 14 who reported an AUC of 89.1% (0.891) for predicting TN malignancy, and Jin et al.'s Thy-Wise 15 an interpretable ultrasound-based random forest model that achieved high diagnostic accuracy (82.4%). Crucially, however, these high-performing AI models rely on expert ultrasound features and are designed for different clinical tasks (e.g., malignancy stratification or nodule characterization). In contrast, our model requires no imaging and operates in a euthyroid, general-population context, targeting a fundamentally different clinical question—early metabolic risk stratification rather than nodule characterization or malignancy prediction. Therefore, this model should be viewed as complementary to, rather than competing with, imaging-based artificial intelligence systems.
Despite these strengths, several limitations warrant caution. First, the single-center retrospective design may limit generalizability; multi-center prospective cohorts from diverse regions are needed. Second, although multiple imputation was applied to address missing baseline data, potential bias related to the missing-at-random assumption cannot be fully excluded. Third, causality cannot be inferred from observational data. Finally, external validation remains essential before clinical deployment.
Looking ahead, future work should integrate this metabolic risk profile with ultrasound features, thyroid autoantibodies, or inflammatory biomarkers to build a multimodal prediction system. Prospective validation across ethnicities and development of a digital health application for real-time risk calculation represent key steps toward precision prevention of TNs in at-risk euthyroid populations.
Conclusions
Female sex, low HDL-C, and MetS are key predictors of TN development and growth in euthyroid individuals. Using LASSO-Cox modeling on routine clinical data, we developed a practical risk stratification tool—modest in discrimination but potentially valuable in settings lacking imaging resources. As the first longitudinal model to jointly predict incident and enlarging nodules in this population, it bridges metabolic risk assessment and thyroid surveillance. Validation in diverse cohorts and integration with imaging or biomarker data will be crucial for translation into precision prevention strategies.
Footnotes
Acknowledgments
The authors thank Blackstone Studios Chengdu (Yunjian Technology) for their valuable contributions to data support and technical assistance. Additionally, the authors acknowledge the use of the generative AI tool ChatGPT to assist with language editing and improving the clarity of the manuscript. All AI-assisted content was critically reviewed and validated by the authors to ensure accuracy and integrity.
Ethical approval
This study was conducted in accordance with the principles of the Declaration of Helsinki (2013 revision). All procedures involving human participants were reviewed and approved by the Sichuan Provincial People's Hospital IRB (Approval No. 487, 2024). Informed consent was obtained from all participants, and data were processed in an anonymized manner to protect participant privacy. The authors confirm that no unapproved procedures involving human participants were conducted, and all ethical considerations (including data privacy, consent documentation, and participant protection) have been fully addressed in the conduct of this study.
Consent to participate
Written informed consent was obtained from all individual human participants included in this study. For participants unable to provide written consent due to physical or cognitive constraints, verbal informed consent was obtained in the presence of a neutral witness, with the witness signing a verification form to document the consent process. All consent procedures complied with the requirements of the Sichuan Provincial People's Hospital IRB and the Declaration of Helsinki.
Authors’ contributions
YT contributed to the acquisition of study data, including the retrieval and verification of clinical and laboratory information. HPY was responsible for data collation, preprocessing, and statistical analysis. FZD and LHL drafted the original version of the manuscript, including the formulation of the research framework and interpretation of the main findings. SP critically reviewed and edited the manuscript for important intellectual content and provided guidance on the organization and clarity of the text. All authors contributed to the conception and design of the study, approved the final version of the manuscript, and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The dataset used in this study is the property of Sichuan Provincial People's Hospital. De-identified data may be made available to qualified researchers upon reasonable request, pending approval by the hospital's Data Governance Committee and execution of a formal data use agreement. The R code used for LASSO-Cox modeling and nomogram development is available from the corresponding author upon reasonable request (email: 13689015288@163.com, hepeiyuan@med.uestc.edu.cn).
Clinical trial number
Not applicable. This study is a retrospective cohort study focused on analyzing existing health management data, not an interventional or observational clinical trial requiring registration.
Guarantor
He Peiyuan (hepeiyuan@med.uestc.edu.cn) and Fan Zhongdou (13689015288@163.com) are the guarantors of this work. They had full access to all the data in the study, take responsibility for the integrity of the data and the accuracy of the data analysis, and ensure that all aspects of the study comply with ethical and academic standards.
