Abstract
Background:
Hepatocellular carcinoma poses a significant public health burden in China, necessitating the economic evaluation of new therapeutic strategies for policy-makers and clinicians. The international, randomized phase 3 trial CARES-310 revealed that camrelizumab plus rivoceranib provided a substantial clinical benefit in patients with advanced hepatocellular carcinoma, but the economic outcome remains unclear. This study aimed to evaluate the cost-effectiveness of camrelizumab plus rivoceranib versus sorafenib as first-line treatment for unresectable hepatocellular carcinoma (CARES-310) from the perspective of the Chinese health care system.
Methods:
A partitioned survival model was developed to estimate the lifetime cost and clinical outcomes of camrelizumab plus rivoceranib versus sorafenib in first-line treatment of advanced hepatocellular carcinoma. Survival data from the CARES-310 trial were used to create a hypothetical cohort of 543 patients with advanced hepatocellular carcinoma for modeling disease progression. The life-year, quality-adjusted life-year (QALY), incremental cost-effectiveness ratio (ICER) was used to measure the model’s outcome, with the willingness-to-pay threshold set at 3 times China’s gross domestic product (GDP) per capita (US$36 780). Univariate, multivariable probabilistic sensitivity analyses, and subgroup analysis were performed to assess parameter uncertainty, complemented by a scenario analysis using health utilities reported in literature.
Results:
The camrelizumab group yielded an additional 0.239 QALYs at an added cost of US$8340 compared with sorafenib, resulting in an ICER of US$34 897/QALY. Univariate sensitivity analysis indicated that the model results were most sensitive to the utility of progression-free survival in the camrelizumab group, sorafenib cost, and camrelizumab cost. Probabilistic sensitivity analysis revealed a 56% probability of cost-effectiveness of camrelizumab plus rivoceranib among all patients. The results of the subgroup analysis demonstrated camrelizumab plus rivoceranib was the most cost-effective in the subgroup with albumin-bilirubin grade 2.
Conclusions:
At a willingness-to-pay threshold of US$36 780/QALY, camrelizumab plus rivoceranib is likely to be a cost-effective option compared with sorafenib as first-line treatment for advanced hepatocellular carcinoma in China.
Introduction
Since 1998, China has been developing a public health care system centered on basic medical insurance. 1 By 2011, this insurance covered more than 95% of the population, with the number of insured individuals reaching 1.334 billion.2,3 The insurance fund is financed through contributions from employers, the government, and individuals, with treatment costs shared between the insurance fund and patients. Medication costs are a major component of cancer treatment expenses, with anticancer drugs presenting the most severe insurance coverage challenges. Since 2016, the National Healthcare Security Administration has conducted annual negotiations based on comprehensive value assessments with manufacturers of innovative and anticancer drugs. These negotiations establish standardized national prices and include successfully negotiated drugs in the National Reimbursement Drug List (NRDL), allowing the insurance fund to proportionally reimburse patients for these drugs. Prices for these drugs are re-negotiated every 2 years. Nevertheless, the inclusion of high-priced antineoplastic drugs in the NRDL remains limited. Although China accounts for 18% of the world’s population, it bears 30% of global cancer deaths, indicating a severe cancer burden. 4 The high cost of cancer treatment drugs imposes an enormous financial strain on both patients and the limited medical insurance fund.
Primary liver cancer is the sixth most common cancer and the third leading cause of cancer-related death worldwide. 5 China has the largest number of primary liver cancer cases in the world, with hepatocellular carcinoma accounting for 75% to 85% of these cases.5,6 Hepatocellular carcinoma stands as the second most common malignant tumor in China, claiming the lives of more than 300 000 patients annually. 7 Most patients are diagnosed at the intermediate or advanced stages of hepatocellular carcinoma, leading to a poor prognosis and an approximate 5-year survival rate of merely 18%.7,8 Tyrosine kinase inhibitors (TKIs), such as sorafenib, have served as the standard first-line treatment for advanced hepatocellular carcinoma for over a decade. Recent studies indicate that immune checkpoint inhibitors (ICIs) like atezolizumab and sintilimab, combined with targeted drugs, can effectively improve patients’ prognosis.9,10 Thus, ICI-based combination therapies are emerging as the standard first-line treatment for advanced hepatocellular carcinoma. 8
Camrelizumab (SHR-1210) is a humanized monoclonal antibody against progressive disease (PD)-1. 11 Rivoceranib, also known as apatinib, is a small-molecule TKI targeting VEGFR2. 12 The randomized, open-label, phase 3 trial CARES-310 assessed the efficacy and safety of camrelizumab in combination with rivoceranib versus sorafenib as first-line treatment for unresectable hepatocellular carcinoma. Results showed that camrelizumab plus rivoceranib substantially prolonged the median progression-free survival (PFS) compared with sorafenib (5.6 vs 3.7 months, hazard ratio = 0.52, P < .0001) and median overall survival (OS = 22.1 vs 15.2 months, hazard ratio = 0.62, P < .0001). However, the combination of camrelizumab and rivoceranib significantly increased the risk of grade 3 or higher treatment-related adverse events (81% vs 52%). 13
Hepatocellular carcinoma poses an enormous public health burden globally, especially in China, and effective and affordable treatment options are desperately needed for both policy-makers and patients. The disparities in survival time, quality of life, incidence of adverse events, drug acquisition cost, and treatment cost between the camrelizumab arm and sorafenib arm led to uncertainty in the cost-effectiveness of the 2 groups of patients. Despite 2 previous studies have conducted relevant cost-effectiveness analyses,14,15 these evaluations lacked sufficient inclusion of various cost parameters and clinical conditions. In addition, their selection of utility values and the methods for subgroup analysis may also have introduced considerable bias into the results. Therefore, the aim of this study was to provide a thorough and systematic evaluation of the cost-effectiveness of camrelizumab plus rivoceranib versus sorafenib as a first-line treatment for unresectable hepatocellular carcinoma from the perspective of the Chinese health care system.
Methods
Model overview
A 4-week cycle (28 days) partitioned survival model was developed in Microsoft Excel 2019 (Microsoft, Redmond, Washington) to simulate a cohort of patients with advanced hepatocellular carcinoma who are presenting for systemic therapy. The model consisted of 3 mutually exclusive states: PFS, PD, and death (Figure 1). The number of patients in each state at each time point was calculated from the survival probabilities depicted by the OS curve and the PFS curve. Notably, the total number of patients in the 3 states remained constant during the model simulation. The time horizon was approximately 13 years given that 99% of the cohort died. Two first-line regimens were evaluated in this model: (1) camrelizumab plus rivoceranib and (2) sorafenib. Costs and health outputs were discounted in this study using a discount rate of 5% per year. 16 The study was conducted from a health care system’s perspective and adhered to the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) guidelines 17 .
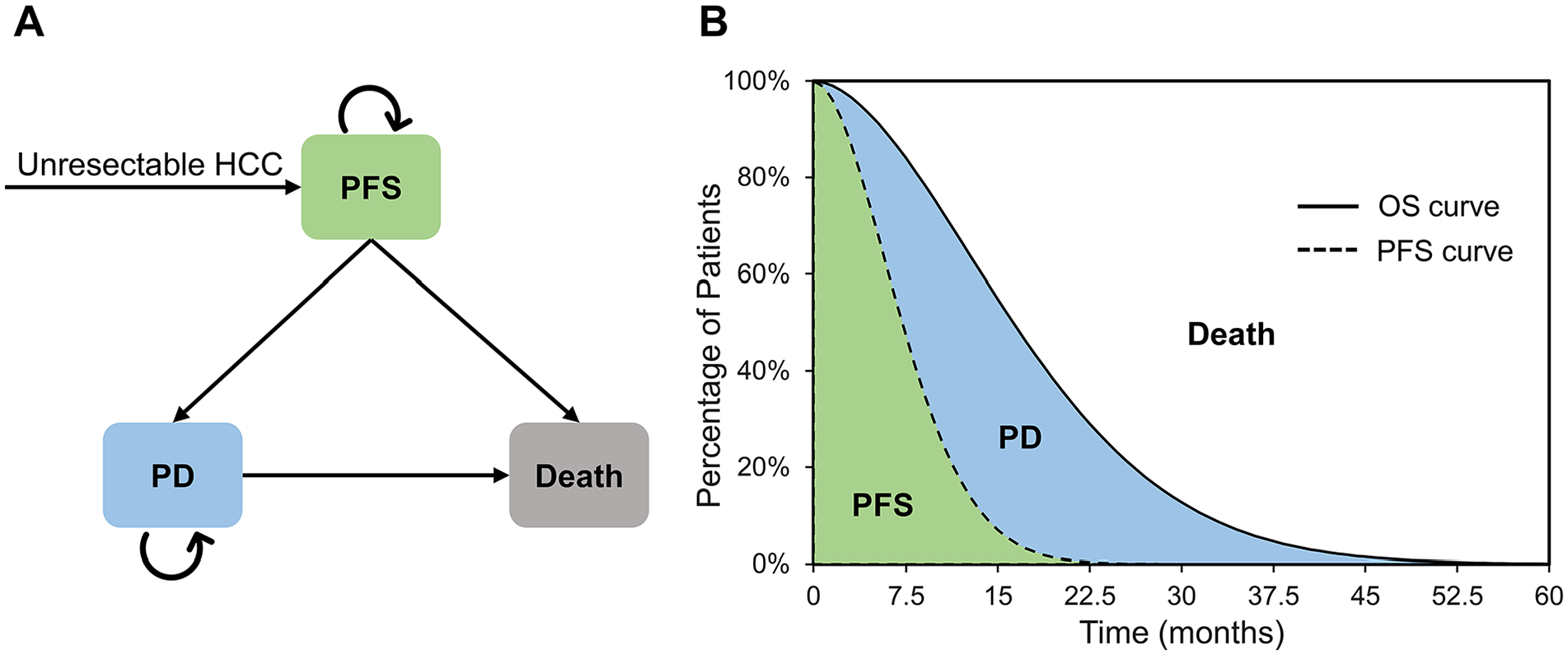
Cost-effectiveness model diagrams. (A) Decision tree; (B) Partitioned survival model conceptual diagram. The time shown in the partitioned survival model in Figure B is for illustration only and is not the final time horizon for this study. PFS indicates progression-free survival; PD, progressive disease; HCC, hepatocellular carcinoma; OS, overall survival.
Patients and intervention
Patient data and treatment protocols for this study were primarily derived from the CARES-310 trial, and patient baseline characteristics were assumed to be consistent with those in the CARES-310 trial (baseline characteristics of patients are shown in Supplemental Table S1). 13 In total, 543 adult patients (⩾18 years) with unresectable or metastatic hepatocellular carcinoma who had not received systemic therapy were randomized in a 1:1 ratio to the camrelizumab group (n = 272) and the sorafenib group (n = 271). Patients in the camrelizumab group received camrelizumab 200 mg intravenously every 2 weeks plus rivoceranib 250 mg orally once daily. In the sorafenib group, patients received sorafenib 400 mg orally twice daily. Both groups received 28-day cycles of therapy until death or disease progression, and after disease progression, both groups of patients received 5.6 months of second-line treatment along with best supportive care. After completing second-line treatment, patients continued to receive best supportive care until death. The second-line therapy drugs used and patient proportions were shown in Supplemental Table S2.
Clinical efficacy data inputs
The survival data of patients in this study were mainly derived from the results of the CARES-310 trial and the extrapolation of survival curves. WebPlotDigitizer software 4.6 was used to extract the survival data of the Kaplan-Meier survival curve, and R software 4.3.0 was used to generate the pseudo-individual patient data to reconstruct the Kaplan-Meier survival curve. Then, the exponential, Weibull, gamma, Gompertz, log-logistic, and lognormal distributions were fitted to the Kaplan-Meier curve. Based on the Akaike information criterion and Bayesian information criterion as well as visual inspection, the best-fitting distribution for each Kaplan-Meier curve was selected (Supplemental Table S3), and the reconstructed Kaplan-Meier curves and model-fitted curves for the first 100 months are shown in Figure 2. Survival curves were extrapolated to 99% of patient deaths using the best-fitting distribution. In this study, survival data from the results of the CARES-310 trial were used for the first 28 months of the partitioned survival model, and survival data extrapolated from the parametric model were used for the subsequent 128 months. The parameters of the best-fitting distribution were presented in Supplemental Table S4.
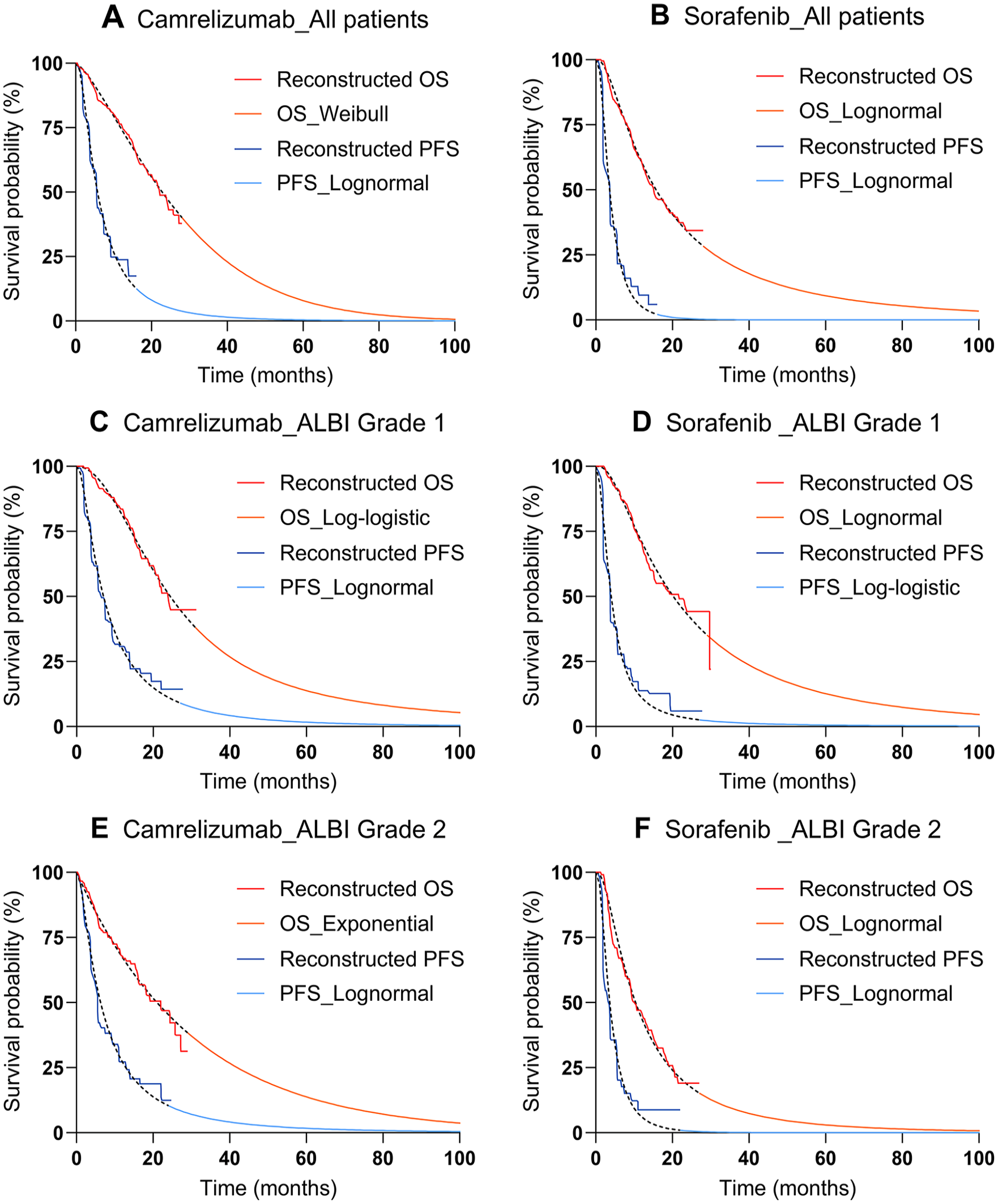
Reconstructed Kaplan-Meier curves and model-fitted curves. ALBI indicates albumin-bilirubin; OS, overall survival; PFS, progression-free survival.
Cost inputs
We estimated the lifetime direct medical costs of the simulated patients from the health care system’s perspective, costs of drugs, enhanced computed tomography (CT), laboratory testing, disease management, best supportive care, adverse event costs, and cost of end-of-life care were included. The drug price was obtained from the average national drug bidding and purchase price in 2023 on the YAOZH database. 18 Laboratory tests comprised hematology, urinalysis, blood biochemistry, coagulation parameters, thyroid function test, and alpha-fetoprotein test. The average price of several provinces in China in the literature was used for the laboratory tests, and the test cycles adhered to the schedule used in the CARES-310 trial.19 -23 In addition, tumor imaging evaluation were conducted using enhanced CT every 8 weeks for the first 48 weeks and every 12 weeks thereafter, focusing on the chest, abdomen, and pelvic. 22 Drug administration costs were inclusive of the costs of diagnosis, intravenous fluids, and beds. Grade 3 or higher adverse events with an incidence of ⩾5% were considered in the model; the incidence of adverse events was shown in Supplemental table S5. The cost of adverse events was derived from the literature.22 -24 The cost of drug administration, best supportive care, and end-of-life care were derived from the recently published literature.21,22,24,25 A mean patient weight of 65 kg was assumed for this study. The exchange rate between the US dollar and the Chinese yuan used for this calculation was 1 US dollar to 6.99 Chinese yuan. 26 Detailed costs are presented in Table 1.
Model input parameters including costs and utilities.
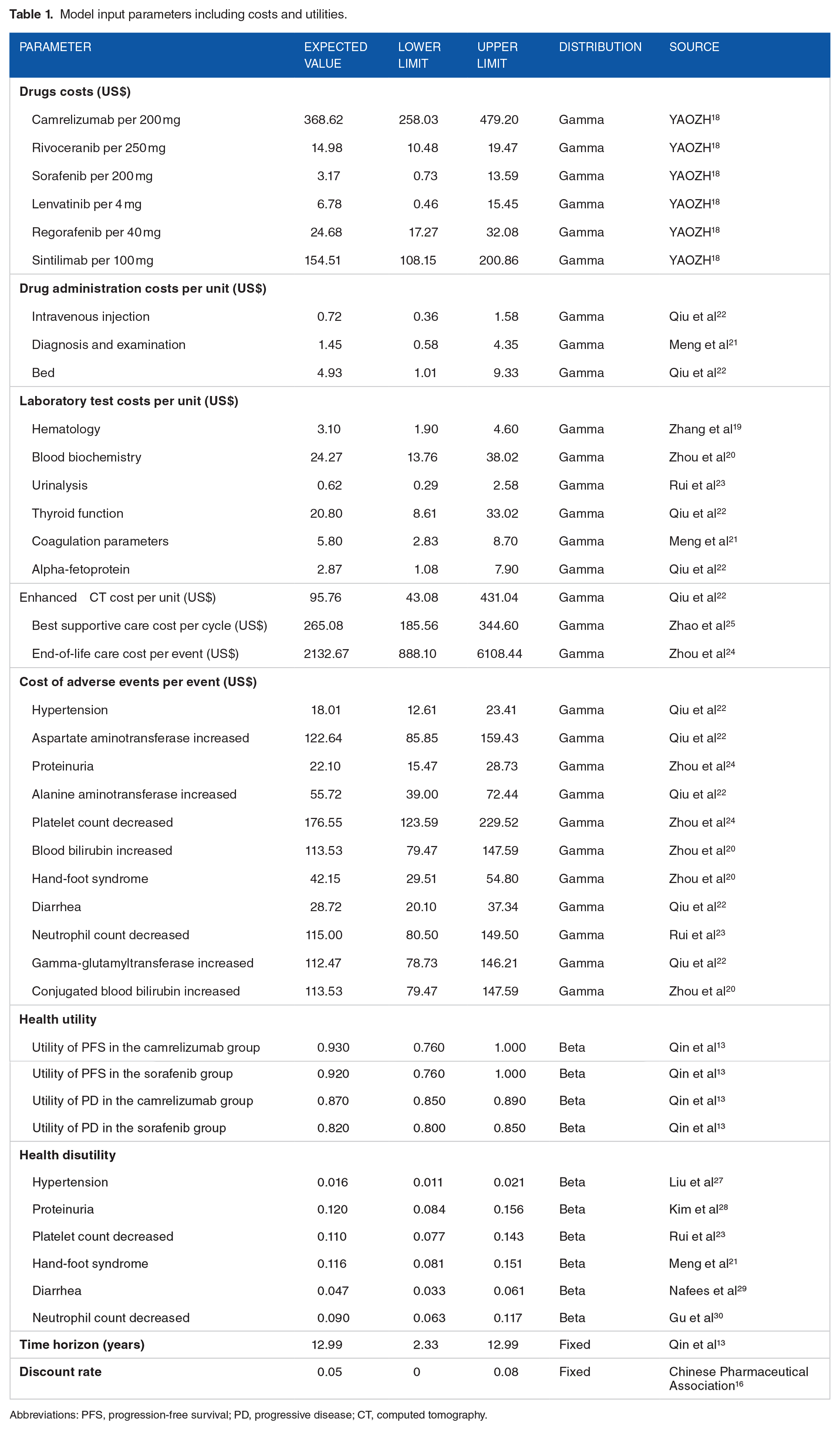
Abbreviations: PFS, progression-free survival; PD, progressive disease; CT, computed tomography.
Quality of life inputs
Utility values ranging from 0 to 1 were assigned to each health state based on the results of the EuroQol-5D 5-level (EQ-5D-5L) questionnaire analysis of the CARES-310 trial. The utility values of PFS and PD in the camrelizumab group were 0.93 and 0.87, respectively, while those in the sorafenib group were 0.92 and 0.82, respectively. 13 Meanwhile, based on the published literature, we assigned utility values of 0.76 and 0.68 for the PFS and PD states of the 2 groups in the scenario analysis.20,31,32 As adverse events affect the quality of life of patients, we also considered the disutility of grade 3 and higher adverse events, which was derived from the published literature.21,23,27 -30 To measure patients’ health outcomes, we calculated the initial QALYs by multiplying the duration each patient spent in each health state by the corresponding utility value and then summing these values. We then subtracted the QALY losses from all adverse events to obtain the final QALYs for each patient. Health outcomes were discounted at an annual rate of 5% during the calculation process. The utility parameters were specified in Table 1.
Cost-effectiveness analysis
The ICER was calculated as the incremental cost per additional QALY gained between the camrelizumab group and the sorafenib group. The willingness-to-pay (WTP) threshold was set at 3 times gross domestic product (GDP) per capita, that is, 36 780 US dollars per QALY, and this study also explored the cost-effectiveness of the camrelizumab group versus sorafenib group for the first-line treatment of hepatocellular carcinoma under the WTP thresholds of 1 time and 2 times GDP per capita (US$12 260/QALY and US$24 520/QALY). Data visualization in this study was performed in Microsoft Excel 2019, Microsoft PowerPoint 2019, R software 4.3.0, and GraphPad Prism 10.
Sensitivity analysis
Univariate and probabilistic sensitivity analyses were performed to evaluate the robustness of the model and the impact of uncertainty in the variables. To investigate the effect of changes in individual parameters on ICER values, we conducted a univariate sensitivity analysis for all parameters in the model. Parameters such as lenvatinib cost, laboratory testing costs and drug administration costs, end-of-life care cost, and utilities varied within the actual range. Specifically, we set the time horizon range from the survival time reported in the CARES-310 trial’s Kaplan-Meier curve to the time when 99% of patients in the model had died, that is, 2.33 to 12.99 years. In addition, the discount rate varied within the range of 0% to 8%. The lower limit of sorafenib cost was the lowest price of generic sorafenib (US$0.73/200 mg), and the upper limit was the price of brand-name sorafenib (US$13.59/200 mg). Other parameters varied by ±30% of the expected value, as shown in Table 1. In the probabilistic sensitivity analysis, 1000 Monte Carlo simulations were performed to explore the impact of simultaneous changes in all parameters on the model, in which the cost parameters followed a gamma distribution, and the utility and the incidence of adverse events followed a beta distribution. 33
Scenario and subgroup analysis
The albumin-bilirubin (ALBI) grade is a measure of liver function in patients with hepatocellular carcinoma. 34 A subgroup analysis of patients with ALBI grade 1 and ALBI grade 2 hepatocellular carcinoma was performed to explore the cost-effectiveness of camrelizumab versus sorafenib in different ALBI grade subgroups. Furthermore, because the utility values set in the base-case analysis may have overestimated the quality of life for PFS and PD states, we assigned utility values of 0.76 and 0.68 for PFS and PD states, respectively, to the 2 treatment groups in the scenario analysis.20,31,32 The cost-effectiveness of camrelizumab versus sorafenib under this utility value scenario was analyzed. In addition, we explored the cost-effective price of camrelizumab in the scenario analysis.
Results
Base-case analysis
For all patients with hepatocellular carcinoma, the expected survival times for the sorafenib and camrelizumab groups were 2.150 and 2.323 life-years, respectively, and the camrelizumab group gained 0.173 more life-years than the sorafenib group. The lifetime treatment costs were US$13 154 and US$21 494, respectively. The camrelizumab group resulted in 0.239 more QALYs than the sorafenib group after accounting for quality of life, with an ICER of US$34 897 per QALY. The results of the base-case analysis were shown in Table 2.
Results of the base-case and scenario analyses.

Abbreviations: QALY, quality-adjusted life-year; ICER, incremental cost-effectiveness ratio; ALBI, albumin-bilirubin.
Sensitivity analysis
The results of the univariate sensitivity analysis for the base case were given in Figure 3. For all patient populations, the ICER values for the camrelizumab plus rivoceranib regimen versus the sorafenib regimen varied in the range of US$28 646/QALY to US$74 237/QALY as the parameter values changed. The ICER values were most sensitive to the utility value of PFS in the camrelizumab arm, followed by the price of sorafenib and the price of camrelizumab. When the utility value for PFS in the camrelizumab group decreased to 0.76 from the baseline value, the ICER value increased to US$72 255/QALY (Figure 3). When the cost of sorafenib was set at US$13.59/200 mg, the ICER in the camrelizumab group was significantly lower than the WTP threshold at US$9004/QALY.
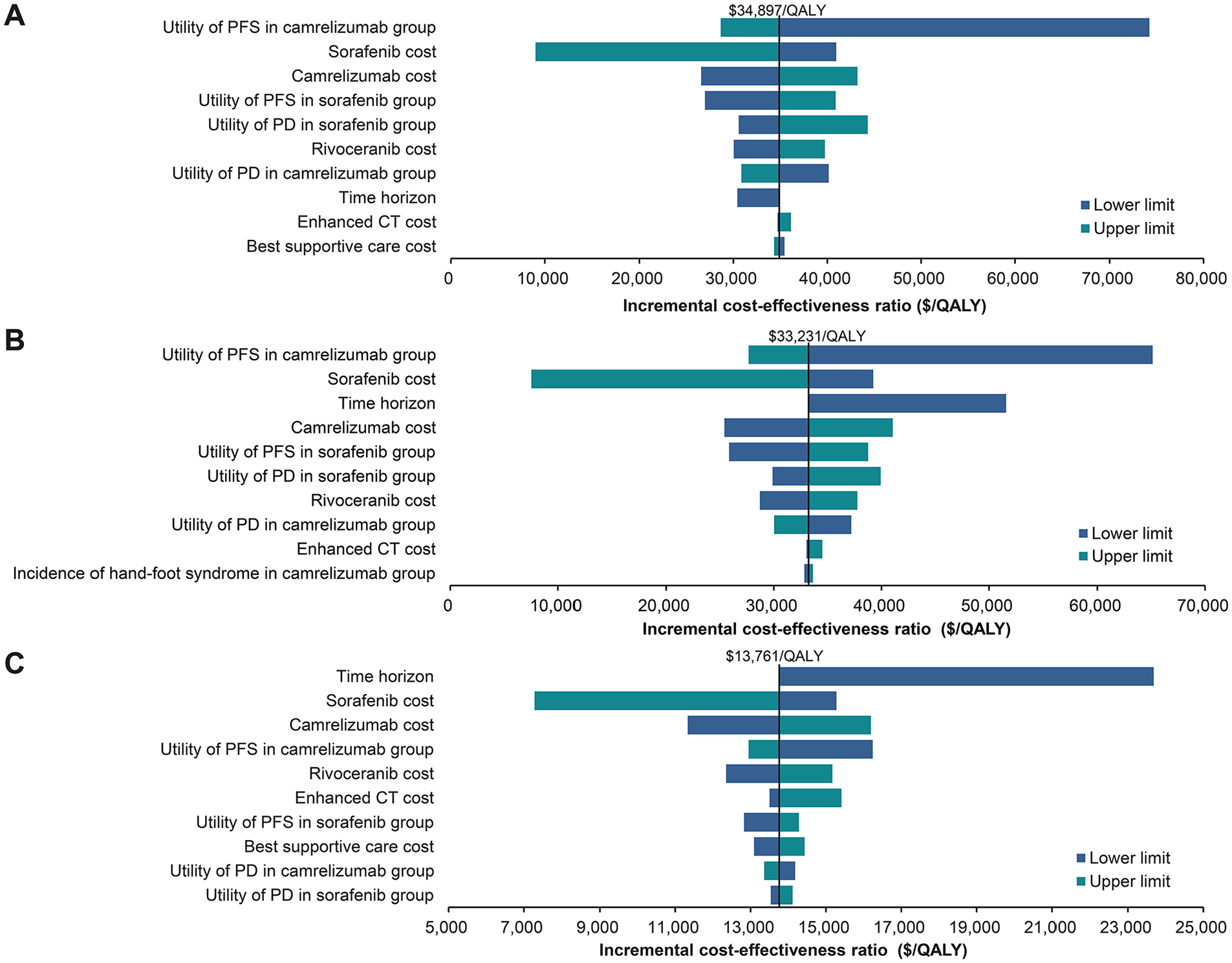
Tornado diagram of the univariate sensitivity analysis of the ICER of camrelizumab plus rivoceranib over sorafenib. (A) For all patient populations; (B) For ALBI grade 1 subgroup; (C) For ALBI grade 2 subgroup. ICER indicates incremental cost-effectiveness ratio; QALY, quality-adjusted life-year; EV, expected value; PFS, progression-free survival; PD, progressive disease; CT, computed tomography; ALBI, albumin-bilirubin.
The results of the probabilistic sensitivity analysis were shown in Figures 4 and 5. The cost-effectiveness acceptability curves showed that the sorafenib regimen was more cost-effective in all patients under the WTP thresholds of 1 to 2 times GDP per capita. At the WTP threshold of US$36 780/QALY, the probability of camrelizumab regimen being cost-effective was 56%. The incremental cost-effectiveness scatter plot showed the incremental cost and incremental effectiveness results of 1000 Monte Carlo simulations. For most simulations, ICER values fell within the first quadrant and below the WTP line of US$36 780/QALY, indicating that the camrelizumab regimen had a higher probability of cost-effectiveness at a threshold of 3 times GDP per capita (Figure 5). The cost-effectiveness probabilities for the camrelizumab regimen and the sorafenib regimen at different payment thresholds were presented in Supplemental Table S6.

Cost-effectiveness acceptability curves. (A) For all patient populations; (B) For ALBI grade 1 subgroup; (C) For ALBI grade 2 subgroup. QALY indicates quality-adjusted life-year; GDP, gross domestic product; ALBI, albumin-bilirubin.

Incremental cost-effectiveness scatter plot. QALY indicates quality-adjusted life-year; GDP, gross domestic product; ALBI, albumin-bilirubin.
Subgroup analysis
Camrelizumab plus rivoceranib yielded ICERs of US$33 231/QALY and US$13 761/QALY in ALBI grade 1 and ALBI grade 2 subgroups, respectively, compared with sorafenib monotherapy. Thus, the ICERs of the camrelizumab group versus sorafenib group were below the WTP threshold of US$36 780/QALY for all patients and both subgroups. Patients with ALBI grade 2 had the smallest ICER, which was below 2 times GDP per capita (Table 2). In the ALBI grade 1 and ALBI grade 2 subgroups, in addition to the utility value of the PFS of the camrelizumab group and the price of sorafenib, time horizon also had a pronounced impact on the ICER as well. Notably, in the ALBI grade 1 subgroup, the camrelizumab regimen would no longer be cost-effective when the time horizon was less than 7.20 years (Figure 3B and C). Probabilistic sensitivity analysis showed that the probability of cost-effectiveness for the ALBI grade 1 and ALBI grade 2 subgroups was 58% and 100%, respectively, at the willing-to-pay threshold of US$36 780/QALY (Supplemental Table S6). The results of 1000 Monte Carlo iterations showed that most ICER results of the 2 subgroups were below the WTP threshold of 3 times GDP per capita [Figures 4 and 5).
Scenario analysis
When utility values were set at 0.76 for PFS and 0.68 for PD in both the camrelizumab and sorafenib groups, the results of the scenario analysis showed a substantial increase in the ICER value of the camrelizumab regimen compared with the base-case analysis (Table 2). More specifically, the ICER of all patients and ALBI grade 1 subgroup in the scenario analysis rose to US$68 103/QALY and US$58 623/QALY, respectively, both above the WTP threshold of US$36 780/QALY and no longer cost-effective. Only in the ALBI grade 2 subgroup, the ICER value of camrelizumab regimen was lower than that of WTP, indicating that camrelizumab was cost-effective.
The results of the univariate sensitivity analysis for this scenario showed that the parameters that had a great impact on the model were mainly utility of PFS, sorafenib cost, camrelizumab cost, utility of PD, and time horizon, and that the ICERs were inversely proportional to the utility of PFS, and sorafenib cost (Supplemental Figure S1). The ICER of sorafenib ranged from US$79 925/QALY to US$17 572/QALY when the cost of sorafenib ranged from US$0.73/200 mg to US$13.59/200 mg. At the WTP threshold of US$36 780 per QALY, the probability of the camrelizumab regimen being cost-effective was 1% for all patients, 3% for ALBI grade 1 subgroup, and 100% for ALBI grade 2 subgroup (Supplemental Figure S2). The incremental cost-effectiveness scatter plot showed that the ICER of camrelizumab versus sorafenib was less than 3 times GDP per capita only in the ALBI grade 2 subgroup (Supplemental Figure S3).
We varied the cost of camrelizumab while keeping other parameters constant to determine the cost-effective price of camrelizumab in the scenario analysis. The results showed that only when the cost of camrelizumab was reduced from US$368.62/200 mg (baseline price) to US$155.51/200 mg did the ICER equal the WTP threshold of US$36 780/QALY. This indicated that camrelizumab needed to be discounted by 58% from its baseline price to be cost-effective for all patients (Supplemental Figure S4).
Discussion
Based on the CARES-310 trial, we developed a partitioned survival model to assess the lifetime costs and effectiveness of camrelizumab plus rivoceranib as first-line therapy for patients with advanced hepatocellular carcinoma. The results indicated that patients in the camrelizumab group and the sorafenib group had expected survival times of 2.323 and 2.150 years, respectively. Compared with sorafenib monotherapy, camrelizumab plus rivoceranib cost US$8340 more but allowed patients to gain an additional 0.239 QALYs. Consequently, the cost per QALY gained for patients in the camrelizumab group was US$34 897, which was below the WTP threshold of US$36 780/QALY. This finding suggested that camrelizumab in combination with rivoceranib was cost-effective as first-line treatment for advanced hepatocellular carcinoma in China and was even more cost-effective in patients with ALBI grade 2. Nevertheless, the results of the scenario analysis differed significantly from the base-case results, showing that camrelizumab plus rivoceranib was cost-effective only in patients with ALBI grade 2. Furthermore, the camrelizumab regimen would only be cost-effective for all patients if the price of camrelizumab is reduced by 58% from its current value.
Our study found that, in addition to the health utilities of PFS and PD, the costs of sorafenib and camrelizumab significantly impacted the model. ICERs decrease with increasing health utilities, and enhanced supportive care for hepatocellular carcinoma patients may improve their quality of life. The camrelizumab arm had a marked cost-effectiveness advantage over the sorafenib originator in both the base case and scenario, but it would no longer be cost-effective if sorafenib was priced below US$2.43/200 mg. At present, there are 4 major generic versions of sorafenib sold in China, all of which have passed therapeutic equivalence evaluations and have been added to the National Centralized Drug Procurement List, with prices as low as US$0.73 per 200 mg, which makes generics an alternative to the originator. 18 This fact also suggests that there is still a large room for price reduction for sorafenib originator. Probabilistic sensitivity analysis showed that at a WTP threshold of 3 times GDP per capita, camrelizumab plus rivoceranib had a 56% and 100% probability of cost-effectiveness in all patients and ALBI grade 2 subgroups, respectively. In scenario analysis, the cost-effectiveness probability of camrelizumab plus rivoceranib dropped to 1% in all patients but remained 100% in the ALBI grade 2 subgroup. Therefore, camrelizumab combined with rivoceranib may be more cost-effective for the first-line treatment of advanced hepatocellular carcinoma patients with ALBI grade 2, whereas sorafenib might be more cost-effective for patients with ALBI grade 1. For patients with poor affordability, generic sorafenib can be considered at a price of less than US$2.43/200 mg.
Currently, ICIs targeting PD-1/L1 in combination with targeted drug regimens, such as atezolizumab plus bevacizumab and sintilimab plus bevacizumab, have been approved in China for the first-line treatment of advanced hepatocellular carcinoma. Several studies have evaluated the cost-effectiveness of atezolizumab plus bevacizumab compared with sorafenib from the perspective of the Chinese health care system. The results showed that the ICERs for atezolizumab plus bevacizumab ranged from US$61 613/QALY to US$160 049/QALY, all of which were higher than the WTP of 3 times China’s GDP per capita. 25 ,35 -39 Three studies reported ICERs of US$20 968 and US$51 877.36 per QALY for sintilimab plus bevacizumab compared with sorafenib, while another reported an ICER of US$75 150/QALY from the Chinese patient’s perspective.20,25,36 In addition, other investigations indicated that the ICERs of sintilimab plus bevacizumab biosimilar (IBI305) compared with sorafenib were US$23 352 per QALY, US$39 766 per QALY, and US$115 760/QALY.38 -40 Most studies concluded that sintilimab plus bevacizumab or bevacizumab biosimilar was not cost-effective. The ICER of camrelizumab plus rivoceranib compared with sorafenib was US$34 897 per QALY, which was below the WTP threshold of US$36 780 per QALY. Moreover, our study found that the lifetime treatment cost of US$21 494 per patient in the camrelizumab group was lower than the cost of US$59 018 for patients treated with the abovementioned cost-effective regimens (sintilimab plus bevacizumab biosimilar, US$59 018; and sintilimab plus bevacizumab, US$33 766).20,40 Therefore, compared with atezolizumab plus bevacizumab, sintilimab plus bevacizumab, or a bevacizumab biosimilar, camrelizumab plus rivoceranib remained a potentially cost-effective treatment option for advanced hepatocellular carcinoma in China. As this study is based on the perspective of the Chinese health care system, we did not evaluate the cost-effectiveness from the perspectives of European and American countries. However, several studies have assessed the cost-effectiveness of first-line treatments for hepatocellular carcinoma in different countries. These studies found that, compared with classic small-molecule targeted therapies, novel anticancer drugs produced by multinational pharmaceutical companies still need to reduce their prices to be considered cost-effective.32,41
Wei et al 14 and Lang et al 15 have conducted cost-effectiveness analyses of camrelizumab for the treatment of advanced hepatocellular carcinoma, obtaining ICER values of US$30 410.56/QALY and US$33 619.98/QALY, slightly lower than the results of this study. Both studies concluded that camrelizumab plus rivoceranib is cost-effective as a first-line treatment for advanced hepatocellular carcinoma in China, and this supports our findings. However, compared with Wei and Lang et al.’s research, our study used derived survival data from the Kaplan-Meier curve during the trial follow-up period and extrapolated data beyond the follow-up period. We also conducted subgroup analyses based on subgroup survival data reported from the CARES-310 trial, avoiding assumptions about subgroup survival data when the proportional hazards assumption is violated. This approach reduced errors in both overall and subgroup survival data. Following progression after first-line treatment, patients in our model received treatments such as lenvatinib, regorafenib, and sintilimab, closely aligning with the trial protocol. In addition, this study included end-of-life care costs, drug administration costs, and major laboratory tests mentioned in the trial protocol to comprehensively reflect the real-world costs that patients might incur. We also accounted for the disutility of all adverse events with an incidence greater than 5%, enhancing the reliability of our results. Furthermore, we conducted scenario analyses with different utility values and explored the impact of time horizons on the model results, thereby enriching the cost-effectiveness analysis and enhancing its practical relevance.
This study has several limitations. First, as the CARES-310 trial only reported 28 months of survival data, we used mathematical modeling to extrapolate the survival curves and estimate survival data beyond 28 months. This was necessary to simulate the lifetime health outcome and costs for patients. However, the extrapolated survival data may differ from real-world outcomes, potentially leading to an ICER deviation. Future cost-effectiveness analyses using real-world data are needed to validate these results. Then, because the CARES-310 trial did not report the duration of the various subsequent regimens, we assumed a mean duration of subsequent therapy of 5.6 months, which may have been inconsistent with trial and thus biased the model results. In addition, we only considered grade 3 and higher adverse events with an incidence ⩾5% in our study. Moreover, we only took into account subsequent treatment regimens where the proportion of treated patients was at least 5%. These restrictions could lead to an underestimation of treatment costs and an overestimation of the health outputs for both treatment groups. Finally, due to the large number of tests in the CARES-310 trial, only the main tests were included in this study, which may also underestimate the total cost of treatment and yield inaccurate results.
Conclusions
From the perspective of the Chinese health care system, camrelizumab plus rivoceranib is a cost-effective regimen compared with sorafenib for the first-line treatment of unresectable hepatocellular carcinoma at a WTP threshold of US$36 780 per QALY. The ALBI grade 2 subgroup is more likely to be cost-effective after considering the ALBI scores of patients with hepatocellular carcinoma. When the cost of sorafenib drops below US$2.43/200 mg, camrelizumab plus rivoceranib is no longer cost-effective. For Chinese hepatocellular carcinoma patients with poor affordability, low-priced generic sorafenib that has completed the therapeutic equivalence evaluation may be a therapeutic alternative.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241299393 – Supplemental material for First-Line Camrelizumab Plus Rivoceranib in Advanced Hepatocellular Carcinoma: A China-Based Cost-Effectiveness Analysis
Supplemental material, sj-docx-1-onc-10.1177_11795549241299393 for First-Line Camrelizumab Plus Rivoceranib in Advanced Hepatocellular Carcinoma: A China-Based Cost-Effectiveness Analysis by Guiyuan Xiang, Yueyue Huang, Ni Zhang, Xinyu Du, Yuanlin Wu, Lanlan Gan, Yanping Li, Tingting Jiang and Yao Liu in Clinical Medicine Insights: Oncology
Footnotes
Author Contributions
GX and YL were responsible for the conception and design of the study. GX drafted the article. YH, NZ, XD, YW, LG, YL, and TJ contributed to the data collection, analysis and interpretation. YL supervised the project. All authors were involved in the study, critically revised the article, and gave final approval.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Chongqing Clinical Pharmacy Key Specialties Construction Project.
Data Availability
All the data are presented in the article and the supplemental material; further inquiries can be directed to the corresponding author.
Ethics approval
This study was based on published data and did not contain personal information; therefore, ethical approval was unnecessary.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
