Abstract
Background:
Hepatocellular carcinoma (HCC) is the third leading cause of cancer-related mortality globally. Recent advancements in targeted therapies have improved outcomes for advanced HCC, yet therapeutic options remain limited. The CARES-310 trial demonstrated that camrelizumab plus rivoceranib significantly improves survival compared to sorafenib for advanced HCC.
Objectives:
This study aimed to evaluate the cost-effectiveness of camrelizumab plus rivoceranib as a first-line treatment for unresectable HCC from the Chinese health system perspective.
Design:
The cost-effectiveness analysis.
Methods:
A partitioned survival model was constructed to estimate clinical and economic outcomes for patients with unresectable or metastatic HCC. The model included three health states: progression-free, progression disease, and death. The hypothetical cohort consisted of patients aged ⩾18 with HCC who had not received systemic therapy, reflecting the CARES-310 trial. Clinical data were derived from the CARES-310 trial and extrapolated using standard parameter distributions. Direct medical costs and utilities were sourced from the CARES-310 trial and published literature.
Results:
The 10-year cost of camrelizumab plus rivoceranib was higher than sorafenib (USD 28,148.01 vs USD 20,997.86). Camrelizumab plus rivoceranib yielded an additional 0.26 quality-adjusted life-years (QALYs) with an incremental cost of USD 7150.15, resulting in an incremental cost-effectiveness ratio of USD 27,633.75/QALY. Sensitivity analyses confirmed the robustness of the base-case results.
Conclusion:
Camrelizumab plus rivoceranib is likely a cost-effective first-line treatment for unresectable HCC from a Chinese health system perspective. This study highlights the need for additional real-world data to validate these findings and guide clinical decision-making for HCC.
Plain language summary
Why was the study done? Hepatocellular carcinoma (HCC), a type of liver cancer, is a major cause of cancer deaths globally, and treatment options are often limited. New therapies, like the combination of camrelizumab and rivoceranib, may improve patient outcomes, but it is important to understand whether they are worth the cost. What did the researchers do? The research team analyzed the costs and health benefits of using camrelizumab combined with rivoceranib compared to sorafenib, the standard treatment, over a 10-year period. They aimed to find out if the new treatment offers better value for money and improves the quality of life for patients with unresectable HCC. What did the researchers find? The study found that while the total cost of camrelizumab and rivoceranib was higher than sorafenib, it provided additional years of quality-adjusted life for patients. The cost-effectiveness ratio indicated that this combination therapy is likely a good investment for the healthcare system in China. What do the findings mean? These results suggest that camrelizumab combined with rivoceranib could be an effective and financially reasonable option for treating patients with unresectable HCC. The study also emphasizes the need for further real-world data to validate these findings and inform future treatment decisions.
Keywords
Introduction
Hepatocellular carcinoma (HCC) stands as the third leading cause of cancer-related mortality worldwide, constituting 8.9% of the global neoplasm burden. 1 This sobering statistic is underscored by the unfortunate reality that merely 30%–40% of patients receive diagnoses during early stages amenable to potentially curative interventions. 2 Over the past decade, significant strides have been made with the advent of novel agents, including lenvatinib- and sorafenib-based targeted therapies, substantially improving outcomes for those with advanced HCC and prolonging median overall survival (OS) to 10–15 months.3,4 Building on these advancements, the combinations of targeted therapy and immunotherapy have been rigorously tested in both preclinical and clinical trials, demonstrating even greater potential to enhance treatment efficacy for advanced HCC. 5
The combinations of targeted therapies, which inhibit key signaling pathways involved in tumor growth, and immunotherapies, particularly immune checkpoint inhibitors (ICIs) such as PD-1/PD-L1 antibodies, work synergistically by addressing both tumor-specific pathways and the immune evasion mechanisms employed by cancer cells.6–9 This approach aims to overcome the limitations of individual therapies, offering improved clinical outcomes, especially in advanced HCC.10,11
Notably, the CARES-310 trial, an open-label, phase III randomized clinical study conducted from June 28, 2019, to March 24, 2021, evaluated the efficacy and safety of camrelizumab combined with rivoceranib versus sorafenib for the treatment of advanced metastatic or unresectable HCC. 12 The results demonstrated a significant improvement in median progression-free survival (PFS) with camrelizumab plus rivoceranib compared to sorafenib (5.6 months vs 3.7 months; hazard ratio (HR) 0.52; 95% confidence interval (CI): 0.41–0.65; one-sided p < 0.0001). Additionally, median OS was significantly extended in the camrelizumab-rivoceranib group relative to sorafenib (22.1 months vs 15.2 months; HR 0.62; 95% CI: 0.49–0.80; one-sided p < 0.0001). Based on these findings, the “camrelizumab-rivoceranib” combination was approved by the National Medical Products Administration in early 2023 as a first-line treatment for patients with unresectable or metastatic HCC.
However, health decision-makers and clinicians must incorporate cost-effectiveness considerations into healthcare decisions to optimally allocate limited resources, which is especially crucial for high-cost treatment for oncology. 13 Given the rapid progression of HCC, more rigorous and thoughtful cost-effectiveness analyses (CEAs) are required. Currently, two CEAs comparing camrelizumab plus rivoceranib with sorafenib—utilizing a Markov model and a partitioned survival model in China and the United States, respectively—have yielded inconsistent results.14,15 These discrepancies are likely due to methodological differences in factors such as cycle length, clinical effectiveness inputs, utility inputs, and cost inputs, aligning with our systematic review of the challenges in decision modeling methodology for economic evaluation of HCC interventions. 16
Thus, this study aimed to examine the cost-effectiveness of camrelizumab plus rivoceranib as a first-line therapy for advanced HCC from the Chinese health system perspective, taking into account the aforementioned challenges. The findings are expected to provide economic evidence for health decisions about camrelizumab plus rivoceranib for advanced HCC in China. In addition, the findings could be references for conducting economic evaluations of camrelizumab plus rivoceranib for HCC in other countries.
Methods
Analytical overview and model structure
A partitioned survival model was constructed to estimate the clinical and economic outcomes of patients with unresectable or metastatic HCC who did not previously receive systemic therapy from a Chinese healthcare system perspective. The model consisted of three discrete health states: progression-free (PF), progression disease (PD), and death (see Figure 1). The proportion of patients in different health states was estimated by the area under the curves of a group of non-mutually exclusive survival curves in each cycle. 17

Model structure of a decision tree combining the partitioned survival model with the three health states.
We assumed that all patients started in the PF state, following which they could remain in the PF state or move to the PD state or the death state at the end of each cycle. Once in the PD state, we assumed those patients were prevented from returning to the PF state. According to the treatment cycle in the CARES-310 trial, the model cycle duration was set to 28 days. A 10-year horizon was simulated, capturing more than 99% of patients in the death state, about a life-long time for advanced liver cancer. 18 This study followed the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022; Supplemental Table S1). 19 Statistical analyses were conducted using TreeAge Pro 2022 (TreeAge Software Inc.; Williamstown, MA) and R version 4.1.2 (R Foundation for Statistical Computing; Vienna, Austria).
Patient cohort
The hypothetical cohort of the population for this study was patients aged ⩾18 years old with histopathologically or cytologically confirmed unresectable or metastatic HCC, no previous systemic therapy, a baseline Child-Pugh class A liver function, and an Eastern Cooperative Oncology Group performance status of 0 or 1, which was in line with the patient characteristics of CARES-310 trial. 12 The intervention group received camrelizumab intravenously (200 mg) every 2 weeks and rivoceranib orally (250 mg) once a day. The control group received sorafenib orally (400 mg) twice a day. The subsequent treatment regimens referred to the CARES-310 trial and the NCCN Guidelines in Hepatocellular Carcinoma. 20 After disease progression, 108 of 272 patients (39.7%) in the camrelizumab plus rivoceranib group and 144 of 269 (53.5%) patients in the sorafenib group were treated with subsequent active therapy. The patients received second-line treatments, including lenvatinib orally (12 mg) once a day in patients weighing ⩾60 kg or best supportive care. Based on the National Healthcare Security Administration submission guideline, we assumed that a typical patient in China weighed 60 kg.
Clinical data inputs
The survival data in the camrelizumab plus rivoceranib and sorafenib groups were derived from the PFS and OS data in the CARES-310 trial 12 and extrapolated beyond the observation period using the methodology depicted by Guyot et al. 21 GetData Graph Digitizer version 2.26 (GetData Pty Ltd.; Kogarah, Australia) was performed to extract the data points from the PFS and OS curves, which were then employed to fit the six standard parameter distributions: Exponential, Gamma, Gompertz, Weibull, log-Logistic, and log-Normal. The eligible distribution was chosen based on the minimizing Akaike information criterion and Bayesian information criteria values, as well as visual fit (Supplemental Table S2 and Supplemental Figures S1–S4). Log-Normal distribution was finally selected as the best-fitted distribution for PFS curve of the camrelizumab plus rivoceranib group and PFS and OS curves of the sorafenib group. In addition, Weibull distribution was selected for the OS curve extrapolation of the camrelizumab plus rivoceranib group. The final shape and scale parameters are presented in Table 1.
Model parameters.

AE, adverse event; ALT, alanine transaminase; AST, aspartate transaminase; OS, overall survival; PD, progressive disease; PFS, progression-free survival; US, United States.
Cost and utility inputs
This study was conducted from the healthcare system perspective in China, only considering direct medical costs, including drug acquisition costs, costs of follow-up and monitoring, costs of drug administration, costs of end-of-life care, and costs of management for adverse events (AEs; Table 1). The costs were reported in US dollars (USD 1 = CNY 6.73), and all costs were adjusted for prices in 2022 using the Chinese consumer price index for medical care.
The prices of camrelizumab, rivoceranib, sorafenib, and lenvatinib were collected from the negotiation documents of the National Reimbursement Drug List by the National Healthcare Security Administration and local charges. At the same time, other costs were estimated using data from Chinese economic studies on the treatments for advanced HCC. According to the CARES-310 trial report, drug costs per cycle were obtained using the local price multiplied by dose schedules. Treatment-related grade ⩾3 AEs with an incidence rate of over 3% were included in this model. The costs of these AEs were calculated by multiplying the cost of managing per event by the corresponding incidence rate of AE.
A health utility preference was assigned for each health state on a scale of 0 (death) to 1 (perfect health). The European Quality-of-Life 5-Dimension 5-Level utility scores of PF and PD states in the model were derived from the CARES-310 trial. 12 The disutility values due to grade 1–2 and grade ⩾3 AEs were extracted from a previously published study (Table 1). 22 Each AE disutility value was calculated by multiplying the given disutility value by the AE incidence rate. We assumed all AEs would occur during the first cycle of the model. The duration-adjusted disutility was subtracted from the baseline PF utility.
Base-case analysis
The incremental cost-effectiveness ratio (ICER) was used as the cost-effectiveness result between camrelizumab plus rivoceranib against sorafenib and defined as the incremental cost per additional quality-adjusted life-year (QALY) gained. According to the Chinese Pharmacoeconomics Recommendation, when the ICER was lower than the prespecified willingness-to-pay (WTP) threshold of $35,856.95/QALY (three times of per capita gross domestic product (GDP) of China in 2022) in China, the cost-effectiveness was assumed. 25 Costs and health outcomes were discounted at an annual rate of 5%. 25
Sensitivity analysis
To test the robustness of the base-case result, one-way sensitivity analyses and probabilistic sensitivity analyses (PSA) were conducted. In the one-way sensitivity analyses, the variation range of each parameter was based on either the 95% CIs reported in the referenced studies or determined by a ±20% change from the base-case value (Table 1).
In the PSA, a Monte Carlo simulation with 10,000 iterations was performed by simultaneously sampling the main model inputs from the specific distributions. Gamma distributions were selected for cost parameters, and beta distributions were selected for utility and probability parameters. After the simulation, a cost-effectiveness plane and an acceptability curve were created to verify the likelihood that camrelizumab plus rivoceranib would be cost-effective at the prespecified WTP threshold.
Results
Base-case analysis
The base-case results of camrelizumab plus rivoceranib compared with sorafenib were summarized in Table 2. The 10-year cost of camrelizumab plus rivoceranib was higher than that of sorafenib (USD 28,148.01 vs USD 20,997.86). The health outcomes of camrelizumab plus rivoceranib and sorafenib were 1.72 and 1.46 QALYs, respectively. In comparison with sorafenib therapy, camrelizumab plus rivoceranib treatment yielded an additional 0.26 QALYs with an incremental cost of USD 7150.15, which was associated with an ICER of USD 27,633.75/QALY. Given the WTP threshold of three times of per capita GDP of China in 2022 (USD 35,856.95/QALY), the results suggested that camrelizumab plus rivoceranib was a cost-effective strategy.
Summary of cost and health outcome results in the base-case analysis.
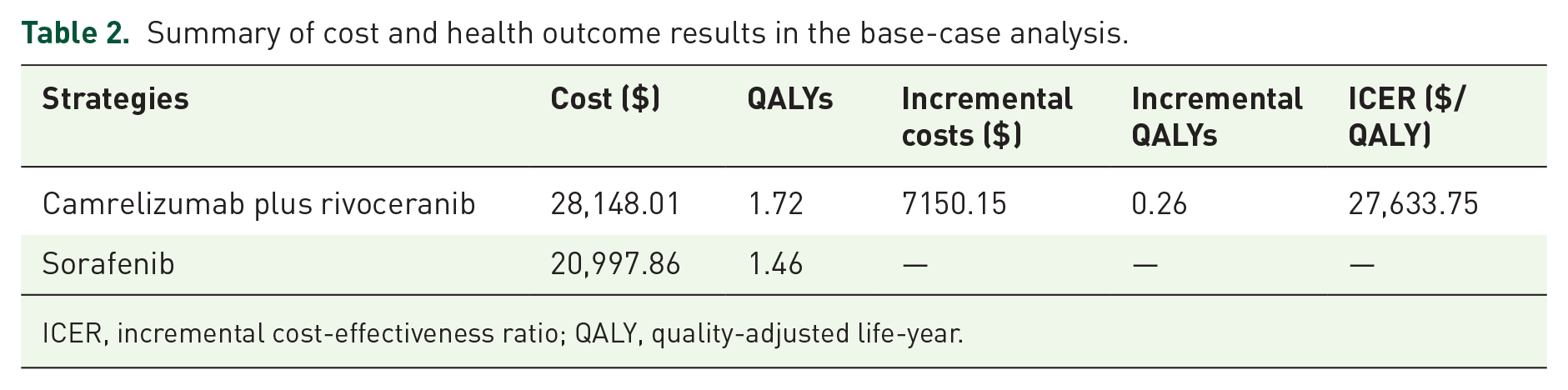
ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life-year.
Sensitivity analysis
One-way sensitivity analyses revealed that the ICER values were most sensitive to the price of camrelizumab, the proportion of patients receiving subsequent active therapy in the sorafenib group, and the utility of PD state in the sorafenib group (Figure 2). All the ICER results in the one-way sensitivity analyses ranged from USD 22,343.88/QALY to USD 33,431.57/QALY, which were lower than three times of per capita GDP of China (USD 35,856.95/QALY). The results were consistent with that in the base-case analysis.

Tornado diagram of one-way sensitivity analyses.
The cost-effectiveness plane of PSA showed that most of the ICERs were located in the northeast quadrant of the plane, suggesting camrelizumab plus rivoceranib with a higher cost but a better effect than sorafenib (Figure 3). In combination with the cost-effectiveness acceptability curve, it was illustrated that the camrelizumab plus rivoceranib regimen had a 63.61% probability of being cost-effective at the WTP threshold of three times of per capita GDP of China (USD 35,856.95/QALY, Figures 3 and 4).

Cost-effectiveness plane.

Cost-effectiveness acceptability curves.
Discussion
This economic assessment, based on aggregate data from the CARES-310 study, employs a partitioned survival model to evaluate the cost-effectiveness of camrelizumab plus rivoceranib versus sorafenib as a first-line treatment for unresectable or metastatic HCC from the perspective of the Chinese healthcare system.
Previous research has highlighted inconsistencies in the cost-effectiveness of ICIs as first-line therapies for advanced HCC.24,26–28 Our analysis found that camrelizumab plus rivoceranib, compared to sorafenib, provided an additional 0.26 QALYs at an incremental cost of USD 7150.15, resulting in an ICER of USD 27,633.75 per QALY gained. Given current pricing and WTP thresholds, the camrelizumab plus rivoceranib regimen appears to be a cost-effective alternative to sorafenib for unresectable HCC patients. Sensitivity analyses further confirmed the robustness of the base-case findings.
Methodologically, this study addresses the challenges identified in two previously published CEAs,14,15 and enhances the model in line with earlier recommendations. 16 For instance, the cycle length used by Wei et al. was inconsistent with the medication cycle of the CARES-310 trial. 15 As recommended in prior literature, the model cycle length should ideally align with the treatment cycle. 16 To address this, our model employs a 28-day cycle, corresponding to the treatment cycle in the CARES-310 trial.
Regarding clinical effectiveness inputs, Lang et al. utilized a state transition model but did not provide a clear method or formula for calculating state transition probabilities. 14 This omission underscores a common methodological challenge with state transition models, as highlighted in our previous research. 16 By contrast, this study uses a partitioned survival model, which estimates the proportion of patients in each health state by calculating the area under the survival curves, thus avoiding the need for detailed transition probabilities. 29
In terms of utility inputs, Lang et al. failed to account for the disutility caused by AEs, while Wei et al. did not use utility values representative of the CARES-310 trial population.14,15 Our study addresses these issues by optimizing the utility inputs. Furthermore, Wei et al. did not consider the costs associated with subsequent active therapy, a key cost driver in advanced HCC treatment. 15 In contrast, this study provides a more comprehensive cost analysis, estimating these costs based on data from the CARES-310 trial and the NCCN Guidelines in Hepatocellular Carcinoma.
Therefore, future model-based health economic evaluations of HCC therapies should carefully follow best practices in modeling to ensure robust and reliable outcomes. 16 Specifically, models should align their structure with clinical practices, as demonstrated by adjusting the cycle length to match the treatment schedule, as in the case of the CARES-310 trial. Additionally, selecting an appropriate modeling technique—such as the partitioned survival model used in this study, which avoids the limitations of state transition models—will ensure a more accurate representation of patient health states. Furthermore, clear explanations of key parameters, including transition probabilities and time-to-event distributions, are essential for transparency. Proper consideration of utility values, particularly by incorporating disutilities related to AE, along with comprehensive cost analyses, is also crucial. Finally, thorough sensitivity analyses and model validation should be prioritized to enhance the credibility and applicability of the findings.
The strengths of this study are noteworthy and deserve attention. Although therapies involving PD-1 blockade, either as monotherapy or in combination with other regimens, are increasingly favored for the treatment of advanced HCC, there remains a significant lack of data on the economic outcomes of ICIs in this setting. 30 To address this, the study integrates the latest evidence by optimizing the partitioned survival modeling approach, providing a comprehensive assessment of the economic outcomes of camrelizumab plus rivoceranib for treating unresectable HCC. Additionally, the study utilizes European Quality-of-Life 5-Dimension 5-Level data for patients receiving either camrelizumab plus rivoceranib or sorafenib in PF and PD states, primarily sourced from the CARES-310 trial. Notably, one-way sensitivity analyses highlighted the significant influence of PF and PD utilities on the ICER, underscoring the importance of these factors in determining economic outcomes.
However, several limitations were identified in the analysis. First, extrapolating health benefits beyond the observation period of the CARES-310 trial involved fitting parametric distributions to the reported Kaplan-Meier PFS and OS data. While efforts were made to validate the modeled and observed data, this approach may introduce uncertainty to the model outputs. Second, as per the CARES-310 trial and relevant guidelines, subsequent treatment regimens were simplified as lenvatinib and best supportive care. However, the complexity of second-line treatment regimens has a profound impact on experimental outcomes. Third, due to the absence of time-series data, the current analysis did not account for the fluctuations in costs over time, including costs related to follow-up time. Fourth, the evaluation excluded costs associated with grade 1 or 2 AE, potentially leading to overestimating the economic outcomes linked to camrelizumab plus rivoceranib. Despite these limitations, the insights provided by this analysis align with typical clinical practices for managing advanced HCC, offering valuable guidance for both physicians and policymakers.
Conclusion
The estimations indicated that camrelizumab plus rivoceranib was likely to emerge as a cost-effective first-line choice for patients with unresectable HCC from the Chinese healthcare system perspective, potentially providing a valuable reference to clinicians in treatment decision-making for advanced HCC. However, acknowledging the methodological constraints in this study, it underscores the necessity for additional high-quality real-world data, both clinical and economic, to be obtained.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241310314 – Supplemental material for Cost-effectiveness of camrelizumab plus rivoceranib versus sorafenib as first-line treatment of unresectable hepatocellular carcinoma
Supplemental material, sj-docx-1-tag-10.1177_17562848241310314 for Cost-effectiveness of camrelizumab plus rivoceranib versus sorafenib as first-line treatment of unresectable hepatocellular carcinoma by Huimin Zou, Yunfeng Lai, Xianwen Chen, Carolina Oi Lam Ung and Hao Hu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
