Abstract
Background:
This study aimed to examine severe bone marrow suppression risk factors during radiotherapy in patients with cervical cancer and to develop and validate a visual evaluation tool for predicting the risk of severe bone marrow suppression during radiotherapy in these patients.
Methods:
A total of 300 patients with cervical cancer who underwent radiotherapy were retrospectively included in this cohort study. Patients were randomly divided into a model group (n = 240) and a validation group (n = 60) at a ratio of 8:2. Univariate and multivariate logistic regression analyses were performed to explore and establish a nomogram prediction model. The feasibility of this nomogram model in predicting the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer was assessed in the validation cohort. The discrimination ability, accuracy, and clinical utility of the model were evaluated via receiver operating characteristic (ROC) curves, calibration curves, and decision curve analysis (DCA).
Results:
Menopausal status, Karnofsky performance score (KPS), clinical stage, concurrent chemotherapy status, and pre-radiotherapy creatinine level were identified as independent risk factors for severe bone marrow suppression during radiotherapy in patients (P < .05). DCA revealed that the nomogram model had a greater net benefit in predicting the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer when the patient’s threshold probability was between 0.20 and 0.93.
Conclusion:
The nomogram model based on these independent risk factors exhibited good predictive performance, assisting in individualized risk assessment and facilitating early intervention to benefit patients during radiotherapy.
Introduction
Cervical cancer, one of the four most common malignant tumors in women globally, accounts for 6.5% of the total incidence and 7.7% of the mortality of female cancers.1,2 Radiotherapy is an essential treatment modality for cervical cancer because it enhances local tumor control and improves patient prognosis. Although radiotherapy has therapeutic benefits, it is frequently associated with several acute side effects, with suppression of the bone marrow being one of the most common adverse reactions. 3 Bone marrow suppression is evident primarily as a reduction in blood cell counts, including white blood cell (WBC) counts, blood platelet (PLT) counts, red blood cell counts, and hemoglobin (Hb) levels. Bone marrow suppression during radiotherapy results in a decline in patients’ physical function and impairs their ability to tolerate further treatment, potentially leading to treatment disruption and adversely affecting the overall effectiveness of radiotherapy. 5,6 The incidence of bone marrow suppression is relatively high, with its severity being closely related to factors such as the dose of radiotherapy, irradiated volume, and individual patient differences in patients with cervical cancer.4,5 This study aims to analyze clinical risk factors (such as age, body mass index [BMI], Karnofsky performance score [KPS], pathological stage, surgical history, chemotherapy status, and pre-radiotherapy creatinine levels) and radiotherapy factors (such as immobilization methods, radiotherapy dose per fraction, irradiation of para-aortic lymph nodes, and dose and volume of radiation to the spinal cord and pelvic bones) to establish a prediction model for severe bone marrow suppression risk during radiotherapy in patients with cervical cancer. The goal is to provide scientific evidence for clinical practice, enabling early intervention during radiotherapy, assisting in the regulation of treatment strategies, improving patient compliance, reducing the incidence of bone marrow suppression, and ultimately benefiting patients.
Materials and Methods
Case enrollment and exclusion criteria
This retrospective cohort study was performed on patients with cervical squamous cell carcinoma who underwent radiotherapy from August 2020 to February 2023 at the Sun Yat-sen University Cancer Center. According to the inclusion and exclusion criteria, a total of 300 patients were selected from records for the study. The identified patients were then randomly divided into model and validation groups at a ratio of 8:2. The model group (n = 240) and the validation group (n = 60) were used to construct the prediction model and assess its performance, respectively. The ages of the individuals in the model and validation groups ranged from 26 to 78 years and 34 to 74 years, respectively. There were no statistically significant differences in clinical characteristics or bone marrow suppression between the two groups. All identified patients received chemotherapy based on platinum drugs. The reporting of this study adheres to the STROBE guidelines (Supplementary File 1).
The inclusion criteria were as follows:
The attending physician assessed liver and kidney function and routine blood investigations before radiotherapy;
Patients who had good compliance with radiotherapy;
Patients who successfully completed all treatments with comprehensive clinical data.
The exclusion criteria were as follows:
Severe liver or kidney dysfunction;
Concurrent autoimmune or hematologic diseases;
Concurrent psychiatric disorders hamper the cooperation of patients with clinical treatment;
A history of prior radiotherapy;
Incomplete clinical data.
The research strictly adhered to the ethical principles outlined in the Declaration of Helsinki (revised in 1975). Ethics Committee of the Sun Yat-sen University Cancer Center approved this study.
Positioning and immobilization
The patients were instructed to raise both arms and lie supine on the vacuum bag in the vacuum bag immobilization group. The pelvic region was fixed after the air was evacuated to mold the bag. The patients lay prone on the Orfit frame with both arms raised in the Orfit frame + body membrane mask immobilization group. A thermoplastic mask was used to fix the pelvic region.
CT scanning and target delineation
A Philips 16-slice large-aperture spiral CT simulator (Philips, Netherlands) with a slice thickness of 3 mm was used to perform positioning. Patients were instructed to retain urine before the positioning scan, and the bladder volume was measured using a bladder measurement device, which required a volume between 200 and 350 mL. The scanning range extended from the lower edge of the 10th thoracic vertebra to the lower edge of the ischial tuberosity. The Monaco treatment planning system (version 5.11.03, Elekta) was used to perform target delineation and normal organ contouring. The target volumes were delineated according to the American Radiation Oncology Group consensus on target delineation for cervical cancer, as recommended by experienced radiation oncologists. The clinical target volume encompasses the uterus, cervix, parametrium, upper two-thirds of the vagina, and pelvic lymphatic drainage areas, including the internal iliac, obturator, external iliac, and common iliac lymph nodes. The organs at risk included the pelvis, spinal cord, rectum, bladder, small intestine, colon, and kidneys.
Data collection and processing
The medical record system was used to obtain data on age, height, weight, KPS, pathological stage, surgical history (whether surgery was performed), chemotherapy status (divided into non-concurrent and concurrent chemotherapy groups based on chemotherapy timing), and pre-radiotherapy creatinine levels. BMI was calculated using the following formula: BMI = weight (kg)/height² (m²).
The MOSAIQ Integration Platform 2.0 was used to obtain data regarding the immobilization method, irradiation of para-aortic lymph nodes, single radiation dose (with doses of 1.8 and 2.4 Gy), and dose to critical organs (such as the spinal cord and pelvis). The evaluation parameters for critical organs included the following spinal cord and pelvic bone doses: Dmin, Dmax, Dmean, V10, V20, V30, V40, and V50.
Routine blood tests were monitored weekly during radiotherapy. Daily monitoring was conducted if the blood counts demonstrated a significant decline. The Hb, neutrophil (NE), WBC, and BLT values were recorded. Based on the National Cancer Institute-Common Terminology Criteria for Adverse Events version 4.0, hematologic toxicity was graded with grades 0-II, indicating mild suppression, and grades III-IV, indicating severe suppression. The degree of bone marrow depression was classified according to the most severe level of toxicity among Hb, NE, WBCs, or BLT. The thresholds for severe inhibition of Hb, NE, WBCs, and BLT were not higher than 75 g/L, 0.9 × 10⁹/L, 1.9 × 10⁹/L, and 49 × 10⁹/L, respectively.
Statistical methods
IBM SPSS Statistics 28.0 software was used to perform the statistical analysis. The median (interquartile range) was used for non-normally distributed continuous data, whereas categorical data were expressed as frequencies (percentages) (n [%]). For intergroup comparisons, independent samples non-parametric tests were used. Multivariate logistic regression analysis was used to identify factors influencing the risk of severe bone marrow suppression during radiotherapy. Based on the selected variables, a nomogram risk prediction model was constructed. The model’s predictive performance was validated via receiver operating characteristic (ROC) curves. Statistical significance was set at P < .05.
Results
Analysis of factors affecting the risk of severe bone marrow suppression
Among 240 patients with cervical cancer, 55 developed severe bone marrow suppression during radiotherapy. The incidence rate was 22.92%. Further evaluation revealed that 34 patients had severe leukocytotoxicity, 10 had severe Hb toxicity, 17 had severe neutropenia, and 14 had severe thrombocytopenia (Figure 1). When the clinical risk and radiotherapy factors were compared between the severe bone marrow suppression (n = 55) and non-severe bone marrow suppression groups (n = 185), statistically significant differences were revealed in menopausal status; KPS; clinical staging; concurrent chemotherapy; pre-radiotherapy creatinine levels; V10, V20, and V30 of the pelvic bone; and V10, V20, V30, and V40 of the spinal cord (P < .05). No statistically significant differences in factors such as the fixation device; single radiation dose; irradiation of para-aortic lymph nodes; BMI; Dmin, Dmean, Dmax, V40, or V50 of the pelvic bone; or Dmin, Dmean, Dmax, or V50 of the spinal cord (P > .05) were noted (Tables 1 and 2).

Incidence of severe bone marrow suppression and hematologic toxicity during radiotherapy in 240 patients. (kps: Karnofsky performance score).
Analysis of factors affecting the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer (clinical risk factors) (n [%], M [IQR]).

Analysis of the risk of severe myelosuppression during radiotherapy in patients with cervical cancer (radiotherapy factors) (n [%], M [IQR]).

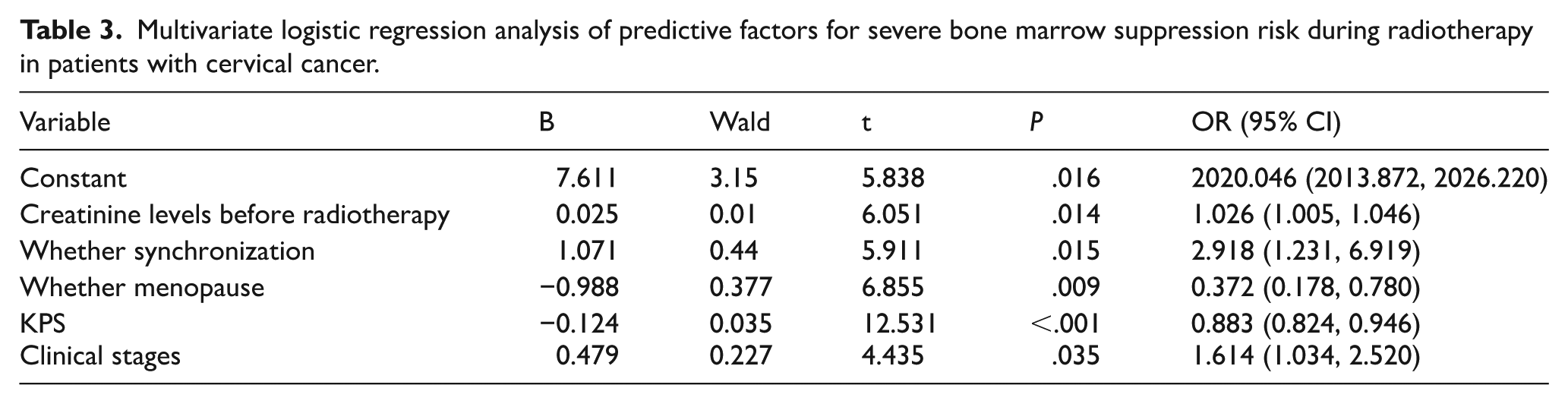
Multivariate logistic regression analysis of predictive factors for severe bone marrow suppression risk
The significant variables in the univariate analysis were included in the multivariate logistic regression analysis. The results indicated that the independent predictive factors for the risk of severe bone marrow suppression are menopausal status, KPS, clinical stage, concurrent chemotherapy, and pre-radiotherapy creatinine levels (Table 3).
Multivariate logistic regression analysis of predictive factors for severe bone marrow suppression risk during radiotherapy in patients with cervical cancer.
Establishment and validation of the nomogram model for predicting the risk of severe bone marrow suppression
A nomogram model was established based on the five independent predictive factors identified in the multivariate logistic regression analysis to predict the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer (Figure 2). The validation results of the nomogram model demonstrated that the area under the ROC curve (AUC) for the model group was 0.759 (95% confidence interval [CI] [0.690–0.828]), with a diagnostic cutoff, sensitivity, and specificity of 0.308, 56.4%, and 82.7%, respectively. The ROC curve AUC for the validation group was 0.741 (95% CI [0.574–0.909]), with a diagnostic cutoff of 0.081, a sensitivity of 100.0%, and a specificity of 47.2% upon validation with an external dataset (Figure 3). The AUC for the validation group was 0.741, whereas it was 0.759 for the model group, with a difference of 0.018. These findings indicate good discriminatory power of the prediction model for the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer in both the model and validation groups.

Nomogram for predicting the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer. (A) Receiver operating characteristic (ROC) curve of the model group. (B) ROC curve of the validation group.

Receiver operating characteristic (ROC) curve of the nomogram model for predicting the risk of severe bone marrow suppression. (A) Model group. (B) Verification group.
Calibration curve for the risk of severe bone marrow suppression
The calibration curve of the nomogram model for the model group and Hosmer–Lemeshow goodness-of-fit test indicated that the model was not significant (x² = 8.814, P = .358). This exhibited good agreement between the predicted and actual outcomes. Good concordance between the predictions and observations was demonstrated through validation via data from the validation group. The Hosmer–Lemeshow goodness-of-fit test for the validation group did not demonstrate any significance (x² = 10.311, P = .244), further confirming that the model aligned well with the actual outcomes (Figure 4).

The calibration curve of the nomogram prediction model for predicting the risk of developing severe myelosuppression. (A) Model group. (B) Verification group.
Decision curve analysis (DCA) for the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer in the model and validation groups
Using the variables selected by the nomogram prediction model, the decision curve for the risk of severe bone marrow suppression during radiotherapy was plotted. The DCA demonstrated that using the nomogram model to predict the risk of severe bone marrow suppression resulted in greater net benefit when the threshold probability for patients was between 0.20 and 0.93 (Figure 5).

Decision curve analysis (DCA) of the nomogram model for predicting the risk of severe bone marrow suppression (model group).
Discussion
A common adverse effect during radiotherapy in patients with cervical cancer is severe bone marrow suppression.6,7 The goal of radiotherapy is to predict the risk of severe bone marrow suppression and reduce its incidence during treatment throughout the process. This study aimed to establish a prediction model for bone marrow suppression during radiotherapy in patients with cervical cancer, evaluate the risk of severe bone marrow suppression, and identify high-risk groups for bone marrow suppression. These findings provide a theoretical foundation for efficiently reducing the risk of severe bone marrow suppression in these targeted groups.
Bone marrow suppression is closely related to the radiation dose and volume received by the pelvic region during cervical cancer radiotherapy. Studies have demonstrated that the incidence of severe bone marrow suppression is correlated with the dose and volume of the irradiated pelvis. 8 Similarly, our study found that the V10, V20, and V30 volumes of the irradiated pelvis were positively correlated with the occurrence of severe bone marrow suppression, indicating that pelvic V10, V20, and V30 are significant factors influencing the occurrence of severe bone marrow suppression in patients with cervical cancer undergoing intensity-modulated radiotherapy (IMRT). Thus, effectively reducing the radiation volume and dose to the pelvis can decrease the incidence of severe bone marrow suppression, which has positive clinical implications.9-12
In addition, the literature regarding the impact of the radiation dose to the spinal cord on severe bone marrow suppression is limited. This study revealed a positive correlation of V10, V20, V30, and V40 of the spinal cord with severe bone marrow suppression. These findings indicate that the V10, V20, V30, and V40 of the spinal cord also influence severe bone marrow suppression in patients with cervical cancer undergoing IMRT. The spinal cord is an essential radiation-sensitive organ. During radiotherapy, the radiation field includes the spinal cord, which increases the risk of neurological damage and affects nearby bone marrow tissue, potentially intensifying hematopoietic dysfunction and further aggravating the suppression of bone marrow. 13
Based on both univariate and multivariate logistic regression, this study revealed that the independent factors influencing the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer are pre-radiotherapy creatinine levels, concurrent chemoradiotherapy (CCRT), menopausal status, KPS, and clinical staging.
BMI reflects nutritional status and chronic inflammation, both of which may influence bone marrow resilience during radiotherapy. A low BMI (<18.5 kg/m²) is associated with a reduced hematopoietic reserve due to malnutrition, whereas a high BMI (⩾ 25 kg/m²) is correlated with pro-inflammatory cytokines (e.g., interleukin 6 and tumor necrosis factor alpha) that impair hematopoietic recovery.14,15 Gupta et al 16 reported that a low BMI increased the risk of hematologic toxicity during chemoradiation in patients with cervical cancer (OR = 1.8, P = .03). Although BMI did not reach statistical significance in our final model (possibly due to sample size limitations), its clinical relevance warrants consideration in risk assessment frameworks.
Several domestic and international studies12,17,18 have shown that CCRT is an important factor affecting the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer, and the results of this study are consistent with those findings. Compared with radiation therapy alone, CCRT improves local control and survival rates, but also increases the incidence of severe bone marrow suppression. Common chemotherapy drugs, such as cisplatin, temporarily damage the hematopoietic function of the bone marrow. Previously, quiescent cells entered the proliferative phase, and bone marrow function gradually recovered to normal when the drug lost its effect through metabolism or excretion.12,18
In addition, this study demonstrated that pre-radiotherapy creatinine levels serve as an independent risk factor for severe bone marrow suppression. A positive correlation was observed between elevated creatinine levels prior to radiotherapy and the increased risk of developing severe bone marrow suppression. Consistent with these findings, Yan and Yawei, 19 through a retrospective analysis, reported a significantly higher incidence of grade 3–4 bone marrow suppression among patients with pre-radiotherapy creatinine levels ⩾ 65 μmol/L. Moreover, higher pre-radiotherapy creatinine levels were found to be an independent factor influencing the risk of severe bone marrow suppression during treatment. The pre-radiotherapy creatinine level is related to the rate of creatinine clearance, which can moderately reflect glomerular filtration function. A decrease in glomerular filtration function slows the excretion of chemotherapy drugs, thereby increasing the incidence of bone marrow suppression. 20 Previous studies have demonstrated21,22 that postmenopausal patients with cervical cancer tend to experience a greater incidence of bone marrow suppression during radiotherapy, following the findings of this study. This may be associated with a decrease in estrogen levels after menopause. Estrogen has a moderate influence on the hematopoietic system, exerting various regulatory effects on bone marrow hematopoiesis, including the promotion of hematopoietic stem cell proliferation and differentiation, as well as the regulation of immune cell function. Insufficient estrogen levels may result in a decline in bone marrow hematopoietic function, affecting blood cell production and impairing the repair of active bone marrow. 21
This study demonstrated that clinical stage is an independent factor influencing the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer. Previous studies have demonstrated 22 that patients with advanced-stage cervical cancer who undergo CCRT have a significantly greater incidence of bone marrow suppression than those with early-stage cancer. The clinical staging of cervical cancer is an important basis for evaluating the severity of the disease and determining treatment plans. Moreover, patients with different clinical stages demonstrate variations in the incidence and severity of bone marrow suppression during radiotherapy.
Studies have shown15,23,24 that KPS is a major factor influencing the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer. In this study, the KPS was also an independent factor influencing the risk of severe bone marrow suppression. The KPS is a tool used to evaluate the overall health and functional status of patients with cancer, and it is often employed to predict survival and treatment response. Patients with lower KPS scores may have a certain degree of physical weakness or comorbidities, making their bone marrow suppression more pronounced and leading to more severe adverse reactions during treatment. KPS scores can help predict patients’ tolerance to cervical cancer radiotherapy and the potential for severe bone marrow suppression during treatment. Therefore, doctors should consider the patient’s KPS score, age, overall condition, and other relevant factors when formulating a radiotherapy plan to optimize the treatment approach.
Nomogram models are widely used in clinical practice, particularly for analyzing treatment efficacy and survival rates.25-28 There has been a growing trend of applying these models to predict adverse effects during radiotherapy. 25 ,29-31 These models exhibit high predictive efficacy and can serve as valuable guides in clinical settings. Our nomogram model demonstrated robust predictive performance, with its primary clinical value residing in the facilitation of personalized interventions for high-risk patients. Numerous studies have highlighted the potential of risk prediction models to enhance clinical decision-making in patients undergoing radiotherapy for cervical cancer. For instance, Li et al 4 implemented a bone marrow-sparing radiotherapy (BMSRT) approach guided by pretreatment risk stratification, achieving a significant reduction in grade 3–4 hematologic toxicity from 46% to 28% (P < .01) without compromising target volume coverage. Similarly, Fullerton et al 5 employed a nutritional-inflammation risk model to tailor supportive care measures, including prophylactic administration of granulocyte-colony stimulating factor (G-CSF) and nutritional supplementation, which led to a 35% reduction in treatment interruptions among high-risk patients. Based on independent factors selected from multivariate logistic regression analysis, this study developed a nomogram model to predict the risk of severe bone marrow suppression during radiotherapy for patients with cervical cancer. The ROC curve results demonstrated that the model had high predictive performance. The nomogram model incorporates multiple predictive indicators, transforming complex regression equations into simple and visual graphs, thereby facilitating clinicians’ ability to assess patients.
Additionally, it provides personalized risk assessments and treatment recommendations, thereby enhancing the quality and efficiency of clinical decision-making. This reduces the risk of severe bone marrow suppression during radiotherapy, ensuring that treatment proceeds as scheduled. The AUC was 0.759 in the model group. When the prediction model was externally validated, both the ROC and calibration curves demonstrated good performance, indicating that this model is an effective tool for predicting the occurrence of bone marrow suppression during radiotherapy in patients with cervical cancer.
As a single-arm cohort analysis, this study was limited in its ability to control for all potential confounding variables through comparison with a control group. Future research should address the following considerations: (1) pretreatment hematologic reserves, including baseline NE counts; (2) distribution of functional bone marrow, such as ABMV assessed via SPECT or MRI; and (3) comorbidities that influence drug metabolism, particularly renal dysfunction extending beyond elevated creatinine levels. Accounting for these variables will enhance the ability to delineate the causal relationships between identified predictors—such as creatinine and KPS) and the development of severe bone marrow suppression.
The impact of severe bone marrow suppression on prognosis has not been analyzed in depth because long-term follow-up is still ongoing, and the data are not yet complete. Thus, this topic will be the focus of future research. The study revealed that the volume of the irradiated pelvis and spinal cord was associated with the occurrence of severe bone marrow suppression during radiotherapy in patients with cervical cancer; however, it was not an independent predictive factor. Future research should focus on integrating radiomics and dosimetry to explore this topic. In addition, the study has several limitations, including a relatively small sample size and its single-center design, this may affect the statistical. results. Therefore, multicenter studies should be conducted in the future with larger sample sizes to improve the predictive accuracy of the model.
In conclusion, the volume of the irradiated pelvis (V10, V20, and V30) and spinal cord (V10, V20, V30, and V40) influences the risk of severe bone marrow suppression during radiotherapy in patients with cervical cancer. The pre-radiotherapy creatinine level, CCRT, menopausal status, KPS, and clinical stage were found to be independent factors in the nomogram model for predicting severe bone marrow suppression. Based on these indicators, the nomogram model can provide a quantitative assessment of the risk of severe bone marrow suppression with high predictive efficacy and significant clinical application.
Conclusion
This study successfully constructed and validated a nomogram for predicting severe bone marrow suppression risk during cervical cancer radiotherapy. Five independent predictors were identified for multivariate analysis: menopausal status, KPS, clinical stage, concurrent chemotherapy, and pre-radiotherapy creatinine levels. Moreover, these indicators are available prior to treatment and can be utilized to identify high-risk groups before the initiation of radiotherapy, thereby allowing a window of opportunity for intervention. The model demonstrated robust discriminative ability (AUC: 0.759 in the training cohort; 0.741 in the validation cohort), good calibration (Hosmer–Lemeshow P > .05), and significant clinical utility according to DCA (net benefit threshold: 20%–93%). Additionally, the pelvic bone (V10–V30) and spinal cord (V10–V40) irradiation volumes were correlated with the risk of bone marrow suppression. This tool enables personalized risk stratification, facilitating early interventions such as BMSRT or supportive care optimization. Implementation may reduce treatment interruption, improve patient compliance, and enhance radiotherapy outcomes in patients with cervical cancer.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251380662 – Supplemental material for Construction of a Risk-Prediction Model for Severe Bone Marrow Suppression During Radiotherapy in Cervical Cancer Patients
Supplemental material, sj-docx-1-onc-10.1177_11795549251380662 for Construction of a Risk-Prediction Model for Severe Bone Marrow Suppression During Radiotherapy in Cervical Cancer Patients by Zongtai Li, Zhiyue Lin, Peishan Qin, Runnan Xiao, Jindi Liu, Wenlong Zhu, Senkui Xu, Huilang He and Jiaxiu Luo in Clinical Medicine Insights: Oncology
Footnotes
Ethical Considerations and Consent to Participate
This study was approved by the Ethics Committee of Sun Yat-sen University Cancer Center, and ethics approval was obtained on 12/10/2024 (approval no. B2024–690–01). In accordance with the Declaration of Helsinki, all methods were carried out in accordance with relevant guidelines and regulations. This study has obtained the exemption of patient informed consent from the ethics committee of Sun Yat-sen University. All patients were selected according to the Case Enrollment and Exclusion Criteria.
Consent for Publication
Not applicable.
Author Contributions
Jiaxiu Luo, Zongtai Li, and Zhiyue Lin made a substantial contribution to the concept or design of the work. Peishan Qin, Runnan Xiao, Jindi Liu, and Wenlong Zhu were involved in the acquisition, analysis, or interpretation of data. Huilang He and Senkui Xu guided the data analysis and processing. Jiaxiu Luo and Zongtai Li drafted the article or revised it critically for important intellectual content. All authors read and approved the final article. Each author should have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
