Abstract
Introduction
To assess the impact of cisplatin cycles on overall survival (OS) and disease-free survival (DFS) in cervical cancer (CC) patients undergoing concurrent chemoradiotherapy (CCRT), and to build a nomogram-based prognostic stratification to identify CC patients who might benefit from ≥ 5 cycles of cisplatin.
Methods
This study retrospectively analyzed data from 918 patients who were treated with external beam radiotherapy and brachytherapy. Weekly cisplatin was the concurrent regimen. The difference in survival outcomes between the < 5 cycles and ≥ 5 cycles groups were compared. Subgroup analysis was further conducted. Univariate and multivariate Cox regression analyses were performed in the <5 cycles, and a nomogram was developed accordingly. The patients were divided into two risk subgroups and the survival outcomes were compared.
Results
The 5-year OS and DFS were 76.7% and 86.1% (p = 0.002), 68.7% and 78.3% (p = 0.0016) for the < 5 and ≥ 5 cycles, respectively. In subgroup analysis, the survival benefit of ≥ 5 cycles could be maintained in patients with > 50 years old, squamous disease, squamous cell carcinoma antigen (SCC Ag) > 1.5 ng/mL, tumor size > 4 cm, International Federation of Gynecology and Obstetrics (FIGO) stage I/II, and no para-aortic metastatic lymph nodes, whereas no significant benefit was observed in FIGO stage III/IV. A nomogram incorporating size, SCC Ag, and stage was constructed, and patients were divided into two risk groups (low-risk and high-risk). Receiving ≥ 5 cycles showed superiority in OS (p = 0.0025) and DFS (p = 0.008) over < 5 cycles in the low-risk subgroup.
Conclusion
Receiving ≥ 5 cycles of cisplatin was associated with OS and DFS in patients with CC who received CCRT. A nomogram was constructed and the newly defined low-risk patients might gain significant OS and DFS benefit from receiving ≥ 5 cycles
Introduction
Cervical cancer (CC) is one of the most common malignancy of the female reproductive system; it is highly prevalent in developing countries. 1 Concurrent chemoradiotherapy (CCRT) is one of the treatment options for CC. 2 Several large randomized trials have demonstrated that cisplatin-based CCRT can improve the survival rate of patients with CC than radiation therapy alone.3,4 Weekly cisplatin is widely accepted as the standard chemotherapy regimen owing to its convenience and effectiveness. 5
Although cisplatin-based CCRT remains a cornerstone of treatment for locally advanced cervical cancer (LACC), contemporary systemic treatment strategies increasingly incorporate immunotherapy in selected populations. For high-risk LACC, the addition of pembrolizumab to CCRT has shown significant survival benefit and has been incorporated into the contemporary treatment paradigm.6,7 Meanwhile, other primary treatment strategies, including neoadjuvant chemotherapy followed by surgery and induction chemotherapy followed by CCRT, have also been investigated in selected settings and may further broaden therapeutic options for CC management.8-10
In resource-constrained settings and regions with limited healthcare infrastructure, CCRT remains the most widely adopted standard treatment for patients with LACC. 11 However, the optimal number of cisplatin cycles administered during CCRT continues to be a clinically important and unresolved question. Cisplatin has dose-limiting hematological, gastrointestinal, and nephrotoxicity. 12 Currently, the recommended dose of cisplatin for CCRT is 40 mg/m2 per week, 13 however, there is currently little consensus regarding the optimal number of cisplatin dosage cycles for CCRT. 14 A previous study demonstrated that five or more cycles of weekly cisplatin resulted in survival benefits in elderly patients with cervical cancer. 15 In addition, our preliminary study found that receiving ≥ 5 cycles of cisplatin may be required for clinical benefit. The optimal number of cycles of cisplatin during CCRT seemed to be five. 16
The objective of this study was to further confirm the prognostic value of ≥ 5 cycles of cisplatin during CCRT for CC in long-term follow-up. Furthermore, subgroup analyses were conducted to assess the value of cisplatin cycles in different patients with CC. Additionally, we attempted to predict the beneficiary population using a nomogram.
Materials and Methods
Patient Cohort
From Jan 2011 to Dec 2015, the medical records of cervical cancer patients treated with CCRT in our hospital were retrospectively reviewed. The study included patients who met the following eligibility criteria:1) histologically confirmed CC by punch biopsy, cone biopsy, and endocervical curettage; 2) Federation International of Gynecology and Obstetrics (FIGO) stage IB-IVA; 3) Karnofsky’s performance score > 70; 4) the completion of CCRT; 5) weekly cisplatin as chemotherapy regimen. Patients would be excluded when the following criteria were met: 1) patients with Karnofsky’s performance score ≤ 70; 2) patients received prior chemo or radiation therapy before CCRT; 3) patients underwent hysterectomy at any period of treatment; 4) cisplatin was not subscribed as the chemo regimen during CCRT; 5) patients diagnosed with organ dysfunction; 6) patients with history of malignant tumors; 7) patients without complete follow-up information. All patients were reclassified based on the 2018 FIGO staging system, utilizing clinical evaluations and imaging results. Eligible patients who met the predefined inclusion and exclusion criteria during the study period were consecutively enrolled to minimize selection bias. Patient data were anonymized and de-identified prior to analysis to ensure confidentiality. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 17
This study was approved by the institutional ethics committee of the participating hospital. The requirement for informed consent was waived due to the retrospective nature of the study. The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024).
Radiotherapy
The delineation of the clinical tumor volume (CTV) was based on imaging results and clinical findings. The CTV general encompassed the primary tumor, cervix, uterus, paracervical tissues, part of the vaginal canal, and pelvic lymphatic drainage regions. Patients with positive common iliac or para-aortic lymph nodes underwent expanded-field radiotherapy. In cases where the primary tumor extended to the lower 1/3 of the vagina, the CTV incorporated the inguinal lymphatic area. The planning target volume (PTV) was created by enlarging the CTV by 0.7–1.0 cm uniformly, with an additional 0.5–1.0 cm of margin on the uterus and cervix. Metastatic lymph nodes (MLNs) in imaging were defined as lymph nodes with a short diameter ≥1 cm on computed tomography (CT) and magnetic resonance imaging. 16 MLNs were also identified based on 18F-fuorodeoxyglucose positron emission tomography/computed tomography, as previously described. 18 Notably, the MLNs would be covered by gross tumor volume nodes, which generated planning target volume nodes (PTVn) through 0.5 cm external expansion. Prescribed dosage for PTV was 50.4 Gy with 28 fractions while 59-61 Gy was delivered for PTVn with simultaneous integrated boost. The external beam radiation therapy (EBRT) was delivered using 6MV-X rays in a linear accelerator. Image guidance was provided using either daily megavoltage CT or weekly cone-beam CT/CT-on-rail. To match tumor regression during treatment, a second CT simulation and radiotherapy planning session were performed for all patients. Typically, the second simulation and planning were performed after approximately 20 fractions of EBRT. If significant tumor regression was observed on images before the completion of 20 fractions, a second CT simulation was performed.
Brachytherapy using 192Ir was administered 3–4 weeks after the start of EBRT. One to two fractions of brachytherapy was delivered in this period; however, administration of EBRT and brachytherapy on the same day were not permitted. The rest fractions of the brachytherapy was performed after the end of EBRT without concurrent chemotherapy. Before December 2012, our institute followed a standard brachytherapy protocol of 36 Gy delivered in 6 fractions to point A. We reduced the brachytherapy dose to 30 Gy administered in 5 fractions from January 2013.
Chemotherapy
Within days 1 through 5 after the first radiation fraction was administered, patients would receive the first cycle of chemotherapy. Complete blood cell counts and renal functions were monitored at least once every week during active treatment. The additional cycles of chemotherapy were repeated weekly. Concurrent chemotherapy typically continued until the end of EBRT. If the patient experienced unacceptable side effects or requested to stop treatment, the concurrent chemotherapy was either delayed or discontinued. EBRT continued if cisplatin was withheld. For patients who had already received six cycles of chemotherapy, if EBRT was ongoing, an additional cycle of chemotherapy would be administered concurrently; if EBRT had been concluded, no additional chemotherapy would be given.
The most common cisplatin-based chemotherapy with 40mg/m2 was weekly delivered during the radiotherapy. For a minority of patients exhibiting weakness, comorbidities, or malnutrition, cisplatin at a weekly dosage of 30 mg/m2 was conducted. Furthermore, no additional chemotherapy regimens were administered.
Study Covariates and Cohort Definition
The information on patients’ age, histology of disease, FIGO stage, tumor size, pr\etreatment squamous cell carcinoma antigen (SCC Ag), pelvic MLNs, para-aortic MLNs, and cycles of cisplatin during CCRT, survival time and vital status were collected and analyzed. Of note, age was dichotomized by X-tile software (version 3.6.1; Yale University, New Haven, CT, USA). 12 Meanwhile, the tumor size was divided into two subgroups (≤4cm VS > 4cm) according to the staging scheme of National Comprehensive Cancer Network guidelines 2 while pretreatment SCC Ag were dichotomized as > 1.5ng/ml and ≤ 1.5ng/ml, which was recognized as the threshold value to differentiate normal from abnormal SCC Ag level.
The included patients were divided into two groups based on the number of chemotherapy cycles they received during CCRT, specifically, patients were assigned into < 5 cycles and ≥ 5 cycles group according to the previous research from our team. 10
Comparison of Survival Outcomes Between the <5 Cycles and ≥5 Cycles Group in Different Subgroups
The primary end point compared between the <5 cycles and ≥5 cycles group were overall survival (OS) and disease free survival (DFS), which were defined as the time from diagnosis to the day of death or last follow-up and the period from treatment to the date of recurrence, metastases, death or last follow-up, respectively. Any loss to follow-up was treated as a censored observation in our analysis. The OS, DFS curves would be plotted and compared between the <5 cycles and ≥5 cycles group using the Kaplan–Meier method and Log-rank test, respectively, in different subgroups including age, histology, tumor size, SCC Ag, FIGO stage, pelvic MLNs, and para-aortic MLNs.
Comparison of Adverse Effects Between the <5 Cycles and ≥5 Cycles Group
Treatment-related adverse effects were graded according to the Common Terminology Criteria for Adverse Events 5.0. Acute toxicity was defined as adverse events occurring during treatment or within 3 months after treatment initiation, whereas late toxicity was defined as adverse events occurring more than 3 months after treatment initiation.
Nomogram to Select Patients Benefiting From <5 Cycles Chemotherapy
The patients in the <5 cycles group were divided into training and validation cohort with a ratio of 7:3. The training cohort was used to identify variables and develop the model. The validation cohort was used to test and validate the performance of the model obtained using the training cohort. 19
Univariate and multivariate Cox regression analysis were performed in the training cohort. Variables with P < 0.05 in univariate Cox regression analysis were entered into the multivariate Cox regression model, and the identified independent risk factors were subsequently incorporated into the development of the predictive nomogram. Of note, the model was constructed based on the <5 cycles population, which was consistent with the theory proposed by Coate et al; according to the theory, the untreated or less treated patients were the optimal population to determine risk factors. 20 The C-index was estimated to evaluate the predictive ability of the model. Additionally, The calibration curves were plotted both in the training and validation cohort to visualize the relationship between predictive and observed survival probability, where the closer was the predictive curve to the observed curve, the better was the predictive accuracy of the nomogram.
The X-tile software was used to identify the optimal cut-off value to stratify the patients into low-risk and high-risk group based on the total points derived from the nomogram. The OS and DFS in each risk subgroup were compared between the <5 cycles and ≥5 cycles group.
Statistical Analysis
Categorical variables were listed as frequencies or percentages while medians with interquartile ranges (IQR) was used to describe continuous variables. Categorical and continuous variables would be compared using the Pearson χ2 test and Wilcoxon test, respectively. Standardized mean differences (SMDs) were calculated to assess baseline balance between the <5 cycles and ≥5 cycles groups, with |SMD| < 0.10 indicating negligible imbalance. All analyses were performed using the R software (version.3.6.1; https://www.r-project.org). A two-tailed p < 0.05 was considered as statistically significant. All statistical procedures, variable definitions, and modeling steps have been described in sufficient detail to allow independent replication.
Results
Patient Characteristics
Demographic and Clinico-Pathological Characteristics in <5 Cycles and ≥5 Cycles Group

FIGO, Federation International of Gynecology and Obstetrics; SCC Ag, Squamous cell carcinoma antigen; MLNs, metastatic lymph nodes; SMD, standardized mean difference; |SMD| < 0.10 indicates negligible imbalance between groups.
Cisplatin Cycle Characteristics
Respectively, 343 and 575 patients were assigned to the <5 cycles and ≥5 cycles group. The min and max cycles of weekly infusions of cisplatin were one and seven, respectively. The number of patients with LACC who received one, two, three, four, five, six, and seven cisplatin cycles were 15(1.6%), 33 (3.6%), 82 (8.9%), 213 (23.2%), 269 (29.3%), 300(32.7%), and 6 (0.7%), respectively. The median cisplatin cycles of the <5 cycles group was 4 cycles [IQR, 3–4 cycles]. The median cisplatin cycles of the ≥5 cycles group was 6 cycles (IQR, 5–6 cycles).
Survival Outcomes
The 5-yr OS was 76.7% (95%CI: 74.3%–79.9%) and 86.1% (95%CI: 84.6%–87.6%) for the <5 cycles and ≥5 cycles group, respectively (p = 0.0017; Figure 1a ). The survival benefit of ≥5 cycles did not wane with the prolongation of the follow-up time.
16
Similarly, the 5-yr DFS was 68.7% (95%CI: 66.1%–71.3%) for the <5 cycles group and 78.3% (95%CI: 76.6%–80.0%) for the ≥5 cycles group (p = 0.0016; Figure 1b). Moreover, five cycles showed consistent superiority to four cycles for OS (85.5% vs. 77.6%, p = 0.040), while five cycles could not benefit patients in DFS (77.3% vs. 72.4%, p = 0.239). The comparison of OS (a) and DFS (b) between < 5 cycles and ≥ 5 cycles group in the total cohort. OS, Overall survival; DFS, Disease free survival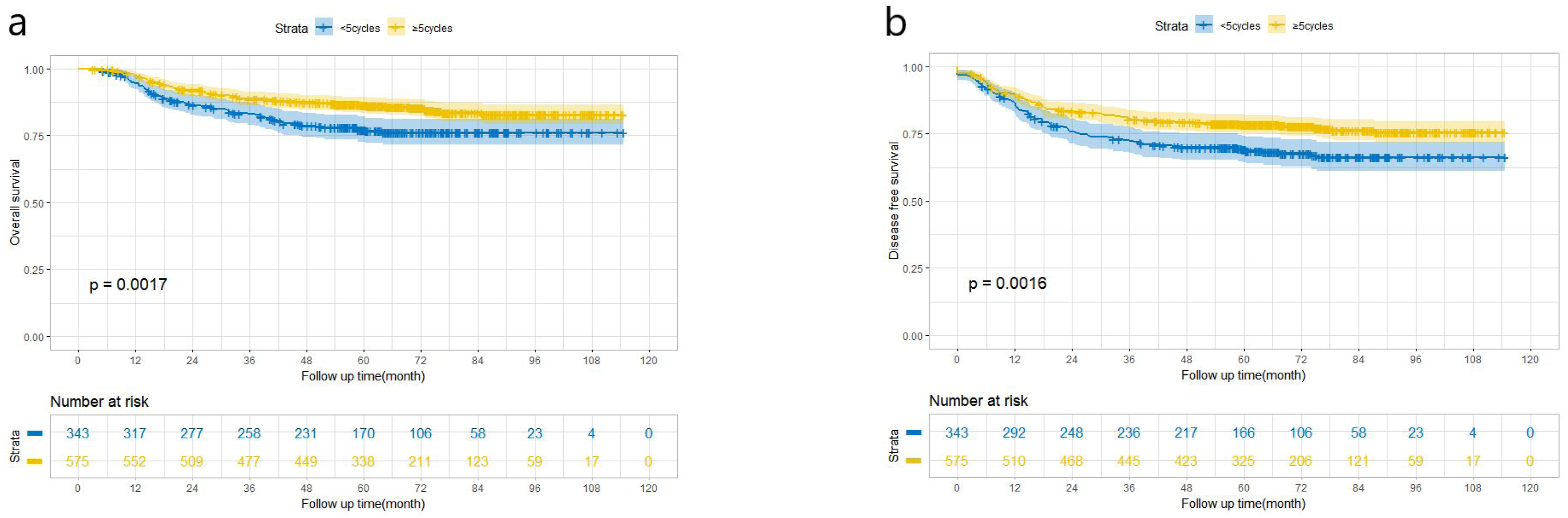
Treatment-Related Adverse Effects
Comparison of Acute Toxicities Between the <5 Cycles and ≥5 Cycles Groups

Subgroup Analysis
The 5-yr OS and DFS were compared between the <5 cycles and ≥5 cycles groups in different subgroups including age (≤50 vs. >50), histology (squamous vs. non-squamous), FIGO stage (I/II vs. III/IV), tumor size (≤4 cm vs. >4 cm), pretreatment SCC Ag (≤1.5 ng/mL vs. >1.5 ng/mL), pelvic MLNs (with vs. without) and para-aortic MLNs (with vs. without). Specifically, ≥5 cycles showed consistent superiority to <5 cycles for OS whether in terms of age ≤50 (89.3% vs. 81.0%, p = 0.039) or age >50 (85.0% vs. 74.6%, p = 0.013), while ≥5 cycles could only benefit patients aged >50 (76.6% vs. 66.8%, p = 0.011) and not those aged ≤50 (77.5% vs. 72.4%, p = 0.053) in DFS. Strikingly, patients with squamous disease could benefit from ≥5 cycles both in OS (91.9% vs. 77.2%, p = 0.0031) and DFS (89.0% vs. 69.6%, p = 0.0019). However, this benefit did not extend to the patients with non-squamous disease whether in OS (80.4% vs. 74.7%, p = 0.370) or DFS (71.5% vs. 66.9%, p = 0.530). Concerning SCC Ag, ≥5 cycles had advantages for patients with SCC Ag >1.5 ng/mL both in OS (84.9% vs. 75.9%, p = 0.0096) and DFS (76.3% vs. 68.7%, p = 0.019), while patients with SCC Ag ≤ 1.5 ng/mL could not benefit more from ≥5 cycles compared to <5 cycles in OS (86.3% vs. 79.0%, p = 0.073). Similarly, ≥5 cycles could benefit patients with tumor size >4 cm instead of those with tumor size ≤4 cm (Figure 2a-d). Surprisingly, patients with early stage such as stage I/II were able to benefit from ≥5 cycles whether in OS (Figure 2a) or DFS (Figure 2b); however, ≥5 cycles were unable to benefit patients with advanced stage (stage III/IV) who were supposed to gain survival benefits from ≥5 cycles (Figure 2c and d). Meanwhile, ≥5 cycles showed consistent superiority to <5 cycles for OS whether in pelvic MLNs (73.7% vs. 60.9%, p = 0.047) or no pelvic MLNs (91.8% vs. 84.7%, p = 0.012), while ≥5 cycles benefitted not only patients with pelvic MLNs (64.8% vs. 50.3%, p = 0.032) but also patients without pelvic MLNs in DFS (84.5% vs. 77.8%, p = 0.020). Finally, patients without para-aortic MLNs benefited from ≥5 cycles both in OS (87.8% vs. 80.6%, p = 0.011) or DFS (80.4% vs. 72.8%, p = 0.009); however, ≥5 cycles could not benefit patients with para-aortic MLNs (OS: 51.5% vs. 36.3%, p = 0.476; DFS: 34.8% vs. 26.4%, p = 0.741). The comparison of OS between < 5 cycles and ≥ 5 cycles group in stage I/II (a) and stage III/IV (c); The comparison of DFS between < 5 cycles and ≥ 5 cycles group in stage I/II (b) and stage III/IV (d). OS, Overall survival; DFS, disease free survival; HR: Hazard ratio; CI: Confidential interval
Construction and Validation of the Nomogram
Univariate and Multivariate Cox Regression Analysis of OS for Patients in <5 Cycles Group

OS, Overall survival; HR, Hazard ratio; CI, Confidential interval; FIGO, Federation International of Gynecology and Obstetrics; SCC Ag, Squamous cell carcinoma antigen; MLNs, metastatic lymph nodes. For categorical variables, HRs are presented for non-reference categories only (reference category = Ref). Blank cells in the multivariable Cox regression columns indicate variables not included in the multivariable model.

(a) Prognostic nomogram for 1-yr, 3-yr and 5-yr OS in < 5 cycles group. Calibration curves of 1-yr (b), 3-yr (c) and 5-yr OS (d) for patients in < 5 cycles group in the training cohort; Calibration curves of 1-yr (e), 3-yr (f) and 5-yr OS (g) for patients in the < 5 cycles group in the validation cohort. OS: Overall survival
Clinical Application of the Model
The risk score for each patient was calculated based on the nomogram. The optimal cut-off value was identified using X-tile software (Figure 4a ) and was determined to be 101.0 (Figure 4b). Based on this cut-off value, patients were stratified into low-risk and high-risk groups, and the corresponding Kaplan–Meier curves demonstrated significant survival differences between the two subgroups (Figure 4c). The group with ≥5 cycles maintained its superiority over that with <5 cycles for patients in low-risk group both in OS (p = 0.0052) (Figure 4d) and DFS (p = 0.008) (Figure 4e). Specifically, the 5-yr OS for < 5 cycles and ≥ 5 cycles in low-risk group were 83.5% (95%CI: 81.0%–86.0%) and 91.3% (95%CI: 89.8%–92.8%), respectively. And the 5-yr DFS for <5 cycles were 77.1% (95%CI: 74.3%–79.9%) and 89.2% (95%CI: 87.7%–90.7%) in ≥5 cycles. However, in the high-risk group, ≥5 cycles was unable to hold its superiority whether in OS (p = 0.130) (Figure 4f or DFS (p = 0.093) (Figure 4g). Specifically, the ≥5 cycles had a 5-yr OS of 61.6% (95%CI: 58.0%-65.2%) and < 5 cycles had a 5-yr OS of 57.5% (95%CI: 52.2%-62.8%). Additionally, the 5-yr DFS for ≥ 5 cycles and < 5 cycles were 51.4% (95%CI: 47.5%-55.3%) and 47.2% (95%CI: 42.0%-52.4%), respectively. (a) Identification of the optimal cut-off value for the nomogram score using X-tile software; (b) the optimal cut-off value was determined to be 101.0; (c) Kaplan–Meier curves illustrating the survival differences between the low-risk and high-risk subgroups stratified by the cut-off value. The comparison of OS between < 5 cycles and ≥ 5 cycles in low-risk (d) and high-risk subgroup (f); the comparison of DFS between < 5cycles and ≥ 5 cycles in low-risk (e) and high-risk subgroup (g). OS, Overall survival; DFS, disease free survival
Discussion
Previously, we analyzed the OS and DFS in patients with CC based on different cisplatin cycles during CCRT; five cycles appeared to be optimal. 16 In this study, we focused on the prognosis of five cycles of CC with different clinicopathological features for long-term follow-up. Patients in the ≥ 5 cycles group showed significantly increased 5-yr OS and DFS than those in the < 5 cycles group. The < 5 cycles and ≥ 5 cycles groups were classified by age, histology, FIGO stage, tumor size, SCC Ag, pelvic MLNs, and para-aortic MLNs. The incidence of grade ≥3 acute and late toxicities was comparable between the two groups. After subgroup analysis, ≥ 5 cycles of cisplatin was found to benefit the OS and DFS of patients aged > 50 years, or with squamous disease, SCC Ag > 1.5 ng/mL, tumor size > 4 cm, early stage, and no para-aortic MLNs. Using univariate and multivariate Cox regression analyses, stage, tumor size, and SCC Ag levels were incorporated into our nomogram to predict OS and DFS. We divided the overall population into two risk categories using the nomogram’s total points and found that individuals in the low-risk group experienced significant survival improvements with ≥ 5 cycles of cisplatin. To the best of our knowledge, this is the first nomogram to predict the OS and DFS of patients with CC undergoing CCRT based on different cisplatin cycle groups.
The use of cisplatin-based CCRT for CC has been shown to improve survival compared with radiation therapy and has been the standard treatment approach for CC.4,21,22 Cisplatin enhances radiotherapy sensitivity by inhibiting the repair of radiation-induced sublethal damage, making hypoxic cells more sensitive to radiation. 23 Weekly cisplatin at 40 mg/m2 for 5-6 cycles during radiotherapy for CC according to a clinical practice guideline. 14 Meanwhile, several studies have investigated the effects of the cumulative dose and number of cycles of cisplatin on clinical survival during CCRT in CC. Torres et al 24 analyzed data from 302 CC patients treated with CCRT and suggested that a total cisplatin dose of at least 200 mg/m2 was associated with better disease-specific survival. A randomized trial studied 268 patients with CC treated with CCRT in an adjuvant setting and found that treatment with > 210 mg/m2 of cisplatin resulted in a survival benefit. 25 Shaverdian et al 26 demonstrated that fewer than five chemotherapy cycles predicted inferior DFS and OS. Kotha et al 27 found an increased 2-yr OS in patients who received ≥ 5 cisplatin cycles (91.4% vs 78.3%; p = 0.020). This finding is consistent with that of our previous study. Current evidence suggests that receiving ≥ 5 cycles during CCRT for CC is an appropriate option.
However, several factors should be considered when comparing our findings with previous studies. Recent studies have refined the concept of cisplatin dose intensity during CCRT using different metrics. A large multicenter study reported that a cumulative cisplatin dose ≥200 mg improved OS, particularly in patients with MLNs and stage III disease, suggesting that the benefit of intensified platinum may depend on metastatic risk and treatment tolerance. 28 In contrast, our exposure was defined by the number of weekly cisplatin cycles. Although clinically intuitive, cycle count may not fully capture cumulative dose variability, limiting comparisons with dose-based analyses. Differences in endpoints and baseline risk profiles across cohorts may also contribute to variations in observed benefit. Consistently, randomized trials comparing weekly and tri-weekly cisplatin have reported similar survival outcomes, suggesting that cumulative dose and treatment completion may be more clinically relevant than schedule. 29
Beyond these comparability issues, our findings should be interpreted in the context of evolving systemic therapies for LACC. Although the integration of pembrolizumab into CCRT has begun to reshape treatment strategies, 6 radiotherapy with concurrent platinum remains the therapeutic backbone. Adequate delivery of concurrent cisplatin therefore remains clinically important, as it functions primarily as a radiosensitizer and contributes to overall treatment intensity. In this context, our finding that ≥5 weekly cisplatin cycles are associated with improved long-term OS and DFS remains clinically relevant, particularly in real-world settings where immunotherapy may be unavailable or contraindicated. Importantly, the cycle-based threshold and nomogram proposed here should be viewed as complementary tools to optimize backbone treatment rather than alternatives to systemic intensification. Future studies are needed to determine whether these cisplatin-intensity thresholds and risk-stratification models retain predictive value in immuno-chemoradiotherapy or other intensified regimens. In addition, recent advances in imaging-based predictive modeling and artificial intelligence have demonstrated promising potential in predicting treatment response and facilitating individualized therapeutic strategies in cervical cancer. 30
These findings highlight the importance of identifying CC patients who may benefit from ≥5 cycles of cisplatin during CCRT. Patients with high SCC Ag levels, large tumors, and certain early-stage disease appeared to derive greater benefit. Elevated SCC Ag levels may be associated with poor OS and higher rates of local, regional, and distant metastases.31,32 For these patients, additional chemotherapies such as neoadjuvant chemotherapy and consolidation chemotherapy should be considered.33,34 The results of our study also confirmed that high-dose cisplatin could be used in patients with SCC Ag levels > 1.5 ng/mL. Furthermore, findings from a randomized controlled study indicated that increased doses of cisplatin could enhance the rate of complete remission and improve the 1- and 2-yr OS rates, while concurrently reducing the rate of distant metastases in CC patients with large tumor masses. 23 We further confirmed that CC patients with a tumor diameter greater than 4 cm required five or more cycles of cisplatin during CCRT. In this study, we observed that in the ≥ 5 cycles group chemotherapy improved the OS (p = 0.090) and DFS (p = 0.058) of patients with stage I/II, but not of those with stage III/IV. This could be because patients with stage III/IV cervical cancer require high-intensity chemotherapy, such as doublet chemotherapy, as these patients have a higher risk of metastasis. 35 Weekly cisplatin could increase sensitivity to radiation, which could improve the local control rate, but not distant metastasis.16,36 Our previous study also showed that five cycles appeared to be the optimal number of cisplatin cycles during CCRT for CC. Moreover, there was no significant difference between the patients with CC in the five cisplatin cycles and six or seven cisplatin cycles by subgroup analyses. 16 In the previous reports, to combine this study on the issue, we speculated that patients with stage III/IV only require five cisplatin cycles. This scheme not only enhances radiosensitivity but also maintains the physical condition necessary for receiving high-intensity chemotherapy after CCRT. Additionally, treatment-related toxicities during concurrent chemoradiotherapy continue to be a clinically relevant concern, as recent evidence suggests that hematological and gastrointestinal adverse events may still occur, even with optimized supportive care strategies. 37 However, no significant differences were observed in grade ≥3 gastrointestinal, urinary, or hematological toxicities between patients receiving <5 and ≥5 cycles.
To identify patients with CC who might benefit from ≥ 5 cycles of cisplatin, univariate and multivariate cox regression analysis were performed to select the clinicopathological factors significantly associated with the prognosis of CC patients. We then developed a novel nomogram to predict the 1-, 3-, and 5-yr OS. Surprisingly, we found that receiving ≥ 5 cisplatin cycles provided a significant improvement in OS and DFS in selected low-risk patients. Low-risk patients who received ≥ 5 cisplatin cycles achieved a 7.8% improvement (from 83.5% to 91.3%) in the 5-yr OS rate (p = 0.0052) and a 12.1% improvement (from 77.1% to 89.2%) in the 5-yr DFS rate (p = 0.008) compared with those who received < 5 cisplatin cycles. Most patients in the high-risk group were at a later stage, and had larger tumors and higher SCC Ag levels, indicating that patients with these features were more likely to obtain a survival benefit from aggressive chemotherapy; this is consistent with previous studies. 38 Ma et al 38 analyzed 1503 patients with CC and showed that CCRT with cisplatin-based doublet chemotherapy significantly improved the OS (p = 0.010) and progression-free survival (p = 0.010). The OUTBACK trial revealed that administering adjuvant chemotherapy (ACT) following standard carboplatin-based CCRT did not enhance OS or progression-free survival among patients with CC. 39 However, ACT may be required in some patients to reduce local or distant disease recurrence. Tangjitgamol et al 40 conducted a randomized controlled trial investigating patients with higher stages of CC and demonstrated a notably reduced incidence of systemic recurrence in the ACT group, which received paclitaxel plus carboplatin, than the CCRT group. A randomized controlled trial conducted by Duenas-Gonzales et al using subgroup analysis showed that patients with large tumors benefited the most from ACT. 41
This study has several strengths that support the validity and clinical relevance of the findings. This study included a large real-world cohort with long-term follow-up, enabling evaluation of OS and DFS. Baseline clinicopathological characteristics were comparable between the <5 and ≥5 cycles groups, and confounders were further adjusted using multivariable Cox regression. Subgroup analyses and nomogram-based risk stratification helped identify patients who may derive differential benefit from cisplatin cycle intensity during CCRT. The relatively large sample size (n = 918) provided sufficient statistical power to detect clinically meaningful differences in survival outcomes.
This study had several limitations, particularly its retrospective design. This could have resulted in a selection bias regarding the diagnostic approaches and arrangements for follow-up. Although major baseline clinicopathological characteristics were comparable between the <5 cycles and ≥5 cycles groups with small absolute SMDs, propensity score-based methods (propensity score matching or inverse probability of treatment weighting) were not performed; therefore, residual confounding due to non-random treatment allocation cannot be completely excluded. In addition, the nomogram was developed using data from patients at a single institution and its generalizability was not assessed through external validation. To reduce potential bias related to incomplete or heterogeneous data availability, this study did not analyze important factors affecting the prognosis of CC, such as tumor differentiation grade, lymphovascular invasion and radiotherapy dose.42,43
Conclusions
In summary, we confirmed that patients with CC who received ≥ 5 cycles of cisplatin showed significantly improved OS and DFS compared with those receiving < 5 cycles by long-term follow-up. Patients were divided into two risk subgroups according to the total points derived from the nomogram (low-risk: total points < 101; high-risk: total points ≥ 101). We found that receiving ≥ 5 cycles of cisplatin may be associated with improved OS and DFS advantages for patients classified in the newly defined low-risk subgroup. Importantly, grade ≥3 toxicities were similar between groups. These findings offer initial support for future prospective clinical trials and are expected to be validated by different institutions.
Supplemental Material
Supplemental Material - Effect of Cisplatin Cycles on Prognosis for Cervical Cancer Patients Treated With Concurrent Chemoradiotherapy: A Retrospective Cohort Study
Supplemental Material for Effect of Cisplatin Cycles on Prognosis for Cervical Cancer Patients Treated With Concurrent Chemoradiotherapy: A Retrospective Cohort Study by Zheng Zeng, Xilin Yang, Weiping Wang, Xiaoliang Liu, Junfang Yan, Ke Hu and Fuquan Zhang in Technology in Cancer Research & Treatment.
Supplemental Material
Supplemental Material - Effect of Cisplatin Cycles on Prognosis for Cervical Cancer Patients Treated With Concurrent Chemoradiotherapy: A Retrospective Cohort Study
Supplemental Material for Effect of Cisplatin Cycles on Prognosis for Cervical Cancer Patients Treated With Concurrent Chemoradiotherapy: A Retrospective Cohort Study by Zheng Zeng, Xilin Yang, Weiping Wang, Xiaoliang Liu, Junfang Yan, Ke Hu and Fuquan Zhang in Technology in Cancer Research & Treatment.
Footnotes
Ethical Considerations
This study was approved by the Ethics Review Committee of Peking Union Medical College Hospital, Chinese Academy of Medical Sciences (Approval No.: I-25PJ2348, approved on June 16, 2025). The requirement for informed consent was waived due to the retrospective nature of the study. The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China, Ministry of Science and Technology of the People’s Republic of China (Grant No. 2022YFC2407100 and 2022YFC2407101) and National High Level Hospital Clinical Research Funding (grant number 2025-PUMCH-A-029)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed for the present study are available from the corresponding author on reasonable request.
Use of Artificial Intelligence
The authors declare that no generative artificial intelligence tools were used in the writing, analysis, or preparation of this manuscript.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
