Abstract
Background:
Thyroid nodules, although mostly benign and symptomless, have a small chance of being cancerous, necessitating accurate diagnosis. This study aims to develop and validate a nomogram for differentiating malignant and non-malignant thyroid nodules in individuals with type 2 diabetes.
Methods:
The study included 484 patients with both thyroid nodules and type 2 diabetes who underwent thyroid gland lobectomy at Wenzhou Medical University Hospital. Optimal cutoff values for continuous variables were determined using ROC curve analysis. Significant factors identified in univariable analysis were used to construct the nomogram. The monocyte-to-high-density lipoprotein cholesterol ratio (MHR) was visualized through a histogram and scatter diagram. Discriminatory power was assessed using ROC analysis, and calibration curves ensured consistency. Decision curve analysis (DCA) evaluated clinical benefits.
Results:
The cohort was divided into a training group (70%) and an internal validation group (30%). The scatter diagram revealed a correlation between MHR levels and the proportion of goiter cases, with higher MHR levels associated with increased goiter incidence. The histogram showed higher average MHR levels in goiter patients compared to those with papillary thyroid carcinoma (PTC) in both groups. Multivariate logistic regression identified age, total cholesterol (TC), triglyceride (TG), fasting blood sugar (FSG), fibrinogen, lymphocyte-to-monocyte ratio (LMR), and MHR as independent predictive factors for malignancy in thyroid nodules with type 2 diabetes. The nomogram achieved high discrimination, with C-index values of 0.901 (training data set) and 0.760 (internal validation data set). Calibration curves displayed good agreement, and DCA demonstrated significant net clinical benefits.
Conclusions:
MHR is associated with sex, serum cholesterol levels, and peripheral blood cell counts, making it a potential novel biomarker for differentiating between PTC and goiter in type 2 diabetes patients.
Keywords
Introduction
Thyroid nodule (TN) is present in more than 50% of the randomly chosen population, and it is quite a common disorder of the endocrine system. 1 According to the existing data, approximately 7% to 15% of TNs are malignant, and 5% cause thyroid dysfunction or pressing symptoms, though most TNs are benign. 2 More recently, except for some well-known factors including age, gender, iodine deficiency, and thyroid-stimulating hormone (TSH), the role of systemic inflammatory response in the formation and progression of TN has also attracted the interest of researchers.3-6
Diabetes mellitus is nowadays known as another common disorder of the endocrine system complicated with high incidence and mortality. It has been discovered that the levels of inflammatory cytokines in patients with diabetes are much higher than that of the non-diabetes population.7,8 And the values of neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) are detected to be much higher in the diabetic group than in the control group.9,10 Thus, the discovery aforementioned raises an interesting question about whether the complication of type 2 diabetes mellitus (T2DM) will influence the level of monocyte-to-high-density lipoprotein cholesterol ratio (MHR), lymphocyte-monocyte ratio (LMR), and other markers of systemic inflammatory response in TN patients.
Presently, MHR has been mostly discovered and established links with some cardiovascular diseases. 11 A rare study has been found focusing on the relationship between MHR and TNs. Only Liu et al found that MHR is strongly associated with the presence and size of TN irrespective of gender. 12 However, there is no data on the role of MHR in differentiating the malignancy of TN in patients complicated with T2DM. To address these research gaps aforementioned, this retrospective case-control study was conducted.
Methods
Patients’ collection and exclusion criteria
Patients who were diagnosed with TN and T2DM and treated with lobectomy of the thyroid gland at the First Affiliated Hospital of Wenzhou Medical University from January 2012 to April 2021 were finally enrolled in this study. The total number is 484, of which the postoperative paraffin pathology results showed 347 were papillary thyroid carcinoma (PTC) and 137 were nodular goiter.
The related information on blood test results was partially missing in the medical records.
Patients who could not be scheduled for surgical treatment after admission due to the evaluation of unstable fasting serum glucose condition.
Patients with an unknown or unrecorded course of diabetes and specific medication regimen.
Patients with a history of thyroid surgery or complicated with other malignant tumors.
Patients with hyperthyroidism or baseline infectious or known inflammatory conditions (except for thyroid autoimmune inflammation, such as Hashimoto thyroiditis (HT)).
To randomly divide all 484 patients into two groups, the R software (package, seed 1988, 70%) was used: the training group (339 patients, 70%) and the internal validation group (145 patients, 30%). The integration of the data and comparison between the two groups are shown in Table 1. There was no statistical difference between the two groups in terms of clinicopathological characteristics.
Baseline characteristics of the training data set and the internal validation data set.

Abbreviations: AGR, albumin-globulin ratio; BMI, body mass index; FSG, fasting blood sugar; HDL, high-density lipoprotein; LDL, low-density lipoprotein; LMR, lymphocyte-to-monocyte ratio; MHR, monocyte-to-high-density lipoprotein cholesterol ratio; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PNI, prognostic nutrition index; TC, total cholesterol; TG, triglyceride; TSH, thyroid-stimulating hormone; Y, years.
Data collection
All patients underwent a well-established preoperative assessment within the first week after being admitted to the hospital, including blood routine examination, blood biochemical examination, coagulation function test, cervical computed tomography (CT) scan, thyroid ultrasound, etc. And all the needed clinicopathological information of patients enrolled in this study was collected: baseline demographic characteristics were obtained including age, sex, BMI, duration of diabetes, and so were medical history (containing the diabetes medicine), the largest nodule size, laterality, multifocality, and presentation of chronic lymphocytic thyroiditis according to the thyroid ultrasound together with cervical CT We also collected the surgical records (including postoperative paraffin pathology results). The formula for calculating MHR and LMR:
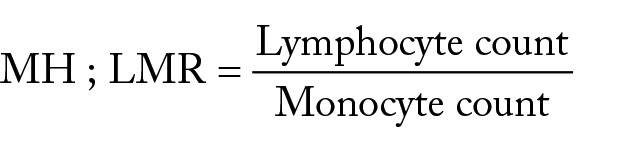
Statistical analysis method
The R software (package, seed 1988, 70 %) was used to randomly divide all patients into two groups. Continuous variables were expressed as medians and quartiles, and categorical variables were indicated with the number of cases, and percentages (%). T-test or Mann-Whitney U test was used for the comparison of data between two groups (depending on whether the data are normally distributed). And the chi-squared test was used to compare the rates. We then performed the receiver operating characteristic curve (ROC) to obtain the optimal cutoff values of the continuous variables. Furthermore, we stratify the patients by the most objective cutoff value calculated to simplify the nomogram. Based on the P-value of variables in the univariate analysis (P < .1), multivariate analysis using the Logistic regression model with forwarding elimination was performed. Odds ratios (ORs) and 95% confidence intervals (CIs) along with P-value were calculated afterward. Variables with a P-value smaller than .05 in the multivariate analysis were then selected for constructing the nomogram. The area under the curve of the ROC curve was calculated in the training group and the internal validation group respectively to quantify the prediction efficiency of the nomogram. We then performed the calibration curves to evaluate and visualize the consistencies between the predicted ones and the actual observed situation. To determine the clinic value of applying the nomogram in daily medical practice, the DCA curve was plotted to quantify the net benefit patients could get at disparate threshold probabilities. A criterion of two-sided P < .05 was considered to be statistically significant. R software (version 4.0.3) was used to finish all the statistical analyses aforementioned.
Results
In our research, a total of 484 participants with T2DM were included. The collected data were then randomly divided into two groups: the training data set consisting of 339 cases (70%), and the internal validation data set consisting of 145 cases (30%). Among the patients in the training data set, approximately 41% were 55 years old or younger, while the internal validation data set had 35.9% of patients in the same age group. The ratio of males to females in both the training and internal validation data sets was similar, with approximately 1:2. No significant differences were observed between the two groups. The summarized data are presented in Table 1.
The choice of optimal cutoff value
To achieve the best balance between sensitivity and specificity, ROC analysis was conducted to determine the optimal cutoff points for several variables, namely fibrinogen, LMR, MHR, TC (total cholesterol), TG (triglyceride), and FSG (fasting blood sugar). The objective cutoff values obtained for these variables, indicating the point at which they are most effective in predicting the malignancy of TNs, were found to be 2.835, 6.225, 0.707, 4.5, 4.14, and 8.25, respectively.
Selection of predictive factors associated with the malignancy of TN
Based on the results of the univariate analysis, several factors were found to be statistically associated with the malignancy of TNs. These factors include age (P = .008), largest tumor size (P = .000), low-density lipoprotein cholesterol (LDLC; P = .015), TC (P = .004), TG (P = .001), glucose (P = .000), fibrinogen (P = .009), LMR (P = .017), and MHR (P = .009), as shown in Table 2. However, due to strong collinearity with TC, LDLC was excluded from the multivariable COX analysis.
Univariate and multivariate analysis of logistic regression in the training data set.

Abbreviations: AGR, albumin-globulin ratio; BMI, body mass index; CI, confidence interval; FSG, fasting blood sugar; HDLC, high-density lipoprotein cholesterol; LDLC, low-density lipoprotein cholesterol; LMR, lymphocyte-to-monocyte ratio; MHR, monocyte-to-high-density lipoprotein cholesterol ratio; NLR, neutrophil-lymphocyte ratio; OR, odds ratio; PLR, platelet-lymphocyte ratio; PNI, prognostic nutrition index; TC, total cholesterol; TG, triglyceride; TSH, thyroid-stimulating hormone; Y, years.
P-value of this variable is statistically significant.
To simplify the regression model and the resulting nomogram, the significant continuous variables identified in the univariate analysis were converted into categorical variables based on the cutoff values obtained through ROC analysis.
Subsequently, a multivariate logistic regression analysis was performed, incorporating these factors. The results indicated that age (> 55 years; OR = 2.253, 95% CI: 1.264-4.017, P = .006), high level of TC (> 4.5; OR = 2.005, 95% CI: 1.089-3.690, P = .025), high level of TG (> 4.14; OR = 2.335, 95% CI: 1.092-4.994, P = .029), high level of fasting blood glucose (> 8.25 mmol/L; OR = 2.758, 95% CI: 1.590-4.785, P = .000), fibrinogen (> 2.835; OR = 2.461, 95% CI: 1.048-5.776, P = .039), LMR (> 6.225; OR = 0.424, 95% CI: 0.234-0.767, P = .005), and MHR (> 0.707; OR = 0.225, 95% CI: 0.090-0.562, P = .001) were identified as independent predictive factors for malignancy in TNs among patients with T2DM, as shown in Table 2.
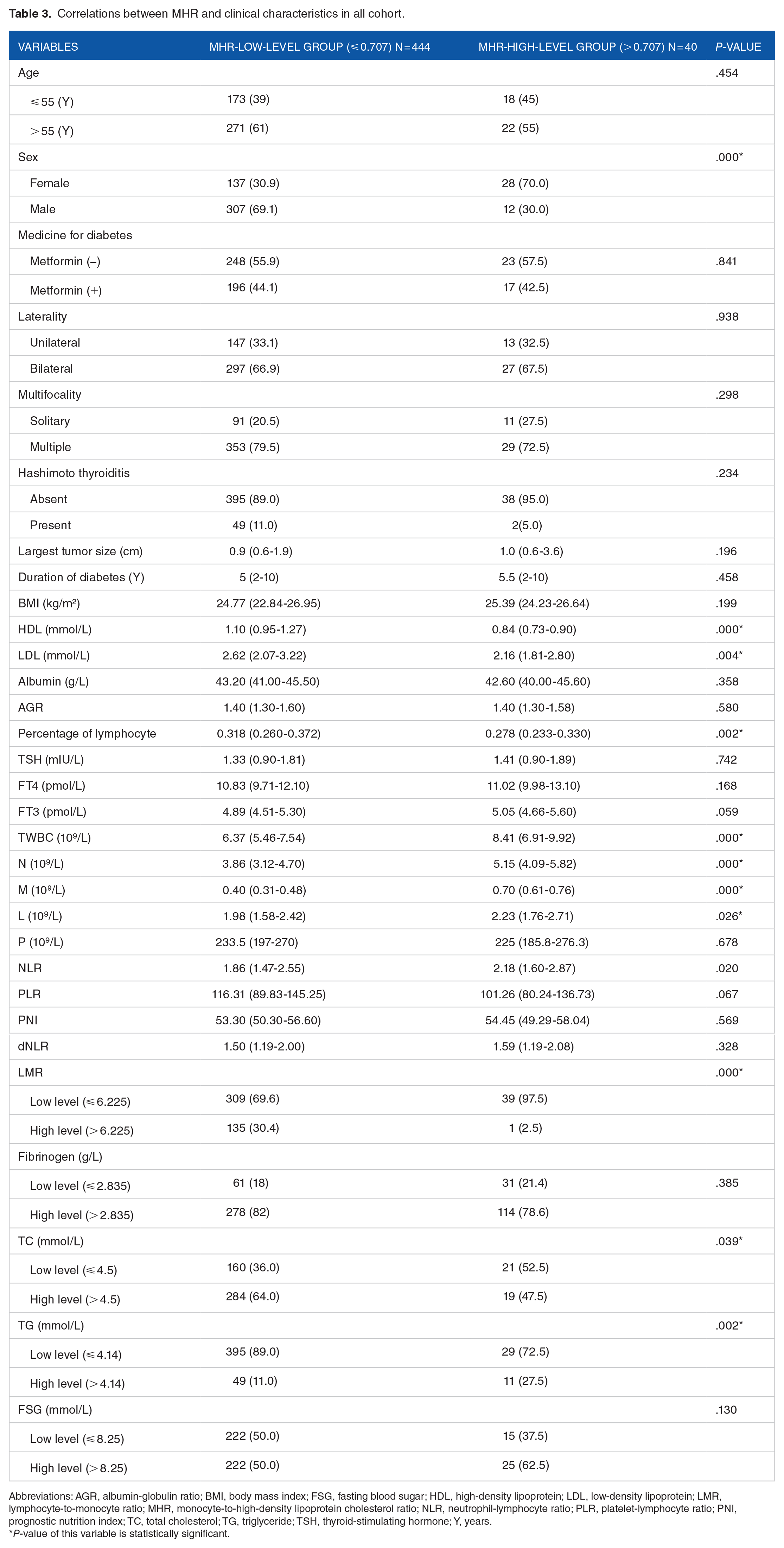
Correlation between clinical characteristics and MHR in all cohorts
Based on the optimal cutoff value of MHR, we obtained using ROC analysis, we then divided the whole cohort into two groups: Group A, low MHR level (⩽ 0.707), including 444 patients (91.74%); Group B, high MHR level (> 0.707), including 40 patients (8.26%); Table 3 indicates that the level of MHR was significantly associated with sex (P = .000*), high-density lipoprotein (HDL; P = .000*), LDL (P = .004*), Percentage of lymphocyte (P = .002*), TWBC (P = .000*), N (P = .000*), M (P = .000*), L (P = .026*), TC (P = .039*), TG (P = .002*). Male patients reported a bigger proportion in the low-MHR-level group, which is contrary to the situation in the high-MHR-level group. The higher MHR level was correlated with a higher level of TWBC, N, M, L, and TG, as well as a lower level of TC, HDL, LDL, and percentage of lymphocytes.
Correlations between MHR and clinical characteristics in all cohort.
Abbreviations: AGR, albumin-globulin ratio; BMI, body mass index; FSG, fasting blood sugar; HDL, high-density lipoprotein; LDL, low-density lipoprotein; LMR, lymphocyte-to-monocyte ratio; MHR, monocyte-to-high-density lipoprotein cholesterol ratio; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PNI, prognostic nutrition index; TC, total cholesterol; TG, triglyceride; TSH, thyroid-stimulating hormone; Y, years.
P-value of this variable is statistically significant.
In addition, we distributed all cohorts into quarters according to the value of MHR. As a result, we found the proportion of goiter patients rose from about 20.66% to 34.71% along with the elevation of MHR level and that of PTC patients decreased reversely from approximately 79.34% to 65.29%, as shown in Figure 1A. To take a step further, we constructed the histogram to compare the MHR level between PTC and goiter in the training data set and internal validation data set, respectively. And the results showed that the average level of MHR in goiter patients is higher than that in PTC patients in both groups, though the P-values (.500, .374, respectively) showed there were no statistically significant differences in both groups, as shown in Figure 1B.

(A) The proportion of goiter patients rose from about 20.66% to 34.71% with the elevation of MHR level and that of PTC patients decreased reversely from approximately 79.34% to 65.29%. (B) The average level of MHR in goiter patients is higher than that in PTC patients in both groups, P-values (.500, .374, respectively). (C) The independent predictive factors in distinguishing the malignancy of thyroid nodules were further incorporated into the nomogram.
Evaluation of the nomogram
The independent predictive factors identified for distinguishing the malignancy of TNs were used to construct a nomogram, as shown in Figure 1C. To assess the performance of the nomogram, ROC analysis was conducted, resulting in a C-index of 0.901 in the training data set and 0.760 in the internal validation data set, as depicted in Figure 2A and B, respectively. The calibration curves demonstrated strong agreement between the predicted values and the actual outcomes, as shown in Figure 3A and B. In addition, decision curve analysis (DCA) indicated that utilizing this nomogram would provide greater clinical net benefit within a threshold probability range of approximately 10% to 90% in the training data set and 10% to 75% in the internal validation data set, as illustrated in Figure 3C and D.

(A and B) ROC analysis and the C-index of the nomogram in the training data set was 0.901, and 0.760 in the internal validation data set.

(A and B) The predictive values and actual situation represented in the calibration curves. (C and D) DCA showed nomogram will help patients get more clinical net benefit within a range of the threshold probability between 10% and 90% in the training set and 10% and 75% in the internal validation set.
Discussion
In this research, we identified the MHR as a potential biomarker for distinguishing the malignancy of TNs in patients with T2DM. This finding, which is being proposed for the first time, suggests that MHR might reflect the interplay between host oxidative stress and malignancy.
TG is converted from fatty acids and stored as the major form of circulating energy in adipose tissues. It is nowadays acknowledged as a key component of metabolic syndrome and discovered to have a strong connection with obesity. Obesity, which is a growing public health concern, has been reported in the observational study as being a risk factor for malignancy in patients presenting with TNs. 13 Significant associations between the high levels of TG and an increased risk of malignant tumors in studies regarding esophageal, colorectal, breast, kidney, and pancreatic have been discovered.14,15 In this study, we found TG high level is an independent risk factor for malignancy in patients with TN, which is consistent with the former study.
T2DM, formerly known as non-insulin dependent diabetes (NIDDM) or adult-onset diabetes, is a chronic metabolic disorder, which usually occurs after the age of 35 to 40, accounting for more than 90% of diabetes patients. The patient is characterized by high blood sugar, a relative lack of insulin, and insulin resistance. Common symptoms include polydipsia, frequent urination, unexplained weight loss, and may also include overeating, fatigue, or soreness. Long-term complications caused by hyperglycemia include heart disease, stroke, and diabetic retinopathy, which may lead to blindness, kidney failure, and even limb amputation due to poor blood flow. Diabetic ketoacidosis is rare.
An area of recent interest is the possible role of cholesterol in carcinogenesis and tumor progression. Moses et al demonstrated the causative link between increased LDL and a higher probability of positive prostate biopsy in black Americans. 16 Several in vitro or in vivo studies have implied that statins do have the antitumor potential.17,18 Investigators consequently postulated that this is due to a reduction in the downstream targets of hMG Co-A reductase metabolism. 19 However, all the studies concerning stain drugs cannot exclude the confounding effect of hypercholesterolemia, and the antitumor potential may be explained by the benefit of LDL reduction. In this study, we suggested that the high level of TC (> 4.5 mmol/L) is also an independent risk for malignancy in TN patients complicated with T2DM.
The former study suggested the PTC risk was positively associated with the FSG levels in all study participants. 20 Furthermore, Jonathan et al demonstrated in their study that the odds of thyroid cancer increased in the highest quartile for genetic liability of T2DM. And the genetic variants that are assigned at conception, unlike observed risk factors, are not susceptible to confounding by other risk factors. 21 In this study, we found that the higher level of fasting serum glucose (> 8.25 mmol/L) is also an independent risk for malignancy in TN patients complicated with T2DM.
The coagulation cascade and systemic inflammatory response play important roles in the process of tumor progression and metastasis. 22 Hu et al reported that hyperfibrinogenemia was a risk factor for PTC recurrence and more aggressive tumor characteristics. 23 Patients with high fibrinogen levels had a significantly worse prognosis than those with low fibrinogen levels in advanced esophageal squamous cell carcinoma. 24 PD (Progressive Disease group) had a significantly higher fibrinogen level than the non-PD group in advanced gastric cancer after first-line chemotherapy or chemoradiotherapy. 25
In a recent study, certain simple as well as cost-effective systemic inflammatory markers, including the lymphocyte-to-monocyte ratio (LMR), have been developed and established links with various human cancers.26-28 Song et al found that the lower level of preoperative LMR was an independent risk factor for a worse prognosis. 29 In this study, we did not focus on the postoperative prognosis of patients, however, we discover a significant difference in the LMR levels in the goiter group and PTC group and the lower LMR level is a validated sign of the higher incidence of malignancy.
Monocytes, critical participants in the inflammatory pathway, are also acknowledged as having an important role in constructing the bridge between the inborn and adaptive immunity. 30 On the contrary, the effects of monocytes on pro-inflammatory and pro-oxidants can be counteracted by HDLC. 31 Thus, monocytes and HDLC together can be combined as a single ratio representing a promising indicator of inflammation and oxidative stress. Most of the existing research focuses on the association between MHR and cardiovascular events, which differs from LMR. 32 Only one study can we find focusing on the link between MHR and TN, of which the result demonstrated the presence of TN and the bigger size of existing nodules can increase the level of MHR in both genders. 12 Intriguing questions are raised by the good performance of MHR concerning the association between inflammation and TN. Because patients with goiter were not usually recommended at outpatient to be cheated with surgery intervention immediately unless the nodule is big enough to be a causative role for clinical symptoms including trachea suppression or dysphagia. Thus, the average nodule largest sizes of patients with goiter enrolled in this study were significantly bigger than those of patients with PTC, which led to a selection bias. And in this study, the average level of MHR in the goiter group is much higher than that in the PTC group, which might be the goiter group’s bigger average nodule size. Furthermore, to exclude the affection of selection bias, all the significant factors in the univariate analysis were further incorporated into the multivariate analysis apart from the largest tumor size.
It is important to acknowledge certain limitations of this study. First, there may be a selection bias concerning the size of the tumor, as the surgery decision differs between PTC and goiter cases. In addition, some crucial information related to diabetes was unavailable, such as HbA1C (%) and proinsulin C-peptide, as they are not routinely tested in admitted patients before thyroid-related surgery. Moreover, to confirm the findings of our study, a larger prospective study is required.
Conclusions
To sum up, the association of MHR with sex, serum cholesterol levels, and peripheral blood cell counts has been observed. MHR shows potential as a novel biomarker to differentiate between PTC and goiter in patients with T2DM. However, further confirmation of these findings and a deeper understanding of the underlying mechanisms are needed through prospective studies involving larger participant numbers.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
The article was written by YH and YS. Raw data collection and analysis were carried out by EX and SH. AB provided assistance in revising the article. The overall study design was conceptualized by AB and ZJ.
Ethics Approval and Informed Consent
The clinical ethics committee approval was given by the ethics committees of the First Affiliated Hospital of Wenzhou Medical University (YS2021-169). All enrolled individuals have signed the written informed consent including those participating in this study. This study was conducted in light of the Declaration of Helsinki.
