Abstract
Introduction
To develop and evaluate a multi-omics machine-learning model that integrates clinical variables, dose-volume histogram (DVH) metrics, radiomics, and dosiomics from both the rectum and rectal wall regions of interest (ROIs) to improve prediction of acute radiation proctitis (ARP) in cervical cancer patients receiving radiotherapy.
Methods
In this single-center retrospective cohort, 107 cervical cancer patients were randomly split into a training set (n = 85) and a testing set (n = 22) in an 8:2 ratio. Radiomic were extracted from planning CT, and dosiomic features from 3-D RT-dose distributions, for both rectum and rectal wall ROIs. Features were z-score standardized; redundant features were filtered by Pearson correlation, followed by least absolute shrinkage and selection operator (LASSO) for selection. Support Vector Machine (SVM) and Multilayer Perceptron (MLP) classifiers were trained using stratified five-fold cross-validation within the training set. Model performance was assessed on the held-out test set using receiver operating characteristic (ROC) analysis; clinical utility was evaluated with decision-curve analysis (DCA). The primary endpoint was Common Terminology Criteria for Adverse Events (CTCAE,version 5.0) grade ≥2 ARP.
Results
Multi-omics fusion outperformed single-modality models across ROIs and classifiers. The rectal-wall multi-omics SVM achieved the best discrimination with AUC 0.867 (95% Confidence Interval [CI]:0.709-1.000) in the test set; performance for the whole-rectum region of interest (ROI) was lower (AUC 0.714). DVH-only models showed limited discrimination, and no DVH feature was retained after penalized selection in the multi-omics pipeline. DCA demonstrated the greatest net clinical benefit for the rectal-wall multi-omics model across threshold probabilities 0.20–0.50.
Conclusion
A rectal-wall, region-specific multi-omics approach integrating clinical, radiomic, and dose-based descriptors improves prediction of radiotherapy-induced ARP compared with single-modality and whole-rectum analyses. These findings highlight the importance of ROI selection and multi-omics integration for precision toxicity assessment and support future external validation and prospective evaluation.
Keywords
Introduction
Cervical cancer ranks as the fourth most common malignancy among women worldwide. 1 Radiotherapy is a primary treatment modality that has markedly improved survival outcomes; however, radiation-induced injury to adjacent organs at risk (OARs) remains a major clinical challenge.2,3 As a frequent complication of cervical cancer radiotherapy, acute radiation proctitis (ARP) results from inflammatory and structural injury to the rectal mucosa and deeper layers caused by ionizing radiation, with a relatively high incidence depending on treatment and assessment criteria, and it markedly impairs patients’ quality of life4–5. Accurate prediction and timely intervention of ARP are therefore essential for optimizing therapeutic efficacy and patient outcomes.
Current studies on ARP prediction predominantly rely on single-modality approaches. 6 Clinical characteristics capture overall patient status, radiomics extract quantitative features from medical images, and dosiomics describe radiation dose distribution and tissue exposure7–9. While each modality is informative, none alone provides a complete picture, whereas multi-omics integration can yield a more comprehensive assessment and potentially enhance predictive performance. Traditionally, the entire rectum has often been used as the region of interest (ROI) for toxicity prediction.10,11 However, as a hollow organ often containing air and fecal matter unrelated to radiation-induced injury, the whole-rectum ROI may introduce irrelevant information and reduce model robustness11,12. In contrast, the rectal wall-the tissue directly irradiated and primarily affected during ARP-represents a more biologically meaningful ROI. 13 Previous evidence has shown that rectal wall are more reliable predictors of gastrointestinal toxicity than whole-rectum measures based on dose metrics,and that geometric or texture descriptors derived from the rectal wall provide additional predictive value beyond conventional dose-volume histogram (DVH) parameters14–15.
Building upon these insights, this study integrates multi-omics features from both the rectum and rectal wall to evaluate their predictive efficacy and to identify the optimal anatomical region for precision toxicity prediction in cervical cancer radiotherapy.The key focus of this study is to develop a predictive model for ARP in cervical cancer patients by integrating clinical, DVH, radiomics, and dosimetric features. Particular emphasis is placed on comparing the rectal wall with the entire rectum as regions of interest (ROIs), given that the rectal wall more accurately reflects structural and functional changes associated with radiation-induced injury. By adopting a dual-ROI strategy, this study evaluates the added value of rectal wall-based modeling and multi-omics integration for improving prediction accuracy and enhancing the precision of toxicity assessment in clinical applications.
Materials and Methods
Patient Selection and Clinical Data
A total of 107 patients with cervical cancer who underwent external beam radiotherapy (EBRT) at the First Affiliated Hospital of Hengyang City, Hunan Province, between January 2022 and March 2025. Clinical variables collected included age, body mass index (BMI), International Federation of Gynecology and Obstetrics (FIGO) stage, and history of concurrent chemotherapy and diabetes mellitus. Patients were randomly assigned to a training cohort (n = 85) and a test cohort (n = 22) using an 8:2 ratio.
Inclusion criteria: (a) pathologically confirmed cervical cancer; (b) age ≥18 years; (c) development of ARP during EBRT or within 90 days after treatment; (d) availability of complete pelvic CT images, radiotherapy planning data, and follow-up records; (e) no prior history of chronic enteritis or other intestinal diseases.
Exclusion criteria: (a) presence of other pathologically confirmed malignancies; (b) history of pelvic radiotherapy; (c) radiotherapy interruption of ≥1 week during the treatment course.
ROI Delineation
Per the institutional contouring protocol, the rectum ROI was manually delineated in the treatment planning system (TPS, Eclipse,version 10.0,Varian Medical Systems, Palo Alto, CA, USA) by experienced radiation therapists and reviewed by a radiation oncologist. The rectal wall ROI was defined based on the rectum ROI through a three-step process.
Step one, CT images were imported into the RT-Mind (version 1.6; MedMind Technology Co., Ltd, Beijing, China) automated segmentation system, where a 3D convolutional neural network model was used to segment the entire rectum. Standardized preprocessing was performed simultaneously, including window width/level calibration and voxel spacing normalization.
Step two was performed in RT-Mind software, which implemented a distance transform-based morphological erosion in the physical space. Specifically, the rectal contour was eroded inward by 3 mm to generate the rectal lumen, ensuring consistency across images with different voxel sizes. This 3-mm margin was selected based on previously validated physiological measurements of rectal wall thickness and has been widely adopted as a standard assumption in dose-wall histogram and normal tissue complication probability(NTCP) modeling studies to approximate the clinical rectal wall.
Step three, the set difference between the original rectal mask and the contracted lumen to obtain a 3-mm-thick rectal wall mask. All auto-generated contours underwent visual quality control by a board-certified radiation oncologist, with minor edits applied where necessary to ensure clinical accuracy. To prevent observer bias, the delineation of ROIs and the extraction of radiomic, dosiomic, and clinical features were performed without knowledge of the patients’ toxicity outcomes.Representative axial, sagittal, and coronal views of the rectal wall segmentation are shown in Fig. 1
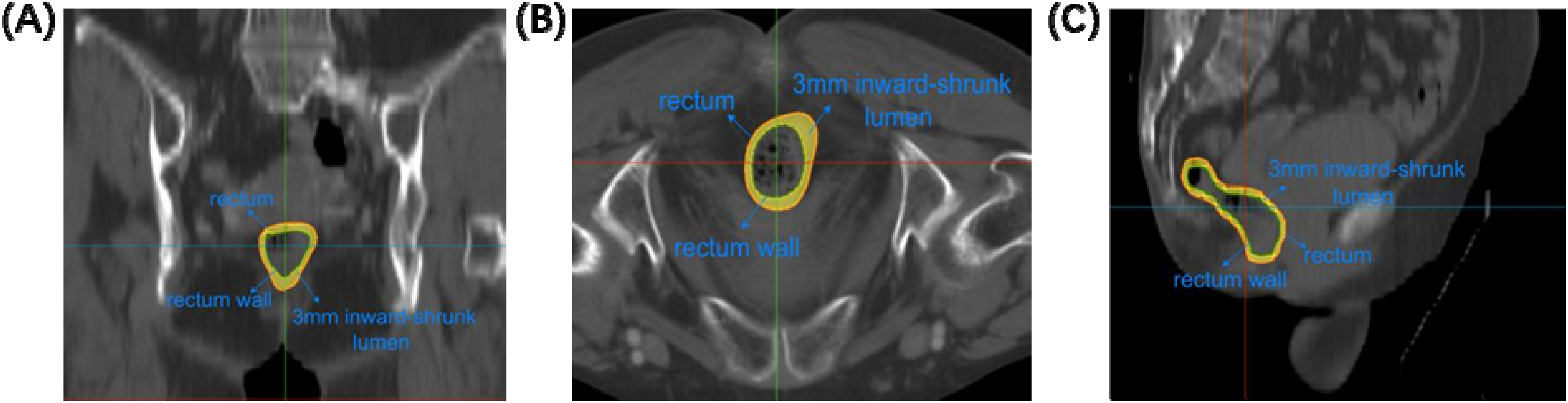
Results of rectal wall ROI extraction: (A) coronal view; (B) axial view; (C) sagittal view. Red contour: original rectum; green contour: 3-mm inward contracted lumen; yellow region: rectal wall (3-mm thickness).
Radiotherapy Protocol and Toxicity Assessment
All patients received EBRT delivered with either intensity-modulated radiotherapy (IMRT) or volumetric-modulated arc therapy (VMAT). The prescribed dose was 45–50 Gy in 25–28 fractions (1.8-2.0 Gy per fraction), five fractions per week, planned in the TPS and delivered on a United Imaging linear accelerator (model 506c) with 6 MV photons. Where applicable, image guidance (cone-beam CT,CBCT) and institutional immobilization protocols were used. Toxicities were assessed during EBRT and within 90 days after completion of EBRT; for each patient, the highest ARP grade observed within this window was recorded as the outcome. For the primary analysis, Common Terminology Criteria for Adverse Events (CTCAE,version 5.0) grade≥2 events were defined as clinically significant endpoints requiring intervention and substantially affecting quality of life, whereas grade < 2 events were classified as non-endpoints. This dichotomization served as the reference standard for subsequent predictive modeling.The assessment of radiation proctitis severity was performed by oncologists who were blinded to the radiomic and dosiomic features extracted for this study.
Feature Extraction and Preprocessing
Feature extraction from the rectum and rectal wall ROIs was performed using the PyRadiomics (version 2.2.0) library in Python (version 3.8) with SimpleITK (version 2.1.1). A total of 1561 radiomic features were extracted from CT images, including first-order statistics, texture, shape and morphology, wavelet-transformed features, and local binary patterns. Similarly, 1561 dosiomic features were derived from three-dimensional dose distribution matrices, including first-order statistics, dose-texture features, and descriptors of high-dose area morphology.A total of 74 DVH-based features were extracted, and the complete list and definitions of these features are provided in the Appendix.
All features were standardized using z-score normalization (mean and standard deviation estimated from the training set). Missing values were imputed using nearest-neighbor interpolation fitted on the training data.Feature stability was evaluated using the intraclass correlation coefficient (ICC), and unstable features with ICC < 0.75 were excluded. To reduce multicollinearity, pairwise Pearson correlation coefficients were computed on the training set; when |r| > 0.90, one feature from each highly correlated pair was removed, retaining the feature with stronger univariable association to the endpoint .Univariate statistical tests were then applied to identify features significantly associated with the outcome. For normally distributed features, the Student's t-test was used; for non-normally distributed features, the Mann-Whitney U test was applied. Features with p ≤ 0.05 were retained for further analysis.Subsequently, Least Absolute Shrinkage and Selection Operator (LASSO) regression with stratified 5-fold cross-validation was performed for feature selection. The regularization parameter (λ) was optimized using the one-standard-error rule to identify a parsimonious subset of predictive features. All feature preprocessing and selection procedures were performed within the inner folds of a stratified 5-fold nested cross-validation framework to avoid data leakage.
Model Construction
Single-omics classifiers were developed using radiomic, dosiomic, or DVH features individually. A total of 1561 radiomic features were extracted from CT images, 1561 dosiomic features were derived from three-dimensional dose distribution matrices, and 74 DVH features were extracted from the radiotherapy dose-volume histogram.
For the multi-omics approach, radiomic, dosiomic, and DVH features were concatenated after feature selection and used as input for the classification model. This multi-omics fusion allowed the model to leverage complementary information from different data sources, enhancing its predictive power. The overall pipeline of feature extraction, selection, and multi-omics model construction is illustrated in Fig. 2.

The workflow of the combined model construction.
Two representative supervised machine learning algorithms were implemented: Support Vector Machine (SVM) and Multilayer Perceptron (MLP). SVM is a kernel-based method that identifies an optimal separating hyperplane in a transformed feature space, capturing nonlinear decision boundaries. MLP, a feedforward neural network model, learns complex hierarchical representations of input data using one or more hidden layers with nonlinear activation functions.
The entire dataset was stratified and randomly split into a training set and a test set in an 8:2 ratio, with the same test set used for all algorithms to ensure fair comparison between single-omics and multi-omics models. Five-fold cross-validation was employed for hyperparameter tuning within the training set, and optimal parameters were applied to evaluate model generalizability and predictive performance on the test set.
Statistical Analysis
For continuous variables, the Kolmogorov-Smirnov test was used to assess distribution type. Data conforming to a normal distribution were expressed as mean±standard deviation and compared between groups using independent-samples t-tests. Data not following a normal distribution were expressed as median and interquartile range and compared using the Mann-Whitney U test.For categorical variables, data were presented as counts and percentages, and the chi-square test was used for inter-group comparisons. These statistical methods were applied to examine baseline differences in clinical and dosimetric characteristics between the training and test cohorts, confirming dataset comparability.
Model performance was evaluated using receiver operating characteristic (ROC) curve analysis, with the area under the curve (AUC) calculated separately for the training and test sets. The optimal model was identified according to AUC and the trade-off between sensitivity and specificity at the maximum Youden index. Decision curve analysis (DCA) was further performed to assess the clinical net benefit and diagnostic utility of the models.All statistical analyses were conducted using SPSS software (version 23.0).
Result
Patient Characteristics
The baseline characteristics of patients in the training and test cohorts are presented in Table 1. According to the CTCAE grading system, the overall incidence of ARP was 86.0% (92/107). Specifically, 60 patients (56.1%) developed grade 1 ARP, 30 patients (28.0%) developed grade 2, and 2 patients (1.9%) developed grade 3 toxicity. Grade 0 reactions were observed in 15 patients (14.0%).In the training set, no significant differences were observed between the grade≥2 (endpoint) and grade < 2 (non-endpoint) groups for any clinical characteristic (P > 0.05). The mean age was 58.53 ± 9.68 years in the non-endpoint group and 56.24 ± 10.61 years in the endpoint group (P = 0.336). Other clinical variables, including BMI, FIGO stage, chemotherapy, ECOG performance status, and diabetes history, showed no significant differences.
Clinical Feature Analysis of ARP Based on CTCAE Grading.

In the testing set, age was the only clinical feature showing a statistically significant difference (P = 0.025). The non-endpoint group had a mean age of 57.27 ± 8.66 years, whereas the endpoint group was younger (50.43 ± 6.50 years). No other clinical characteristics differed significantly between groups.
These findings indicate that the training set was well balanced in terms of clinical characteristics, while age may be associated with the risk of developing grade ≥2 ARP in the testing cohort, warranting further investigation.In addition,no significant difference in the incidence of ARP was observed between patients treated with IMRT and those treated with VMAT (P = 1.000).
Feature Selqection Results and Correlation Weights
The feature selection results for single- and multi-omics models are presented in Fig. 3. Three types of features - radiomics, dosiomics, and DVH - were initially included in the multi-omics model. After feature selection, only radiomic and dosiomic features were retained, while no DVH feature entered the final model.

Feature selection results for Multi-omics models across different ROIs. (A):Rectal wall; (B):Rectum.
For the rectal wall ROI, selected radiomic features were dominated by texture-related descriptors, whereas dosiomic features were mainly associated with dose gradient and spatial distribution characteristics. A similar pattern was observed for the rectum ROI, although the selected radiomic features placed more emphasis on morphological and intensity-based metrics.In the single-omics models, feature selection from radiomics and dosiomics independently revealed distinct patterns, reflecting the complementary nature of these feature domains. Radiomic features captured anatomical and structural variations, while dosiomic features highlighted dose distribution heterogeneity.
Model Validation
Across different feature sets and ROIs, both classifiers exhibited variable predictive performance. Overall, the SVM classifier consistently achieved higher AUC values compared with the MLP classifier, and multi-omics integration provided clear performance gains over single-modality models.Model robustness was further assessed using five-fold cross-validation, and detailed fold-wise performance results are provided in the Appendix.
For the rectal wall ROI, the SVM multi-omics model demonstrated the best predictive performance, with an AUC of 0.867 (95% Confidence Interval [CI]: 0.7093-1), followed by the MLP multi-omics model with an AUC of 0.857 (95% CI: 0.5771-1). Single-omics models based on radiomics or dosiomics showed moderate performance, whereas DVH-only models provided limited discriminative ability. Similar trends were observed for the rectum ROI, with overall AUCs slightly lower than those for the rectal wall (Table 2).
Classification Performance Metrics of Single and multi-Omics Models Across Different ROIs.

In single-omics settings, dosiomics models generally outperformed radiomics and DVH models across both classifiers, highlighting the stronger predictive signal contained in dose-distribution features. However, radiomics features still contributed complementary structural and textural information, which, when combined with dosiomics, significantly enhanced model performance.
Multi-omics integration combining radiomics, dosiomics, and DVH features yielded the most robust models overall. Although DVH features were not retained after feature selection, their inclusion during model construction improved the optimization process and reinforced the contribution of dosiomics features. The performance improvement was particularly evident in the SVM model for the rectal wall ROI, reflecting the benefit of integrating multiple feature domains.
Decision curve analysis (Fig. 4) further demonstrated that the rectal wall-based multi-omics model provided the greatest net clinical benefit within the threshold probability range of 0.20–0.50, outperforming the single-modality models across the same range ROC curve analysis (Fig. 5) further highlighted the consistent performance advantage of the multi-omics SVM model compared to single-modality and MLP models.

ROC curves of the DVH model, radiomics model, dosiomics model and combined model for Rectum ROI in test cohort.

DCA of DVH model, radiomics model, dosiomics model and combined model in the test cohort
Discussion
Integrating clinical variables, DVH metrics, dosiomics, and radiomics features within a region-specific rectal-wall framework demonstrated superior discriminative performance and greater net clinical benefit in predicting acute radiation proctitis, compared with single-modality approaches and whole-rectum-based analyses. In this study, we developed and validated a multi-regional multi-omics model integrating clinical, DVH, dosiomic, and radiomic features extracted from both the rectum and rectal wall to predict the severity of rectal toxicity. The proposed model outperformed all single-feature-category models, and the rectal wall-based model achieved the highest predictive accuracy, emphasising the importance of regional feature differentiation. This comprehensive multi-omics approach offers a robust and non-invasive framework for individualised risk assessment and clinical decision support in radiotherapy.The present study focuses on pretreatment risk stratification of acute radiation proctitis using baseline planning CT and planned dose distributions, reflecting routine clinical practice in which toxicity risk assessment must be performed before treatment initiation. Although rectal geometry may vary during treatment, baseline imaging-derived features may still capture intrinsic anatomical and dosimetric susceptibilities relevant to early risk identification.
An essential methodological consideration in radiomics and dosiomics modelling is the definition of the ROI. Conventional studies often use the entire rectum as the ROI, which may introduce irrelevant information from the lumen and surrounding tissues, thereby reducing model performance. For example, Wei et al 8 reported only moderate predictive accuracy(AUC=0.6855) when using rectum-based radiomic models for radiation proctitis prediction. In contrast, recent work has highlighted the advantages of rectal wall-based analysis. Abbaspour et al 16 demonstrated that radiomic features extracted from the rectal wall markedly improved the prediction of proctitis compared with rectum-based features, with the model achieving high predictive performance(AUC=0.907). Similarly, Yang et al 17 found that wall-based features better reflect localised dose-response relationships, reinforcing the wall's biological relevance.
From a dosimetric perspective, Tucker et al 18 showed that dose-wall histograms (DWH) outperform traditional DVH in predicting late rectal bleeding, further supporting wall-focused modelling. Qin et al 19 also emphasised that integrating radiomic and dosiomic features enhances model robustness and clinical applicability. In line with these findings, Andree Desrochers et al 20 compared rectum- and rectal wall-based models and demonstrated that wall-based models achieved higher predictive performance across multiple classifiers and feature selection strategies.
The feature importance analysis further supports the superiority of high-throughput radiomic and dosiomic descriptors over conventional DVH parameters. As shown in Figure 3, the final multi-omics models for both the rectal wall and the whole rectum were dominated by wavelet- and LoG-filtered radiomic and dosiomic features. In contrast, no DVH features were retained after feature selection. In the rectal wall model, the top-weighted predictors were mainly texture-based features such as ClusterShade, Correlation, and LowGrayLevelEmphasis, derived from multi-scale wavelet or Laplacian-of-Gaussian transformations. These features capture subtle heterogeneity and spatial dose response variations within the wall region. In contrast, the rectum model exhibited slightly lower feature diversity and smaller coefficient magnitudes, indicating reduced sensitivity to localised textural variations.
The absence of DVH features suggests that traditional dose-volume parameters lack the spatial and textural resolution necessary to describe the complex dose-response behaviour of hollow organs. Similar conclusions have been reported by Qin et al, 19 who demonstrated that dosimetric features containing texture and gradient information outperform DVH metrics in capturing spatial heterogeneity of dose distributions. Abbaspour et al 16 also found that wavelet-based radiomic features provided the strongest predictive performance for radiation-induced proctitis and cystitis, and Wei et al 8 confirmed that radiomics-based models contributed more to prediction accuracy than clinical or DVH-based ones. Further supporting evidence comes from Andree Desrochers et al 20 and several recent investigations employing high-throughput radiomic and dosiomic analyses. For example, Hou et al 21 demonstrated that integrating three-dimensional dosiomic and radiomic features significantly improved the prediction of radiation-induced inflammatory responses compared with traditional DVH models. Huang et al 22 developed a combined CT-based radiomics-dosiomics model that markedly improved the prediction of grade-4 radiation-induced lymphopenia in nasopharyngeal carcinoma, whereas Yang et al 23 established a multiview radiomics-dosiomics risk model that outperformed DVH-based NTCP models in predicting radiation-induced hypothyroidism and clearly distinguished patients with identical DVHs but different spatial dose distributions. These studies collectively highlight that 3D spatially resolved dosiomic and radiomic descriptors better capture dose heterogeneity and toxicity-related patterns, underscoring that conventional DVH metrics - being one-dimensional summaries - fail to represent the complex spatial dose-response relationships in hollow organs.
Previous studies have consistently shown that integrating multi-omics features substantially enhances the prediction of radiation-induced toxicities compared with single-omics or DVH-based models. Huang et al 22 demonstrated that combining radiomic, dosiomic, and clinical features markedly improved the prediction of severe lymphopenia in nasopharyngeal carcinoma, while Hu et al 24 showed similar benefits of radiomics-clinical integration for treatment response assessment in rectal cancer. In thoracic cohorts, Kraus et al 25 and Puttanawarut et al 26 reported that radiomic-dosiomic fusion models achieved higher accuracy for predicting radiation pneumonitis than DVH- or single-omics based approaches.Other investigations across different cancer types have consistently confirmed that multi-omics integration provides complementary spatial, textural, and biological information, yielding more robust and generalizable predictive models.27,28 Collectively, these findings align with our results, demonstrating that integrating clinical characteristics, DVH metrics, radiomics, and dosiomics yields superior predictive power over single-omics models in forecasting radiation-induced rectal toxicity for both rectum and rectal wall ROIs.
Interestingly, the dosiomics model consistently outperformed the radiomics model in predicting ARP. This superiority likely arises from the fact that dose distributions encode direct information on the spatial heterogeneity of radiation exposure, which is inherently linked to tissue injury risk. In contrast, imaging-derived radiomic features mainly capture post-treatment tissue appearance and are more susceptible to factors such as intestinal contents, imaging settings, and patient positioning. Therefore, dosiomics may capture more biologically and treatment-relevant information than radiomics alone. This interpretation aligns with the findings of Zhang et al and Tang et al,29–30 both of which demonstrated that dose-based or combined dosiomic-radiomic models outperform DVH- or radiomics-only approaches, underscoring the higher predictive robustness of dose-derived spatial features.
However, several limitations should be acknowledged.This was a single-centre study with a relatively small sample size and a limited number of severe proctitis cases, which may limit the generalizability and increase the risk of overfitting, even though internal validation strategies were applied.Multi-centre validation with larger and more heterogeneous cohorts is warranted to confirm model robustness.Moreover, rectal geometry may vary during EBRT due to bowel filling and bladder volume changes, whereas only the planning CT was analysed in this study. Although this aligns with the objective of pretreatment toxicity prediction, future studies incorporating longitudinal imaging and delta-radiomics features may better capture inter-fraction anatomical variability and further improve model performance.In addition, alternative sub-region segmentation strategies and explicit spatial descriptors (eg, distance-based or overlap-based metrics) were not evaluated, which may influence feature extraction and model accuracy. 31 Finally, as with most machine learning models, issues related to feature selection and interpretability remain, and addressing them will be essential for clinical translation.
Conclusion
In summary, the proposed multi-regional multi-omics model integrating clinical, DVH, dosiomic, and radiomic features effectively predicts the severity of radiation-induced rectal toxicity. The rectal wall based model achieved the highest predictive accuracy, emphasizing the value of region-specific feature extraction. Feature importance analysis demonstrated that high-throughput radiomic and dosiomic descriptors, rather than traditional DVH metrics, dominated the final model, highlighting their superior capacity to capture spatial dose heterogeneity and tissue microstructural variations. These results underscore the potential of multi-omics approaches as non-invasive, personalized tools to support clinical decision-making and optimize radiotherapy planning.
Supplemental Material
sj-docx-2-tct-10.1177_15330338261424144 - Supplemental material for Region-specific Multi-Omics Modeling for Predicting Acute Radiation-Induced Proctitis in Cervical Cancer Radiotherapy: A Retrospective Analysis
Supplemental material, sj-docx-2-tct-10.1177_15330338261424144 for Region-specific Multi-Omics Modeling for Predicting Acute Radiation-Induced Proctitis in Cervical Cancer Radiotherapy: A Retrospective Analysis by Gaocen Xiao, Kerun Quan, Miaomiao Zeng, Yanxi Ye, Jiabiao Hong, Zhijun Liu and Haibiao Wu in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-3-tct-10.1177_15330338261424144 - Supplemental material for Region-specific Multi-Omics Modeling for Predicting Acute Radiation-Induced Proctitis in Cervical Cancer Radiotherapy: A Retrospective Analysis
Supplemental material, sj-docx-3-tct-10.1177_15330338261424144 for Region-specific Multi-Omics Modeling for Predicting Acute Radiation-Induced Proctitis in Cervical Cancer Radiotherapy: A Retrospective Analysis by Gaocen Xiao, Kerun Quan, Miaomiao Zeng, Yanxi Ye, Jiabiao Hong, Zhijun Liu and Haibiao Wu in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
Thanks to our colleagues and staff at the Department of Radiation Oncology, The First Affiliated Hospital of University of South China.
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of The First Affiliated Hospital of University of South China, located in Hengyang, China (Approval No. 2024ll0902003). Written informed consent was waived by the Institutional Review Board due to the retrospective nature of the study, and all patient data were anonymized prior to analysis.
Author Contributions
Gaocen Xiao:Data collection, model establishment, and manuscript drafting.
Kerun Quan:Conception and design of the study, model establishment,and critically revised the manuscript for important intellectual content.
Miaomiao Zeng:Data collection and critically revised the manuscript for important intellectual content.
Yanxi Ye:Assisted in data analysis and participated in the revision of the final manuscript.
Jiabiao Hong:Assisted in data analysis and participated in the revision of the final manuscript.
Zhijun Liu:Conception and design of the study, critically revised the manuscript for important intellectual content.
Haibiao Wu:Conception and design of the study and critically revised the manuscript for important intellectual content.
Funding
This work was supported by the Joint Funds for the innovation of science and Technology, Fujian province (Grant number: 2021Y9190), Startup Fund for scientific research, Fujian Medical University (Grant number:2024QH1173) and the Hunan Provincial Natural Science Foundation (Grant number:2025JJ81055).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
