Abstract
Objective
To evaluate the clinical outcomes of volumetric modulated arc therapy (VMAT) followed by brachytherapy (BT), combined with chemotherapy, and local hyperthermia (HT) on locally advanced cervical cancer (LACC).
Methods
In total, 40 patients with FIGO stage IB1-IVB cervical cancer from January 2016 to December 2018 were selectively enrolled in this study. All patients were treated with VMAT (50.4 Gy/1.8 Gy/28 f) concurrent with cisplatin-based chemotherapy (40 mg/m2, q1w, 6 cycles) and local HT (40.5-41°C for 60 min, BIW). BT (30-36 y/5-6 f, 2 f/w) was conducted after VMAT. Objective response rate (ORR), local control (LC) time, LC rate, progression-free survival (PFS) rate, cancer-specific survival (CSS) rate, overall survival (OS), median time to tumor progression and treatment-related toxicity were evaluated.
Results
The median follow-up time was 31 months (8-48). The ORR was 100% at 3 months after treatment and 92.1% at 6 months, respectively. The 1-year, 2-year, and 3-year LC rates were 87.4%, 81.9%, and 70.9%, respectively. The average LC time was 31.50 ± 1.89 months (95% CI 27.79-35.21). The 1-year, 2-year, and 3-year PFS rates were 75.85%, 61.2%, and 51.3%, respectively, while the median PFS was 27.07 months. The 1-year, 2-year, and 3-year OS rates were 95%, 84%, and 79.6%, respectively. In total, 12(30%) patients had grade 3/4 bone marrow suppression. One patient had grade 4 leukopenia. In total, 17 patients had grade 1/2 bone marrow suppression. Two patients had grade 3 nausea and grade 3 vomiting reaction, respectively. No grade 3/4 proctitis and bladder reaction were observed. In the late period of treatment, 1 patient had a rectal hemorrhage. In total, 13 patients had vaginal stenosis.
Conclusion
VMAT concurrent with chemotherapy, BT, and local HT had a favorable short-term efficacy and acceptable toxicity on cervical cancer, which was an alternative option for LACC.
Introduction
Cervical cancer is one of the most common types of cancer in women globally and ranks fourth for both incidence and mortality,1,2 with an estimated 570,000 newly diagnosed cases and 311,000 deaths in 2018 worldwide. Due to the lack of obvious clinical manifestations, most patients are already in advanced stages at the time of diagnosis, with local infiltration and lymphatic metastasis, which portends a poor prognosis. For early-stage cervical cancer, radical surgery is an effective method. Besides, in terms of patients with unresected locally advanced cervical cancer (LACC), platinum-based concurrent chemoradiotherapy (CCRT) is the recommended treatment option.3,4 However, for patients with specific risk factors, the clinical efficacy of CCRT is limited.3,5,6,7,8 Thus, new treatment modality combinations are needed to improve the outcomes of LACC with high-risk factors.
Hyperthermia (HT) acts as a sensitizer for EBRT and chemotherapy by heating the local tumor up to 42 °C for about 60 min to increase the intratumoral blood perfusion and oxygenation, which inhibit cancer cell DNA repair and increase drug uptake. 9 Studies in the Netherlands reported that HT + RT increased local control (LC) rate after 12-year follow up from 37% to 56% in LACC patients, compared to EBRT alone. 10 Additionally, a multicenter randomized clinical trial in LACC reported a better 5-year overall survival (OS), DFS and local relapse-free survival (LRFS) rate in CCRT + HT group (77.8%, 70.8%, and 80.1%, respectively) compared with CCRT group (64.8%, 60.6%, and 71.0%, respectively) without significant difference. 11 Thus, more study was required to further confirm the clinical efficacy of this trimodality combination.
In this study, we treated patients with cervical cancer with CCRT + HT. In order to improve the precision of RT, we selected volumetric modulated arc therapy (VMAT) which is one of the most advanced RT techniques. We retrospectively analyzed the short-term efficacy, survival, and adverse reactions of VMAT plus brachytherapy (BT) combined with chemotherapy and deep HT in the treatment of unresectable massive LACC, so as to provide basis for further study of the effectiveness and feasibility of this therapeutic strategy.
Materials and Methods
Patients
In total, 40 patients, diagnosed with cervical cancer (FIGO 2009 stages IB2 to IVA) from January 2016 to December 2018 in our center, were selectively enrolled in this study. All patients provided written informed consent before enrollment (Table 1). The detailed information of all patients was de-identified.
Baseline Characteristics of Patients.

The inclusion criteria included (1) Histologically and radiologically confirmed cervical cancer; (2) No major organ dysfunction; (3) KPS score > 70; (4) Expected survival time beyond 6 months; (5) Tolerable to CCRT + HT; (6) Written informed consent.
The exclusion criteria included (1) there are contraindications of radiotherapy and chemotherapy; (2) patients with liver and kidney diseases, cardiovascular and cerebrovascular diseases, etc; (3) age < 18 or >85; (4) pregnancy or lactation; (5) unable to cooperate with the whole research or have participated in other experiments; (6) refusing to sign the informed consent.
Radiotherapy
To ascertain the accuracy of treatment, target volume delineation was based on the fusion image of computed tomographicand magnetic resonance imaging images. According to ICRU62 report, gross tumor volume (GTV) includes cervical lesions, paracteral invasion and metastatic lymph nodes. Clinical target volume (CTV) of primary cervical cancer includes cervical tumor, cervix, uterus, parauterine, ovary, upper vagina, as well as lymph node drainage area (common iliac, bilateral iliac, presacral, and obturator lymphatic drainage area). The planning target volume (PTV) was defined as CTV plus a 3–15 mm margine, while the PGTVnd was defined as GTV of nodal metastases (GTVnd) plus a 3 mm. The 95% PTV volume was to receive 54 Gy in 1.8 Gy fractions up to 5 times a week with the EQD2 was about 49.56 Gy. The 95% PGTVnd volume was to receive 60 Gy in 2.14 Gy fractions. The VMAT contained a total of 90 dose control points which was designed from 182° To 178° with 2 full arc irradiation. Reversed dose optimization was required to satisfy the prescription dose. For organs at risk (OARs), the V40 and V50 of the rectum and bladder were less than 40% and 50%, respectively; the V40 and Vmax of small intestine and colon were less than 25% and 53 Gy; the V50 of the femoral head was less than 5%. HDR-BT was started after 36–45 Gy of EBRT (prescription dose of 192Ir postloading point a ranged from 30 to 36 Gy/5-6f, 2f/W, EQD2: 31.25-48 Gy). The totally bioequivalent dose (EQD2) of point A was about 80.8 to 97.5 Gy. The treatment period of EBRT and BT was controlled within 55 days (Figure 1).
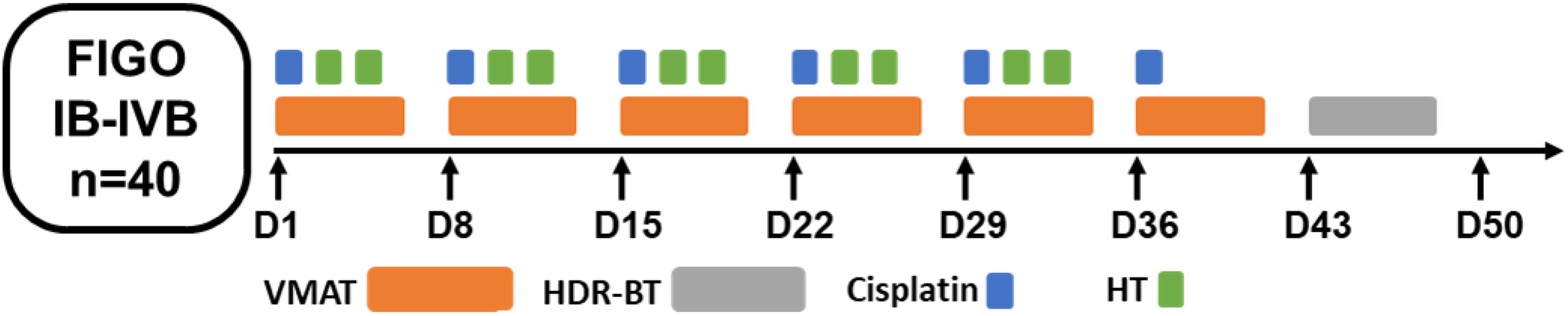
Patient flow diagram.
Chemotherapy and HT
All patients received weekly cisplatin-based chemotherapy with 40 mg/m2 for 6 cycles, concurrent with pelvic EBRT and HT (SR1000 RF applicator). Deep HT was performed at 40.5–41 °C for 40 min every time, 2f/w, with an interval of more than 72 h. Each HT was performed within 1 hour before EBRT. The temperature is monitored by a sensor placed in the vagina. The median treatment number of HT was 6 times (2-10 times) (Figure 1).
Evaluation of Treatment
The primary endpoint was OS rate. The second endpoint included objective response rate (ORR), LC, progression-free survival (PFS), cancer-specific survival (CSS) and acute or chronic toxicity. The short-term efficacy evaluation was divided into complete remission (CR), partial remission (PR), stable disease (SD), and disease progression (PD) according to the WHO objective efficacy evaluation criteria for solid tumors. The objective effective rate was calculated by CR and PR. The adverse reaction was evaluated according to the common terminology criteria for adverse events (CTCAE 5.0) issued by the National Cancer Institute (NCI) to evaluate acute (3 months after treatment) and chronic (6 months after treatment) toxicity. Acute reactions included leukopenia, anemia, nausea and vomiting, diarrhea, cystitis, rectal mucositis, frequent urination, and skin reactions related to HT. Chronic reactions included vaginal stricture and rectal bleeding.
Follow-up
All patients were followed every 3 months for the first 2 years and then, every 6 months thereafter. ORR and acute toxicity were evaluated and OS, LC, PFS, and CSS were assessed during follow-up. The deadline for follow-up was September 31, 2019. The median follow-up time was 23 months (range, 6-38).
Statistical Analysis
SPSS version 22 (IBM SPSS Statistics, 2013) was used for statistical analysis. The Kaplan–Meier method was used to analyze LC, OS, and CSS. GraphPad prism 7.0 was used to plot the survival curve. The reporting of this study conforms to STROBE guidelines. 12
Ethical Approval Statement
This study was approved by Peking University Third Hospital Medical Science Research Ethics Committee (Approval number: 2019-IRB-494-01) on 25th Nov 2019. All patients provided written informed consent prior to enrollment in the study.
Results
Patient Characteristics
The median age was 55 years old (range, 29-83). The median KPS score was 90. The median diameter of the tumor was 4.55 cm (2-7.3), but the tumor diameter of 33 patients was bigger than 4 cm. At the end of the follow-up, all patients had completed the course of EBRT and postloading treatment. In total, 80% of the patients completed 4–6 cycles of CCRT. 4 patients were intolerable to chemotherapy due to poor physical conditions such as heart disease and severe myelosuppression, and 2 patients stopped chemotherapy after 2 to 3 cycles because of myelosuppression. In total, 36 patients completed HT for more than 6 times, and 2 cases completed 4–6 times of HT (Table 1).
Clinical Efficacy
At the end of follow-up, 33 patients (82.5%) survived and 7 patients (17.5%) died. One patient recurred in pelvic 17 months after treatment and received local reradiotherapy. After treatment, the patient gradually developed leukopenia, anemia, gastrointestinal reaction, and hematochezia. The ending was systemic failure and death. One patient died of leukemia (confirmed by bone marrow pathology) after remission. One patient died of pelvic recurrence. One patient died of systemic failure. Four patients died of distant metastasis. The 1-year LC for all patients was 87.4%, while the 2-year and 3-year LC were 81.9% and 70.9%, respectively. The 1-year, 2-year, and 3-year CSS were 95%, 84%, and 79.6%, respectively. The 1-year, 2-year, and 3-year survival rate were 95%, 84%, and 79.6%, respectively (Figure 2).

The PFS rate (A), OS rate (B) and LC rate (C) of VMAT combined with chemotherapy and local HT. Abbreviations: PFS, progression-free survival; OS, overall survival; LC, local control.
Four weeks after EBRT, the ORR, CR, and SD before BT were 85% (CR + PR, 28/40), 15% (6/40), and 15% (6/40), respectively. The 1-month ORR and CR after CCRT was 100% (CR + PR, 40/40) and 67.5% (27/40), respectively. The 3-months ORR and CR after CCRT were 95% (CR + PR, 39/40) and 75% (27/40), respectively. The 6-months ORR and CR after CCRT were 92.11% (CR + PR, 35/38) and 84.21% (32/38), respectively. The median LC time were 31.504 ± 1.89 months (95% CI, 27.79-35.21), and the median time to tumor progression was 18 months (range, 6-38 months) (Table 2).
The Short Term Effects.
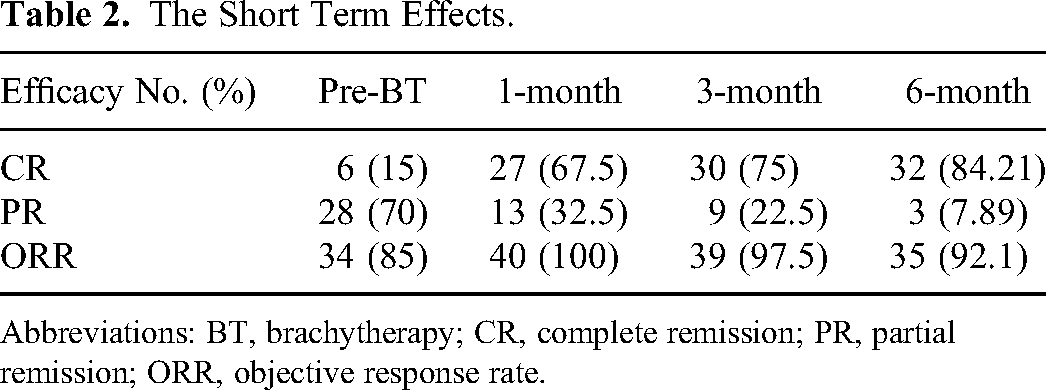
Abbreviations: BT, brachytherapy; CR, complete remission; PR, partial remission; ORR, objective response rate.
Treatment Related Toxicity
Twenty patients (50%) had acute side effects of grade 1-2 leukopenia. The occurrences of grade 1-2, grade 3, and grade 4 leukopenia were 50% (20/40), 25% (10/40) and 2.5% (1/40), respectively. In total, 17 patient had grade 1-2 anemia and 1 patient had grade 3-4 nausea and vomiting. No grade 3-4 proctitis and bladder reaction were observed. In terms of late toxicities, 1 patient had rectal bleeding and 13 patients had vaginal stenosis (Tables 3,4).
The Acute Toxicity of VMAT Combined with Chemotherapy and Local HT.

The Late Toxicity of VMAT Combined with Chemotherapy and Local HT.

Abbreviations: VMAT, volumetric modulated arc therapy; HT, hyperthermia.
Discussion
Currently, the standard regimen for LACC is cisplatin-based CCRT and BT. 3 The results of multiple randomized trials have shown that CCRT can improve DFS and OS of these patients,3,5–7,13 which confirmed the benefits of CCRT. The 5-year OS and DFS were increased by 6% and 8%, respectively, compared with the RT alone, whereas the prognosis in stage III/IVA was only 3% which was higher than RT alone. In order to break through the treatment bottleneck, increasing attention fell on RT combined with different chemotherapy drugs, targeted drugs, and HT, which put forward individualized treatment suggestions aiming at different stages of tumor, as well as to improve the control of LACC. Most studies have reported the short-term efficacy, but few reported the temporal changes (Table 5).14–17 We analyzed the objective remission rate at various times within half a year after the completion of treatment. In this study, we retrospectively analyzed the tolerance and clinical efficacy of CCRT + HT. We hoped to further explore combination therapy on the basis of existing research, especially for Asian women. Most patients with anemia can only complete RT alone, but are unable to bear the adverse effects of multiple drug combinations or increased chemotherapy dose. For cervical cancer patients with advanced stage, unresectable and lymph node metastasis, deep HT with good tolerance combined with CRT is a favorable option.
Summary of Clinical Trial Comparing CCRT and CCRT + HT in Patients With LACC.
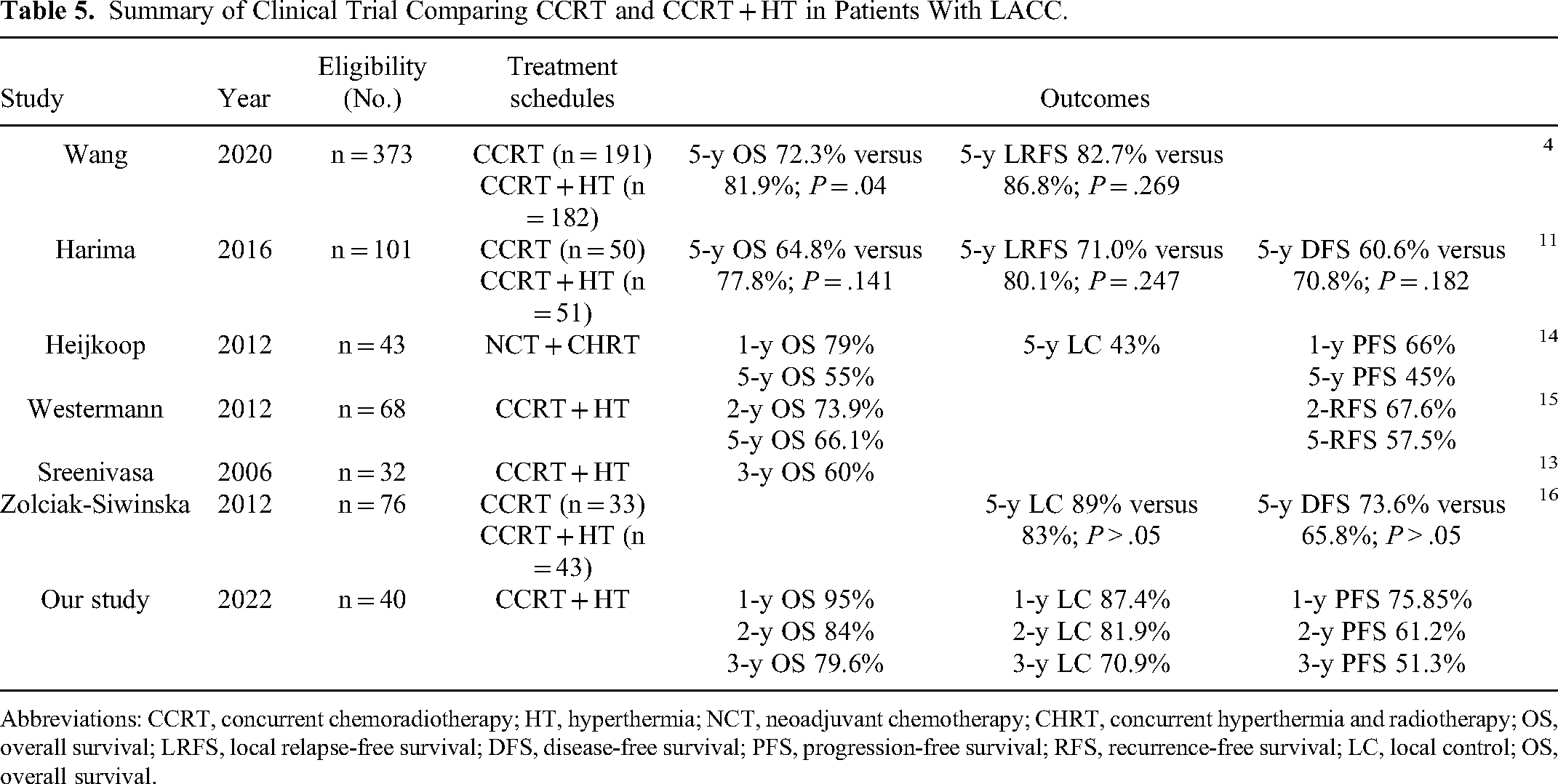
Abbreviations: CCRT, concurrent chemoradiotherapy; HT, hyperthermia; NCT, neoadjuvant chemotherapy; CHRT, concurrent hyperthermia and radiotherapy; OS, overall survival; LRFS, local relapse-free survival; DFS, disease-free survival; PFS, progression-free survival; RFS, recurrence-free survival; LC, local control; OS, overall survival.
The biological rationale of HT to sensitize RT and chemotherapy lies on the inhibition of DNA repair and increase of blood supply inside tumor. 9 Deep HT could suppress DNA repair to cause cell cycle arrest and subsequent cell apoptosis. Furthermore, the increased temperature can improve blood perfusion, which leads to elevated oxygenation and chemotherapy drug delivery. Increased oxygen level promotes irradiation-induced DNA double stand break and makes it difficult to repair. 18 A prospective, randomized, multicenter trial conducted by the Dutch Deep HT Group trial reported that RT + HT 3-year OS from 27% to 51% and 3-year LC from 41% to 61%, compared with RT alone. 19 This study confirmed the enhanced effect of HT on RT. Additionally, Lutgens et al 20 reported that HT + RT can significantly reduce the LC rate (HR, 0.48; 95% CI, 0.37-0.63; P < .001) and improve OS (HR, 0.67; 95% CI, 0.45-0.99; P < .05) of LACC, compared with RT alone. However, there is no significant difference in acute and late toxicity. These results showed that RT + HT had better efficacy without increasing toxicity. However, since CCRT has always been the standard strategy for LACC, a multicenter randomized study in Japan explored the effect of CCRT + HT on LACC compared with CCRT. They suggested that 5-year OS, DFS, and LRFS (77.8%, 70.8%, and 80.1%, respectively) in the CCRT + HT group were better than those in the CCRT group (64.8%, 60.6%, and 71.0%, respectively). 11 However, in our study, as for short-term efficacy, 88.0% of patients achieved ORR at the end of treatment, while that was 100% at 3 months after treatment. However, the LC and OS rates were not better than that in the Japanese study. In our study, 50% (20/40) of patients with lymph node positivity and 75% of patients in stage IIB—IVA, including 50% of IIIC and 7.5% of IVA. Although patients in our study generally had an advanced stage, the 3-year survival rate was 79.6%, better than that of CCRT group. Furthermore, ORR reached 100% in 3 months after treatment. It was preliminarily considered that the evaluation time of LACC can be extended to 3 months to observe the control effects, while we need a lot of case data later to confirm the evaluation time of CR and ORR after treatment, as well as the correlation with OS and PFS.
One highlight of this study is the utility of VMAT, which is a dynamic rotation intensity modulation technology. VMAT could potentially modulate the radiation beam with high precision, reduce the positioning error within fractions via shortening the treatment time from 8 min to about 2 min, and make the radiotherapy target more conformal and protect the OAR, which is more accurate than traditional intensity-modulated radiation therapy and improves the patients’ tolerance and compliance.2,21
Radically CCRT + HT can improve the LC and reduce recurrence. The early adverse reactions may prolong the treatment time and reduce the control rate. 22 Grade ≥ 3 toxicities may recede the curative effect. In our study, early toxicities are mainly hematologic reactions, while the incidence of grade 3 leukopenia and anemia were 25% (10/40) and 12.5% (5/40), respectively. For gastrointestinal toxicities, both the incidences of grade 3 nausea and vomiting were 2.5%. No grade ≥ 3 acute proctitis and cystitis were observed. For late adverse reactions, there were 2 patients with rectal bleeding, accounting for 5% of the total number, and 13 patients with vaginal stenosis with an incidence of 32.5%, indicating the effects on sexual life. No vaginal fistula or intestinal obstruction was found. There were few reports of late adverse reactions tof vaginal stricture recently. In our analysis, we found that 13 cases (32.5%) suffered vaginal stricture. The relationship between local dose of radiotherapy and vaginal adhesion and stenosis will be further discussed, so as to reduce the incidence and improve the quality of life. Due to the short observation time, the study of late side effects needs further observation.
Moreover, there were still some limitations in this study, such as the small number of cases and short follow-up time as well as the retrospective nature of the study. Given this was a retrospective study, the calculation and justification of the sample size selection were lacking in this study. Therefore, future randomized prospective studies with larger samples need to be conducted to obtain more objective results.
Conclusion
Our study suggested that VMAT combined with concurrent cisplatin-based chemotherapy, BT plus deep HT is one of the radical therapies for inoperable cervical cancer with late stage, lymph node metastasis and anemia, which has good safety and short-term curative effect.
Footnotes
Acknowledgments
The authors thank the funding support from the National Natural Science Foundation of China (NSFC), Natural Science Foundation of Beijing Municipality, Clinical scientist training program of Peking University and National Clinical Key Specialty Construction Program of China.
Author Contribution
Ping Jiang and Shuhua Wei collected all material and wrote the manuscript. Bai Yan, Ang Qu, and Weijuan Jiang participated in the collection of material. Haitao Sun was responsible for the design of the treatment plan. Junjie Wang and Ping Jiang conceived the core idea of the study, designed the article framework, and supervised this study.
Ethical Approval Statement
This study was approved by Peking University Third Hospital Medical Science Research Ethics Committee (2019-IRB-494-01) on 25 November 2019. All patients provided written informed consent prior to enrollment in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82073335 to J. Wang), the Key Specialty program of Natural Science Foundation of Beijing Municipality (Z20008 to P.Jiang), Clinical scientist training program of Peking University (BMU2023PYJH009 to Ping Jiang), Clinical key project of Peking University Third Hospital (BYSYZD202011 to Ping Jiang), Innovation and Translation project of Haidian District (HDCXZHKC2021215 to Ping Jiang), and the Special Fund of National Clinical Key Specialty Construction Program of China (2021).
