Abstract
Background:
The HIMALAYA trial found that durvalumab plus tremelimumab significantly prolonged progression-free survival and overall survival in patients with unresectable hepatocellular carcinoma (HCC) compared with sorafenib.
Objective:
This study aimed to investigate the cost-effectiveness of durvalumab plus tremelimumab compared with sorafenib in the first-line HCC setting.
Design:
A Markov model-based cost-effectiveness analysis.
Methods:
We created a Markov model to compare healthcare costs and clinical outcomes of HCC patients treated with durvalumab plus tremelimumab in the first-line setting compared with sorafenib. We estimated transition probabilities from randomized trials. Lifetime direct healthcare costs, quality-adjusted life-years (QALYs), and incremental cost-effectiveness ratios were calculated for first-line durvalumab plus tremelimumab compared with sorafenib from a US payer’s perspective.
Results:
In the base case, first-line durvalumab plus tremelimumab was associated with an improvement of 0.29 QALYs compared with sorafenib. While both treatment strategies were associated with considerable lifetime expenditures, first-line durvalumab plus tremelimumab was less expensive than sorafenib ($188,405 vs $218,584). The incremental net monetary benefit for durvalumab plus tremelimumab versus sorafenib was $72,762 (valuing QALYs at $150,000 each). The results of durvalumab plus tremelimumab were better in terms of costs and health outcomes in patients with HBV-related HCC and high alpha-fetoprotein levels.
Conclusion:
First-line durvalumab plus tremelimumab was estimated to be dominant for the treatment of unresectable HCC compared with sorafenib from a US payer’s perspective.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the most common type of primary liver cancer and is a significant and growing cause of cancer-related death in the United States. 1 Previously approved agents for first-line therapy, such as sorafenib, lenvatinib, and bevacizumab combined with atezolizumab, have focused on angiogenesis 2 . In the last few years, immune-checkpoint inhibitors (ICIs) have revolutionized cancer therapy and have gained an increased interest in the treatment of HCC. 3
Durvalumab (programmed death ligand 1 inhibitor) plus tremelimumab (cytotoxic T lymphocyte associate protein-4 inhibitor) showed high efficacy and low toxicity in phase I and II clinical trials. 4 The randomized, open-label, sponsor-blind, multicenter, global, phase III HIMALAYA trial 5 was therefore designed to evaluate Single Tremelimumab Regular Interval Durvalumab (STRIDE) versus sorafenib in patients with unresectable HCC who had not been previously treated with systemic therapy. The median overall survival (OS) was 16.43 months (95% CI, 14.16–19.58) with STRIDE and 13.77 months (95% CI, 12.25–16.13) with sorafenib. 5 The frequency of grade 3/4 treatment-emergent adverse events (AEs) is comparable in STRIDE patients (50.5%) and sorafenib patients (52.4%). The first combination of anti-PD-L1 and anti-CTLA4 antibodies for HCC treatment showed an acceptable and manageable toxicity profile. Based on these exciting results, FDA approved this combined therapy as the first-line treatment for advanced HCC. 6
Evidence-based treatments are limited for advanced HCC, 7 once a new intervention is on the market, the availability and cost-conscious priority for patients’ choice become an essential issue. Hereby, this study aimed to investigate the cost-effectiveness of durvalumab plus tremelimumab compared with sorafenib in advanced HCC from a US payer’s perspective.
Methods
Target population and treatment
Based on the HIMALAYA study, 5 a total of 393 patients were assigned to receive STRIDE, and 389 patients received sorafenib. The STRIDE regimen contained 300 mg of tremelimumab for one dose plus 1500 mg of durvalumab every 4 weeks. The 400 mg of sorafenib was given twice daily. Treatment continued until progression, unacceptable toxicity, consent withdrawal, or other discontinuation criteria were met. 5 After treatment discontinuation, 40.7% and 45.0% of patients in the STRIDE and sorafenib arms, received subsequent second-line anticancer therapy, respectively. This cost-effectiveness study followed the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) reporting guideline, 8 shown in Supplemental Table 1.
Markov model
Long-term costs and health outcomes were simulated with a Markov model. The Markov model (Figure 1) simulated three health states: progression-free, progression, and death. The model assumed that patients transitioned from progression-free status to death via a progression state, as performed in previous cost-effectiveness analyses.9,10 Transitions between the health states were based on 1.5-month cycles. All the costs and quality-adjusted life-years (QALYs) were discounted at 3% per year, 11 with a lifetime horizon of 10 years.
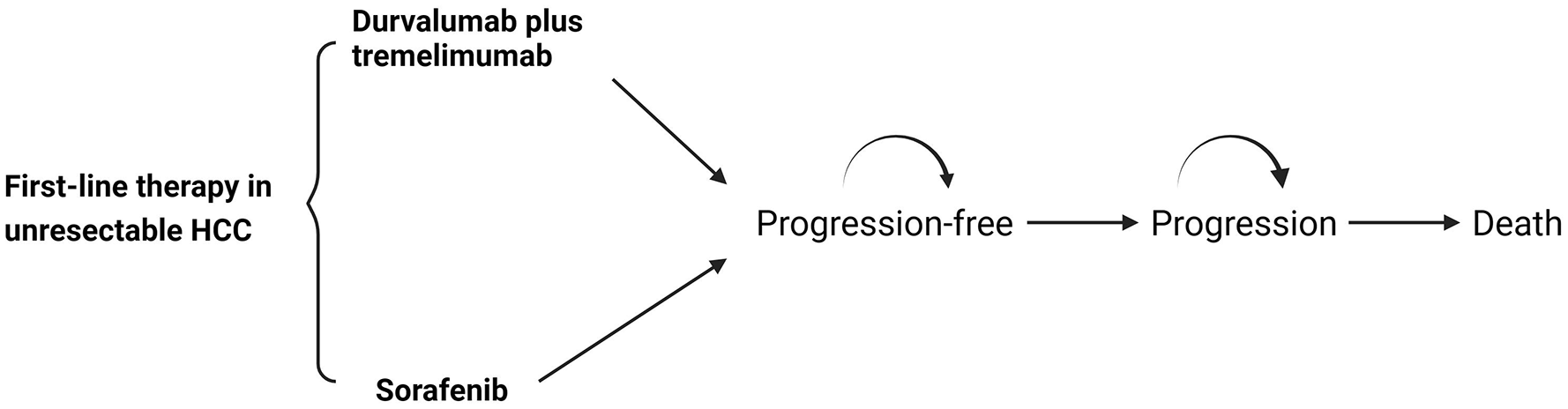
Markov model.
Cost estimate
Only direct costs were considered including the costs for the regimen, costs for AE management, costs of drug administration, costs of subsequent active treatment, costs of best supportive care, and costs of terminal care per patient, displayed in Table 1. The costs for subsequent active treatment, best supportive care, and terminal care per patient came from a cost-effectiveness analysis of first-line treatments in advanced HCC. 12 The cost for drugs was based on the Red Book accessed in 2023. 13 The cost for durvalumab was $466.79 per 50 mg for a 6-week cost of $21,005.55. The cost for tremelimumab was $3120 per 20 mg, for a 300 mg dose cost of $46,800. The cost for sorafenib was $210.83 per 200 mg for a cost of $35,419.44 per 6 weeks. We considered costs for the following adverse effects: diarrhea, palmar–plantar erythrodysesthesia syndrome, and hypertension because these side effects were the most frequently and clinically relevant grade 3 or 4 events in the trial. Costs of treating AEs 14 as well as drug administration 15 came from the published papers. All the costs were inflated to 2023 values using the Medical-Care Inflation data set. 16
Input parameters.

AE, adverse event; OS, overall survival; PFS, progression-free survival.
Transition probability estimate
The GetData Graph Digitizer 17 was used to extract the data points on the Kaplan–Meier curve. The Kaplan–Meier OS and progression-free survival curves of durvalumab plus tremelimumab and sorafenib obtained from the Weibull regression and empirical Kaplan–Meier curves in the HIMALAYA trial are shown in Supplemental Figure 1.18,19 We used the Weibull distribution in our analysis as it is a flexible method widely utilized in survival analysis for cancer patients.20–23 Transition probabilities were calculated using the formula: p(t) = 1 − exp[λ(t − 1)γ − λtγ] where λ represented the scale of the distribution, γ represented the shape of the distribution, and t was the Markov cycle. The survival curves for each strategy are depicted in the modeled survival curve. The input survival parameter in either group is shown in Table 1.
Utility estimate
Health state utility scores were derived from previously published literature: 0.76 for the progression-free state and 0.68 for the progressed disease.14,24,25
Outcomes
We measured costs in 2023 dollars and QALYs. For an intervention that was both more expensive and had higher QALYs, we calculated an incremental cost-effectiveness ratio (ICER) which is the incremental cost divided by the incremental effectiveness. We assumed the willingness-to-pay for QALYs was $150,000 from a US payer’s perspective. 26 In cases where an intervention may be less expensive with higher QALYs, the interpretation of the magnitude of the ICER becomes problematic, so we calculated outcomes in terms of incremental net monetary benefit (INMB) which was computed as the increased QALYs multiplied by the willingness-to-pay and subtract the incremental costs. Using this approach, if an INMB ⩾ 0, the intervention of interest would be considered cost-effective relative to the alternative.27,28 This can be particularly useful for sensitivity analysis.
Sensitivity analysis
The hazard ratio (HR) between durvalumab plus tremelimumab and sorafenib that was used to estimate the scale parameters was calculated using the following equation: HR × γRT. 29 We assumed a constant reduced hazard of progression and death with durvalumab plus tremelimumab compared to sorafenib. We varied the HR in sensitivity analysis. Further subgroup analyses were performed for the prespecified subgroups that were reported in the HIMALAYA trial by varying the HRs for OS. During one-way sensitivity analyses, model parameters were varied across the ranges outlined in Table 1 to determine the impact on the NMB. The costs and utility values were varied within a ±30% range, while the survival parameters were varied with a ±20% range. To investigate the uncertainty of cost-effectiveness, we performed a probabilistic sensitivity analysis with 10,000 Monte Carlo simulations, each time randomly sampling from the distribution of model inputs.
Results
Base-case analysis
The results from the base-case analysis are shown in Table 2. The total lifetime costs for durvalumab plus tremelimumab are $188,405, while costs for sorafenib are $218,584. The QALYs for durvalumab plus tremelimumab are 1.75 and that for sorafenib are 1.46. This makes durvalumab plus tremelimumab dominant (cost-saving) when compared to sorafenib. Valuing QALYs at $150,000 each, the value of the durvalumab plus tremelimumab treatment strategy is $73,899, and that for sorafenib is $1137, leading to the INMB of $72,762 for durvalumab plus tremelimumab compared to sorafenib.
Cost and outcome results in the base-case analysis.
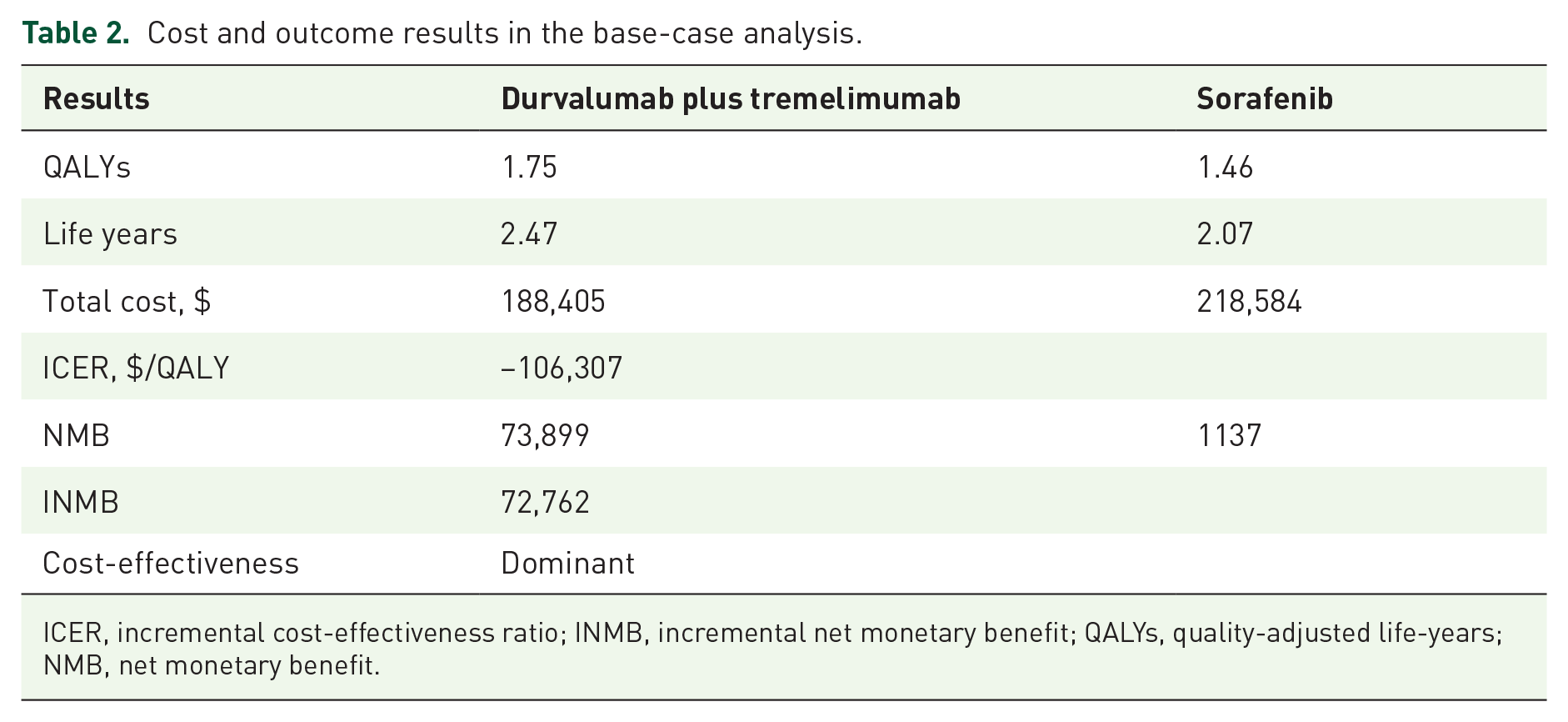
ICER, incremental cost-effectiveness ratio; INMB, incremental net monetary benefit; QALYs, quality-adjusted life-years; NMB, net monetary benefit.
Deterministic sensitivity analyses
The tornado diagram for durvalumab plus tremelimumab is shown in Figure 2. Discount rate, the OS of durvalumab plus tremelimumab, and the cost of durvalumab are the major sensitive parameters comparing durvalumab plus tremelimumab to sorafenib. Subgroup analysis showed that varying the HR of durvalumab plus tremelimumab compared with sorafenib across the subgroup in the HIMALAYA trial did lead to durvalumab plus tremelimumab having a positive INMB when compared to sorafenib (Table 3). Durvalumab plus tremelimumab achieved the most beneficial results in terms of health outcomes and costs, in the group with HBV-related HCC and high AFP level, with the INMB of $88,336.

Tornado diagram of one-way sensitivity analyses.
Subgroup analysis of cost-effectiveness by varying the hazard ratios of overall survival.

BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; ICER, incremental cost-effectiveness ratio; INMB, incremental net monetary benefit; QALY, quality-adjusted-life year; NMB, net monetary benefit; OS, overall survival; WTP, willingness-to-pay.
Probabilistic sensitivity analysis
In 10,000 simulations, durvalumab plus tremelimumab was often dominant and would be considered cost-effective in 86% (valuing QALYs at $100,000) and 91% (valuing QALYs at $150,000) of iterations (Table 3). The majority of simulations fell in the southeast quadrant of the incremental cost-effectiveness plane, indicating durvalumab plus tremelimumab as a dominant strategy (Figure 3). The result for Monte Carlo simulations is shown in Table 4.

Cost-effectiveness scatterplot.
Monte Carlo simulations.

IQR, interquartile range; QALY, quality-adjusted life-year.
Discussion
This study showed a single priming dose of tremelimumab plus once-monthly durvalumab is dominant (cost-saving) in patients with unresectable HCC compared with sorafenib. Generally, the cost-effectiveness of STRIDE versus sorafenib remained consistent across the subgroups.
The most influential parameters in our model were the discount rate, the Weibull OS gamma parameter in the durvalumab plus tremelimumab group, the cost of durvalumab, and the utilities of the progression and progression-free health states. The high cost of tremelimumab could pose a significant obstacle to the widespread utilization of this medication. This may decrease its accessibility and affordability for patients.
To the best of our knowledge, this is the first cost-effectiveness analysis of durvalumab plus tremelimuab in advanced HCC and was the only regimen yet found to be cost-effective or dominant compared with sorafenib in the first-line setting from a US payer’s perspective. Another approved immunotherapy combination therapy of atezolizumab and bevacizumab was not a cost-effective strategy compared with sorafenib for the first-line treatment of unresectable HCC in the USA.12,25 Of note, participants who took STRIDE had a lower relative risk of experiencing a decline in their quality of life compared to those who took sorafenib. 30 Though we assumed equal utility values for both treatments in our model-based analysis due to the lack of STRIDE utility data, STRIDE was found to be the dominant treatment option, making our conclusion solid.
The etiology of HCC affects the immune response and reprograms the unique tumor microenvironment. 31 In the subgroup analysis, the most dominant subgroup to treat with durvalumab plus tremelimumab was HBV-infected patients. This is because highly suppressive PD-1hi Treg cells that are associated with a poor prognosis are selectively enriched in HBV-related versus non-viral HCCs, 32 which could provide an opportunity for anti-CTLA4 blockade to be effective in HBV-related HCC. 33 Another beneficial subgroup for durvalumab plus tremelimumab was patients with AFP levels higher than 400 ng/ml. Intriguingly, AFP is a well-known negative prognostic factor for survival in HCC, but immunotherapy should not be withheld solely due to high AFP level. 34 Based on the results of cost-effectiveness and efficacy, first-line dual immunotherapy with durvalumab plus tremelimumab should be prioritized for HBV-related HCC and patients with AFP high level ⩾400 ng/ml. Of note, for patients with non-viral etiology of chronic liver disease, the only regimen confirmed to significantly improve OS, compared with sorafenib, is durvalumab plus tremelimumab. 35 In this population, the INMB was $76,946 for durvalumab plus tremelimumab, showing good value.
A network meta-analysis of phase III trials showed that anti-PD-1 therapy with nivolumab was associated with a lower proportion of AEs, whereas among ICI combinations, durvalumab plus tremelimumab reported the lowest risk of AEs. 36 The grade 3 or 4 AEs occurred in 50.5% and 52.4% of patients receiving STRIDE and sorafenib in the HIMALAYA study. Based on our study, the total cost of AE management in the durvalumab plus tremelimumab group was also much lower than that in the sorafenib group. Therefore, durvalumab plus tremelimumab brought a lower toxicity-related cost burden.
The HIMALAYA was the first trial to demonstrate the benefit of dual ICIs, representing a new treatment option. 2 Dual immunotherapy is usually presumed to not be cost-effective; however, our model-based cost-effectiveness study indicated that reasonable application of immunotherapy not only brings significant efficacy, fewer AEs, and survival advantages but also can be dominant compared with first-line targeted therapy. Implementing scientific administration orders and reasonable combination therapies wound prolong patient survival and enhance overall cost-effectiveness.
This study has some limitations. First, clinical efficacy inputs were based on the randomized controlled trial, rather than real-world evidence of efficacy, which may limit generalizability. Second, the health-related quality-of-life associated with disease used came from previous literature. Third, although the calibrated survival curves based on the Weibull distribution matched well with the trial, assumptions about survival parameters remain a concern. To mitigate this limitation, we conducted extensive sensitivity analyses, demonstrating that STRIDE remained cost-effective across a broad spectrum of model inputs. Furthermore, the comparator in this cost-effectiveness analysis chose sorafenib, the long-standing standard first-line therapy in advanced HCC. As therapeutic advances have dramatically evolved, 37 future cost-effectiveness analyses should consider comparing to constantly updating first-line therapies in advanced HCC.
Conclusion
Based on this Markov model study, first-line durvalumab plus tremelimumab is dominant compared with sorafenib in patients with unresectable HCC from a US payer’s perspective.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241274625 – Supplemental material for Cost-effectiveness analysis of durvalumab plus tremelimumab as first-line therapy in patients with unresectable hepatocellular carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241274625 for Cost-effectiveness analysis of durvalumab plus tremelimumab as first-line therapy in patients with unresectable hepatocellular carcinoma by Weiting Liao, Huiqiong Xu, David Hutton, Qiuji Wu, Yang Yang, Mingyang Feng, Wanting Lei, Liangliang Bai, Junying Li and Qiu Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
